Abstract
Introduction
Muscle weakness is a common symptom in Parkinson’s and several studies have reported weakness in people with Parkinson’s (PwP). 1 Muscle strength has been measured in PwP using both isokinetic and isometric methods. Isometric assessment is considered to give a more accurate measurement of maximum strength since the lack of required movement reduces the impact of bradykinesia on reported values, provided the participant is given sufficient time to generate the force. 2 Grip strength is an isometric measure of muscle strength and recommended as a simple assessment suitable for use in clinical settings, 3 yet has been relatively little explored in Parkinson’s. Grip strength and leg strength are highly correlated, 4 and weak grip strength is associated with poor current and future health in middle-aged and older adults without Parkinson’s living at home and in acute and rehabilitation hospital settings.5-7 In epidemiological studies, it is recognized that men have stronger grip than women, and other known determinants of low grip strength include older age, smaller body size, impaired physical and cognitive function, and poor nutritional status. 8
However, studies in Parkinson’s to date have simply reported average muscle strength from different groups of PwP, sometimes age and sex matched but always without adjusting for body size, physical function, cognitive function, and nutritional status. Thus, these potential confounding factors can obscure the true relationship between muscle weakness and specific factors associated with Parkinson’s such as disease severity and duration. The lack of adjustment for these factors also prevents accurate comparison of the values reported from studies in different samples. This is important as physical therapy strategies, particularly progressive resistance exercise, are reported to have benefits on muscle strength in Parkinson’s 9 as well as improvements in posture, balance, and gait.10,11
The objective of this novel study was, for the first time, to explore the association of grip strength with disease severity and duration of Parkinson’s after adjustment for the impact of these known influences.
Methods
Participants
This observational cross-sectional study was conducted in one town in England. A letter of invitation was sent to all patients living in the town aged 50 years and over known to the Parkinson’s specialist nurse, including care home residents, with a reply slip to return if they were interested in participating. Participants had had a working diagnosis of Parkinson’s for at least 6 months. Those who did not reply were contacted by telephone once to check whether they wished to participate or not. Exclusion criteria included inability to give written informed consent or hold a dynamometer. The study was approved by the local research ethics committee, and written informed consent was obtained from all participants.
Data Collection
One researcher assessed all participants in the afternoon when they were in the “on” state (determined by participants’ self-report). Participants’ demographic details, comorbidities, and current medications were abstracted from their clinical records. Height was calculated from forearm length (cm),
12
current weight was assessed to the nearest 1 kg in light clothing on calibrated scales, and body mass index (BMI) was calculated. Grip strength was measured 3 times with each hand using a Jamar hand dynamometer (Promedics, Blackburn, UK) according to a standard protocol with standardized encouragement.
13
The overall maximum grip strength was recorded to the nearest 1 kg, as well as the maximum grip of the weaker hand. The test–retest coefficient (95% confidence intervals) for the assessor was 0.8 kg (−2.5, 4.2),
Statistical Analyses
The data were double entered and prepared for analysis with the Stata statistical software package, release 12 (StataCorp, College Station, TX). Descriptive statistics (number, percentage) were used to report participant recruitment rates and reasons for exclusion.
Participants’ characteristics including age, body size, grip strength, UPDRS (motor) score, H&Y stage, duration of Parkinson’s, numbers of comorbidities and medications, Barthel score, MMSE score, and MUST score were described for men and women using summary statistics: means (standard deviations [SDs]), medians (inter quartile ranges [IQRs]), and number (%) were presented. The MUST score was recoded from 5 categories (score of 0 representing low risk of malnutrition, 1 (modest risk), 2 (high risk), 3 and 4 representing extremely high risk) to 3 categories (score 0, 1, and 2-4) since a score of 2 or more is used clinically to denote a high risk of malnutrition: only one male and one female participant scored above 2. The clinical characteristics of men and women were compared using the 2-sample
Scatter plots were produced for grip strength versus UPDRS (motor) score, H&Y stage, and duration of Parkinson’s for men and women separately. Tests for homogeneity suggested that the associations between grip strength and each of UPDRS score, H&Y stage, and duration of Parkinson’s were similar in men and women. As such, men and women were pooled for all subsequent analyses. The associations between grip strength (as the outcome variable) and each of UPDRS motor score, H&Y stage, duration of Parkinson’s, and other participant characteristics in turn, adjusted for gender, were analyzed using simple linear regression. Results were presented using regression estimates with confidence intervals, and statistical significance indicated using
Results
Participants
Fifty-seven of 79 (72%) PwP resident in the town were recruited to the study. The 57 patients comprised 34 men (mean [SD]; age 71.3 [8.0] years, range = 53 to 85) and 23 women (mean [SD]; age 72.6 [7.6] years, range = 61 to 86). Two participants lived in care homes and the remainder lived in their own homes. Twenty-two patients were not recruited: 14 were excluded and 8 declined (Figure 1). The patients not recruited were older with a mean age of 79.3 years.
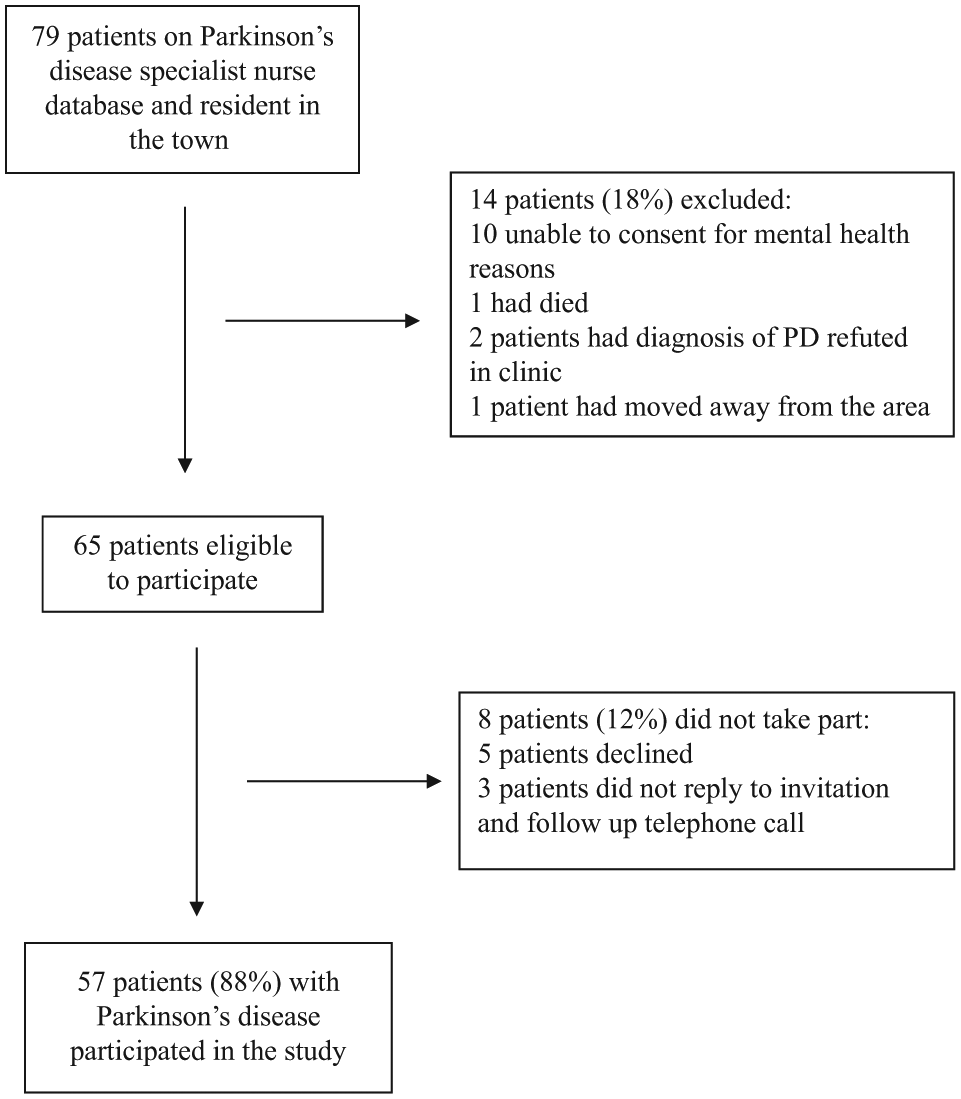
Recruitment of participants.
The male participants were taller and heavier than the women with stronger grip strength (Table 1). The UPDRS motor score, H&Y stage, duration of Parkinson’s, number of comorbidities, number of medications, Barthel score, and MUST score in the last year were similar for both men and women but the MMSE score was significantly lower in men than in women. All participants were taking medication for their Parkinson’s disease. Nine (27%) men and 6 (26%) women were taking levodopa alone, 19 (56%) men and 14 (61%) women were taking levodopa with other antiparkinsonian medication (dopamine agonists, entacapone, selegiline, or rasagiline), and 6 (18%) men and 3 (13%) women were taking dopamine agonists and/or selegiline or rasagiline without levodopa.
Characteristics of Participants.
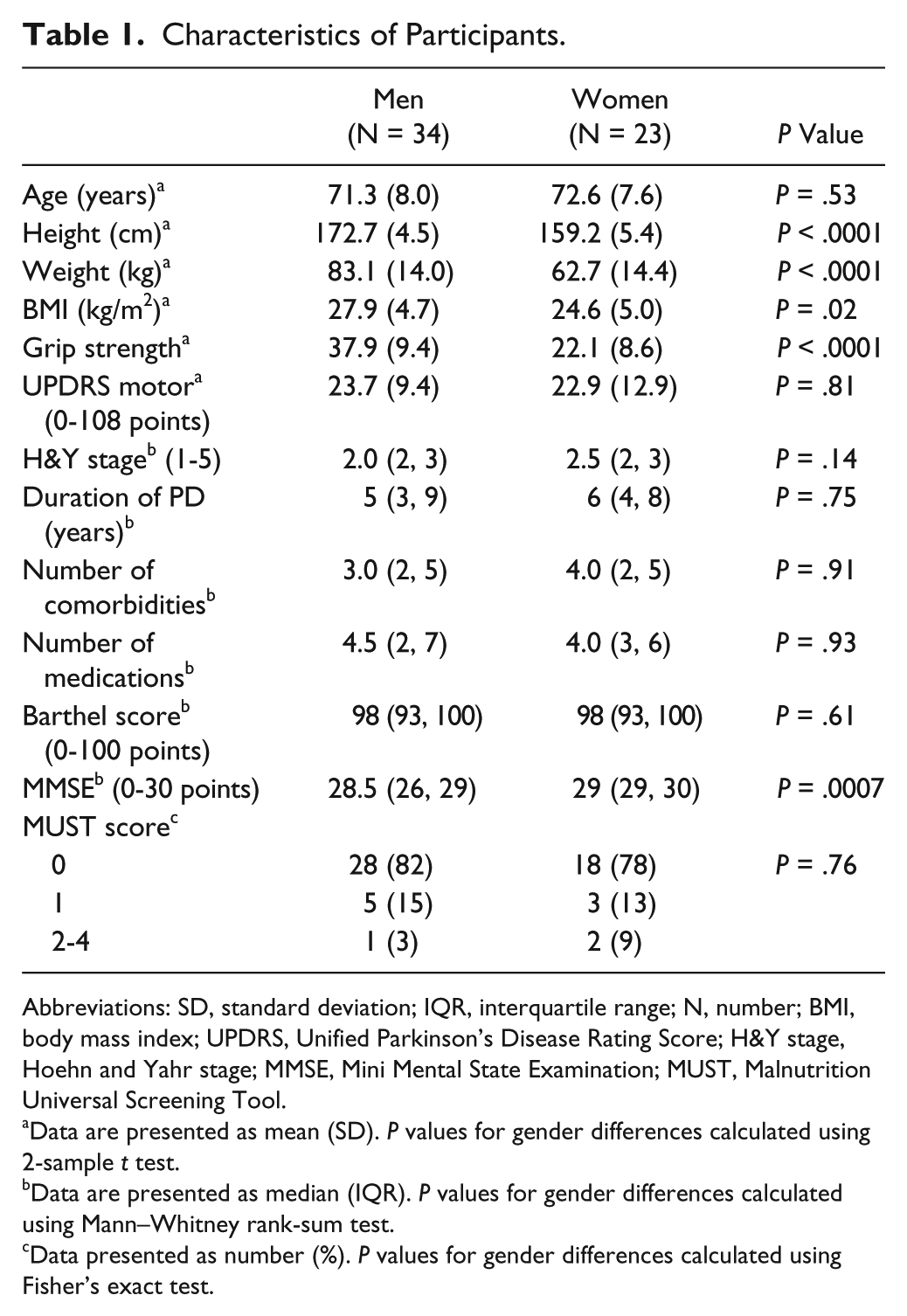
Abbreviations: SD, standard deviation; IQR, interquartile range; N, number; BMI, body mass index; UPDRS, Unified Parkinson’s Disease Rating Score; H&Y stage, Hoehn and Yahr stage; MMSE, Mini Mental State Examination; MUST, Malnutrition Universal Screening Tool.
Data are presented as mean (SD).
Data are presented as median (IQR).
Data presented as number (%).
Grip Strength and Parkinson’s
Figures 2, 3, and 4 show the association between grip strength and each of UPDRS motor score, H&Y stage, and duration of Parkinson’s in men and women separately. Higher UPDRS motor scores and higher H&Y stages were associated with lower overall maximum grip strength but there was no association between duration of Parkinson’s and grip strength. Although associations were somewhat stronger among men than women there was no significant gender difference in the association between grip strength and UPDRS score (
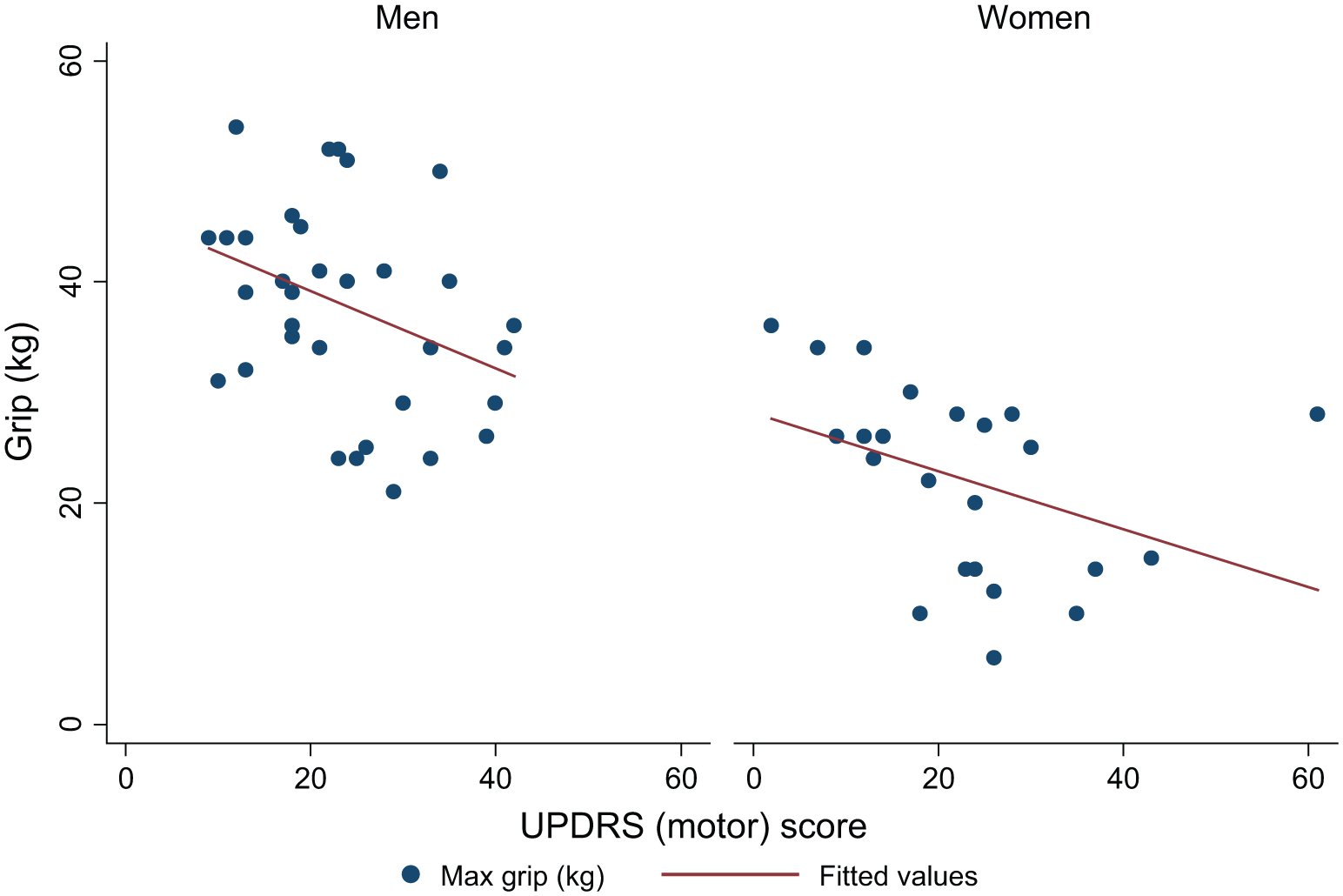
Association between grip strength and UPDRS (motor) score in men and women.
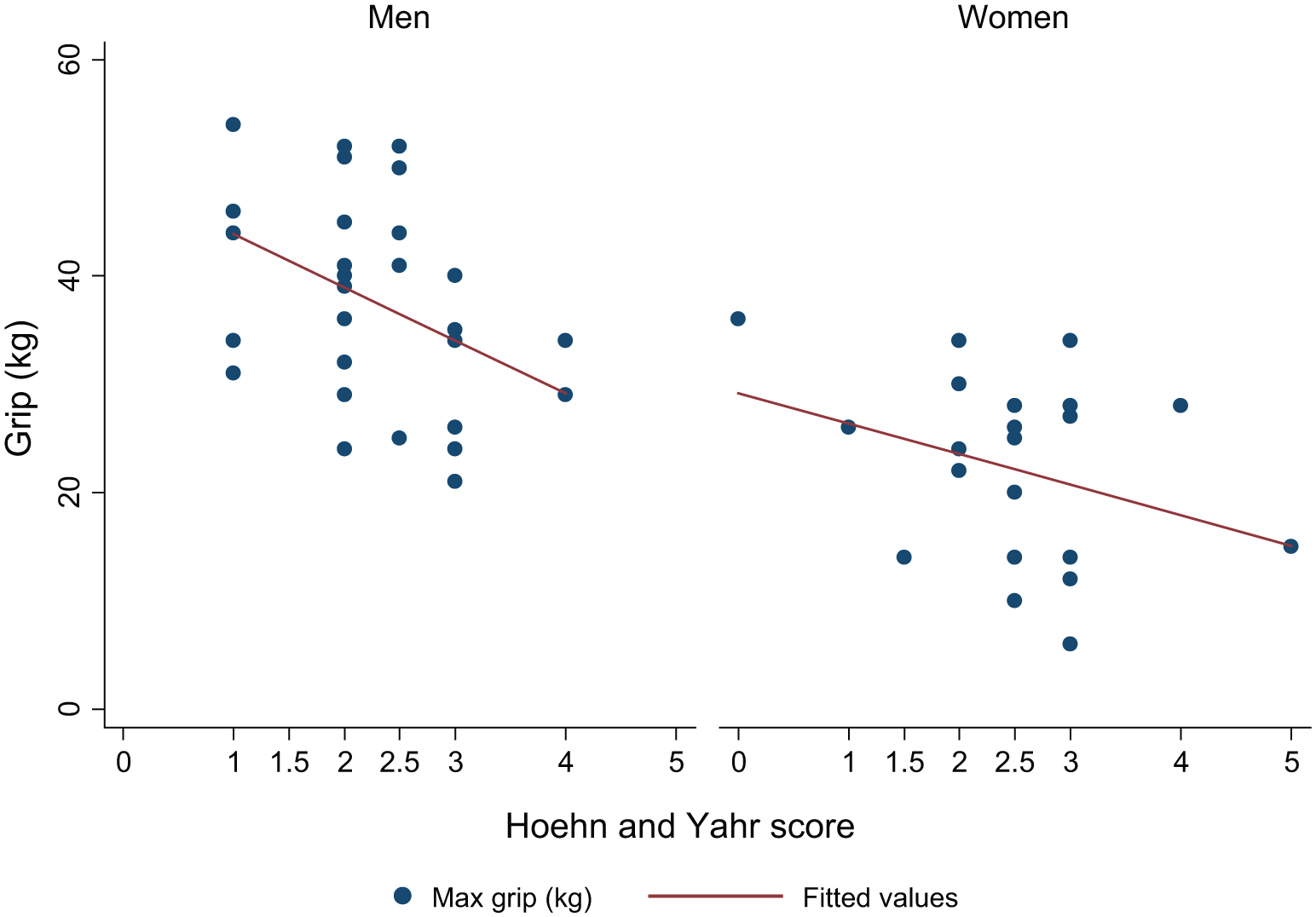
Association between grip strength and H&Y stage in men and women.
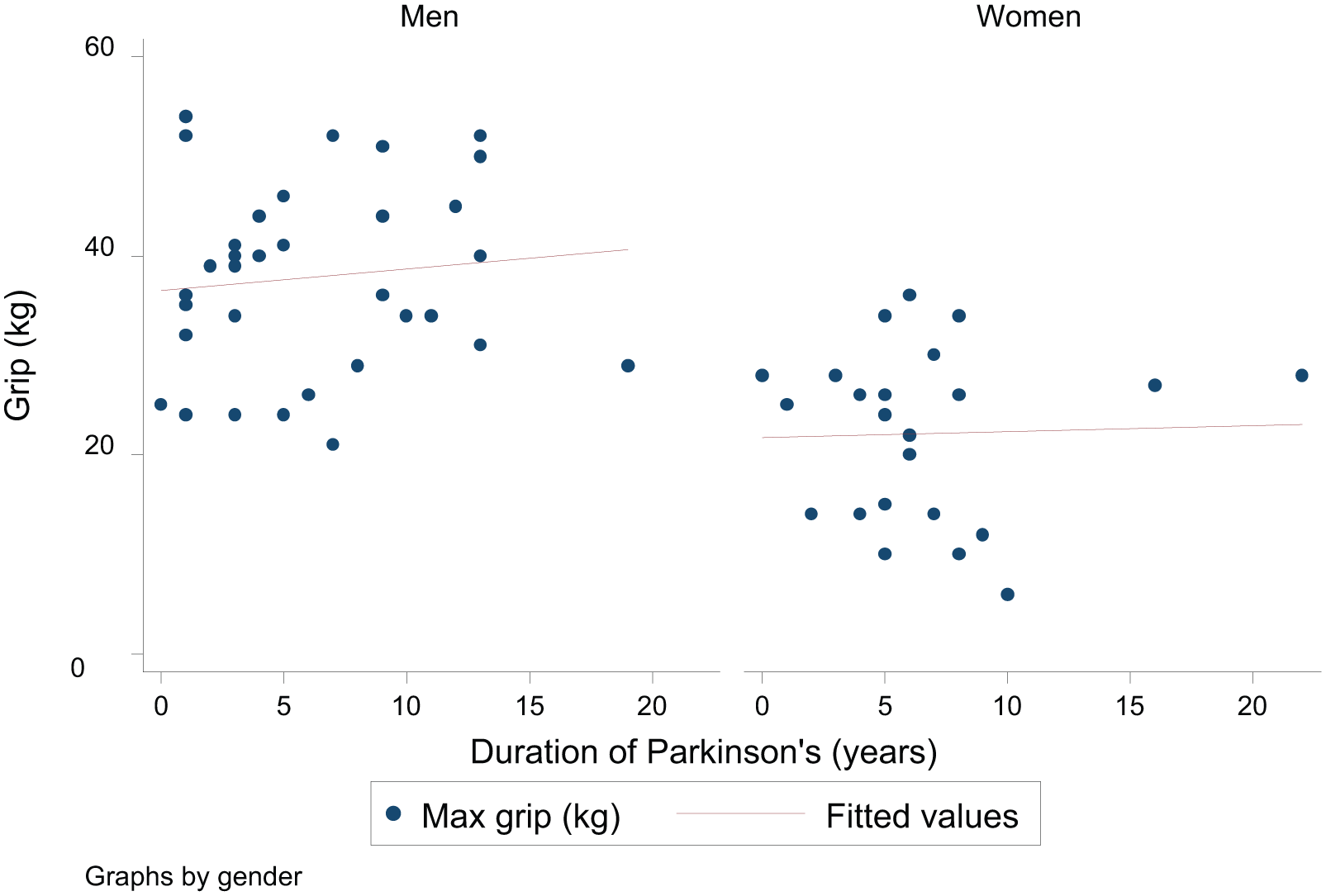
Association between grip strength and duration of Parkinson’s in men and women.
The main associations with grip strength in this study are shown in Table 2. Analysis of each variable in turn, adjusted for gender, confirmed that older age, lower height and weight, higher UPDRS motor score, higher H&Y stage, and lower Barthel score were all associated with weaker grip strength. The other baseline variables including duration of Parkinson’s and MMSE score were not associated with grip strength, and so were not carried forward to adjusted analyses. A mutually adjusted model for UPDRS score versus the characteristics significantly associated with grip strength in the univariate analyses demonstrated that while the associations were attenuated, age, gender, height, and UPDRS score remained the most important determinants of grip strength. A mutually adjusted model using the H&Y stage similarly showed that age, height, the H&Y stage, and gender were the most significant determinants of grip strength.
Determinants of Grip Strength in Participants With Parkinson’s Disease.
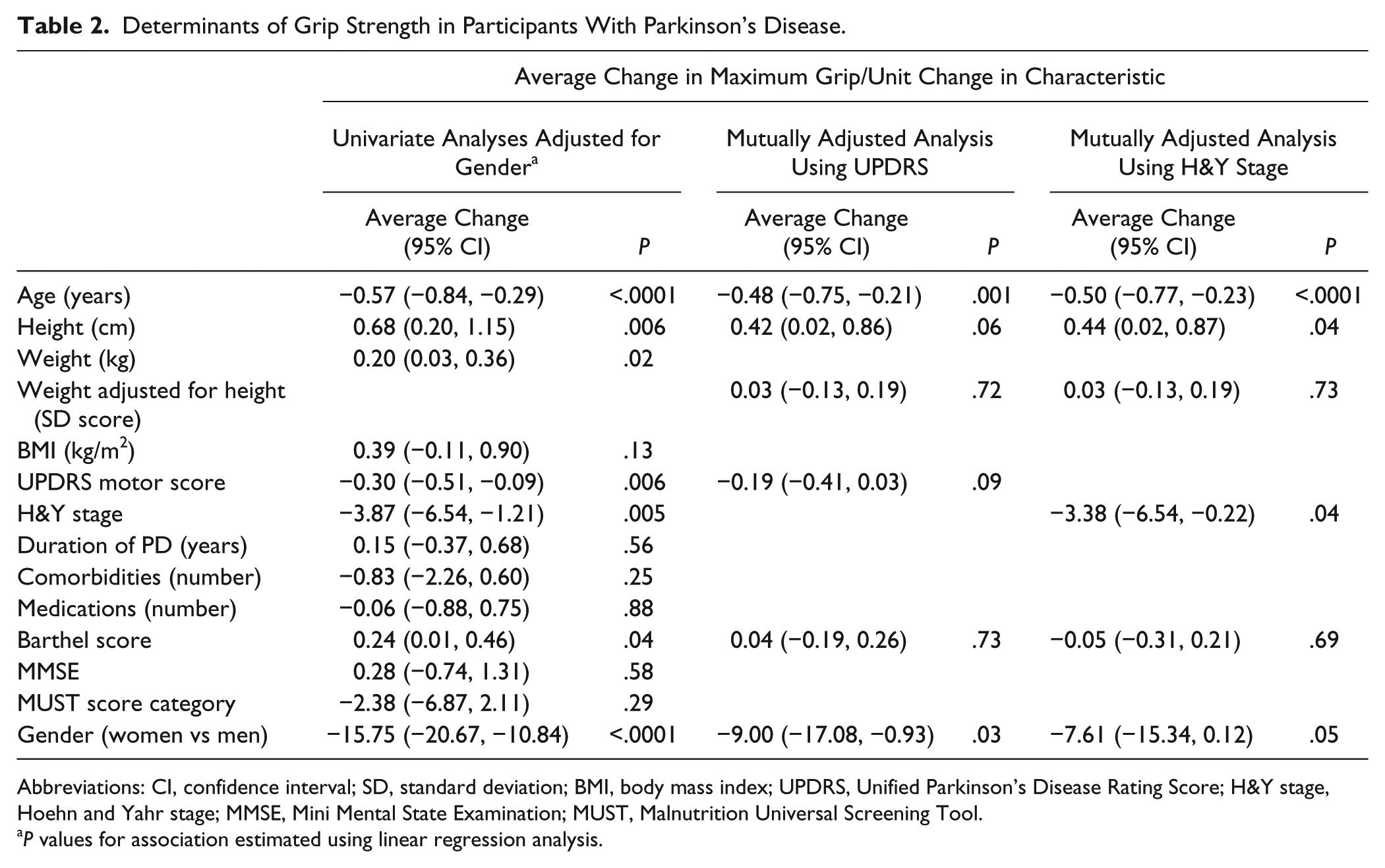
Abbreviations: CI, confidence interval; SD, standard deviation; BMI, body mass index; UPDRS, Unified Parkinson’s Disease Rating Score; H&Y stage, Hoehn and Yahr stage; MMSE, Mini Mental State Examination; MUST, Malnutrition Universal Screening Tool.
Discussion
This cross-sectional study has shown for the first time that increasing severity of Parkinson’s measured using both the UPDRS motor score and H&Y stage was significantly associated with lower grip strength after adjustment for potential confounding influences. In mutually adjusted models the strongest determinants of grip strength were age, gender, body size, and severity of Parkinson’s. This confirms the need to adjust for these factors to allow accurate comparison of grip strength values across different studies. Studies of muscle strength in Parkinson’s are sometimes age and sex matched but have not previously included adjustment for body size, UPDRS score, or H&Y stage. The severity of Parkinson’s was more strongly associated with grip strength than Barthel score, a measure of physical function, which is in contrast to studies of older people in which physical function is typically identified as the most important determinant of grip strength after age, gender, and body size. 19 This may partly reflect the broad domains of physical function in the Barthel score and more specific measures of physical function relevant to people with Parkinson’s may be indicated in future studies. The duration of Parkinson’s was not associated with grip strength.
The results of this study indicate a specific effect of Parkinson’s on muscle strength although the mechanism is unclear and appears to be independent of disease duration. The reduction in nigro-striatal dopamine in Parkinson’s leading to an increase in tonic inhibition of the thalamus and thus reduction in excitation of the motor cortex is well recognized. It is suggested that this may disrupt the corticospinal activation of muscle and abnormal electromyographic (EMG) activation patterns have been described in PwP throughout isometric movements in keeping with impaired muscle activation.20,21 The possibility that the weakness in people with Parkinson’s found in our study reflects impaired activation of muscles is highlighted by a recent study that confirmed a lack of alteration of peripheral motor neuron axonal excitability in Parkinson’s, in contrast to other disorders of the central nervous system such as stroke disease or multiple sclerosis. 22 Thus, it is possible that altered variability, intensity, and frequency of corticospinal activation of the muscle may lead to impaired motor unit recruitment and thus muscle weakness. It is also possible that the association of weakness with increasing severity of Parkinson’s found in this study is due to a myopathy. Few researchers have studied muscle morphology in Parkinson’s but one small study comparing paraspinal muscle biopsies from 14 PwP who had camptocormia with age and sex matched controls reported a consistent myopathic pattern among the PwP, with type I fiber hypertrophy and loss of type II fibers. The degree of myopathic changes was associated with the severity of camptocormia. 23 However, it is unclear if this process affects other muscles in PwP or patients without camptocormia. A recent study of 15 PwP who completed 16 weeks of high-intensity exercise training has reported skeletal muscle adaptations to the training including myofiber hypertrophy, a shift to less fatiguable myofiber profile and increased mitochondrial complex activity. 24
It is possible that PwP become increasingly sarcopenic as the condition progresses, due to a combination of the condition, progressive immobility, and increasing age. Studies on muscle mass in Parkinson’s are few but contradictory. Petroni e al found a reduced mid arm circumference with coexisting adiposity in advanced Parkinson’s 25 in a cross-sectional study of 35 PwP, while an earlier study reports a reduction in body fat rather than muscle in PwP compared to age and sex matched controls. 26 However, the rapid improvement in muscle strength seen in response to medication or deep brain stimulation 27 indicates that a central mechanism rather than low muscle mass is more likely to be responsible for the low grip strength in the short term although chronic changes may occur within the muscle over time. The participants in this study had been living with Parkinson’s for a median of 5 (men) and 6 (women) years, and so the weakness associated with increasing severity of Parkinson’s reported here may reflect both these central mechanisms, for example, impaired neuronal activation of muscle, and chronic changes within the muscle itself, such as a myopathy. However, further research into the mechanisms of muscle weakness in Parkinson’s is required.
The role of inflammatory processes in the causation of muscle weakness is unclear. Sarcopenia is associated with an increase in cytokines such as interleukin-6 (IL-6), and a recent study has demonstrated higher levels of IL-6 among 44 PwP compared to 22 controls, and an association with slower gait speed although muscle strength was not assessed. 28 However, there was no correlation between serum levels of IL-6 and either UPDRS score or H&Y stage.
Few studies have evaluated muscle strength alongside functional performance in PwP. Recent studies of resistance exercise in Parkinson’s have demonstrated improvements in muscle strength 29 and volume, as well as improvements in function among participants with mild to moderate Parkinson’s after 10 to 12 weeks of resistance training. 26 Upper limb function in PwP may be assessed using brief timed motor tests such as the pegboard dexterity test, which has been shown to be highly correlated with the UPDRS motor score. 30 Studies reporting upper extremity tests of motor function in Parkinson’s have typically not reported the results by gender.31,32
This study had a number of strengths. The UPDRS is recognized as the clinical gold standard for measuring motor deficits in Parkinson’s. The association between the UPDRS and grip strength was replicated in this study using the H&Y stage, confirming the association between lower grip strength and more advanced Parkinson’s. Importantly, the wide range of participants’ grip strength (21-54 kg in men and 6-36 kg in women) and of UPDRS scores (2-61 points) and H&Y stages (1-5) allowed an appropriate evaluation of the association between grip strength and Parkinson’s. The Jamar dynamometer is well validated with excellent reliability and repeatability using a standard protocol. 13 The advantage of measuring maximum grip strength as a measure of muscle strength in this study is that it is a quick and simple measurement using inexpensive equipment that can be successfully completed by most people in clinical settings. Additionally, isometric tasks, unlike isokinetic tasks, do not involve visual perception and spatial orientation, which may be affected by cognitive impairment. A single assessor ensured consistency between patients, the participants were clinically stable, and all PwP known to the nurse specialist were contacted.
However, this was a relatively small study, albeit with a similar number of participants to previous studies, and the patients who were ineligible or declined were older and may have had lower grip strength. The participants were all living in one town and this may potentially limit the generalizability of the findings. Participants had mild to moderate Parkinson’s and relatively high median Barthel and MMSE scores, which may have contributed to the lack of association of these scores with grip strength. The impact of medication on movement is well recognized, and our patients were assessed in the “on” state. The influence of this on the mechanism of muscle activation is unknown. Some studies have assessed participants in the “off” state but this was impractical for our patients.
Conclusions
This is the first study of grip strength in Parkinson’s to take into account known potential confounding factors affecting muscle strength. We have demonstrated that higher UPDRS score and H&Y stage was associated with weaker grip. Gender, participants’ age, and body size also had a significant impact on strength. Adjustment of reported values for all these factors is necessary to allow accurate reporting of grip strength values in intervention trials, which will be particularly relevant to trials of physical therapies. It would also allow accurate comparison between studies of different groups of PwP. Further research is required into the causes of muscle weakness in Parkinson’s to determine suitable therapeutic approaches.
Footnotes
Acknowledgements
The authors wish to thank the participants of the study and Tracy McElwaine, Parkinson’s nurse specialist, for her assistance in identifying potential recruits.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Faculty of Medicine and the MRC Lifecourse Epidemiology Unit at the University of Southampton. HCR and AAS receive support from the National Institute for Health Research (NIHR) Southampton Biomedical Research Centre (BRC) and the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) Wessex. This report is independent research funded by the NIHR BRC and the NIHR CLAHRC. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research, or the Department of Health.
