Abstract
Background. Muscle weakness in old Parkinson’s disease (PD) patients has been shown to impair their mobility, although the specific origin of this weakness and its relation to falls has not been well examined in young patients. Objective. This study aimed to analyze the possible contribution of central factors to muscle weakness of the triceps surae and quadriceps femoris muscles in young faller and nonfaller PD patients. Methods. Twenty-six young PD patients (fallers, n = 13 and nonfallers, n = 13) and 15 matched healthy controls performed several isometric maximal voluntary knee extension and plantar flexion contractions (MVC) of the most affected leg on a dynamometer. We estimated the maximal resultant agonist moments, the antagonistic moment of hamstrings and tibialis anterior during MVCs and the activation deficit of the quadriceps femoris and triceps surae muscles. Results. Only the Parkinson fallers showed significantly lower muscle strength, higher antagonistic moments and higher activation deficit compared with controls. Multiple regression analysis showed that the antagonistic moments and the activation deficit explained about 39% and 27%, of the variance in the maximal resultant moments of the knee extensors and the plantar flexors, respectively. Conclusions. Our findings suggest that Parkinson fallers are affected by strength impairments arising from the central nervous system and not from the peripheral muscle contractile capacity, even at early stages of the disease and young age. High-intensity resistance training may help enhance neural drive and decrease unwanted antagonistic moments and reduce the risk of falls.
Introduction
In patients with Parkinson’s disease (PD), the neuromuscular system becomes faulty mainly because of the degeneration of dopaminergic neurons in nigrostriatal systems. 1 Although PD is generally considered to be a disorder of the elderly, it also affects a substantial number of younger individuals. The mean age of onset is around 65 years, although 5% to 10% of cases, classified as early-onset, begin at age less than 51 years. 2 The primary symptoms in this disease have a dramatic impact on the patient’s mobility and quality of life; postural instability and falls are among the most incapacitating features of PD. 3 Several studies have analysed the rate of falling in Parkinson patients. They reported that 50% to 68% of PD patients fell at least once in the previous year,3,4 and 13% of them fell more than once a week. 5 About 42% of the patients suffered injurious falls. 6 Despite advances in pharmacological treatments and surgical techniques, gait and balance deficits still persist and are associated with loss of independence, immobility, and high cost for health care systems. 7
Intrinsic neuromuscular properties of the muscle–tendon unit have been shown to influence the function and performance of the entire musculoskeletal system during locomotion.8,9 The capacity of the human system to generate rapid force for balance corrections after sudden perturbations during locomotion is especially affected by muscle strength and tendon stiffness. 10 Decreased strength has been reported to be a factor contributing to increased incidences of falling in the elderly.10,11 It has been reported that elderly PD patients have shown reduced muscle strength,5,12 decreased rate of force development, 13 impaired ability to maintain constant force, and increased muscle coactivation during balance perturbation tasks.14,15 The aforementioned weakness in muscle capacities related to several mobility tests such as the chair-rising test 13 or the time up and go test. 16 Recently Latt et al 17 reported that muscle weakness in older PD patients was one of the 4 main independent risk factors for falls. They developed an explanatory model using muscle weakness as a factor that correctly classified 77% of the fallers and 82% of the nonfallers. 17 However, the majority of the reported information about muscle weakness in PD patients resulted from studies investigating old participants (between 65 and 85 years old). The Parkinson-related decrease in muscle strength may result from both central (ie, arising from the central nervous system) and peripheral factors (ie, occurring distal to the neuromuscular junction).18,19 To our knowledge, there is no study investigating the contribution of central and peripheral factors to the reported muscle weakness in young PD patients.
The muscles of the lower extremities (ie, triceps surae and quadriceps femoris) are very important during locomotion and their strength significantly affect the risk of falling 20 and the ability of humans to regain balance after disturbances during walking. 11 Therefore, the purpose of this study was to investigate the possible contribution of central factors (ie, activation deficit of the agonist muscles—quadriceps femoris and triceps surae—and coactivation of the antagonist muscles—hamstrings and tibialis anterior) to muscle weakness of the knee extensor and plantar flexor muscle groups in young faller and nonfaller PD patients (39-57 years old) comparing them with healthy matched controls. We hypothesized a reduction of muscle strength in PD patients compared with a matched healthy control group and a higher contribution of the central factors in Parkinson fallers.
Methods
Participants
Fifteen healthy adults and 26 patients with early-onset idiopathic PD (in the I-III stage of the Hoehn and Yahr Parkinson scale) have been recruited for the study (Table 1). The Parkinson patients were divided into 2 groups (nonfallers, n = 13 and fallers, n = 13). The nonfaller group included patients with no fall experience in the past 6 months and the faller group included patients who have experienced more than one fall in the past 6 months. A fall was defined as “inadvertently coming to rest on the ground, floor or other lower level, excluding intentional change in position to rest in furniture, wall or other objects.” 21 A history of any other neurological or orthopedic disorder that could affect their ability to perform a maximal contraction was considered as exclusion criteria for participating in this study. The patients were included in the study if they did not meet these exclusion criteria. Individuals were examined during the ON phase, when they self reported that their medications were working optimally, about 30 minutes to 1 hour after they had taken their usual dose of antiparkinsonian medication. The control group was matched to the Parkinson group regarding age, anthropometrical parameters (weight and height), and sport activity. Sport activity was quantified as hours per week of regular sport activity practiced in the past year and was estimated using a questionnaire enquiring the type of sport activity, frequency of practice (ie, hours per week and weeks per year), and intensity of the activity. The study has been approved by the university ethics committee and the participants gave informed consent to the experimental procedure.
Anthropometric Data, Age at Disease Onset, and Stage in the Hoehn and Yahr (H&Y) Parkinson Scale for the 3 Groups (Means ± Standard Deviation). a
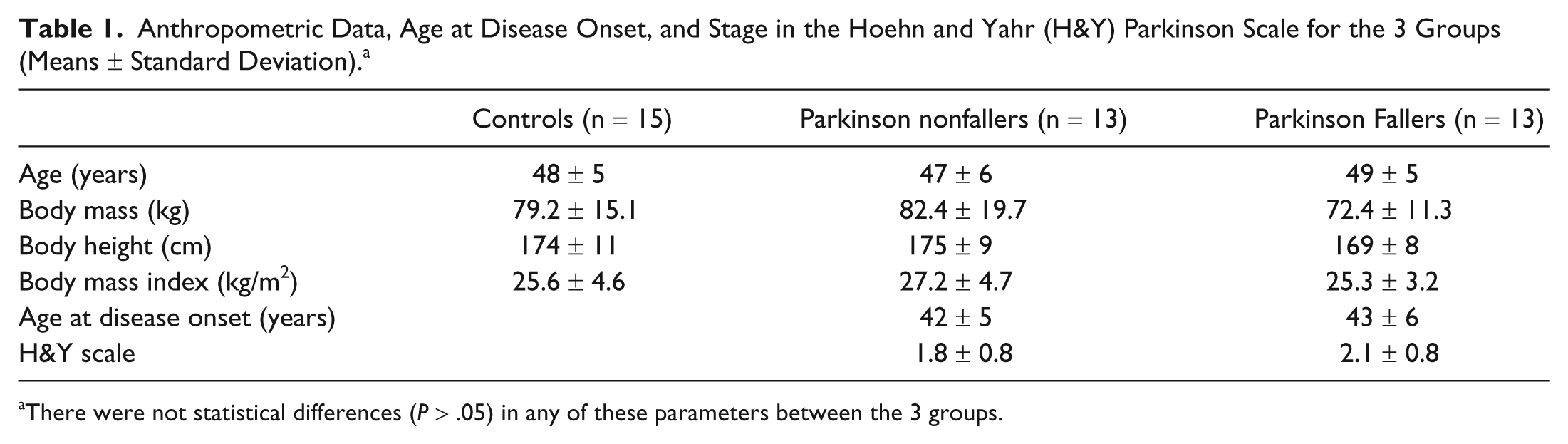
There were not statistical differences (P > .05) in any of these parameters between the 3 groups.
Measurement of the Muscle Strength and Electromyographic Activity
After an initial warm-up consisting of several submaximal and 2 to 3 maximal contractions, the participants performed several isometric maximal voluntary knee extension contractions (MVCs) and ankle plantar flexion contractions with the most affected leg (the one where the symptoms appeared at first). For the knee extension, they were seated with a hip angle set at 140° and knee angles set at 105°, 110°, 115°, and 120°. A straight position at the hip and knee joints corresponded to a 180° joint angle. For the plantar flexion, the knee angle was set at 180° and ankle angles at 75°, 80°, 85°, and 90°. The foot perpendicular to the tibia corresponded to an ankle joint angle of 90°. Different joint angle configurations were chosen to examine triceps surae (TS), that is, soleus, gastrocnemius medialis and lateralis, and quadriceps femoris (QF), that is, rectus femoris, vastus lateralis, medialis and intermedius, muscle strength potential near to the optimal individual joint angle. For the statistical analysis we used the highest value within the contractions. The different joint angle configurations were applied in random order where a 3-minute rest between the contractions was allowed.
We used an inverse dynamic approach to calculate the resultant moments at the knee and ankle joints. With this approach, we corrected the moments measured by the used dynamometer (Biodex Medical Systems, Inc, Shirley, NY) by rectifying the misalignment of the dynamometer and joint axis during the contractions.22,23 For that, kinematic data were recorded using the Vicon 624 system (Vicon Motion Systems, Oxford, UK) with 11 cameras operating at 120 Hz. Reflective markers were fixed on the tuber calcanei, lateral and medial malleolus, lateral and medial femoral condyles, trochanter major, lateral aspect of the spina iliaca, axis of the dynamometer, and on the dynamometer arm at the point of force application to define the distance between the line of action of the exerted force and the axis of rotation of the dynamometer lever. The exact method has been previously described.22,23
To assess the contribution of the antagonist muscles (ie, coactivity) to the examined maximal resultant joint moment we calculated the moments of the antagonist hamstrings (HA) and tibialis anterior (TA) muscles during knee extension and plantar flexion MVCs. For that the antagonist electromyographic (EMG) activity of HA and TA was recorded using autoadhesive preamplified electrodes during the MVCs. The electrodes were positioned above the midpoint of the muscle belly of biceps femoris muscle as a representation of the HA muscles and TA. The interelectrode distance was 2 cm and electrodes were placed parallel to the presumed direction of the muscle fibers. The antagonistic moment of HA and TA was then estimated by establishing a relationship between the EMG amplitude and the exerted moment for HA and TA while working as agonist.24,25 Therefore, the EMG activity of the HA and TA and the corresponding moment were measured in 3 additional trials: (a) in relaxed state, (b) producing knee flexion and dorsiflexion contractions displaying an EMG amplitude of the HA and TA below the maximum amplitude measured during knee extension and plantar flexion, and (c) a second knee flexion and dorsiflexion contractions where the EMG amplitude was slightly above the maximum amplitude registered during the knee extension and the plantar flexion. The moments measured in each of the 3 trials were fitted by a linear regression curve as a function of the corresponding EMG values of the HA and TA. This allowed the estimation of the antagonistic moment during the MVCs. This method is only valid under the condition that the neuromuscular activation is the only factor determining the force generating potential. We assured this condition by keeping the muscle length constant (ie, at a given joint angle) and the muscle shortening velocity equal to zero (ie, isometric contractions) during the trials. The coactivation level was defined as the antagonistic moment of HA or TA muscles during the knee extension or plantar flexion MVCs normalized to the maximal exerted knee extension or plantar flexion resultant moment, respectively.
Assessment of the Voluntary Activation
For the assessment of the voluntary activation during the maximal isometric knee extension and ankle plantar flexion contractions we used the twitch interpolation method. 26 Two carbon rubber electrodes (5.5 × 10 cm and 5.5 × 9 cm), thinly coated with conductive adhesive gel, were secured onto the skin. For the QF, the cathode was placed on the proximal anterior thigh, and the anode was placed over the motor point area of the muscles rectus femoris and vastus lateralis. For the TS, the cathode was above the midway between the two heads of gastrocnemii and approximately 5 cm distal to the crease of the fossa poplitea, and the anode above the soleus’ motor point, along the medial line directly below the belly of medial and lateral gastrocnemius muscles. Each muscle group was stimulated transcutaneously with triplet twitches (square-wave pulses of 500 ms at 200 Hz) by means of a constant-current stimulator (Digitimer DS 7A, Welwyn Garden City, Herdfordshire, UK) at the plateaus of knee extension and plantar flexion contractions as well as at the subsequent resting phases. The current of the twitches was determined in a previous test by successively increasing the current until the maximal rest twitch torque was evoked. This current plus 20% was used during the stimulation trials. Triplet twitches were used instead of single ones to increase the duration of the elicited contraction and this way minimize the influence of tendon compliance on muscle force production. It has further been reported that the multiple twitches decrease the variability of muscle responses. 27 The stimulator delivered a TTL (transistor–transistor logic) output signal (0-5 V) simultaneous to the twitch, which was registered by the Vicon unit to synchronize the systems. Activation deficit was calculated by normalizing the evoked interpolated twitch torque (ITT) 28 to the subsequent resting twitch torque (RTT): activation deficit = (ITT/RTT) × 100.
Statistics
A 2-way analysis of variance was used to check the muscle group (QF, TS) and participant group (controls, Parkinson nonfallers, Parkinson fallers) related differences in the examined parameters (ie, maximal resultant moment, moment of the antagonists, and activation deficit). When significant participant group differences were detected, a post hoc test (Bonferroni) was applied to determine where these differences occurred. Furthermore, we conducted a multiple regression analysis to determine whether the activation deficit of the agonists (QF and TS) and the coactivity of the antagonist muscles (HA and TA) might predict the achieved plantar flexion and knee extension moments. The level of significance was set at α = .05.
Results
The 2-way analysis of variance revealed statistically significant (P < .05) muscle group effects in the resultant moment and in the moments of the antagonistic muscles, but not in the activation deficit (Tables 2-4). The maximal resultant knee joint moments were higher (P < .05) compared with the maximal plantar flexor moments in all 3 participant groups, as well as the moments of the antagonist muscles HA compared with TA. The Parkinson faller group revealed significantly (P < .05) lower maximal resultant knee extension and lower maximal resultant ankle plantar flexion moments compared with the control group (Table 2). However, the Parkinson nonfallers did not show any statistically significant differences (P > .05) in the maximal knee extension and plantar flexion moments compared with the other 2 groups (Table 2). In a similar way the moments of the antagonists HA and TA showed significantly higher values (P < .05) only between the control and the Parkinson faller group (Table 3). Furthermore, the activation deficit in both muscle groups (knee extensors and plantar flexors) was significantly higher (P < .05) in Parkinson fallers compared with controls (Table 4).
Maximal Resultant Knee Extension Joint Moment (MomentKnee) and Maximal Resultant Ankle Plantar Flexion Moment (MomentAnkle) Normalized to Body Weight by the 3 Examined Groups. a
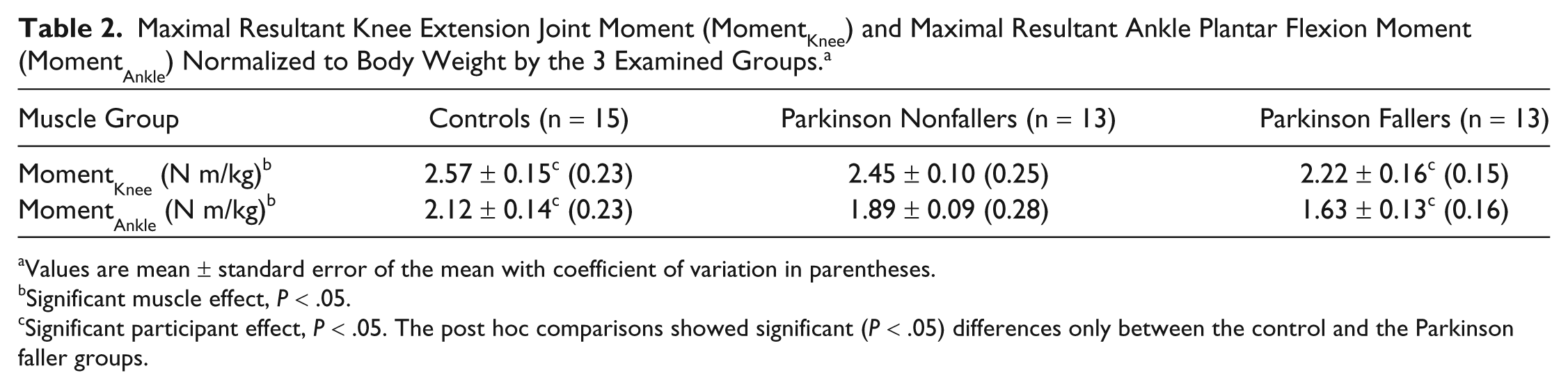
Values are mean ± standard error of the mean with coefficient of variation in parentheses.
Significant muscle effect, P < .05.
Significant participant effect, P < .05. The post hoc comparisons showed significant (P < .05) differences only between the control and the Parkinson faller groups.
Moment of the Antagonist Muscles Hamstrings and Tibialis Anterior During the Maximal Knee Extension and Plantar Flexion Contractions. a

The values are normalized to the maximal resultant knee extension and ankle plantar flexion moments. Values represent mean ± standard error of the mean.
Significant muscle effect, P < .05.
Significant participant effect, P < .05. The post hoc comparisons showed significant (P < .05) differences only between the control and the Parkinson faller groups.
Activation Deficit of the Quadriceps Femoris and Triceps Surae Muscles During the Maximal Knee Extension and Plantar Flexion Contractions (Mean ± Standard Error of the Mean).
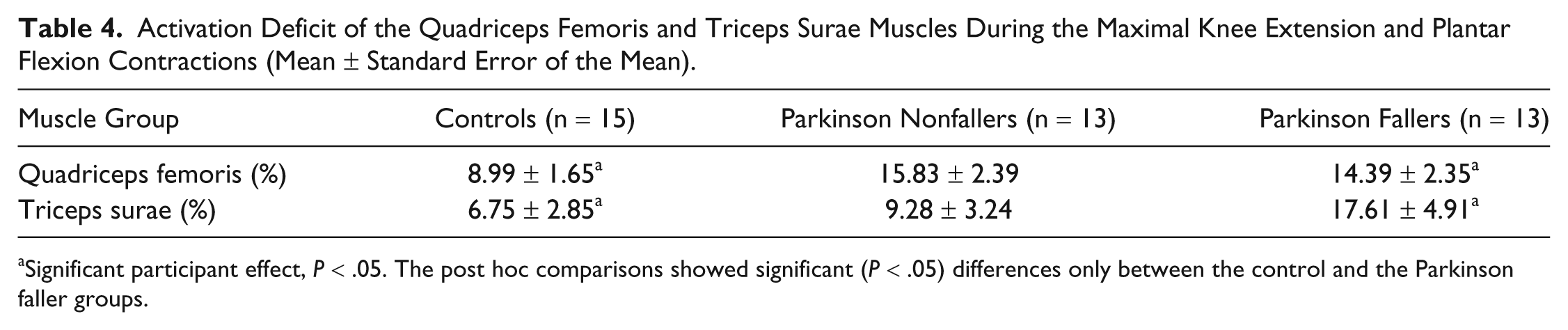
Significant participant effect, P < .05. The post hoc comparisons showed significant (P < .05) differences only between the control and the Parkinson faller groups.
The multiple regression analysis revealed a significant influence of the 2 predictor variables (moment of the antagonists HA and TA and activation deficit of QF and TS) on the maximal resultant moments of the knee extensors (F = 9.742, P = .001, R = 0.627) and the plantar flexors (F = 4.114, P = .03, R = 0.522).
Discussion
Differences in muscle strength between old Parkinson patients (>65 years old) and healthy matched controls are reported by the majority of research studies using maximal isometric contractions.29,30 This weakness has been related to performance deficits by several functional and clinical tests showing a disease-related increased risk of falls. 31 Reduced maximal muscle strength is the major determinant of reduced power in Parkinson patients, whereas other factors such as bradykinesia of the lower limbs make a minor contribution. 32 This means that maximal muscle strength remains one of the most relevant parameters regarding mobility and stability in Parkinson patients. However, the contribution of central and peripheral factors to the found muscle weakness especially in young Parkinson patients (<57 years old) is not well understood. Knowledge regarding this contribution would be very important for planning effective therapeutic interventions aiming to prevent falls in Parkinson patients. The study identified that only the Parkinson faller group showed significantly lower muscle strength, higher antagonistic moments, and higher activation deficit within the examined muscle groups compared with the control group, partly confirming our hypothesis.
To examine whether the variance in the maximal resultant knee extension and plantar flexion moments in our participants could be accounted for by the antagonistic moment of HA and TA and the activation deficit of QF and TS we used a multiple regression analysis for each response variable (ie, knee extension and plantar flexion moments), including the 2 corresponding predictor variables (ie, antagonistic moment of HA or TA and activation deficit of QF or TS). The antagonistic moment and the activation deficit of the agonists explained about 39% and 27% of the variance in the maximal resultant moments of the knee extensors and the plantar flexors, respectively. This relationship demonstrates that the reduced muscle strength of the leg extensors in PD patients can be highly accounted for by the increased antagonistic moment and the increased activation deficit of the agonists.
An important finding of the current investigation was the existence of a central origin of deficits (ie, increased antagonistic moments and activation deficit of the agonists) in muscle strength in young Parkinson fallers. Assuming similar coactivation moments and activation deficits between the 3 groups, the achieved joint moments would be more or less similar between the groups indicating no significant contribution of peripheral factors to the found muscle weakness in our young Parkinson patients. Therefore, we can argue that impairments in the neuromuscular capacities already appear at young ages, thus reducing their ability to exploit the entire muscle potential.
The consequences of our results in the Parkinson fallers may be a decrease in rate of muscle force development, 33 a decrease in postural stability,14,34 and a decrease in reactive adjustments to regain balance after perturbations, 15 increasing the risk of falling. An increase in the coactivation of the antagonist muscles in Parkinson patients during maximal isometric contractions of the upper limb muscles 35 and also in the lower limb muscles during balance perturbation tasks14,15 has also been reported in literature. It can be argued that the Parkinson fallers were not able to selectively activate only the most effective muscles for a given task already in early disease onset, suggesting an important role of the basal ganglia in optimizing muscle synergy patterns. An increase in muscle coactivation has been shown to decrease the postural control of old participants during quiet stance 36 and during downward stepping. 37 Furthermore, the higher activation deficit in TS and QF muscles indicates impairments in the ability of Parkinson fallers to use the entire muscle potential of the most important muscles for locomotion. A limited capacity to sufficiently generate high knee extension and ankle plantar flexion moments increases the risk of falls. 11 Our findings suggest that Parkinson fallers are affected by strength impairments arising from the central nervous system and not from the peripheral muscle contractile capacity, even at early stages of the disease and at a young age, increasing the risk of falls during daily activities.
However, knowing the origin of the impairment, an exercise intervention for muscle strength can be effectively designed and focused the central originated factors for muscle weakness. It has been reported in the literature that short-term traditional strength training in healthy populations shows significant gains in maximal force production without concomitant muscle hypertrophy. 38 It is generally accepted that neural adaptations might be responsible for muscle strength enhancement. 39 Griffin and Cafarelli 40 reported that an increase in neural drive observed subsequent to resistance training may reflect adaptations at the level of the central nervous system. Likewise heavy-resistance training can cause a decrease in the coactivity of the antagonist muscles 41 resulting in an increase of the resultant joint moment and thus affecting the effectiveness of functional tasks. Furthermore, high-intensity strength training shows beneficial effects in denervated muscles as for example in post–polio syndrome patients 42 and among the elderly. In old Parkinson patients, improvements in muscle strength and mobility have been reported following strength training. These improvements were greater in those patients who performed high-intensity strength training compared with those who performed traditional resistance strength training. 43 Therefore, it can be argued that high-intensity resistance training may be an appropriate therapy for Parkinson fallers to enhance neural drive to the agonist muscles and to decrease unwanted antagonistic moments, which are not efficient for the performance of quick postural corrections. Both may contribute to improve strength and movement control and therefore prevent falls. Finally, the current results show the necessity of resistance training interventions for young Parkinson patients already in early stages of the disease to reduce the risk of falls.
Footnotes
Acknowledgements
The authors thank Kiros Karamanidis, PhD, and the Institute of Biomechanics and Orthopedics of the German Sport University of Cologne for their contribution to this investigation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
