Abstract
Introduction
Parkinson disease (PD) is the second most prevalent neurodegenerative disorder, affecting about 1% of the population older than 60 years in industrialized countries and up to 4% of those more than 80 years of age. 1 An estimated 4.1 to 4.6 million people worldwide had PD in 2005, and these numbers are expected to double by the year 2030. 2 Typically, PD is thought of as a disorder characterized by 4 cardinal signs: tremor, rigidity, bradykinesia, and postural instability. 3 Weakness is one symptom associated with PD that has attracted attention and may have substantial functional consequences for individuals.4,5 Although many people with PD complain of weakness, findings have been inconsistent regarding the extent to which weakness occurs and for which patients.5-12 One of the reasons for inconsistent findings is that the perception of weakness can reflect any of a number of factors, including the actual ability of the muscle to produce force,8,10,12,13 the rate at which force can be produced,11,14 the ability of the muscle to relax after force is produced,11,14,15 and the extent to which fatigue occurs with repeated force production. 16 How these factors specifically contribute to deficits in patients with PD is not clear. Cano-de-la-Cuerda and colleagues 5 recently reviewed the available literature on force production among people with PD and concluded that 1) muscle force production is indeed reduced in those with PD compared with age-matched controls, 2) the loss of force production occurs bilat, and 3) the loss becomes more prominent with performance velocity, especially as the disease progresses. Furthermore, deficits in force production vary, depending on the muscle groups investigated. 17 Although much has been learned in recent years about force production among those with PD, the mechanisms underlying these deficits remain unclear. It is possible that muscle weakness, or loss of ability to generate force, reflects general deconditioning that occurs among people with PD who become increasingly sedentary as a result of the functional limitations associated with the disorder. 18 However, an alternative explanation is that the weakness reflects a deficit in central activation of muscles. Central activation deficits have been identified in individuals with a variety of neurological disorders.19-22 As such, it is important to determine whether central activation deficits are present in people with PD.
Furthermore, muscle fatigue with repeated contractions may contribute to muscle weakness reported by patients with PD, especially when performing repetitive movements. Fatigability is defined as any reduction in the force output of a muscle caused by recent activation and can be attributed to peripheral or central nervous system failure.23-25 Individuals with PD may have some mitochondrial dysfunction that may contribute to muscle fatigability,26-28 yet almost no investigation has focused on the contributions of muscle fatigability to account for muscle weakness in individuals with PD. For this reason, it is important to better understand muscle fatigability in people with PD to further interpret the mechanisms underlying impaired movement.
In particular, quadriceps muscle weakness has profound functional consequences and is associated with decreased gait speed 29 and stair-climbing ability 30 as well as increased risk for falls with PD. 31 Therefore, the purpose of this investigation was to characterize quadriceps muscle weakness and activation failure in people with PD and explore whether these deficits were related to disease severity. We further sought to examine quadriceps muscle fatigability in these patients. To this end, we examined quadriceps muscle torque, activation, and fatigability in a sample of people with PD and a sample of healthy controls matched for age, sex, and body mass index (BMI). We were particularly interested in comparisons between PD participants with low-motor severity, high-motor severity, and healthy controls. We hypothesized that individuals with PD would demonstrate greater quadriceps weakness, activation deficits, and fatigue than healthy controls and that these deficits would be related to disease severity as measured by the UPDRS.
Methods
Study Design
This was a cross-sectional comparison of individuals with PD and healthy adults. All testing was performed on both lower extremities, although analysis focused on the less-involved (stronger) lower extremity for more conservative comparisons. The study was approved by the Colorado Multiple Institutional Review Board, and informed consent was obtained from all participants.
Participants
A total of 17 individuals with PD (12 men/5 women) were compared with healthy adult controls matched by age, sex, and BMI to approximate the characteristics of the patient sample (Table 1). Individuals were matched within 5 years of age and 5 BMI units. Recruitment occurred between January 2009 and June 2010. Individuals with PD were recruited through the Movement Disorders Clinic at the Movement Disorders Clinic at the University of Colorado. All patients were diagnosed by a fellowship-trained movement disorders neurologist using UK Brain Bank Criteria, 32 including clinical response to levodopa. Stage of disease was determined using the Hoehn and Yahr (H&Y) criteria (Table 2). 33
Baseline Characteristics of Individuals With Parkinson Disease and Healthy Controls a
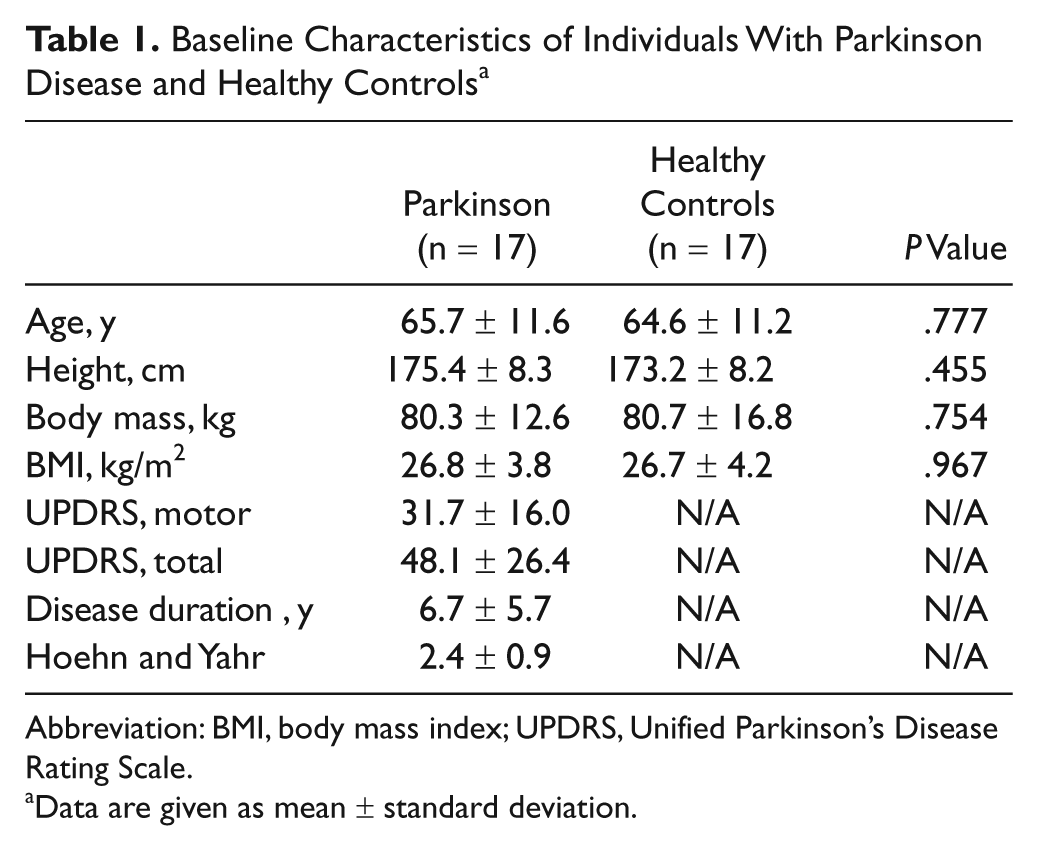
Abbreviation: BMI, body mass index; UPDRS, Unified Parkinson’s Disease Rating Scale.
Data are given as mean ± standard deviation.
Baseline Characteristics of Individuals With PD by Low- and High-PD Motor Signs a
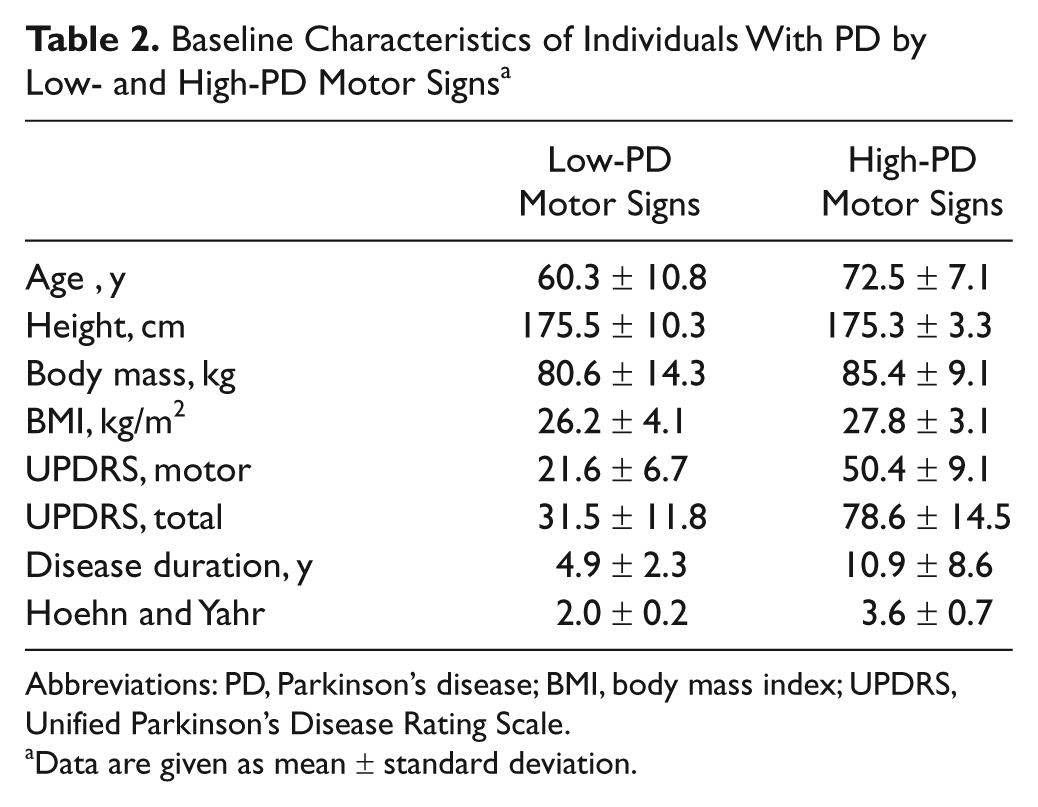
Abbreviations: PD, Parkinson’s disease; BMI, body mass index; UPDRS, Unified Parkinson’s Disease Rating Scale.
Data are given as mean ± standard deviation.
Individuals with PD were able to ambulate safely and independently with or without the use of an assistive device. They were excluded if they were unable to follow directions and/or had a deep brain stimulator implanted. In addition, individuals were excluded if they had any contraindications to exertion with exercise, any other neurological problems, any knee osteoarthritis, or knee pain greater than 2 out of 10 on an intermittent basis or average knee pain greater than 0 out of 10 with daily activities based on self-report. All participants were tested in their optimally medicated state. This was determined by asking patients to report if they felt that they were currently in their “on state” at the time of testing. Optimal medication dosage was previously established by a neurologist who was a movement disorders specialist.
Healthy adults were recruited from the community. They were excluded if they had uncontrolled hypertension, uncontrolled diabetes, significant neurological impairments, unstable lower-extremity orthopedic conditions, and knee pain greater than 2 out of 10 on an intermittent basis or average knee pain greater than 0 out of 10 with daily activities based on self-report.
We recruited people with a wide range of PD disease severity because we were interested in comparing contributions to weakness (Table 2) for those with lower severity (low-PD motor signs, UPDRS motor score < 31.7, n = 11) and those with higher severity (high-PD motor signs, UPDRS motor score ≥31.7, n = 6). The UPDRS motor score cutoff was based on the median score for all participants (31.7 ± 16.0) and related well with H&Y staging.
Outcomes
Isometric Muscle Torque Testing
Participants were seated and stabilized on a HUMAC NORM (CSMi, Stoughton, Massachusetts) electromechanical dynamometer with 60° of knee flexion for isometric quadriceps muscle torque assessments as previously described. 34 Following 2 warm-up contractions, a practice maximal voluntary isometric contraction (MVIC) was performed. Data were acquired using a Biopac data acquisition system (Biodex Systems, Inc, Goleta, California) using a sampling frequency of 2000 Hz and analyzed using AcqKnowledge software, Version 3.8.2 (Biodex Systems, Inc) to allow for gravity correction. Verbal encouragement was given during each maximum attempt. Visual torque targets were set on the feedback monitor at slightly higher torques than those produced during the practice MVIC trial. Data for 3 maximum trials were collected, and the best trial was used for analysis.
Muscle Activation Testing
Voluntary activation of the quadriceps muscle was assessed using the doublet interpolation technique, where a 2-pulse, 600-µs pulse duration, 100-Hz supramaximal stimulus was applied during an MVIC and again, immediately afterward, while the quadriceps muscle was at rest.35,36 The ratio of voluntary activation was quantified with the following formula: Voluntary activation = [1 – (
Fatigability Testing
Following the baseline MVIC and activation testing, individuals performed a quadriceps muscle fatigability test. After initial warm-up and practice, individuals were asked to perform 30 consecutive maximal, isokinetic repetitions of knee extension at 90 deg/s. During the fatigability test, participants were given strong verbal encouragement and visual feedback to help them attain maximal efforts. Peak torque from the highest of the first 3 contractions was used to normalize peak knee extension torque throughout the test. Normalized torque for the 2nd (“initial”) and 29th (“final”) contraction of the 30 repetition isokinetic protocol was used to calculate a fatigability index because the first and last repetitions were less likely to reflect maximal effort during the fatigue test. Using the normalized values, a fatigability index was calculated as [1 − (final normalized torque/initial normalized torque)] for assessment of quadriceps fatigability.
Previous studies have used different methods to evaluate muscle force production. Some studies have focused on isometric measures,7,9,11 and others have used isokinetic measures,8-10,12 both of which accurately assess muscle performance. We chose isometric methods for testing quadriceps torque production because interpolation testing methods for muscle activation require isometric contractions. However, for the fatigability protocol, we chose an isokinetic test to assess how repetitive, fatiguing activity affects quadriceps muscle performance. 38
Many previous studies have compared force production of the more- and less-involved legs. For this investigation, we chose to specifically target our analysis to the less-involved (stronger) leg to minimize the number of comparisons and better focus our interpretation of findings. As such, our conclusions are conservative because the more-affected leg may exhibit even greater deficits in performance.
Unified Parkinson’s Disease Rating Scale (UPDRS)
The UPDRS was used to quantify the signs, symptoms, and disease progression of PD.39,40 The UPDRS characterizes impairments and functional ability using a rating scale from 0 to 4. The UPDRS includes a motor subscale that contains a range of items describing signs and symptoms of PD such as tremor and rigidity and body movements such as foot taps and rising from a chair. The motor subscale (UPDRS motor) was used for analysis. A physical therapist trained by a movement disorders specialist performed UPDRS assessments.
Data Analysis
A sample size estimate for determining differences in quadriceps strength between individuals with PD and healthy controls was calculated using the means and standard deviation (SD) values available for the first 7 participants with PD and 5 healthy controls. We determined that a sample size of 14 per group would detect differences between groups using a power level of 0.9, an α level of .05, and a 2-tailed
Every attempt was made to precisely match healthy controls to individuals with PD; however, the inherent limitations with matching led us to use a conservative analytic strategy (independent samples rather than paired
Results
There were no differences in demographics or anthropometrics between PD and healthy control groups, which was as expected because of the matching criteria used for healthy controls (Table 1). The average UPDRS motor score for all individuals with PD was (mean ± SD) 31.7 ± 16.0, and the total UPDRS score was 48.1 ± 26.4. Characteristics of the 2 PD subgroups (low- and high-PD motor signs) are presented in Table 2.
Quadriceps Torque
A 1-way ANOVA comparing quadriceps torque for healthy controls and the low-PD and high-PD motor signs groups indicated an overall main effect (
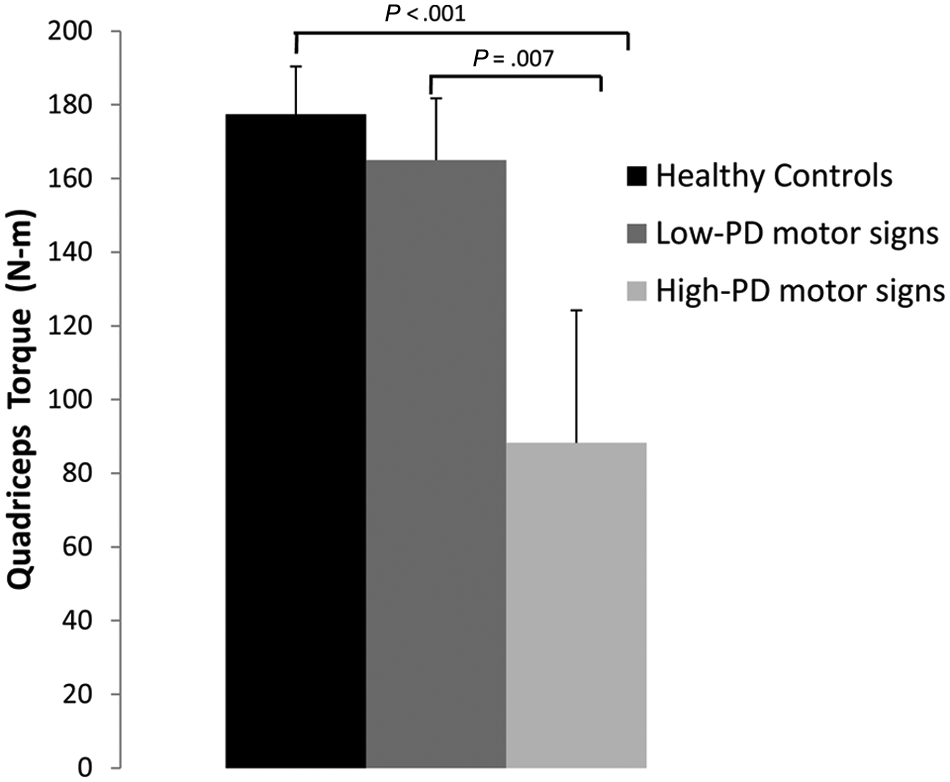
Quadriceps torque: isometric quadriceps torque for healthy controls, individuals with low-PD motor signs, and individuals with high-PD motor signs (mean ± standard error of the mean). Abbreviation: PD, Parkinson disease.
Quadriceps Activation
A 1-way ANOVA comparing quadriceps activation for healthy controls and the low-PD and high-PD motor signs groups indicated an overall main effect for quadriceps activation (
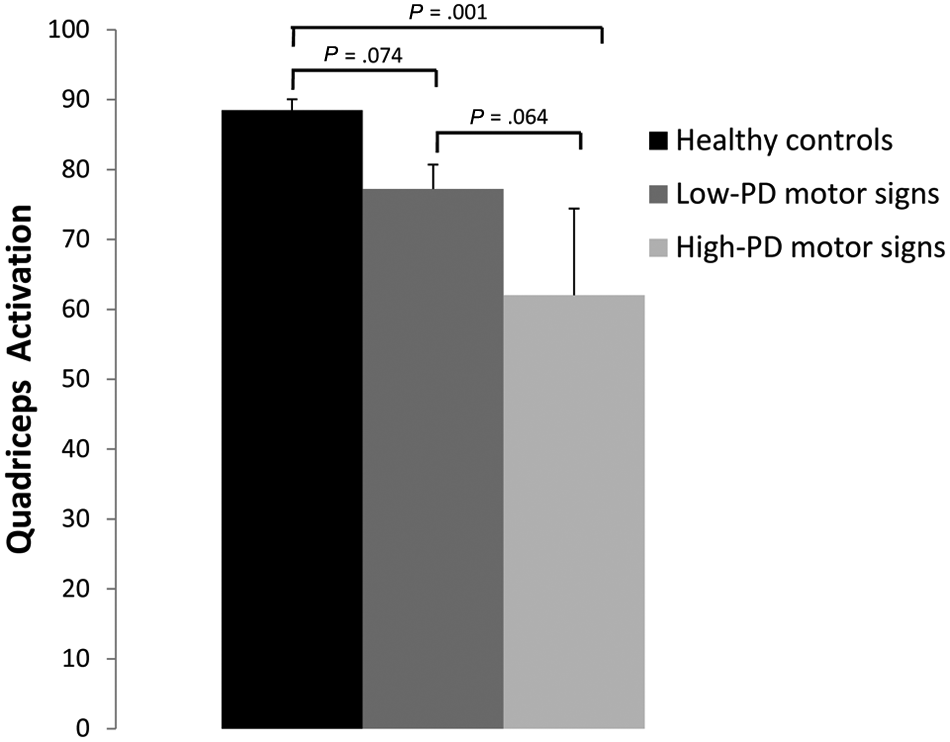
Quadriceps activation for healthy controls, individuals with low-PD motor signs, and individuals with high-PD motor signs (mean ± standard error of the mean). Abbreviation: PD, Parkinson disease.
Quadriceps Fatigability
A 1-way ANOVA comparing quadriceps fatigability for healthy controls and the low-PD and high-PD motor signs groups (Figure 3) indicated an overall main effect for fatigue index (
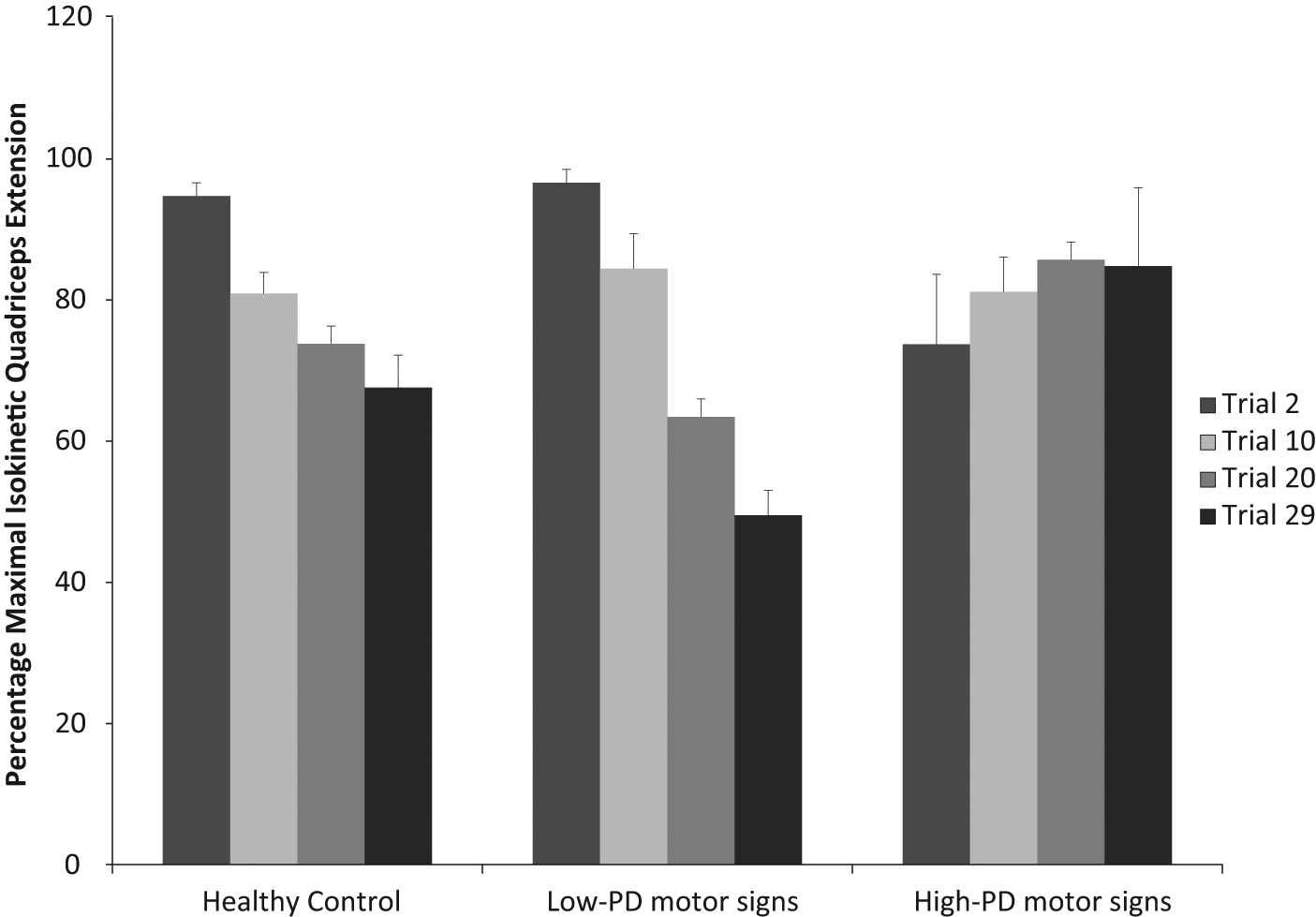
Percentage maximal isokinetic quadriceps extension for healthy controls and the low-PD and high-PD motor signs groups (mean ± standard error of the mean). Abbreviation: PD, Parkinson disease.
Discussion
Results from this investigation provide additional evidence for lower-extremity strength loss and new evidence for quadriceps activation deficits among people with PD. Furthermore, strength and activation deficits correlated strongly with severity of PD as indicated by the UPDRS motor score. In addition, our findings suggest that muscle fatigability in the low-PD motor signs group (42.1%) was greater than that of healthy adults (28.9%) but not statistically significant. Yet for individuals with more PD symptoms, muscle fatigue did not occur, most likely because of insufficient central activation to allow for muscle overload to induce metabolic fatigue.
Our first objective was to examine strength deficits in a sample of individuals with a wide range of UPDRS motor scores. To this end, we recruited participants with UPDRS motor scores from 9.5 to 61.0, including individuals who relied on an assistive device for ambulation. Thus, the sample in this investigation includes individuals with greater motor deficits than are reported in many studies that have investigated lower-extremity muscle strength to date.6,7,9,42-44
Those individuals in our sample who had low-PD motor signs (UPDRS motor scores < 31.7) had a slight reduction in maximum isometric torque production compared with healthy controls (12.4 N m; 7.0% deficit), which was not statistically significant or clinically meaningful.45,46 Yet reductions in torque may become more apparent with disease progression as evidenced by the subset of participants with high-PD motor signs (UPRDRS motor score ≥ 31.7). This group had significant torque deficits compared with both the control group and low-PD motor signs group. Furthermore, torque production was strongly correlated to UPDRS motor scores, suggesting that pronounced deficits in torque production occur with greater disease severity, further clarifying the development of strength deficits among people with PD.
Our results support those of other studies with respect to muscle weakness with PD. Koller and Kase 9 found that isokinetic force production was decreased even in early stage PD (H&Y I and II). Although the authors found statistically significant losses in both the involved and uninvolved legs, the differences were not clinically meaningful for the less-involved side. These results are consistent with our findings that there was no appreciable strength deficit in the less-involved leg in early-stage PD. Nogaki and colleagues10,12 have similarly reported lower-extremity muscle weakness using individuals with unilateral and bilateral PD involvement.
Our second objective was to determine whether people with PD have deficits in central activation. Our results provide direct evidence for central activation deficits and, furthermore, for the fact that these deficits parallel the loss of quadriceps strength in patients with PD. Although a lack of statistical power likely dampened the strength of some of our statistical findings, overall, our results suggest that central activation deficits may begin early but are most pronounced later in the disease. Furthermore, the strong negative correlation of central activation deficits with quadriceps strength suggests that this may be an important mechanism driving strength loss. Further studies will need to be done to compare the contribution of central mechanisms versus peripheral factors such as atrophy from disuse. Studies of other patient populations have also shown that when central activation deficits are present, they often account for a large amount of the muscle weakness.37,47,48 This information is important because of the ramifications for rehabilitation. Muscle atrophy caused by deconditioning (eg, as a result of the sedentary lifestyle that can accompany PD) can be remediated by loading the muscle (eg, progressive resistive exercise).4,49 However, central activation deficits do not allow for sufficient overload of muscle to induce muscle hypertrophy, 50 which may limit the effectiveness of volition-based strengthening programs in the later stages of PD when central activation deficits are most apparent. In fact, individuals with large activation deficits from traumatic knee injuries have demonstrated negligible improvements in force production even after intensive rehabilitation. 50 Therefore, these results suggest that alternative treatment options may be necessary to counter activation deficits to achieve gains in force production.
Our third objective was to examine quadriceps muscle fatigability. Fatigue is a substantial complaint among people with PD. 51 We thought it was possible that physiological fatigability of the muscle might contribute to the overall perception of fatigue for individuals with PD with more advanced disease severity. However, our findings do not support this expectation. This is consistent with prior findings of a dissociation between subjective fatigue ratings and objective muscle fatigability in patient populations, including in those with PD.52,53 Indeed, people with low-PD motor signs (UPDRS motor < 31.7) had some increase in quadriceps muscle fatigability (not statistically significant) that could be considered clinically meaningful. In contrast, and contrary to our expectations, people with high-PD motor signs did not exhibit any fatigability. We believe that this finding is related to deficits in central activation described above. Because people with high-PD motor signs were not able to activate the quadriceps muscle fully, they did not have sufficient muscle overload to reach a level of metabolic fatigue during repeated contractions. The fact that patients demonstrated a trend toward a slight increase in muscle torque with fatigue testing suggests that central activation of muscle might improve during repetitive activities, which should be the focus of future investigations. Similar findings of increased torque during fatigue testing have been reported in patients with spinal cord injury who also have marked central activation deficits (CAD). 54
Findings from this investigation have implications for rehabilitation and suggest that we may need to find alternative strategies to help people with PD improve strength, especially in later stages of the disease when volition-based exercise programs are not likely to be effective. Possibly, treatments designed to improve activation deficits would result in the greatest gains. Recent investigations have demonstrated improvements in quadriceps force when neuromuscular electrical stimulation is used in combination with traditional intervention strategies in patients who have sustained a stroke or have cerebral palsy.21,22,55 Improvements have been demonstrated in both quadriceps force and motor unit recruitment.21,22,55 Taken together, these findings suggest that it would be appropriate to examine whether strengthening exercises in the presence of neuromuscular electrical stimulation would facilitate muscle strengthening for people especially in later stages of PD. This issue is of substantial clinical importance because of the known relationship between quadriceps strength, task performance (eg, stair climbing), and stability,29,56,57 all of which are compromised as PD progresses.
Similarly, eccentric muscle strengthening may offer an alternative option for improving muscle activation and muscle strength. Eccentric muscle strengthening is specific to functional tasks such as stair descent, but it also provides higher absolute forces and thus a greater stimulus for hypertrophy compared with concentric training. 58 Training programs that combine both concentric and eccentric muscle activation produce greater increases in muscle fiber size compared with standard programs that involve only concentric training.59,60 In addition, eccentric training may provide a greater stimulus for neural adaptation compared with concentric training, 61 which may help attenuate central activation deficits. Indeed, preliminary work of Dibble and colleagues4,62,63 found that high-intensity eccentric strengthening was safe for people in the early stages of PD and resulted in better muscle size and strength as well as improved functional performance (stair descent and 6-minute walk time). 63 At present, it is unknown if similar benefits can be achieved in the presence of large activation deficits, such as those reported in the present study for patients with high-PD motor signs.
We acknowledge limitations to the study because of the small sample size, but we feel that our results highlight many clinically meaningful differences that allowed for the accurate interpretation of our results. We also acknowledge that our choice to focus our analyses on the stronger extremity may result in more conservative conclusions than might be obtained by including analysis of the weaker lower extremity. Also, we only tested participants when optimally medicated as assessed by asking them to report if they felt any change in their PD symptoms on the day of testing. Pedersen and Oberg 64 found that when individuals were off medication, they had a significant reduction in extensor muscle strength and prolonged muscle relaxation times, both of which correlated with changes in clinical status. Similarly, it is possible that deficits in central activation would be even greater in the off-medication state and in the more affected leg. Finally, whereas cognitive dysfunction, including impaired attention or poor motor learning, could have also influenced fatigability in our high-PD group, our observations of participants performing this simple motor task did not suggest problems with task learning, execution, or attention.
Findings from this investigation, together with data in the literature, provide direction to future studies designed to better understand the impact of weakness on function in people with PD. First, we recommend that such investigations focus on people with UPDRS motor scores of 30 or above to fully characterize the role of weakness with more advanced PD. Second, data are needed from a larger sample to further clarify the time course of deficits of strength and central activation and how these contribute to weakness and function as PD symptoms increase. Third, we recommend a reconsideration of tasks used to examine the relationship of quadriceps strength to function. To date, most studies have used tasks such as rising from sitting to standing that do not require fast muscle recruitment.6,13,65 Based on data suggesting that velocity influences the magnitude of deficits present in this population, 10 we recommend using tasks of high enough velocity to adequately challenge patients’ ability to generate force rapidly. Such responses are required, for example, when responding to an impending fall. Future investigations are also needed to examine other measures of force production (eg, rate of force production and steadiness of force production) in people with a range of UPDRS motor scores to further characterize deficits. Finally, investigations are necessary while patients are on and off anti-Parkinson medications to better understand performance deficits.
Footnotes
Acknowledgements
The authors thank Emily Hoke, SPT, John Kvale, DPT, Katherine Kulp, SPT, and David Weitzenkamp, PhD, for their contributions to this investigation.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NCRR Colorado CTSI Grant Number UL1 RR025780 and R01HD043770.
