Abstract
Background. The level of consciousness is regulated by the activation of the frontal parietal network since a disruption within this interregional cortical connectivity is associated with a consciousness breakdown. Objective. The primary aim of the present study was to examine the feasibility and the safety of a single session of repetitive transcranial magnetic stimulation (rTMS), delivered over the dorsolateral prefrontal cortex (DLPFC), in patients with unresponsive wakefulness syndrome (UWS). As a secondary aim, we investigated the efficacy of rTMS over DLPFC on cortico-cortical connectivity as tested with paired-pulse and dual-coil TMS techniques. Methods. We enrolled 10 healthy and 10 postanoxic UWS subjects. After clinical assessment, subjects underwent a single/paired pulse TMS paradigm evaluating the inhibitory and facilitatory intracortical circuits. In addition, several interregional interactions between primary motor, dorsal and ventral premotor, supplementary motor, and posterior parietal cortex areas were investigated by means of dual-coil TMS technique. These parameters were evaluated at baseline, immediately after, and 60 minutes after a protocol of 10-Hz rTMS delivered at the level of the DLPFC. Results. rTMS over DLPFC did not cause any adverse events. We showed that rTMS over DLPFC did not induce, at group level, any clinical improvement or intra-/intercortical connectivity changes. Interestingly, in 3 patients rTMS induced a significant, although transient, clinical improvement associated with a short-lasting reshaping of brain connectivity. Conclusions. We demonstrated that a single session of 10-Hz rTMS over the right DLPFC may transiently improve consciousness and partially restore the connectivity within several cortical areas in some patients with UWS.
Introduction
Comatose patients, who survive brain injury (BI) because of the ongoing improvements of intensive care medicine, may recover consciousness within several days or weeks or enter an unresponsive wakefulness syndrome (UWS), previously known as vegetative state (VS). 1 UWS is characterized by wakefulness without awareness and often may progress to long-lasting condition with a poor outcome. 2
Another distinct condition that can emerge after coma has been defined by Giacino and associates as the minimally conscious state (MCS), where consciousness is severely altered, although a minimal behavioral evidence of self- or environmental-awareness can be demonstrated. 3 MCS patients show fluctuating awareness and may appropriately respond to some stimuli,4-7 whereas UWS patients totally lack self-environment perception but are awake (differently from coma in which patients are unawake and unaware).
At present, there are no evidence-based guidelines regarding the treatment of patients with disorders of consciousness (DOC). 8 Nevertheless, some studies have recently aimed to demonstrate the potential therapeutic effect of different pharmacologic interventions. 9 Among the nonpharmacological treatment, a controlled case study assessing the role of thalamic deep brain stimulation in an MCS patient has been reported. 10 The observed improvements in arousal level, motor control, and behavioral persistence, obtained after thalamic stimulation, might reflect an activation of frontoparietal networks, which are interconnected with the anterior intralaminar and adjacent paralaminar regions of thalamic association nuclei, basal ganglia, and brainstem arousal systems. 10
Indeed, the level of consciousness is regulated by activation of the frontoparietal network, 11 since it has been postulated that a disruption within this interregional cortical connectivity is associated with a consciousness breakdown. Moreover, bidirectional corticothalamic loops may have an important role in cons-ciousness generation and maintenance.12,13 Another possibility to modulate frontoparietal network is by means of noninvasive neuromodulation using transcranial magnetic stimulation (TMS). TMS is a painless and noninvasive means of perturbing the excitability of cortical neurons, allowing the assessment of the integrity of motor pathways, and the cortical connectivity. 14 Cortico-cortical connectivity can be evaluated by using the dual-coil TMS (dcTMS), which tests the time course of interactions in a “2-node” neural circuit, using 2 different coils. In this way, it possible to test the supplementary motor area (SMA)–primary motor cortex (M1), premotor–M1, and parietal–M1 connectivity.15-18
The most relevant feature of this technique is that sustained changes in neural excitability can be induced 19 if TMS is applied repetitively (rTMS). Therefore, the primary aim of the present study was to examine the feasibility and the safety of a single session of rTMS delivered over the dorsolateral prefrontal cortex (DLPFC) in patients with postanoxic UWS. A secondary aim was to investigate the efficacy of rTMS over DLPFC in improving connectivity within the frontoparietal network (as tested with dcTMS), in an attempt to facilitate the recovery of consciousness. We chose to stimulate the right DLPFC because it has been linked to the maintenance of awareness and attention. 20 In addition, the right DLPFC is thought to play a central integrative function in motor control and behavior and is a critical component of the decision-making network. 21
Materials and Methods
Subjects
We enrolled 10 healthy (HC; 5 females and 5 males, mean age 35.3 ± 7.2 years) and 10 postanoxic UWS subjects (7 females and 3 males, mean age 58.9 ± 5.3 years), attending the long-term rehabilitative unit of the IRCCS Centro Neurolesi “Bonino-Pulejo” (Messina, Italy). All the patients met the international criteria for VS diagnosis,4,5,22 with mean disease duration of 12.2 ± 1 months. Of the eligible 17 postanoxic UWS patients, 2 were excluded since in a UWS condition lasting less than 3 months after BI, 2 had other severe neurological or systemic diseases or were in critical conditions (ie, inability to breathe independently, hemodynamic instability), and 3 assumed cortical excitability-modifying drugs.
The demographic and clinical characteristics at baseline are reported in Table 1. The postanoxic condition was a consequence of a prolonged cardiac arrest following acute myocardial infarction or ventricular fibrillation. Nuclear magnetic resonance imaging (MRI) showed diffuse cerebral cortical and white matter T2-hyperintensities (ie, at thalamus and basal ganglia). Electroencephalographic (EEG) examination evidenced a continuous slowing in theta and/or delta frequency ranges. Moreover, in each patient we determined the bilateral presence of the somatosensory evoked potential N20 component. In addition, all the enrolled patients had no focal lesions in frontal lobes at MRI scans, epileptic history or EEG epileptiform activity, pacemaker, aneurysms clips, neurostimulator, or brain/subdural electrodes.
UWS Subjects’ Baseline Clinical, CRS-R, and Demographic Characteristics a .
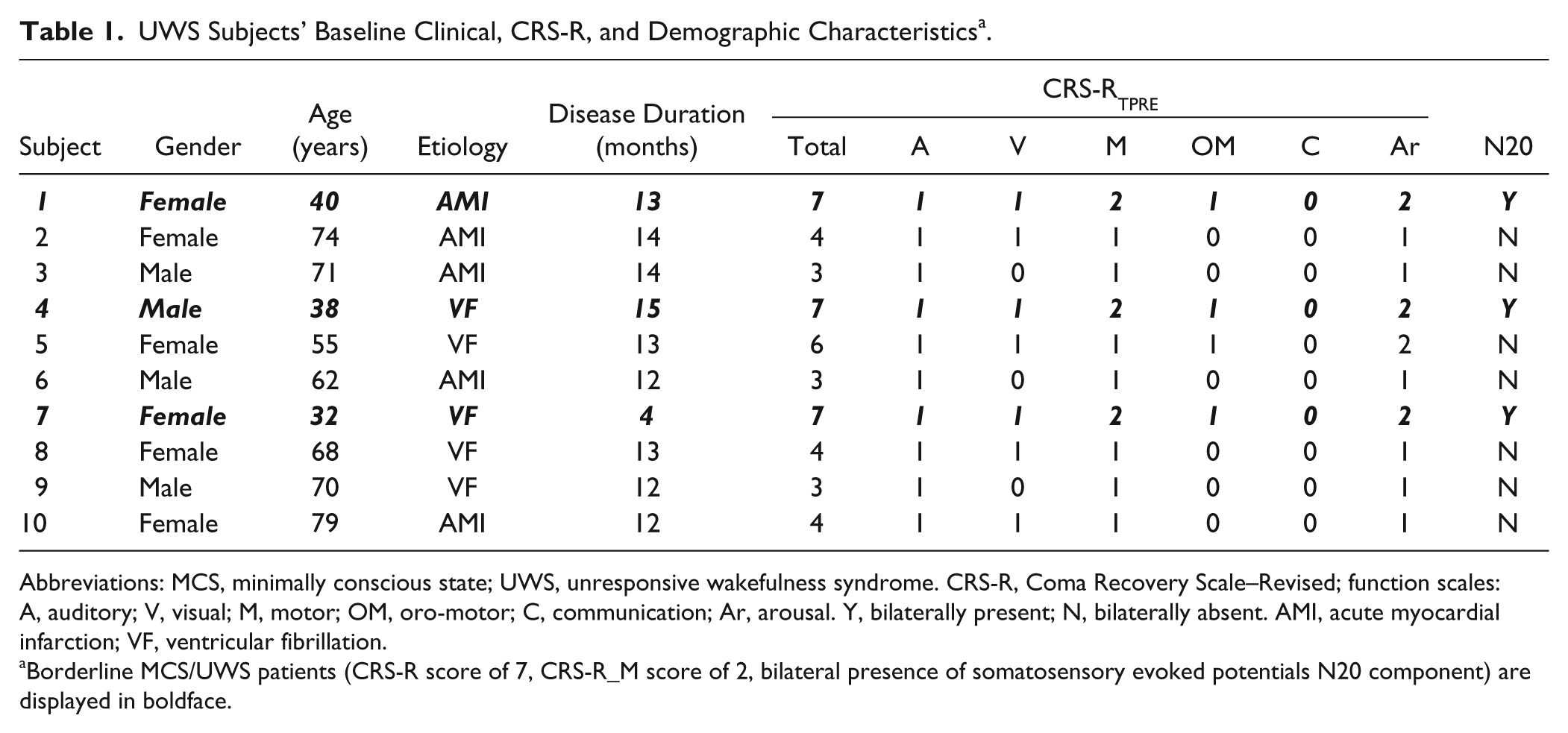
Abbreviations: MCS, minimally conscious state; UWS, unresponsive wakefulness syndrome. CRS-R, Coma Recovery Scale–Revised; function scales: A, auditory; V, visual; M, motor; OM, oro-motor; C, communication; Ar, arousal. Y, bilaterally present; N, bilaterally absent. AMI, acute myocardial infarction; VF, ventricular fibrillation.
Borderline MCS/UWS patients (CRS-R score of 7, CRS-R_M score of 2, bilateral presence of somatosensory evoked potentials N20 component) are displayed in boldface.
The present study was approved by our research institute ethics committee (registration number: 43/2013), and written informed consent was obtained either from HC or the legal guardian of each patient.
Study Design
After clinical evaluation, including neurological examination and JFK Coma Recovery Scale–Revised (CRS-R) 23 (daily administered for 30 days, at different times, before the study enrollment in order to assess a truly and stable UWS condition), subjects were tested for the following single-pulse TMS or paired-pulse TMS parameters at baseline (TPRE): resting motor threshold (rMT), motor evoked potentials (MEP) amplitude, short (SICI) and long (LICI) intracortical inhibition, intracortical facilitation (ICF). Furthermore, we tested the interregional interactions between M1 and dorsal premotor (PMd), ventral premotor (PMv), SMA, and posterior parietal (PPC) cortices by means of dcTMS (the time implied was around 20 minutes). Once the arousal was steadily maintained (ie, the eyes remained opened across the length of the entire examination with no need of stimulation), we applied the real_rTMS on the right DLPFC (the time implied was around 8 minutes). Then, we performed the same baseline measures and the CRS-R assessment immediately after the end (T0, around 60 minutes), and after 1 hour (T60, ~60 minutes) of the conditioning protocol.
As control experiment, after a week from the main protocol, we performed a sham rTMS conditioning in the UWS “responder” patients (ie, UWS individuals with higher CRS-R score, who had presented a motor behavioral improvement after real stimulation) and in 5 HC, in an attempt to confirm that such findings (ie, the positive response to rTMS) were related to our stimulation protocol.
Clinical Assessment
The neurological examination predominantly showed a pattern of spastic tetraparesis. Patients were clinically evaluated also through the JFK CRS-R, which was daily administered for 30 days before the study enrollment by at least 2 independent DOC skilled neurologists in order to assess a truly and stable UWS condition. CRS-R is a reliable and standardized tool, which integrates behavioral and clinical assessments, and includes the current diagnostic criteria for coma, VS, and MCS, allowing the patient to be assigned to the most appropriate diagnostic category. It consists of 29 hierarchically organized items divided into 6 subscales addressing auditory, visual, motor, oro-motor, communication, and arousal processes. The total score ranges from 0 to 23. A score of ≤2 on the auditory, motor (CRS-R_M), and oro-motor/verbal subscales and ≤1 on the visual subscale, and of zero on the communication subscale is consistent with the diagnosis of VS. Thus, the CRS-R is an appropriate measure for characterizing the level of consciousness and for monitoring the neurobehavioral function recovery.
Single-Pulse and Paired-Pulse TMS Measures
MEPs were obtained through magnetic monophasic stimuli delivered by a high-power Magstim200 Stimulator (Magstim, Whitland, Dyfed, UK). The coil was placed tangentially to the scalp with the handle pointing backwards and laterally, at a 45° angle to the sagittal plane, approximately perpendicular to the central sulcus of the left hemisphere, on the optimal site on the scalp to get the wider MEP amplitude (motor hot-spot), from the right abductor pollicis brevis muscle (APB). The rise time of the magnetic monophasic stimulus was about 100 µs with a to-zero of about 800 µs. The current flowed in handle direction during the rise-time of the magnetic field, thus with a posterior-anterior direction. We preliminarily evaluated the RMT, defined as the smallest stimulus intensity able to evoke a peak-to-peak MEP of 50 µV in rest right APB, in at least 5 out of 10 tracks consecutively. 24 Then, we applied an intensity of stimulation of 120% of rMT. Electromyographic activity (EMG) was recorded through Ag–AgCl surface electrodes applied to right APB using a classic muscle belly-tendon montage. Signals were amplified and filtered (from 32 Hz to 1 kHz) via a Digitimer D150 Amplifier (Digitimer Ltd, Welwyn Garden City, Herts, UK), and stored using a sampling frequency of 10 kHz on a personal computer for off-line analysis (Signal Software, Cambridge Electronic Design, Cambridge, UK).
SICI and ICF were determined according to the Kujirai paired-pulse method. 25 The intensity of the CS was set at 70% of rMT. 26 The intensity of the test stimulus was set at 120% of rMT. Such intensities were kept constant throughout the experiment. SICI and ICF were assessed at an interstimulus interval (ISI) of 2 and 12 ms, respectively. LICI was elicited by suprathreshold CS and TS applied at an ISI of 150 ms. 27 At ISIs longer than 50 ms LICI is mediated within M1 rather than subcortical structures. 28
Mean amplitude of the conditioned MEP was expressed as percentage of the amplitude of the unconditioned MEP and was taken as a measure of corticospinal excitability. We registered 15 MEPs, 10 SICI, 10 LICI, and 10 ICF, randomly intermingled in a single trial, at a frequency of 0.2 Hz. All data are given as mean or percentage difference in comparison to baseline values ± standard error (se).
dcTMS Measures
Before and after rTMS protocol on right DLPFC, we measured the effects on MEP amplitude following dsTMS on PMdright-M1left and PMvleft-M1left (ISI of 7 ms, CS at 70% rMT, TS as per unconditioned MEP generation on M1), 19 SMA-M1left (CS at an ISI of 6 ms, 3 cm anterior to Cz-preSMA at 70% rMT, TS as per unconditioned MEP generation on M1),29,30 and PPCleft-M1left (CS at an ISI of 10 ms, at 90% rMT, TS as per unconditioned MEP generation on M1). 20 The stimulation setup used was nearly the same to the one for MEP elicitation, differing only in the application of 2 figure-of-eight monophasic coils. We registered, in different trials, 10 conditioned MEPs for each non-M1–M1 interaction, intermingled with 10 MEPs, delivered at a frequency of 0.2 Hz. Mean amplitude of the conditioned MEP was expressed as percentage of the amplitude of the unconditioned MEP and was taken as a measure of non-M1–M1 excitability. All data are given as mean or percentage difference in comparison to baseline values ± se.
Conditioning Protocol
According to the safety guidelines reported by Rossi and associates, 31 real rTMS consisted of 1000 biphasic pulses delivered in trains of 50 stimuli in 5 seconds (10 Hz frequency) repeated every 20 seconds on the right DLPFC, targeted by putting the coil to the F4 position (according to the international 10-20 system for electroencephalography electrode placement). Magnetic pulses were delivered through a standard figure-of-eight biphasic coil connected to a Magstim Rapid2 stimulator (Magstim Company, Whitland, Dyfed, UK). The mean loop diameter of the wings of the coil was 9 cm. The intensity of TMS pulses was set at 90% rMT. The magnetic stimulus had a biphasic waveform with a pulse width of approximately 300 µs. The junction region of the coil pointed backwards and laterally at a 45° angle away from the midline. The sham rTMS protocol had the same experimental setup but differed for the application of a sham-coil for rTMS.
Data Acquisition and Statistical Analysis
EMG was recorded with Ag–AgCl surface electrodes from the right APB muscle using a belly tendon montage. Signal was amplified and band-pass filtered (32 Hz to1 kHz) by a Digitimer D-360 amplifier and stored at a sampling rate of 10 kHz on a personal computer for off-line analysis (Signal Software; Cambridge Electronic Design, Cambridge, UK). During the experiments, EMG activity was continuously monitored with visual (oscilloscope) and auditory (speakers) feedback, to ensure the complete muscle relaxation.
The effects of rTMS on each dependent variable (CRS-R, CRS-R_M, RMT, MEP, SICI, ICF, LICI, PMd-M1, PMv-M1, SMA-M1, PPC-M1) were evaluated in separated 3-way repeated-measures analysis of variance (rmANOVA), implying time (3 levels: TPRE, T0, and T60) and protocol (2 levels: sham rTMS and real rTMS) as within-subjects factor and group (2 levels: UWS and HC) as between-subjects factor. The Greenhouse–Geisser method was used if necessary to correct for nonsphericity. Conditional on a significant F-value, post hoc t tests (Bonferroni test) were performed to explore the strength of main effects and the patterns of interaction between the experimental factors. All statistical tests were applied 2-tailed. A P value of <.05 was considered significant. All data are given as means or percent changes ± se. A Pearson’s correlation analysis was carried out to assess an eventual relationship among age, disease duration, CRS-R, CRS-R_M, and each electrophysiological parameter.
Results
No side effects were observed for each of our patients either during or after the entire experiment.
UWS/HC Electrophysiological Differences at Baseline
RMT in UWS was similar to HC (RMT: 55 ± 5 vs 52 ± 3, P > .05). On the other hand, SICI, LICI, and ICF were significantly different from HC (SICI: 91 ± 11 vs 29 ± 5, P < .001; ICF: 102 ± 13 vs 129 ± 20, P = .03; LICI: 76 ± 16 vs 39 ± 6, P = .01). UWS patients had an alteration of cortico-cortical interactions in comparison to HC (PMd-M1: 98 ± 10 vs 59 ± 9, P = .01; PMv-M1: 102 ± 13 vs 124 ± 19, P = .03; SMA-M1: 105 ± 18 vs 69 ± 18, P = .04; PPC-M1: 93 ± 11 vs 121 ± 11, P = .03). Interestingly, 3 patients showed a partially preserved SICI (Figure 2).
rTMS Effects on Clinical Assessment
rTMS after-effects concerning CRS-R score are reported in Table 2. UWS patients showed at baseline a mean CRS-R score of ≤7 (Table 1). No patients showed a CRS-R_M score >2. Indeed, in response to the pain stimuli applied on limbs they showed a flexion withdrawal of the stimulated limb (2 points; “there is isolated flexion withdrawal of at least one limb; the limb must move away from the point of stimulation”) or an abnormal posturing (1 point; “slow, stereotyped flexion, or extension of the upper and/or lower extremities occurs immediately after the stimulus is applied”), or no response (0 points; “there is no discernible movement following application of noxious stimulation, secondary to hypertonic or flaccid muscle tone”). rmANOVA did not show any significant time * protocol interactions concerning either CRS-R or CRS-R_M. Nevertheless, after the application of real rTMS, 3 patients (nos. 1, 4, and 7) showed a short-lasting increase of the CRS-R_M scoring, from 2 to 3 points (only at T0; Table 2), thus showing a cortical motor behavior (ie, “the nonstimulated limb must locate and make contact with the stimulated body part at the point of stimulation on at least 2 of the 4 trials”; this denotes an MCS condition). These patients did not show such behavior at baseline (ie, CRS-R_M ≤ 2). At T0, such patients showed also a MEP amplitude increase, a remodulation of SICI and ICF, and a normalization of interregional interactions. There were no effects concerning LICI and PMv/M1, as well as in HC.
UWS Subjects’ CRS-R_M Changes After Real_rTMS a .
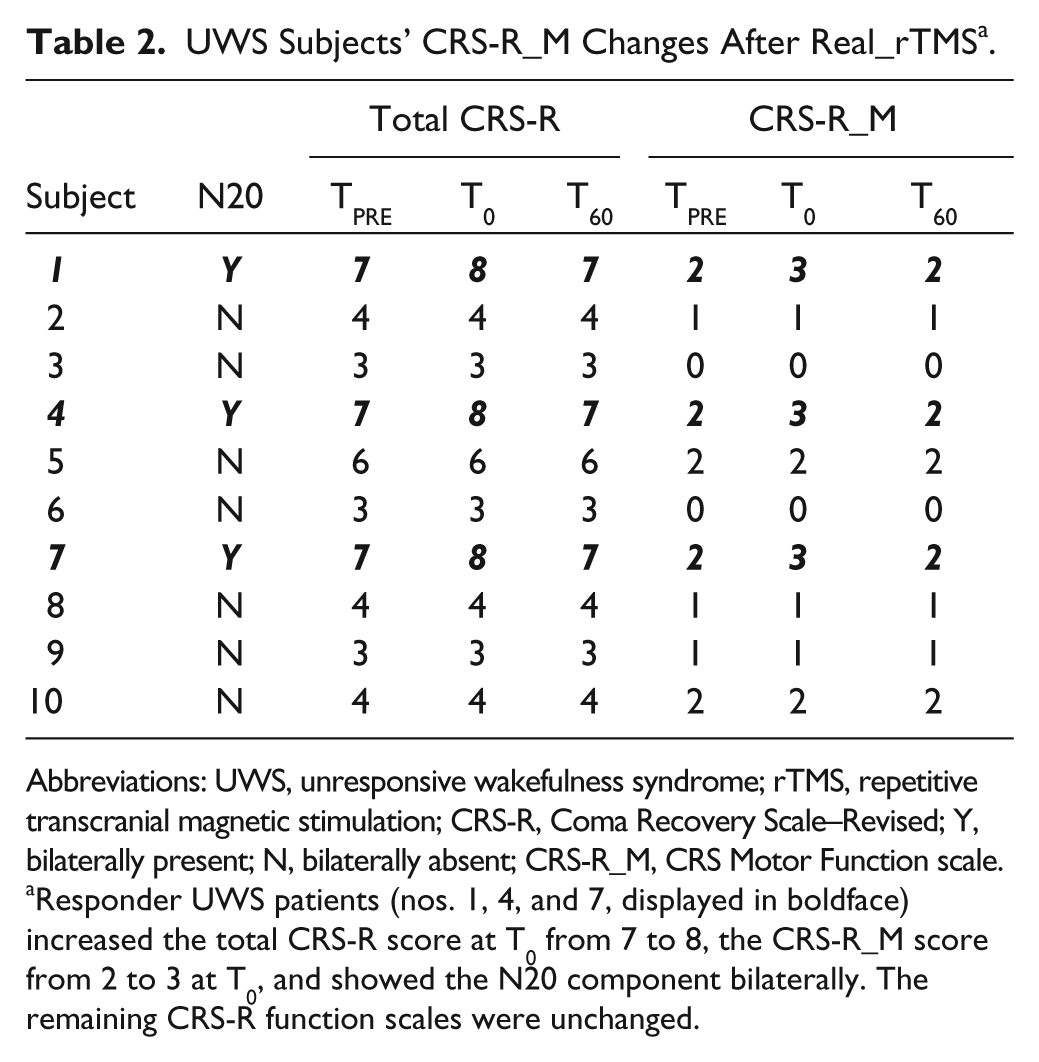
Abbreviations: UWS, unresponsive wakefulness syndrome; rTMS, repetitive transcranial magnetic stimulation; CRS-R, Coma Recovery Scale–Revised; Y, bilaterally present; N, bilaterally absent; CRS-R_M, CRS Motor Function scale.
Responder UWS patients (nos. 1, 4, and 7, displayed in boldface) increased the total CRS-R score at T0 from 7 to 8, the CRS-R_M score from 2 to 3 at T0, and showed the N20 component bilaterally. The remaining CRS-R function scales were unchanged.
rTMS Electrophysiological Effects
rmANOVA showed a significant main group * protocol * time (F(2,38) = 4.7, P = .03) and a significant time * protocol interaction in HC (F(2,18) = 6.9, P < .001), but not in UWS (P > .3).
In HC, one-way ANOVA showed a strong effect of factor time after real rTMS (Figure 1) concerning MEP amplitude (F(2,18) = 6, P = .007), SICI (F(2,18) = 8.5, P < .001), and ICF strength (F(2,18) = 4.1, P = .02), and SMA-M1 (F(2,18) = 3.2, P = .04), PMd-M1 (F(2,18) = 4.8, P = .01) and PPC-M1 (F(2,18) = 5.1, P = .009) interactions. Post hoc analysis showed a significant change at T0 concerning MEP (t(1,9) = 4.53, P = .001), ICF (t(1,9) = 4.36, P = .002), SICI (t(1,9) = 4.1, P = .003), PMd-M1 (t(1,9) = 4.34, P = .002), SMA-M1 (t(1,9) = 5.5, P < .001), PPC-M1 (t(1,9) = 4, P = .003). Hence, sham rTMS produced no effects (P > .05) in the 5 HC who also underwent the sham protocol (data not shown).
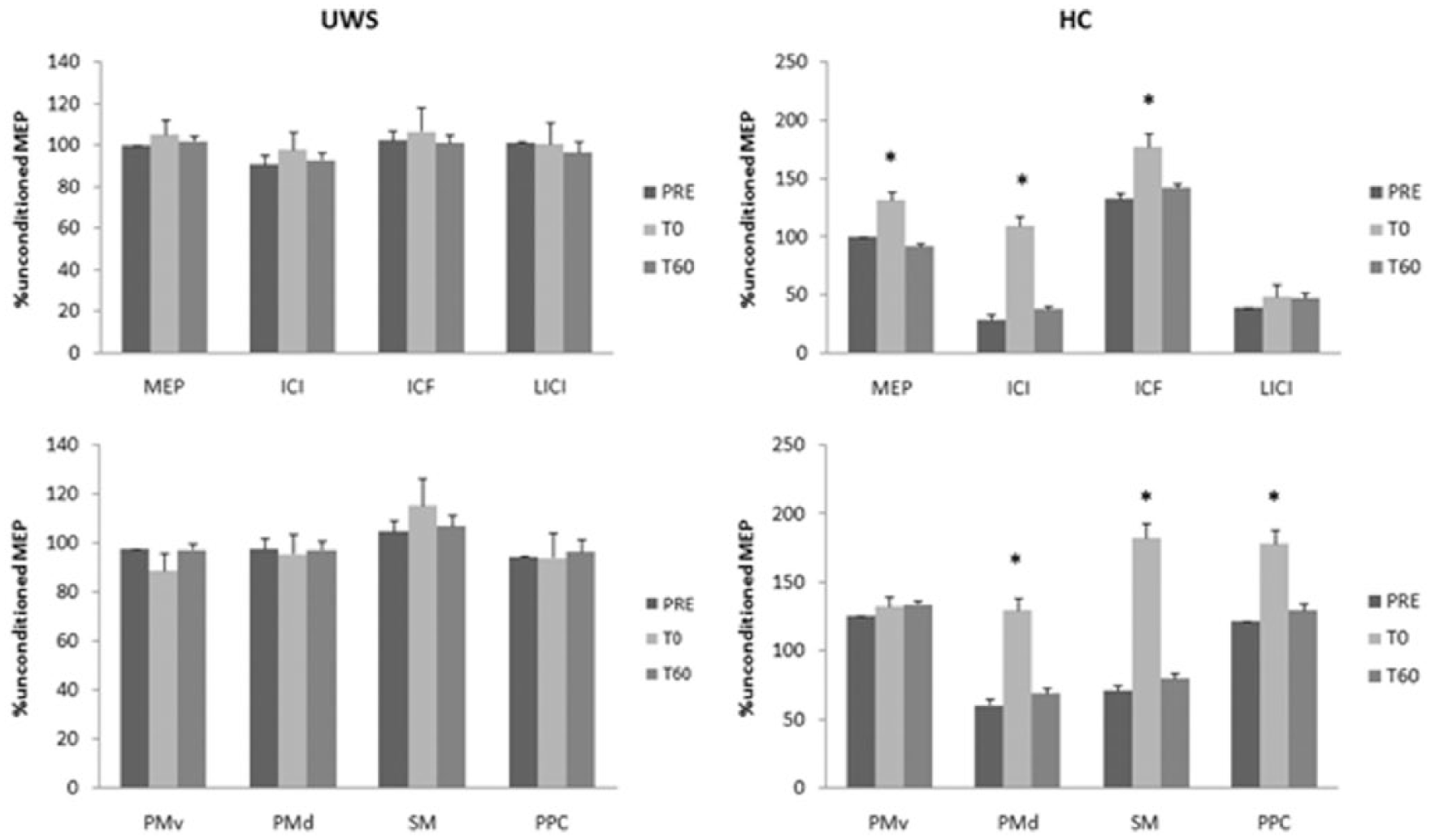
We show the time course of real_rTMS after-effects in UWS (left column) and HC (right column).
Although we did not find any real rTMS effect at UWS group level (Figure 1), we found only in 3 UWS subjects, that is, at UWS individual level, a modulation of MEP, SICI, and ICF (but not of LICI, as well as in HC; Figure 2). Moreover, they showed a restored normal pattern of cortico-cortical interactions (with the exception of PMv/M1), even though less evident in comparison to HC. These effects were short-lasting, as they were absent at T60 (Figure 2). We also found a short-lasting increase of CRS-R_mot scoring (from 2 to 3) in the same subjects (Table 2). Sham rTMS did not induce any effect in the 3 patients who were responsive to the real protocol (data not shown).
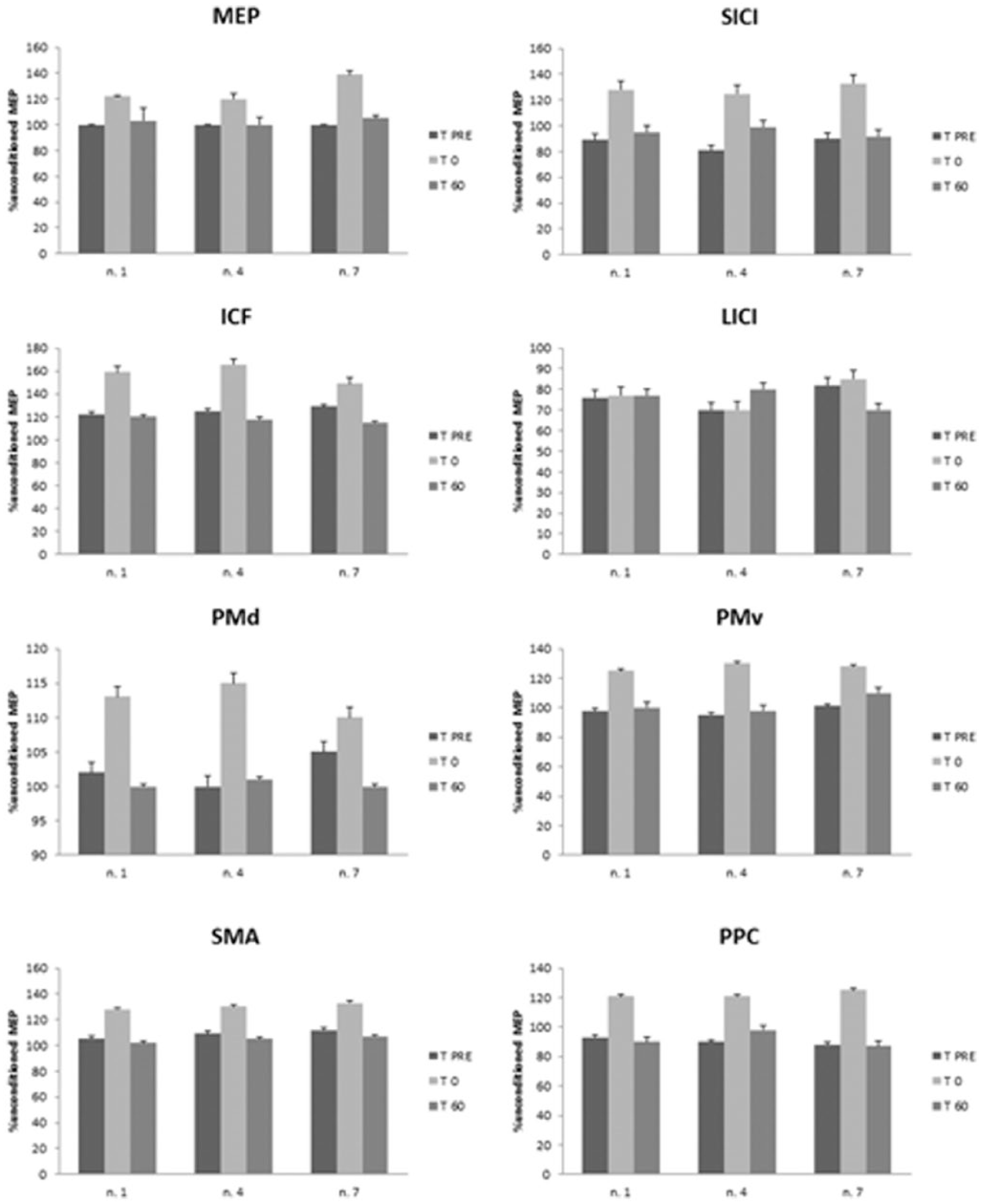
We show the time course of real_rTMS after-effects in the 3 “responder” UWS patients (nos. 1, 4, and 7), who also showed a CRS-R_M amelioration.
Correlations
We assessed the correlation between electrophysiological parameter at T0 in HC, in order to determine the direction of the effects induced by real rTMS. The main factors related to MEP amplitude increase at T0 were the SICI (r = .932, P < .001), and the PMd/M1 (r = .889, P < .001) and PPC/M1 (r = .890, P < .001) interactions. Moreover, in HC, we found that SICI, determined at TPRE, could be considered as a potential predicting factor of the rTMS effects at T0, as expressed by significant correlations with MEP (r = −0.906, P < .001), ICF (r = −.807, P = .006), and PMd-M1 (r = .895, P = .001) and PPC-M1 (r = −.904, P < .001) interactions.
Discussion
Our study raises 3 main issues:
DLPFC rTMS represents a safe brain-stimulation protocol, at the specified dose, since no adverse events were caused in both HC and UWS patients.
Paired pulse and dcTMS techniques may be helpful in better understanding the pathophysiology of cortical excitability and connectivity abnormalities in UWS patients.
rTMS over the right DLPFC might induce a transient improvement of CRS-R_M score in some postanoxic UWS patients, associated with a short-lasting reshaping of brain excitability and connectivity.
Safety Considerations
An important premise is that the protocol of stimulation was designed taking into account the safety guidelines recommended by the International Federation of Clinical Neurophysiology. 31 Unfortunately, only few studies have addressed the safety of a single or multisession rTMS approach in patients with DOC.
Notably, no side effects were observed for each of our patients either during or after the entire experiment.
Louise-Bender Pape and coworkers 32 applied a 6-week rTMS protocol (trains of paired stimuli, ie, 100 ms interpulse interval, 5-second intertrain interval) over the DLPFC in a 26-year-old man who remained in a VS around 9 months after severe traumatic BI. The authors did not report any adverse events related to the provision of rTMS treatment.
On the other hand, Cavinato and coworkers 33 reported the occurrence of a partial and secondarily generalized tonic–clonic seizure in a 31-year-old right handed male, 3 hours after 20-Hz rTMS stimulation applied to the DLPFC. Generally, this event is unlikely to occur in healthy subjects and has been rarely described in patients with different neurological or psychiatric disorders. 31 However, it is well known that subjects with history of traumatic or anoxic damage could have a potentially increased risk of seizure during or immediately after rTMS. 31 Other rTMS studies (Manganotti and coworkers, 34 Piccione and coworkers 35 ) did not report any side effect. Therefore, we cannot exclude that the stimulation dose of our rTMS could be safe for every UWS patients, especially using a multisessional approach; this issue needs to be better elucidated in larger post-anoxic individual samples.
Cortical Excitability and Connectivity
An important result of the present study was that, in comparison to HC, UWS patients showed an abnormal intracortical inhibition/facilitation balance within M1 and a lack of cortico-cortical connectivity, as previously reported.12,34-39 Interestingly, some UWS patients (nos. 1, 4, and 7) showed a partially preserved cortical excitability at baseline (as per SICI), in comparison to the other patients. Only these subjects showed real_rTMS after-effects.
Our findings further support the hypothesis that cortico-cortical disconnection and impaired cortical excitability may have a pivotal role in DOC pathophysiology, since patients with impaired premotor–motor and parietal–motor connectivity and cortical dysexcitability showed only “subcortical” behaviors, as per CRS-R assessment. On the other hand, such factors may represent the basis for consciousness recovery, since patients showing an at least minimal preservation of cortical excitability at baseline (SICI) improved their motor responsiveness after real_rTMS.
In addition, cortico-thalamic deafferentation may also have a critical role in DOC pathophysiology, since it has been hypothesized that a diffuse cortico-subcortical postanoxic injury may lead to either thalamic or cortico-thalamo-cortical loops damage, which in turn produces a long-term alteration of cortical excitability.34-40 Indeed, we can only indirectly hypothesize an impairment of cortico-thalamic projections, since we did not directly assess thalamo-cortical connectivity in our study.
Thus, our results may indicate that, in addition to the well-known structural postanoxic brain damage, functional disturbances of intracortical and cortico-cortical (as tested by TMS) may play a significant role in DOC pathophysiology.37-42
A limiting factor in our study may be represented by the younger age of HC in comparison to UWS patients, since age-related differences in cortical excitability have been reported.43 -45 Nevertheless, we found electrophysiological parameter differences between HC and UWS that are greater than those expected for age-related reasons. Another limiting concern may be the fact that experimenters knew what type of stimulation was delivered. Finally, a time series design (ie, an ABAB design with A being the TMS session, and B the sham one) could be useful to strengthen the validity of our findings.
rTMS Effects
The present study illustrates the residual capacity for neural plasticity and transient recovery of “minimal signs of consciousness” in some postanoxic UWS patients. Indeed, at the individual level, 3 patients were responsive to our stimulation protocol, with a transient improvement of CRS-R_M score from 2 to 3, associated with a modulation of cortical excitability and connectivity (as per MEP, SICI, and ICF modulation and premotor/parietal-motor connectivity restoration).
Interestingly, rTMS behavioral effects were limited to the CRS-R_M domain, as we did not detect any change in the remaining items. Thus, we could hypothesize that our rTMS protocol could have induced some specific, although transient, restoration of the connectivity of the UWS cortico-subcortical “residual islands” involved in the sensory-motor control. However, the electrophysiological effects of DLPFC rTMS over other brain areas need to be further investigated.
Noteworthy, at TPRE, responder UWS patients (nos. 1, 4, and 7) were not able to show any “cortical” behavior (according to CRS-R subitems), were borderline between MCS and UWS (according to CRS total and subitems scores), and had a partially preserved cortical excitability (SICI). These characteristics further raise the issue that cortical plasticity and connectivity are key points of consciousness recovery in severe DOC patients, as suggested by rTMS responsiveness. Indeed, we may argue that some “clinically defined” UWS patient could be able to show some “cortical” behavior, that is, to improve their level of consciousness, if their residual plasticity potentiality is properly triggered. In fact, patients with a partial preservation of connectivity and excitability at baseline improved their level of consciousness through plasticity mechanism,34-40 as well as those induced by rTMS protocol, even though short-lasting.
In addition, responder patients showed the N20 component bilaterally and were younger than other patients. Hence, age and bilateral N20, which are well-known prognostic factors for DOC outcome, 4 seem to have an important role in predicting rTMS response. Future studies with larger postanoxic sample size should be fostered in order to assess whether rTMS might induce after-effects also in patients lacking of the N20 component. On the other hand, disease duration, anoxic etiology, and gender seem not to be relevant.
To date, rTMS and tDCS neuromodulation studies have been performed over M1 and DLPFC, showing behavioral improvement only in MCS, but not in UWS patients32,34-36 (except for one UWS who had a MCS diagnosis at follow-up). 37 More in detail, only one DLPFC-rTMS study showed a trend to neurobehavioral improvement in a UWS patient following traumatic BI. 32 Hence, in comparison to previous studies implying tDCS or rTMS, we can speculate that the partial behavioral effectiveness of our rTMS protocol could depend on different DOC pathophysiology (ie, postanoxic vs posttraumatic BI), different cortical–subcortical mechanisms triggered by rTMS or tDCS, and different rTMS/tDCS stimulation site and duration. Nevertheless, to our knowledge, this is the first ever study extensively investigating the electrophysiological after-effects of a brain-stimulation protocol in a postanoxic UWS sample.32-35
rTMS in DOC: Putative Mechanism of Action
Since we found rTMS effects in only 3 of 10 UWS patients, we may argue a putative mechanism of action of rTMS in UWS patients, although the specific mechanisms are still unclear. 42
In vitro studies have demonstrated that oxygen and glucose deprivation exerts long-term effects on the synaptic efficacy via the induction of a postischemic long-term potentiation (i-LTP). 48 An interesting line of research postulates that the final effect of ischemia-induced neuroplasticity is to permit the reorganization of cortical circuits by which some individuals achieve functional recovery after BI. 49 In keeping with this hypothesis, we can speculate that rTMS can interact and boost up this i-LTP.
Another mechanism of recovery may be mediated by brain-derived neurotrophic factor (BDNF), whose production is upregulated after BI. 50 It is important to consider that BDNF is involved in mechanisms underlying LTP induction and maintenance by activating latent synapses and modulating cytoskeletal functions.51-53 Moreover, it is likely that BDNF expression after BI may enhance neurogenesis and promote sensory-motor recovery.54,55 Therefore, it is likely that rTMS may increase BDNF levels within DLPFC circuits promoting a restorative plasticity within the frontoparietal network. Traumatic BI animal models show a dramatic shift in excitatory/inhibitory dynamics, suggesting a long-term hyperexcitability of the cortical circuits, after an initial suppression that could be linked to the disruption of one or more inhibitory mechanisms of the thalamocortical circuit. 56 Following a BI, NMDA glutamate receptors are upregulated, whereas GABAA receptors are downregulated, in both the ipsilesional and contralesional hemisphere. 57
However, since we used only a single rTMS session, it is likely that our protocol could have modulated the cortical excitatory/inhibitory dynamics 58 (as suggested by the correlation among MEP amplitude, SICI/ICF strength, and premotor/parietal-motor connectivity), thus allowing to reach the threshold necessary to recruit some “dormant circuits” within the frontoparietal networks, in some UWS patients. Indeed, it has been suggested that GABAergic 40 and/or cholinergic 37 inhibitory tone reduction could facilitate the reorganization of the circuits impaired by damage, in UWS patients, allowing activity-dependent plastic changes.
In addition, a possible involvement of the subcortical pathways, including basal ganglia, thalamus, anterior cingulate cortex, and even ascending reticular activating system and cholinergic neurons of the basal forebrain, could be taken into account.
Conclusions and Implications
In conclusion, we demonstrated that even a single session of 10-Hz rTMS over the right DLPFC may transiently improve consciousness and partially restore the connectivity within several cortical areas in some UWS patients and, possibly, thalamocortical networks. Despite this being just a pilot study to show the feasibility and safety of a single session of high-frequency rTMS over DLPFC in UWS patients, these preliminary results may encourage future larger trials using a multisession approach.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
