Abstract
Background:
High-definition transcranial electrical theta burst superimposing direct current stimulation (HD-tDCS-eTBS) not only incorporates the therapeutic advantages of tDCS and TBS but enhances stimulation focality and practicality. However, the applicability of this innovative neuromodulatory device in post-stroke rehabilitation remains uncertain.
Objective:
This study aimed to assess the efficacy and safety of the HD-tDCS-eTBS on upper extremity (UE) motor function in patients with chronic stroke.
Methods:
A patient-blinded, randomized controlled study was conducted. Twenty-four participants were randomly assigned into either the active HD-tDCS-eTBS group or sham HD-tDCS-eTBS group. Both groups received 20 minutes of active/sham HD-tDCS-eTBS combined with 30 minutes of conventional UE rehabilitation each time, 3 times a week for 4 weeks. Outcome measures including the Fugl-Meyer Assessment of Upper Extremity, Wolf Motor Function Test, Jebsen-Taylor Hand Function Test, Finger-Nose Test, and Modified Ashworth Scale were assessed before and immediately after the intervention period.
Results:
Spasticity of shoulder adductor (P = .05), elbow extensor (P = .04), and thumb flexor (P < .01) were significantly reduced in the active HD-tDCS-eTBS group versus the sham group. Nonsignificant trends in the improvements of most other outcome measures were in favor of the active HD-tDCS-eTBS group with moderate to large effect sizes (P = .06–.26, ηp2 = 0.06–0.16). No severe adverse events except for slight skin redness under the stimulus electrode was detected after the HD-tDCS-eTBS.
Conclusions:
Our findings support that HD-tDCS-eTBS is safe and has therapeutic potential for post-stroke UE motor rehabilitation.
Trial registration:
ClinicalTrials.gov (ID: NCT04278105).
Keywords
Introduction
Motor impairment is a common cause of disability after stroke, which place an onerous burden on family. Nearly half of the people with upper extremity (UE) motor impairment do not regain functional ability of their UE, and only a minor proportion achieve full recovery. 1 It is commonly known that recovery from stroke is a neuroplasticity-dependent process and the time window is limited. 2 Investigation on neuromodulatory devices that promote neuroplasticity are to increase the effectiveness of stroke rehabilitation for the UE and to accelerate UE motor recovery.
Transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) are emerging non-invasive techniques for modulating brain plasticity and have revealed significant beneficial effects on motor function post-stroke.3,4 Moreover, researchers have identified that tDCS and rTMS trigger the long-term potentiation- and long-term depression-like effects on post-synaptic transmitters.5,6 Theta burst stimulation (TBS; 3 pulses repeated at 50 Hz), an innovative rTMS protocol, has been demonstrated to be more efficient and efficacious than conventional rTMS, which induces long-lasting effects on cortical excitability after a shorter stimulation duration.7,8 It is acknowledged that non-invasive brain stimulation concurrently applied with rehabilitation training produces an enhanced neuroplastic effect.9 -12 However, rTMS is too bulky to be concurrently implemented during rehabilitation and may accidentally trigger a seizure, particularly in high-risk populations (e.g., stroke). On the contrary, a portable tDCS device can be safely and simultaneously coupled with rehabilitation therapy. A meta-analysis showed that applying stimulation during therapy yielded greater improvement than applying stimulation before therapy. 13 Therefore, tDCS probably offers more safety and flexibility for designing innovative neuromodulatory protocols for stroke rehabilitation.
Transcranial electrical theta burst superimposing direct current stimulation (tDCS-eTBS) was introduced to overcome the flaws of TBS and to enhance the neuromodulatory effect of tDCS. 14 TDCS-eTBS versus conventional tDCS induced larger amplitudes of motor-evoked potential lasting up to 30 minutes after stimulation in an animal study. 14 The advantageous impact of adding ipsilateral tDCS-eTBS to conventional rehabilitation on upper-extremity motor function was supported in patients with chronic stroke. 15 Nonetheless, the tDCS-eTBS using conventional tDCS montage, with normally sized 25–35 cm2 bipolar electrodes, 16 produces diffused distributions of current flow in widespread brain areas. The low spatial precision may induce large electrical field strength falling outside the electrodes.17,18
Recently, a novel high-definition tDCS-eTBS (HD-tDCS-eTBS) device was designed to optimize the therapeutic effect for patients with neuropsychological diseases. 19 The HD-tDCS-eTBS montage uses a 1 × 4 ring configuration with one anodal electrode placed on the cortical regions of interest and four return electrodes on the surroundings, which contributes to increased focality and density of the electrical current. 19 This pilot randomized controlled trial aimed to assess the efficacy and safety of the HD-tDCS-eTBS on UE motor rehabilitation in patients with chronic stroke.
Methods
Participants
Patients with chronic stroke were prospectively recruited from three teaching hospitals in northern Taiwan from March 2020 to March 2021. Patients admitted to the department of rehabilitation were recruited if they met the following five inclusion criteria: (1) aged ≥ 20 years; (2) diagnosed with ischemic or hemorrhagic stroke by a neurologist based on neuroimaging examination; (3) between 6 months and 5 years after a stroke onset; (4) had a Brunnstrom stage between III and V for the affected UE; (5) had no more than moderate spasticity in the affected UE (Modified Ashworth Scale ≤ 3). The exclusion criteria were as follows: (1) contraindications for transcranial electrical stimulation, including history of epilepsy, history of atrial fibrillation, metal implantations, cardiac pacemakers, convexity skull defects, elevated intracranial pressure; (2) skin hypersensitivity, contact dermatitis, allodynia, hyperalgesia, wounds, or sores on the scalp; (3) severe cognitive or psychiatric disorders (such as major depression, schizophrenia, organic brain syndrome, or substance abuse); (4) brain tumor; (5) contracture, rigidity, inflammation, or myositis ossificans on the affected UE; (6) rheumatic diseases (such as systemic lupus erythematosus, rheumatoid arthritis, or ankylosing spondylitis). Written informed consent was obtained from each participant after screening for eligibility. The trial was approved by the institutional review board of the hospitals. A sample size of 12 per group was adopted for this pilot trial. 20
Procedure
A patient-blinded, randomized controlled study was conducted. Participants were randomly assigned into either the active HD-tDCS-eTBS group or the sham HD-tDCS-eTBS group, based on block randomization with age as the stratification factor. Within each age stratum, the allocation ratio was 1:1 and the block size was four. Concealed allocation with opaque envelopes was performed based on a computer-generated random number list. Allocation was conducted by a research assistant who was not involved in the assessments or intervention provision. Patients were assessed by a non-blinded outcome assessor at baseline (pre-intervention) and within 7 days after the intervention finished (post-intervention). When assessing post-intervention outcomes, the outcome assessor did not refer to baseline scores, to minimize the risk of bias.
Interventions
Both groups received 20 minutes of active/sham HD-tDCS-eTBS addition to 30 minutes of UE rehabilitation on the affected side concurrently each time, three times a week for 4 weeks. The anodal electrode of HD-tDCS-eTBS was placed over the ipsilesional M1 (C3/C4) and the remaining four return electrodes over Cz, F3, T7, and P3 on the left hemisphere or Cz, F4, T8, and P4 on the right hemisphere. The schematic representation of HD-tDCS-eTBS intervention combined with UE rehabilitation is shown in Figure 1(a). The active HD-tDCS-eTBS was delivered at an intensity of 1.0 mA intermittent TBS-like waveform superimposed onto an intensity of 1.0 mA anodal direct current waveform. The sham HD-tDCS-eTBS was provided by a 10-second ramp up/down to the desired current intensity (the same as the active HD-tDCS-eTBS group) at the beginning and at the end of the 20-minute period, with other no current delivered during the session (Figure 1(b)). Figure 1(c) illustrates the simulation of the current distribution in the brain cortex of the HD-tDCS-eTBS.21,22
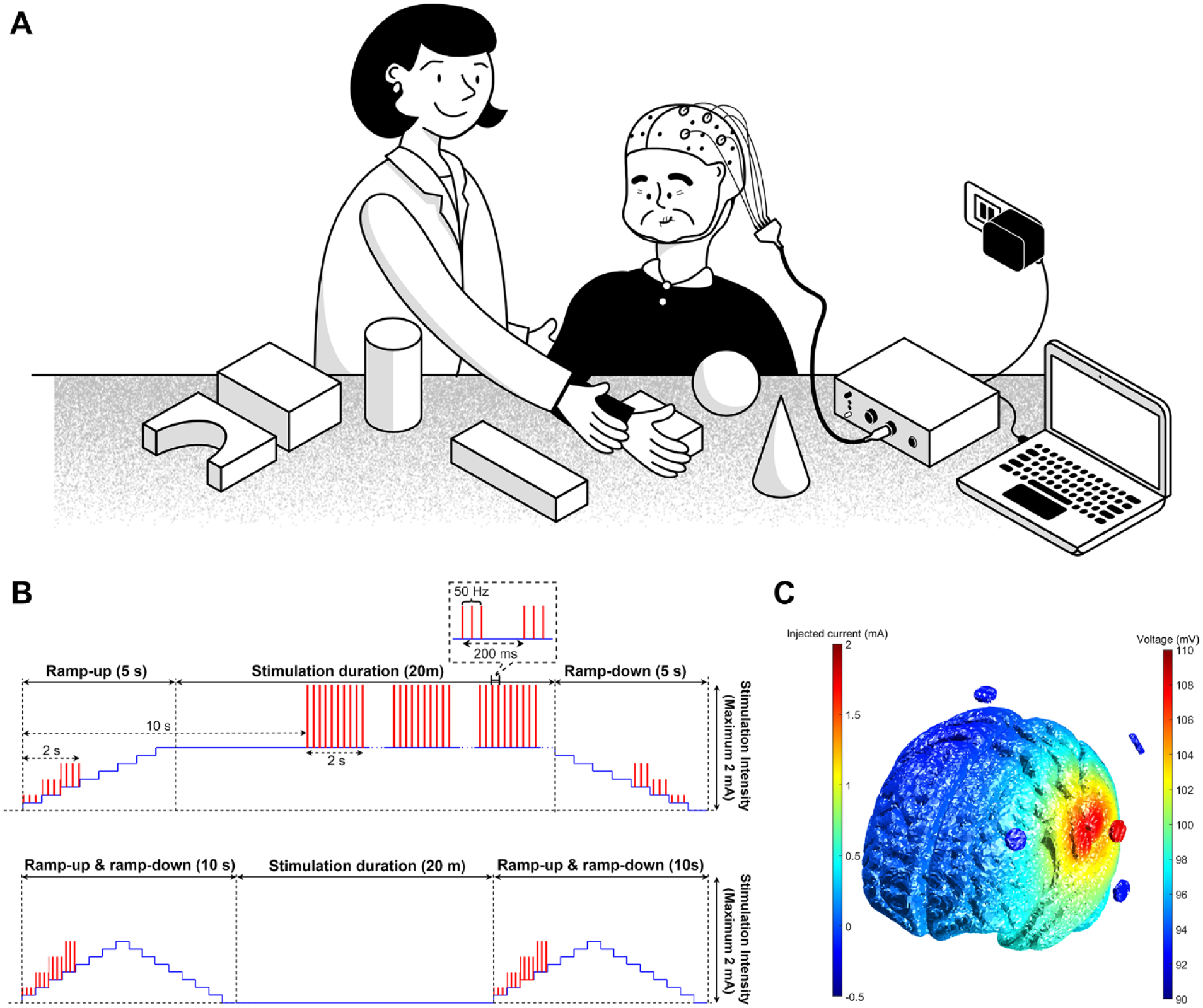
UE rehabilitation treatments included conventional occupational therapy strategies, such as passive or active range-of-motion exercises, muscle strengthening exercises, and gross or fine motor function training, which were conducted by certified occupational therapists unaware of the group assignment. These exercises or training included movements of all UE segments. A repetitive task-oriented training approach was taken, using movements such as reaching and weight bearing, in-hand manipulation skills, and activities related to daily living.
Outcome Measures
Fugl-Meyer Assessment of Upper Extremity (FMA-UE)
The FMA-UE is a stroke-specific and performance-based scale assessing voluntary movement of the UE. 23 The FMA-UE consists of 33 items, and each item is rated on a 3-point scale (0-1-2). The total score of the FMA-UE is 66, with higher scores indicating better movement ability. The FMU-UE is psychometrically sound. 24
Wolf Motor Function Test (WMFT)
The WMFT assesses UE motor ability through timed and functional tasks. 25 All tasks are performed as quickly as possible. The performance time, WMFT-TIME, is the median time required for all timed tasks executed and is truncated at 120 seconds. The functional ability, WMFT-FAS, is rated on a 6-point scale (0-1-2-3-4-5). The total score of the WMFT-FAS is 75, with higher scores indicating higher levels of functioning. The WMFT has sufficient validity, responsiveness, and reliability in patients with stroke. 26
Jebsen-Taylor Hand Function Test (JTT)
The JTT consists of seven items assessing hand functions commonly used in activities of daily living. 27 The items are performed as quickly as possible and are scored according to time taken to complete the task, with a maximum time of 120 seconds for each. Total score is the sum of the time taken for each item, with a shorter time indicating better performance. The reliability, validity, and responsiveness of the JTT are supported in the stroke population.27,28
Finger-Nose Test (FNT)
The FNT assesses the coordination of upper-extremity movement. 29 The patient alternately touches their nose and the assessor’s stationary finger as quickly and accurately as possible. The numbers of complete nose-target movements during a 1-minute period is recorded.
Modified Ashworth Scale (MAS)
The MAS is commonly used in clinical practice for grading spasticity. 30 The MAS is rated on a 6-point scale (0, 1, 1+, 2, 3, 4), and a higher score indicates that the patient has higher muscle tension. The muscles assessed in this study include shoulder abductor, elbow flexor, wrist flexor, and finger flexor. The MAS is responsive in detecting the changes in muscle tone in patients with stroke. 31
Safety
Serious adverse events (e.g., seizure) were monitored during the 12 intervention sessions. At the end of each intervention session, the scalp under the electrode site was checked for redness or skin damage. Patients were asked about the following side effects using a questionnaire regarding the incidence and severity they experienced after each active/sham HD-tDCS-eTBS intervention session: headache, neck pain, scalp pain, tingling, itching, burning sensation, sleepiness, difficulty concentrating, altered mood, or any other problems. The severity was rated as four levels (i.e., none, mild, moderate, or severe).
Statistical Analysis
Statistical analyses were performed using SPSS software ver. 22.0 (IBM, Armonk, NY, USA). The absolute values of skewness and kurtosis of all the outcome variables within each group conformed to the acceptable normality range of < 3 and <10, respectively. 32 The baseline characteristics between groups were compared using chi-squared tests for categorical variables and independent t tests for continuous variables.
The changes within each group were analyzed using paired t tests. The response rate was calculated by the percentage of participants whose improvement exceeded the minimal clinically important difference of the outcome measures. The minimal clinically important difference of the FMA-UE, WMFT-TIME, WMFT-FAS, JTT, and MAS were 5.25 points, 1.64 seconds, 0.33 points, 54.67 seconds, and 0.48 points, respectively.31,33-35 For the FNT, due to the lack of evidence on the minimal clinically important difference, successful treatment was defined as an improvement greater than half a standard deviation of the baseline scores of all participants. 36
The differences in post-intervention scores between the two groups were compared with adjustment for baseline scores using ANCOVA. The significance level was set at 0.05. A small, medium, and large effect was represented by a partial η2 (ηp2) ≥ 0.01, 0.06, and 0.14, respectively. 37
The safety outcome was calculated by the frequency of each serious adverse event and side effect.
Results
Figure 2 shows the flow chart of the participants’ enrollment in the study. A total of 24 eligible and consenting patients with stroke were randomly allocated to either the active HD-tDCS-eTBS group (n = 13) or the sham HD-tDCS-eTBS group (n = 11) and completed the trial. Details on the demographic data and clinical characteristics of the participants are shown in Table 1 and Supplemental Table 1. No significant differences between the two groups were found in terms of age (P = .40), gender (P = .07), time since stroke (P = .64), type of stroke (P = .68), lesioned hemisphere side (P = 1.00), or Brunnstrom stage (P = .43). The baseline scores of the outcome measures were not significantly different between the two groups (P = .10–.79). Several patients were on stable regimens of traditional Chinese acupuncture (6 and 5 patients in the active and sham groups, respectively) or other medications, including the following proconvulsant and CNS-acting agents: baclofen for muscle relaxation (1 patient in the sham group), piracetam for increase of regional cerebral blood flow (1 patient in each group), Seroquel for anti-insomnia (1 patient in the sham group), and sertraline for anti-depression (1 patient in the active group).
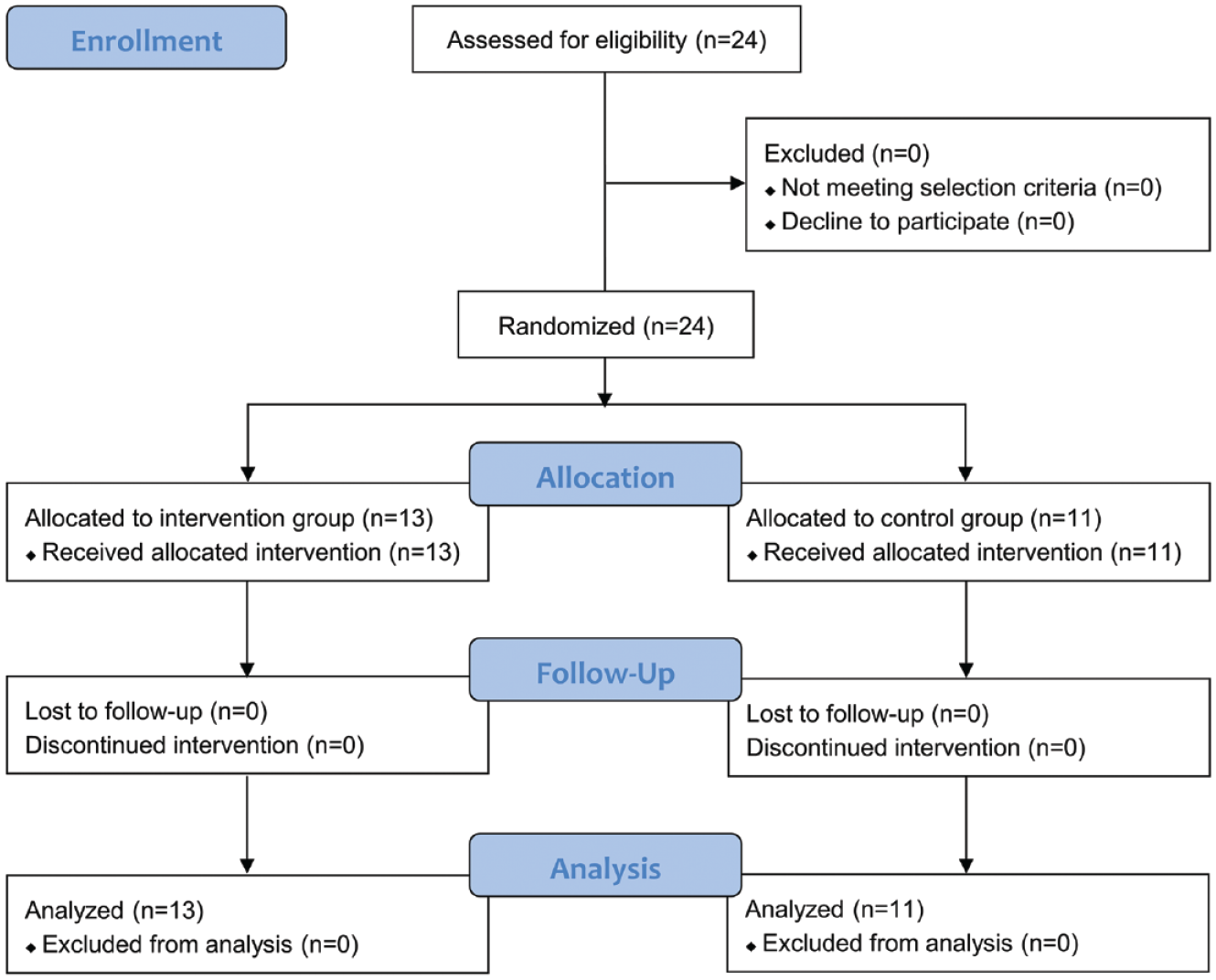
Flowchart of the trial.
Comparison of Demographics and Clinical Characteristics between Treatment Groups at Baseline.
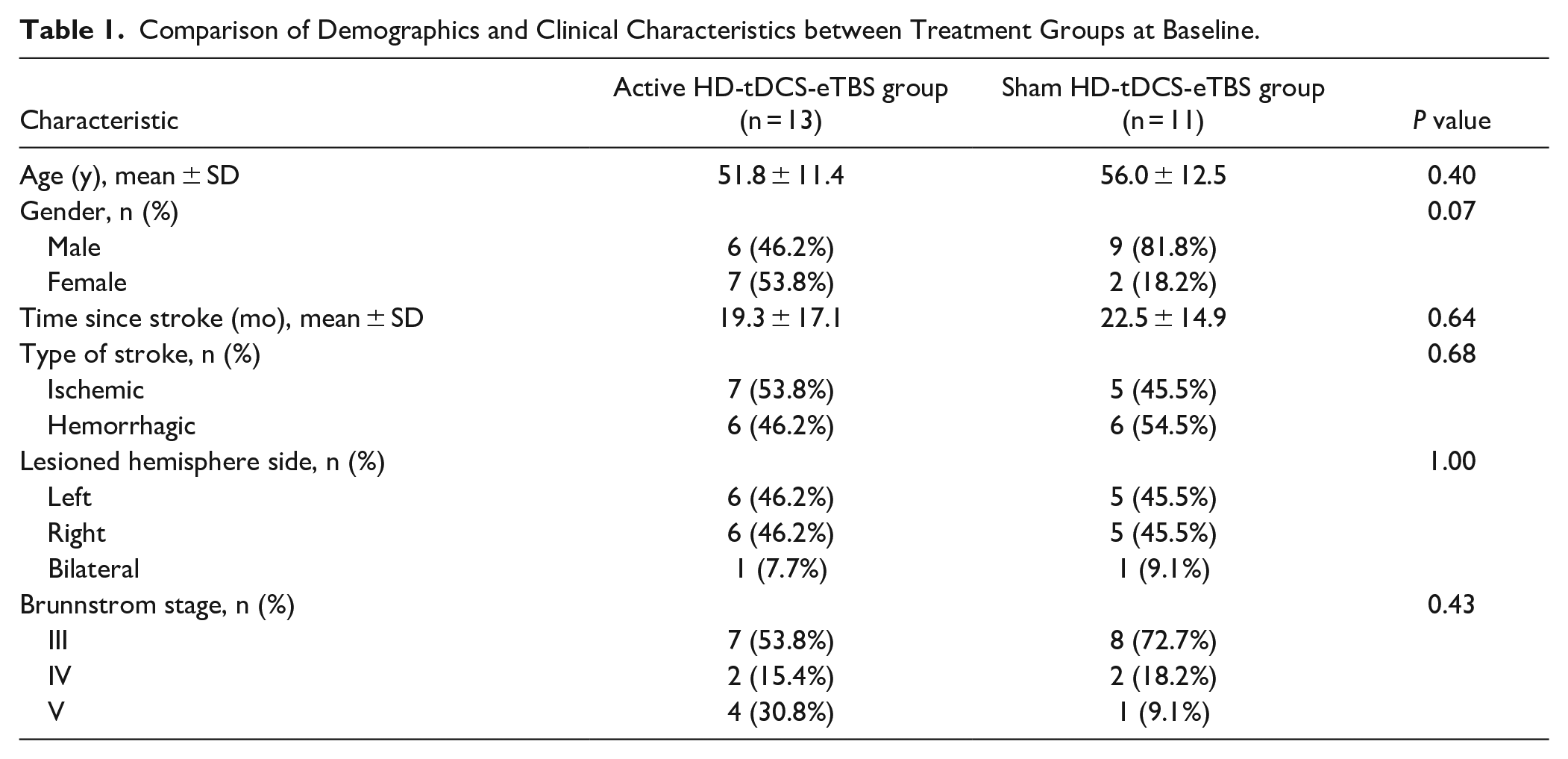
Figure 3 displays the response rates of the two groups, and Table 2 shows the descriptive data of all outcome measures at baseline and post-intervention, as well as the within-group and between-group differences. Each individual’s response pattern across each time point is presented in the Supplemental Figure 1. For the FMA-UE, the active HD-tDCS-eTBS group showed a significant change after intervention while the sham group did not. The response rates of the active group were more than twice that of the control group (38.5% vs. 18.2%). ANCOVA analysis revealed a nonsignificant trend in FMA-UE improvement in favor of the active HD-tDCS-eTBS group with a moderate effect size (P = .09, ηp2 = 0.13).
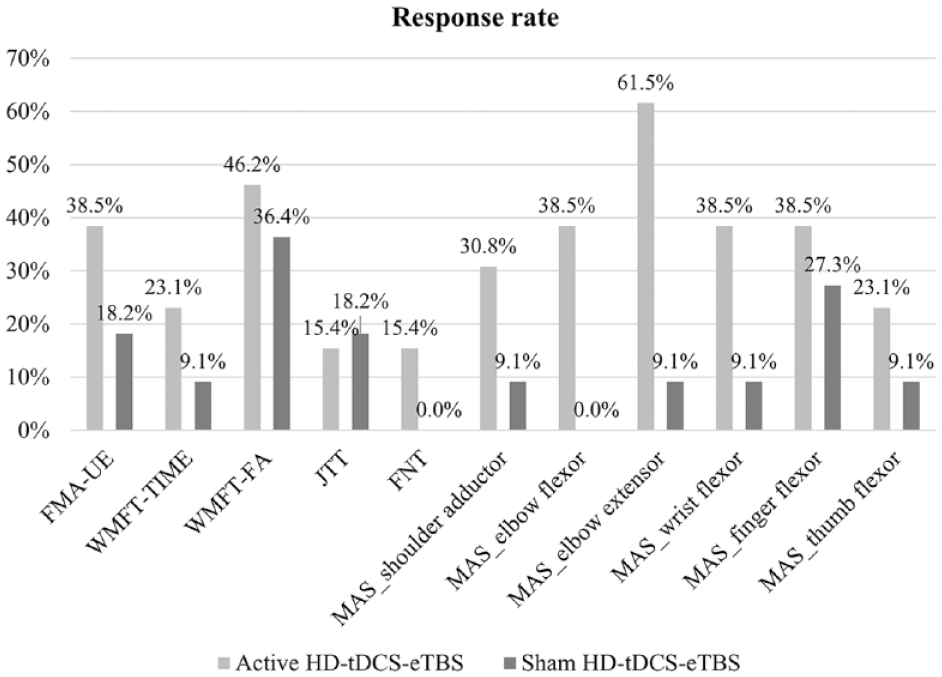
Response Rates of the Two Groups. HD-tDCS-eTBS, high-Definition Transcranial Electrical Theta Burst Superimposing Direct Current Stimulation; FMA-UE, Fugl-Meyer Assessment of Upper Extremity; WMFT-TIME, Wolf Motor Function Test performance time; WMFT-FAS, Wolf Motor Function Test functional ability; JTT, Jebsen-Taylor Hand Function Test; FNT, Finger-Nose Test; MAS, Modified Ashworth Scale.
Efficacy Outcomes.
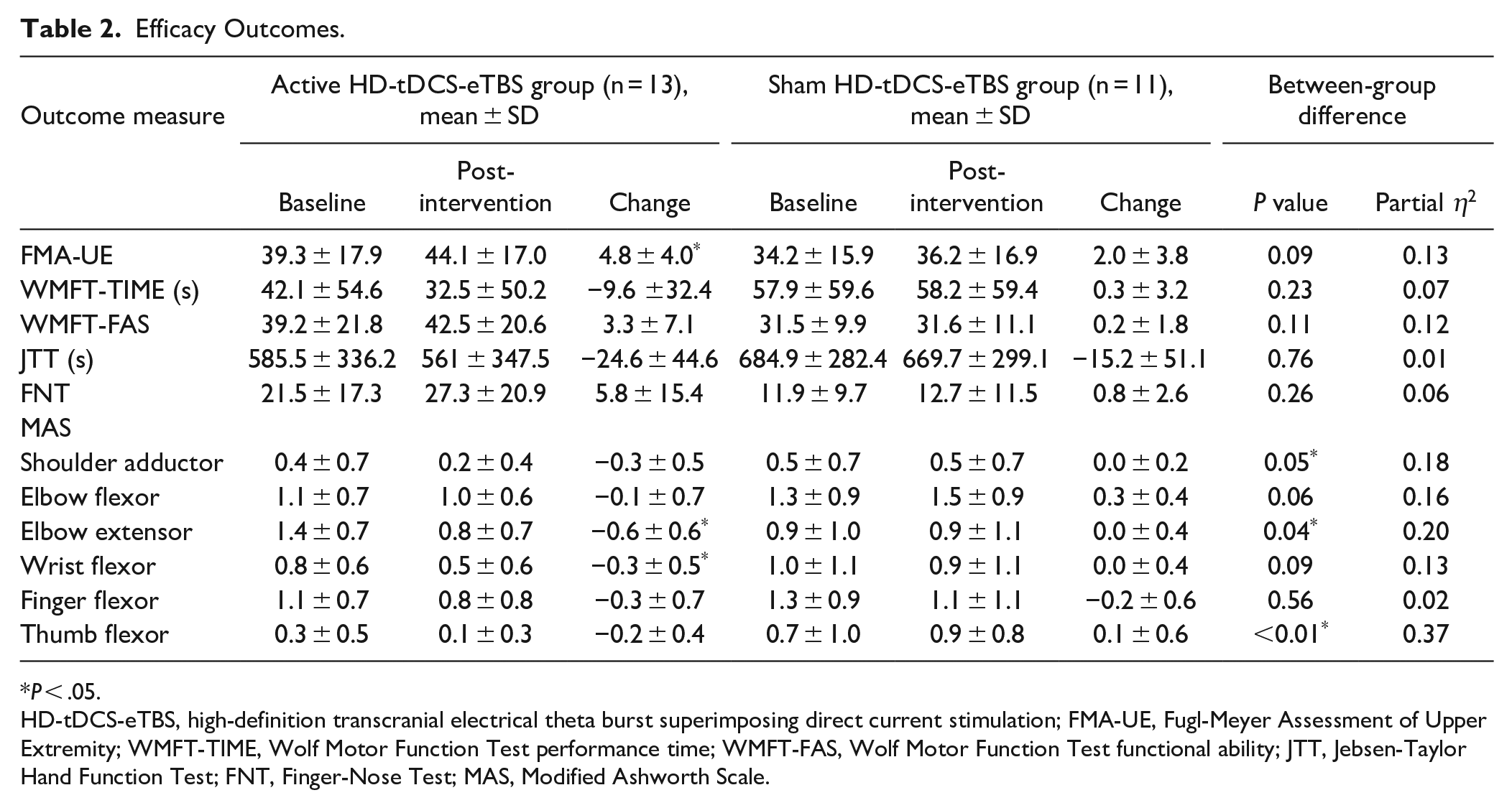
P < .05.
HD-tDCS-eTBS, high-definition transcranial electrical theta burst superimposing direct current stimulation; FMA-UE, Fugl-Meyer Assessment of Upper Extremity; WMFT-TIME, Wolf Motor Function Test performance time; WMFT-FAS, Wolf Motor Function Test functional ability; JTT, Jebsen-Taylor Hand Function Test; FNT, Finger-Nose Test; MAS, Modified Ashworth Scale.
Regarding WMFT, nonsignificant changes after intervention were found in both groups regardless of the performance time or functional ability. The response rates of the WMFT-TIME and the WMFT-FAS were higher in the active group than those in the sham group (23.1% vs. 9.1% and 46.2% vs. 36.4%). Nonsignificant trends in treatment effects favoring the active HD-tDCS-eTBS group on the WMFT-TIME (P = .23, ηp2 = 0.07) and WMFT-FAS (P = .11, ηp2 = 0.12) were found with moderate effect sizes.
Both groups did not show significant reductions in the total time of the JTT after intervention and had similar response rates (15.4% in the active group and 18.2% in the sham group). Between-group differences were nonsignificant (P = .76) and small (ηp2 = 0.01).
The improvement in the FNT did not achieve statistical significance within each group. Patients of 15.4% experienced clinically important improvement in the active HD-tDCS-eTBS group; while the sham HD-tDCS-eTBS group had none. A nonsignificant effect on FNT improvement was found for the active HD-tDCS-eTBS group with a moderate effect size compared with the sham group (P = .26, ηp2 = 0.06).
Concerning the MAS, significant reductions in the spasticity of the elbow extensor and wrist flexor in the active HD-tDCS-eTBS group were found, but the other muscles did not achieve statistical significance in either both groups. The active HD-tDCS-eTBS group, compared with the sham group, showed greater spasticity reduction in all of the UE muscles assessed. The response rates were between 23.1% and 61.5% in the active HD-tDCS-eTBS group and between 0.0% and 27.3% in the sham group. Among them, the treatment effects of the active HD-tDCS-eTBS on the spasticity of the shoulder adductor, elbow extensor, and thumb flexor were significantly superior to those of the sham group, with large effect sizes (ηp2 = 0.18–0.37).
No severe adverse events over the 12 intervention sessions occurred. Four participants (30.8%) in the active HD-tDCS-eTBS group were detected with slight skin redness under the stimulus electrode at the end of intervention. Two of them showed skin redness after each intervention session, and the others showed skin redness after 2 or 5 out of the 12 intervention sessions. In the sham group, slight itching on the scalp was noted in one participant (9.1%) after 1 out of the 12 intervention sessions. Slight skin redness was reported in one participant (9.1%) after 3 out of the 12 intervention sessions. These side effects vanished on their own in a few minutes. The other participants reported no side effects after each intervention session.
Discussion
To the best of our knowledge, this study is the first randomized controlled trial to assess the add-on effect of HD-tDCS-eTBS on post-stoke UE motor rehabilitation. We found that the HD-tDCS-eTBS remediated the spasticity of the shoulder adductor, elbow extensor, and thumb flexor significantly more than the sham stimulation. Nonsignificant trends in treatment effects in favor of the active HD-tDCS-eTBS were shown for voluntary UE movement, UE motor ability, hand function, and coordination of UE movement. Although the modest sample size in this pilot randomized trial may be unpowered to reveal statistical significance, 38 the moderate to large between-group effect sizes in most of the outcomes could support that HD-tDCS-eTBS accompanied with conventional rehabilitation reduces spasticity and improve voluntary UE movement in patients with chronic stroke.
Significant between-group differences on the MAS and a significant improvement on the FMA-UE in the active HD-tDCS-eTBS group were found in this pilot trial. However, there were no significant effects on UE motor ability, hand function, and coordination of UE movement. This finding may have resulted from the therapeutic effect of the 12-session, 2-mA HD-tDCS-eTBS which was insufficiently large to induce changes in UE functional capacity. Thus, the participants showed significant improvements only in basic motor components, such as spasticity and voluntary movements. Further studies are needed to investigate the optimal dosage of the HD-tDCS-eTBS.
Similar to our findings, a previous clinical trial found that tDCS-eTBS combined with conventional rehabilitation had a significant additive effect on post-stroke UE motor rehabilitation. 15 Furthermore, the long-term-potentiation-like after-effect of TBS over the human motor cortex were found to be modulated by concurrent tDCS, 39 and intermittent TBS combined with direction-of-electrical-field-matched tDCS generated greater potentiation in neuroplastic after-effect compared to intermittent TBS alone. 40 An animal study also exhibited that tDCS-eTBS versus tDCS alone induced larger amplitudes of motor-evoked potential. 14 Thus, these evidence supports that tDCS-eTBS may provide a more beneficial effect on neuroplasticity than tDCS alone or TBS alone. Further studies comparing the neuromodulatory effect among tDCS-eTBS, tDCS alone, and TBS alone are needed.
In addition to the tDCS-eTBS, HD montage may further enhance the neuromodulatory effect of electrical stimulation. Previous simulation work proposed that HD montage versus conventional bipolar rectangular-pad configuration enhanced spatial focality, with peak magnitude of electric field observed directly underneath the active electrode. 17 A human study further verified that transcranial electrical stimulation with an HD montage delivered focal cortical stimulation over specific target cortical areas. 18 These evidence support that the tDCS-eTBS with an HD montage may improve the focality of electric fields targeting the cortical area of interest. However, whether the focality of electric fields leads to behavioral effects needs further investigation.
The beneficial effect of HD-tDCS-eTBS on post-stroke UE motor rehabilitation may have resulted from the neuroplasticity induced by the electrical stimulation and burst stimulation. Both anodal tDCS and intermittent TBS have been shown to induce long-term-potentiation-like plasticity.41 -43 Anodal tDCS polarizes neuronal membranes and results in enhanced neuronal excitability. 41 The action potentials in presynaptic neurons generates signal transduction cascades and subsequently induces BDNF- and NMDA-dependent long-lasting synaptic potentiation in the brain cortex.5,44 Whereas intermittent TBS applies high-frequency bursts (3 pulses at 50 Hz) at a low-frequency interval (5 Hz) to depolarize neurons. Repeated bursts stimulating presynaptic neurons at such a high frequency induces sufficient postsynaptic depolarization to activate NMDA receptors and subsequently triggers long-term-potentiation-like changes in synaptic connections. 43 These neuroplastic mechanisms of anodal tDCS and intermittent TBS may produce an accumulative effect on neuroplasticity. However, the physiological effects of HD-tDCS-eTBS require further investigation.
No severe adverse events except for slight skin redness under the stimulus electrode was found after the HD-tDCS-eTBS. Such a side effect is known to be a common physical response immediately after electrical stimulation 16 and vanishes in a few minutes. The incidence of minor side effects were similar in the HD-tDCS-eTBS and sham groups. These results indicate that the HD-tDCS-eTBS can be safely implemented in chronic stroke populations in clinical settings. A systematic review concluded that conventional tDCS at intensities ≤4 mA for up to 40 minutes did not cause any severe adverse events and irreversible brain injury. 16 Moreover, HD-tDCS at up to 3 mA for a duration of 20–30 minutes was safe and tolerable for older adults. 45 Thus, HD-tDCS-eTBS at 2 mA lasting 20 minutes applied in this study is within the safety range.
Although the intervention protocol in this study was designed based on the safety standards of previous studies, the protocol may not yet be optimal for patients with stroke. The optimal dosage of stimulation required to reach a tradeoff between efficacy and safety should be determined. Furthermore, higher dosage may not necessarily result in larger cortical excitability enhancements. Increasing the intensity and/or duration of anodal tDCS beyond certain limits may reduce tDCS-induced excitability or even reverse the long-term potentiation- into long-term depression-like plasticity, as shown in previous studies.46 -48 A meta-analysis of multiple-session tDCS on post-stroke UE function concluded that increasing the number of sessions did not show a beneficial effect. 13 Thus, further studies to identify the optimal combination of stimulation parameters (e.g., intensity, duration, and number of sessions) that maximizes the neuromodulatory effect and safety are needed.
Several limitations were identified in this study. First, this study did not compare the effectiveness and safety of the HD-tDCS-eTBS with those of the tDCS-eTBS, tDCS, rTMS, and intermittent TBS. Thus, whether the HD-tDCS-eTBS has a superior neuromodulatory effect and safety than the other devices remains unknown. Second, the generalizability of the results was restricted by the modest sample size. Third, we excluded patients with severe UE impairment (Brunnstrom stage < III) or those with UE spasticity (MAS > 3) to avoid confounding the effect on voluntary movement recovery. Whether HD-tDCS-eTBS provides clinical gains in these populations is worthy of further investigation. Fourth, although all participants received identical amounts of conventional rehabilitation, the contents of these therapies were not controlled. Thus, the effect of conventional rehabilitation may consist inter-individual variability. Fifth, a single-blinded design was used in this study, which may have caused bias during intervention provision and outcome assessment. Sixth, patients with contraindications for transcranial electrical stimulation or psychiatric disorders were excluded from this study. Thus, the relationship between the incidence of side effects of the HD-tDCS-eTBS and these risk factors remains unknown. Last, we did not follow up on the long-term effect of the HD-tDCS-eTBS, whether the beneficial effect of the HD-tDCS-eTBS on post-stroke UE motor rehabilitation can be maintained remains unclear.
Conclusions
Accompanied with conventional rehabilitation, HD-tDCS-eTBS significantly reduced UE spasticity and had nonsignificant trends in treatment effects on voluntary UE movement, UE motor ability, hand function, and coordination of UE movement in patients with chronic stroke. In addition, the HD-tDCS-eTBS at 2 mA lasting 20 minutes was shown to be safe for clinical application. These findings support the therapeutic potential of the HD-tDCS-eTBS in post-stroke rehabilitation. Large-scale studies with long-term follow-up are needed to further validate the immediate and longitudinal effects of HD-tDCS-eTBS. Future studies optimizing the stimulation protocol and investigating the neurophysiological mechanism of HD-tDCS-eTBS are warranted.
Supplemental Material
sj-pdf-1-nnr-10.1177_15459683221121751 – Supplemental material for High-Definition Transcranial Direct Current with Electrical Theta Burst on Post-Stroke Motor Rehabilitation: A Pilot Randomized Controlled Trial
Supplemental material, sj-pdf-1-nnr-10.1177_15459683221121751 for High-Definition Transcranial Direct Current with Electrical Theta Burst on Post-Stroke Motor Rehabilitation: A Pilot Randomized Controlled Trial by Yi-Jing Huang, Shun-Min Wang, Chieh Chen, Chien-An Chen, Chun-Wei Wu, Jia-Jin Chen, Chih-Wei Peng, Che-Wei Lin, Shih-Wei Huang and Shih-Ching Chen in Neurorehabilitation and Neural Repair
Footnotes
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Chih-Wei Peng and Shih-Ching Chen have patents relevant to the work (EP3106202, US10543367, and I573606). The other authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology (MOST-108-2321-B-038-002 and MOST-109-2321-B-038-002). The Ministry of Science and Technology did not have a role in designing the study or writing the manuscript.
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
