Abstract
Background. Sensory stimulation is often provided to persons incurring severe traumatic brain injury (TBI), but therapeutic effects are unclear. Objective. This preliminary study investigated neurobehavioral and neurophysiological effects related to sensory stimulation on global neurobehavioral functioning, arousal, and awareness. Methods. A double-blind randomized placebo-controlled trial where 15 participants in states of disordered consciousness (DOC), an average of 70 days after TBI, were provided either the Familiar Auditory Sensory Training (FAST) or Placebo of silence. Global neurobehavioral functioning was measured with the Disorders of Consciousness Scale (DOCS). Arousal and awareness were measured with the Coma-Near-Coma (CNC) scale. Neurophysiological effect was measured using functional magnetic resonance imaging (fMRI). Results. FAST (n = 8) and Placebo (n = 7) groups each showed neurobehavioral improvement. Mean DOCS change (FAST = 13.5, SD = 8.2; Placebo = 18.9, SD = 15.6) was not different, but FAST patients had significantly (P = .049; 95% confidence interval [CI] = −1.51, −.005) more CNC gains (FAST = 1.01, SD = 0.60; Placebo = 0.25, SD = 0.70). Mixed-effects models confirm CNC findings (P = .002). Treatment effect, based on CNC, is large (d = 1.88, 95% CI = 0.77, 3.00). Number needed to treat is 2. FAST patients had more fMRI activation in language regions and whole brain (P values <.05) resembling healthy controls’ activation. Conclusions. For persons with DOC 29 to 170 days after TBI, FAST resulted in CNC gains and increased neural responsivity to vocal stimuli in language regions. Clinicians should consider providing the FAST to support patient engagement in neurorehabilitation.
Keywords
Introduction
For persons remaining in states of disordered consciousness (DOC) 1 after a traumatic brain injury (TBI), neurorehabilitation commonly includes sensory stimulation. 1 Evidence1-5 and theory6-8 suggest that sensory stimulation might support neurobehavioral recovery. Therapeutic effect, however, is not established because evidence is derived largely from noncontrolled trials due in part to the research challenges associated with this population.5,9-11 Neurobehavioral effect is important to determine, but neurophysiological evidence 12 is also important because it can support neurobehavioral findings for this noncommunicative population. Neurophysiological evidence can also advance knowledge regarding how sensory stimulation supports neurobehavioral recovery.13-16
We hypothesized that patients with DOC after TBI, who receive the Familiar Auditory Sensory Training (FAST) protocol, 8 compared to the patients who receive Placebo of Silence, would (a) have significantly better global neurobehavioral functioning and/or arousal/awareness and (b) demonstrate increased neural activation in response to familiar and nonfamiliar vocal and nonvocal auditory stimuli in cortical association areas and resemble neural activation of healthy controls. To address these hypotheses, we conducted a double-blind placebo-controlled randomized clinical trial (RCT).
Methods
Persons with DOC for at least 28 days and within 1 year of TBI were recruited for study participation. Patients were recruited from 2 acute rehabilitation programs and from a large urban community (Figure 1). After screening medical records, state of disordered consciousness was confirmed via a 24-hour observation using a previously published consciousness screening 17 that is aligned with current clinical consensus criteria distinguishing between the comatose, 18 vegetative (VS),19-21 and minimally conscious states (MCS), 22 as well as emergence from MCS. All eligible subjects were randomized, stratified by the 3 recruitment sites, to either the Experimental FAST group or the Placebo Silence group (see Supplement Section Ia & Ib for additional details regarding recruitment strategies, screening procedures, and eligibility criteria). The study was approved by each site’s institutional review board. An independent data and safety monitoring board convened quarterly.
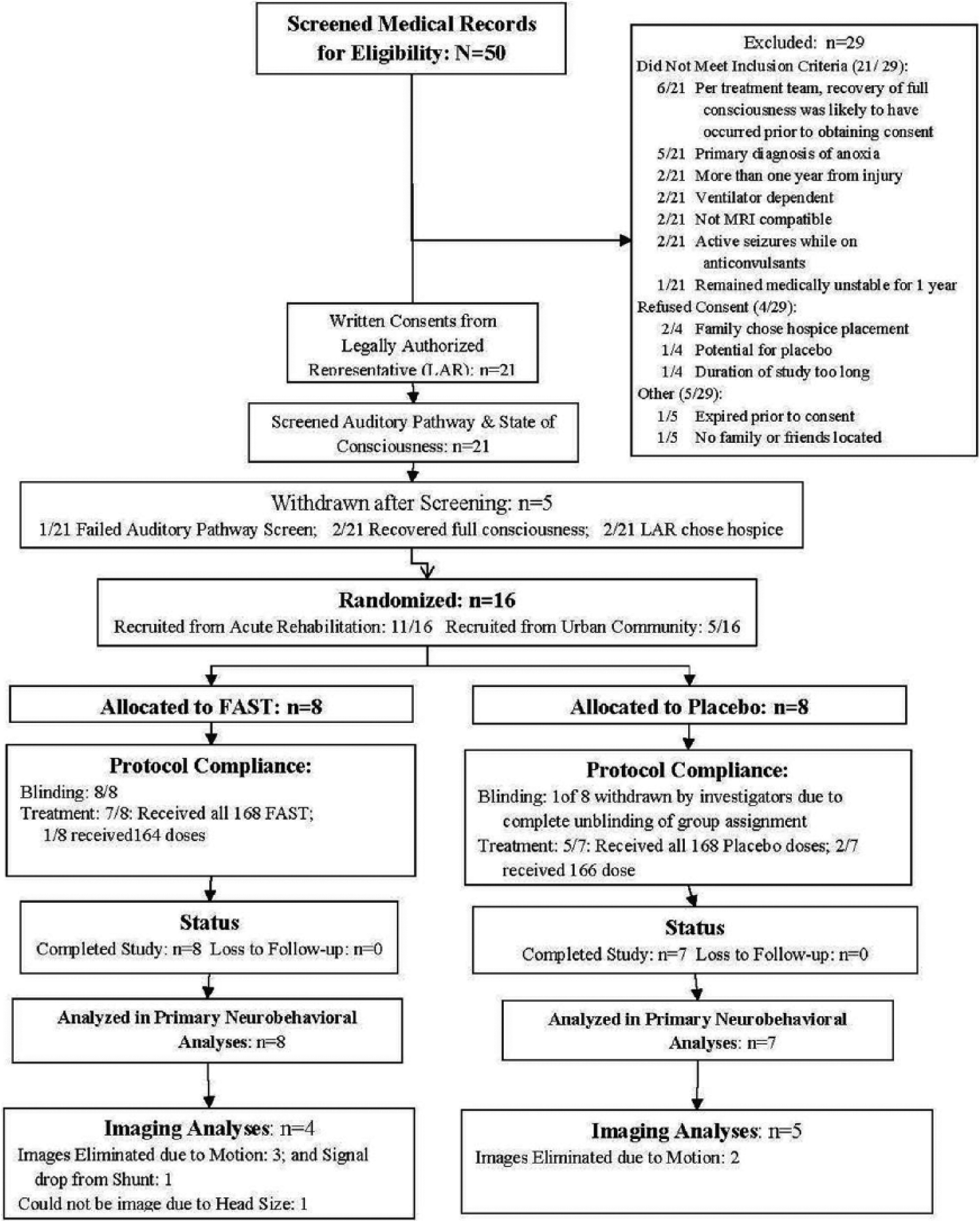
Consort diagram.
Study Procedures
For baseline and endpoint testing, patients were admitted to an acute research bed and were titrated off neuro-stimulants (modafinil, amantadine, methylphenidate, and bromocriptine). All testing was completed 24 hours after stopping these medications.
Researchers tracked informal stimulation, prescribed rehabilitation, and usual-care medications. Baseline doses of usual-care medications were maintained throughout study participation making doses during baseline and endpoint testing identical.
Treatment Protocols and Blinding
FAST is a standardized passive auditory stimulation protocol described elsewhere. 8 In brief, the patient is provided with customized recordings of stories told by people well known to the patient at least 1 year prior to injury. The stories represent specific events experienced by both the patient and the storyteller. The Placebo protocol is silence.
The FAST protocol and Placebo protocol were provided on compact discs (CDs), using portable players and noise-cancelling headphones, while patients were awake (ie, eyes open). Speakers were used for one patient not tolerating his headphones. The CDs were identical according to track duration, labeling, and administration procedures. (For additional details on study and nonstudy interventions, see Supplement Sections Ic & Id.)
To provide patients with the correct assigned intervention, one researcher remained unblinded. All other researchers and visitors were blinded to group assignment. Patients received FAST or Placebo protocols for 10 minutes 4 times per day, with at least 2 hours in between, for 6 weeks.
Neurobehavioral Outcomes and Analyses
The Disorders of Consciousness Scale (DOCS)23,24 and the Coma-Near-Coma (CNC) scale 25 were used to measure neurobehavioral effects and are described here briefly, but additional details are available in the supplement (Section Ie). The primary outcome, the DOCS, is a reliable, valid, and precise measure of global neurobehavioral functioning shown to remain stable over 6 weeks.23,24,26-28 The DOCS-25 starts with a systematic observation followed by administration of 25 sensory stimuli. Best responses to each stimulus are rated on a scale of 0 to 2, and total raw scores range from 0 (worst) to 50 (best).
The CNC scale measures arousal and awareness,25,29-31 and test stimuli are administered to elicit a specified behavior. Presence/absence of this behavior is scored as 0, 2, or 4. Total raw scores range from 0 (consistently responsive) to 36 (extreme coma).25,32
To optimize data quality, the DOCS and CNC were administered independently by different therapists. Given concerns of CNC reliability, 28 22% of CNC tests were randomly selected for readministration within 24 hours by different raters. We examined the paired CNC tests for interrater reliability by comparing mean total CNC raw scores between groups using paired sample t tests and correlations. To account for multiple raters, DOCS scores were transformed to interval-level measures using a many-faceted Rasch model anchored to values from a larger validation data set. 24 The CNC was analyzed using a Rasch partial credit model.33,34 After rescaling Rasch logit measures to 0 to 100, we compared DOCS and CNC baseline scores, as well as change scores (from baseline to endpoint), between groups using 2-sample t tests.
To confirm that t test results on CNC measures were not spurious findings, we conducted mixed-effects longitudinal linear models with random subject intercepts and slopes. 35 Groups were compared by testing the group by time interaction. 36 We used 8 CNC measures obtained during the first 4 weeks of the intervention because 2 patients (1 per group) recovered full consciousness after the eighth CNC. To inform clinical interpretation, we computed 2 effect sizes. We computed Cohen’s d 37 and the number of patients that need to be treated (NNT) for one patient to benefit. Cohen’s d values of .20, .50, and .80 are considered small, medium, and large effect sizes, respectively. 38 NNT values in the low single digits are considered ideal. 39
Functional MRI Testing Paradigm and the Neural Activation Outcome
Functional magnetic resonance imaging (fMRI) was used to examine the effect of the FAST protocol on the brain’s response to 8 different types of auditory stimuli, which is measured in terms of volume of activation to each stimulus (see Supplement, Section If, for additional details regarding fMRI methods). To examine the nature of identified neural responses, we used a fMRI auditory paradigm consisting of 8 individual stimuli with each stimulus presented for 30 seconds a total of 8 times each in random order. We examined volume of neural activation in response to these 8 stimuli individually and 8 combinations of these stimuli to control for and isolate stimuli of interest. To aid interpretation of the fMRI results, 7 healthy control subjects completed 2 fMRIs, 6 weeks apart, and were provided the same auditory paradigm.
The content of the fMRI auditory paradigm, described in detail elsewhere, 8 is different from the FAST protocol, which includes customized stories. For the fMRI auditory paradigm, the short story is derived from Scales of Cognitive Ability for Traumatic Brain Injury (SCATBI). 40 The SCATBI story is read by a familiar voice and a gender-matched nonfamiliar voice. The paradigm also includes a familiar voice and a gender matched nonfamiliar voice calling the patient’s name aloud. These 4 stimuli were chosen because they incorporate familiar voices, nonfamiliar voices, unfamiliar linguistic content (SCATBI story), and familiar linguistic content (patient’s name). The fMRI auditory paradigm also includes 30-second segments of silence and nonvocal sounds (bell, whistle, and clicking) to serve as control stimuli when determining the presence and nature of responses to the familiar and nonfamiliar vocal stimuli.
We chose to measure neural activation in response to the above-specified auditory stimuli because of well-established evidence indicating that (a) auditory information coupled with familiar auditory stimuli increases neural responsivity,13-15 (b) familiar and nonfamiliar auditory stimuli can elicit changes in neural responsivity for persons in states of DOC,15,41,42 and (c) that cortical processing of familiar information enhances subsequent processing of novel information (ie, brain priming).43,44 The selection of these stimuli, therefore, enabled an examination of the nature of the brain’s responses to the FAST protocol.
For this study, we made the neurophysiological assumption that positive neural recovery after severe TBI would be indicated if neural activation of severe TBI patients resembled neural activation of healthy persons. Making this assumption, we can use the healthy control data to interpret the fMRI findings. If the FAST protocol is related to increased neural activation in response to familiar voices, then we would expect increased activation within logical brain regions (eg, temporal poles), 45 but no significant neural activation changes in response to nonfamiliar voices, nonvocal sounds and silence. However, if the FAST protocol is related to increased neural activation in response to language, then we would expect increased neural activation for both the familiar and nonfamiliar voices reading the SCATBI story within a logical region (eg, Wernicke’s), but no activation changes would be expected in response to nonvocal sounds or silence. Finally, if the FAST protocol is related to increased neural activation in response to auditory stimulation in general, then we would expect increased activation for all of the tasks throughout the whole brain.
To test the neurophysiological effect of the FAST protocol, we measured change (from baseline to endpoint) in average neural activation in response to each of the 8 individual stimuli and the 8 combinations of the fMRI testing stimuli. Thus, the measure of change in neural activation in response to the specified auditory stimuli, when compared between treatment groups, reflects how the repeated FAST protocol influences the neurophysiology of the brain’s response to familiar and nonfamiliar vocal and nonvocal auditory stimuli. Furthermore, by using predefined regions of interest (ROIs), we can measure how the FAST protocol affects the location of the brain’s responses to these stimuli.
Functional MRI Analyses Between FAST and Placebo Groups
After standard fMRI preprocessing and image normalization, a first-level Generalized Linear Model using motion parameters as regressors was set up to generate t-maps for auditory stimuli. Mean neural activation in response to stimuli for each group was computed for the whole brain and 15 ROIs. To identify FAST effects on neural activation, we computed mean change between baseline and endpoint for each group for each stimulus or combination of stimuli for the whole brain and each ROI. We then used t-tests to compare mean activation between groups.
For data quality purposes, we sought to determine if nonstudy interventions (ie, rehabilitation, medications, and informal stimulation) influenced the fMRI signal between groups. Therefore, we examined mean neural activation in response to stimuli at baseline using t tests.
Functional MRI Analyses Within TBI Subjects Relative to Healthy Controls
We hypothesized that neural recovery after severe TBI would resemble neural responsivity of healthy persons. To test this, we conducted within TBI subject analyses to determine whether or not neural recovery, measured in terms of neural activation, resembles that of healthy controls.
To conduct within TBI subject analyses, we first defined meaningful functional activation change (FAC) according to the healthy control group using prediction intervals. 46 Next, we computed the average difference in neural activation between baseline and endpoint for the healthy group using all 8 individual auditory stimuli and the 8 combinations of the stimuli for whole brain and each ROI. Utilizing these average differences, we computed 95% prediction intervals (lower and upper), 46 which characterizes the natural variability of the healthy group. Next, we used the parameter estimates of the healthy group to construct a prediction interval for each TBI subject individually. Each TBI subject’s prediction interval incorporates parameter estimation variability for the healthy group and the TBI subject’s own variability. The prediction intervals were constructed utilizing (a) the healthy control group FAC scores, (b) the absolute values of same FAC scores, and (c) the cube root of the absolute value of the same FAC scores. While between-group analyses described in preceding section will maintain directionality, the use of the cube root transformation removes directionality for the within subject analyses but achieves the normal distribution.
We used the prediction intervals to determine whether the FAC of each TBI subject was significantly different from the mean FAC for the healthy group. If a TBI subject’s FAC fell within the mean prediction interval for the healthy group, then their FAC was not significantly (P > .05) different from the average healthy group’s FAC.
Results
Of the 50 patients screened, 21 were enrolled (Figure 1). Five were withdrawn prior to randomization and 16 were randomized to groups with 15 completing the study. Final fMRI samples included 4 FAST, 5 Placebo, and 7 healthy control participants. Six TBI participants were eliminated from imaging analyses due to excessive motion, signal dropout, or head size that prohibited scanning.
Group Comparability
The groups for the total study sample (n = 15) did not significantly differ according to average days required to provide the 168 doses (FAST mean = 44, SD = 4 days; Placebo mean = 46, SD = 10 days; P = .57), demographics (Table 1), prognostic factors including baseline clinical conditions and nonstudy interventions. Relative to the FAST group, the Placebo group did, however, have significantly more contusions,47,48 was enrolled into the study earlier after injury, 47 had a better injury severity score, and fewer subdural hematomas. The FAST group also received more anticonvulsants and antihypertensive medications27,49 (for additional findings, see Supplement Section IIa).
Demographic and Prognostic Factors and Nonstudy Treatments Received.
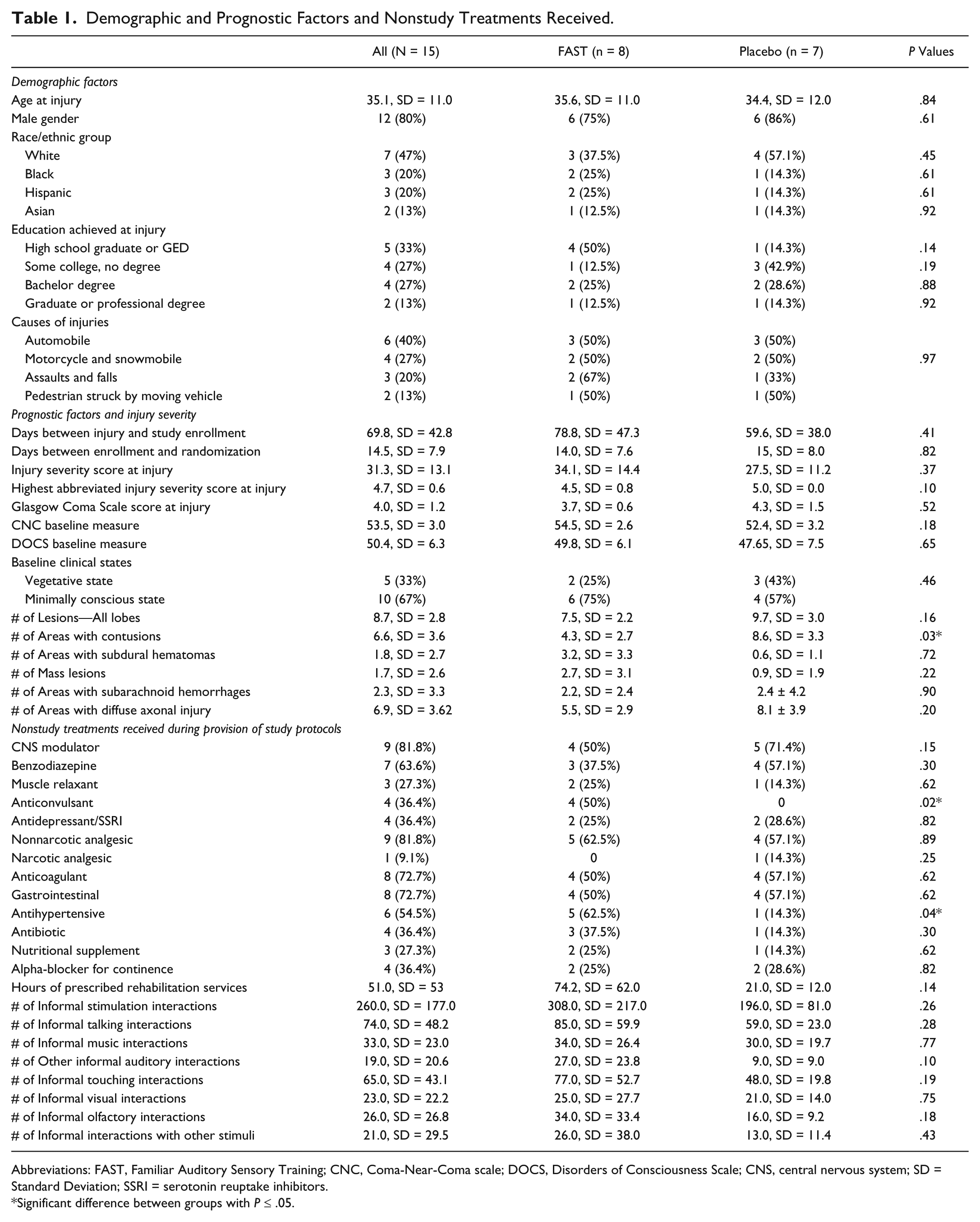
Abbreviations: FAST, Familiar Auditory Sensory Training; CNC, Coma-Near-Coma scale; DOCS, Disorders of Consciousness Scale; CNS, central nervous system; SD = Standard Deviation; SSRI = serotonin reuptake inhibitors.
Significant difference between groups with P ≤ .05.
The final fMRI groups (FAST n = 4; Placebo n = 5) do not significantly differ by demographics, baseline clinical states, causes of injury, time between injury and study enrollment or randomization, injury severity scores, number of lesions, or by nonstudy interventions. FAST and Placebo groups also indicate no differences at baseline according to neural activation in response to the 8 auditory stimuli and 8 combinations of the stimuli within the whole brain and 15 ROIs (All P values >.353) suggesting that nonstudy treatments do not influence fMRI signal (also see Tables S2 and S3 in Supplement Section IIb).
Clinical Findings
The FAST and Placebo groups had an equal number of patients in VS and MCS at baseline (Table 1, P = 0.46). At endpoint, fewer patients in the total sample remained in VS (13%, 2/15) and MCS (53%, 8/15), with 5 patients emerging from MCS (33%). The FAST and Placebo groups each had 1 patient emerge from VS (FAST: 1 of 2; Placebo: 1 of 3), but the FAST group had 4 patients emerge from MCS with only 1 Placebo patient emerging from MCS.
CNC Interrater Reliability
The mean total CNC scores (n = 40 pairs) for each group do not differ according to raters (FAST CNC means: Rater A mean = 16.4, SD = 9.7, Rater B mean = 15.4, SD = 8.6, P = .49, r = .85) (Placebo CNC means: Rater A mean = 19.2, SD = 5.2, Rater B mean = 19.4, SD = 9.7, P = .91, r = .61) (see Supplement Sections IIc and IId for additional data quality analyses).
Neurobehavioral Results
Average change in DOCS measures do not differ between groups (FAST mean = 13.5, SD = 8.2; Placebo mean = 18.9, SD = 15.6; P = .465; 95% confidence interval [CI] = −11.1, 21.9), but the FAST group had significantly greater average change in CNC measures (FAST mean = 1.0, SD = 0.6; Placebo mean = 0.25, SD = 0.7; P = .049; 95% CI = −1.51, −.005).
Findings from the mixed-effect longitudinal analyses (Table 2 and Figure 2) indicate (a) a significant difference with CNC slopes (P = .0022; FAST: Intercept = 53.1, Slope = −0.07; Placebo: Intercept = 53.4, Slope = −0.08), with the FAST group recovering more quickly than the Placebo group (mean slope difference −0.63 points per week), and (b) the largest effect occurring within the first week (ie, second CNC measure in Figure 2) of the intervention with small consistent gains thereafter (see Supplement Section IIe for additional neurobehavioral results).
CNC Means by Groups and Time.
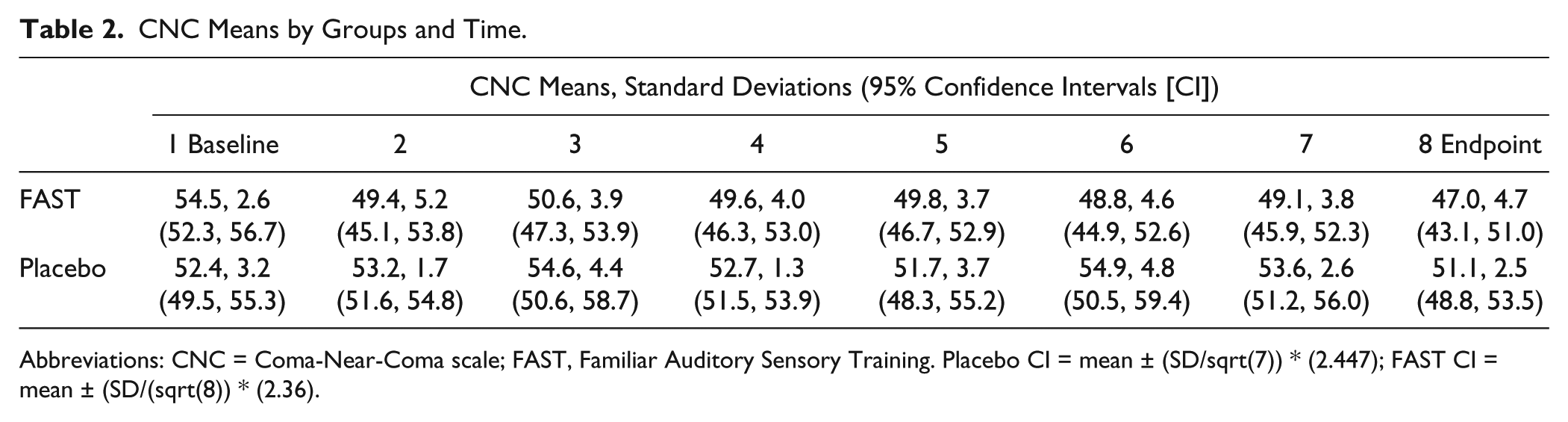
Abbreviations: CNC = Coma-Near-Coma scale; FAST, Familiar Auditory Sensory Training. Placebo CI = mean ± (SD/sqrt(7)) * (2.447); FAST CI = mean ± (SD/(sqrt(8)) * (2.36).
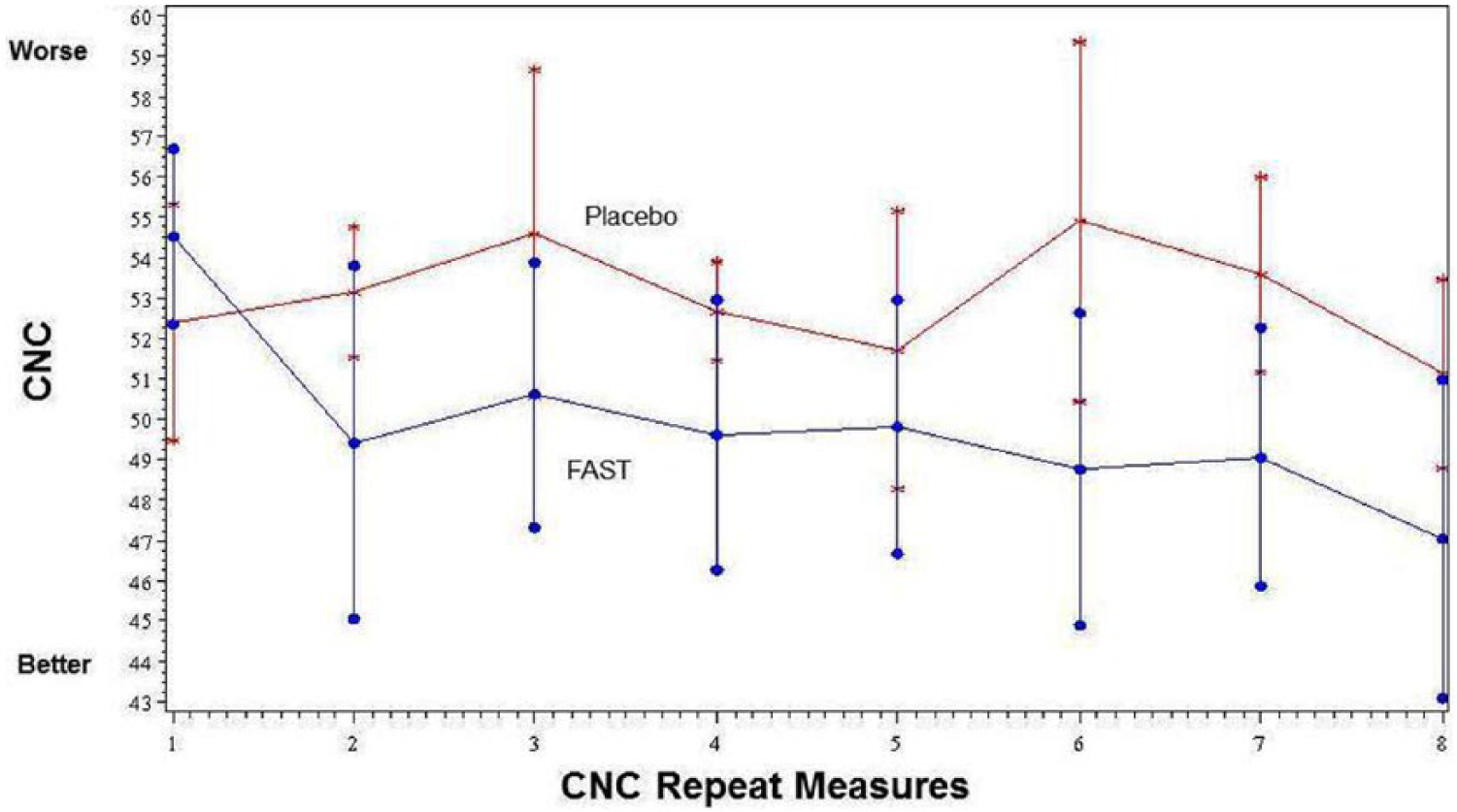
Mean Coma-Near-Coma Scale (CNC) Rasch measures 2 times per week during the provision of study intervention according to study groups.
The hypothesized treatment effect based on DOCS measures was 0.91. 37 We did not detect this hypothesized effect. At endpoint, the 2 groups differed on average by 3.25 DOCS units. Although we recruited only 50% of the planned sample size, which reduced power to 0.51, the observed effect size (d = 0.24; CI = −.52, .99) based on DOCS measures is much smaller than the hypothesized treatment effect (d = .91) indicating that we did not miss a significant treatment effect due to low power (also see Supplement Section IIf). Based on CNC measures, the groups differed at Endpoint, by an average of 4.1 CNC units. The effect size (d = 1.88, 95% CI = 0.77, 3.00), based on CNC measures, is considered large and indicates that 96% of the FAST group will have CNC measures above the mean of the Placebo group; only 37% of the 2 groups will have overlapping measures. Conservatively, the lower CI limit for the effect size based on CNC measures (0.77) is also considered a large effect size. In the absence of an established minimally clinically important difference for the CNC, interpretation of this effect size is challenging. 50 To compute NNT, we assumed that a change of 2 CNC units is important because our CNC Pooled SD is 6, and previous work suggests that .3 SD is important. 50 Two of the 7 placebo patients (0.29) achieved a change of more than 2 CNC units, while 7 of the 8 FAST patients (0.88) made a CNC change of more than 2 units. The NNT then is 1.7 (rounded to 2), 39 which indicates that you would need to treat 2 persons with the FAST for 4 weeks, rather than placebo, in order to find 1 additional patient who would improve more than 2 CNC units.
Functional MRI Results Between Groups
Findings from examinations of neural activation between groups indicate that the FAST group had significant mean activation changes (Figure 3) in response to the following:
Right Heschl’s region for all auditory stimuli combined (P = .039; t = −2.885df; CI = −72.4, −2.99) (µFAST base = 0, SD = 0; µFAST end = 19.3, SD = 27.8; µPlacebo base = 23.8, SD = 28.3; µPlacebo end = 29.8, SD = 36.7)
Right Heschl’s region for a nonfamiliar voice reading a short story (P = .033; t = −3.124df; CI = −50.7, −3.5) (µFAST base = 2.5, SD = 3.5; µFAST end = 13.3, SD = 16.7; µPlacebo base = 17.0, SD = 18.9; µPlacebo end = 22.8, SD = 28.8)
Right Wernicke’s for a nonfamiliar voice reading the short story after accounting for the familiar voice reading the same short story (P = .034; t = 5.2922df; CI = 0.87, 8.5) (µFAST base = 0.0, SD = 0.0; µFAST end = 4.7, SD = 1.5; µPlacebo base = 0.0, SD = 0.0; µPlacebo end = 0.0, SD = 0.0)
Whole brain in response to familiar voice calling subject’s name aloud (P = .040; t = 2.517df; CI = 61.7, 2071.7) (µFAST base = 644.8, SD = 44.4; µFAST end = 1338.3, SD = 698.3; µPlacebo base = 579.8, SD = 640.9; µPlacebo end = 206.6, SD = 172.3)
Left Heschl’s region for bell ringing (P = .028; t = −3.0695df; CI = −117.6, −10.4) (µFAST base = 46.0, SD = 31.1; µFAST end = 0.0, SD = 0.0; µPlacebo base = 13.0, SD = 16.8; µPlacebo end = 23.3, SD = 26.9)
Left prefrontal region for bell ringing (P = .048; t = −2.3967df; CI = −15.4, −0.1) (µFAST base = 5.3, SD = 6.4; µFAST end = 0.5, SD = 1.0; µPlacebo base = 0.2, SD = 0.5; µPlacebo end = 3.2, SD = 4.4)
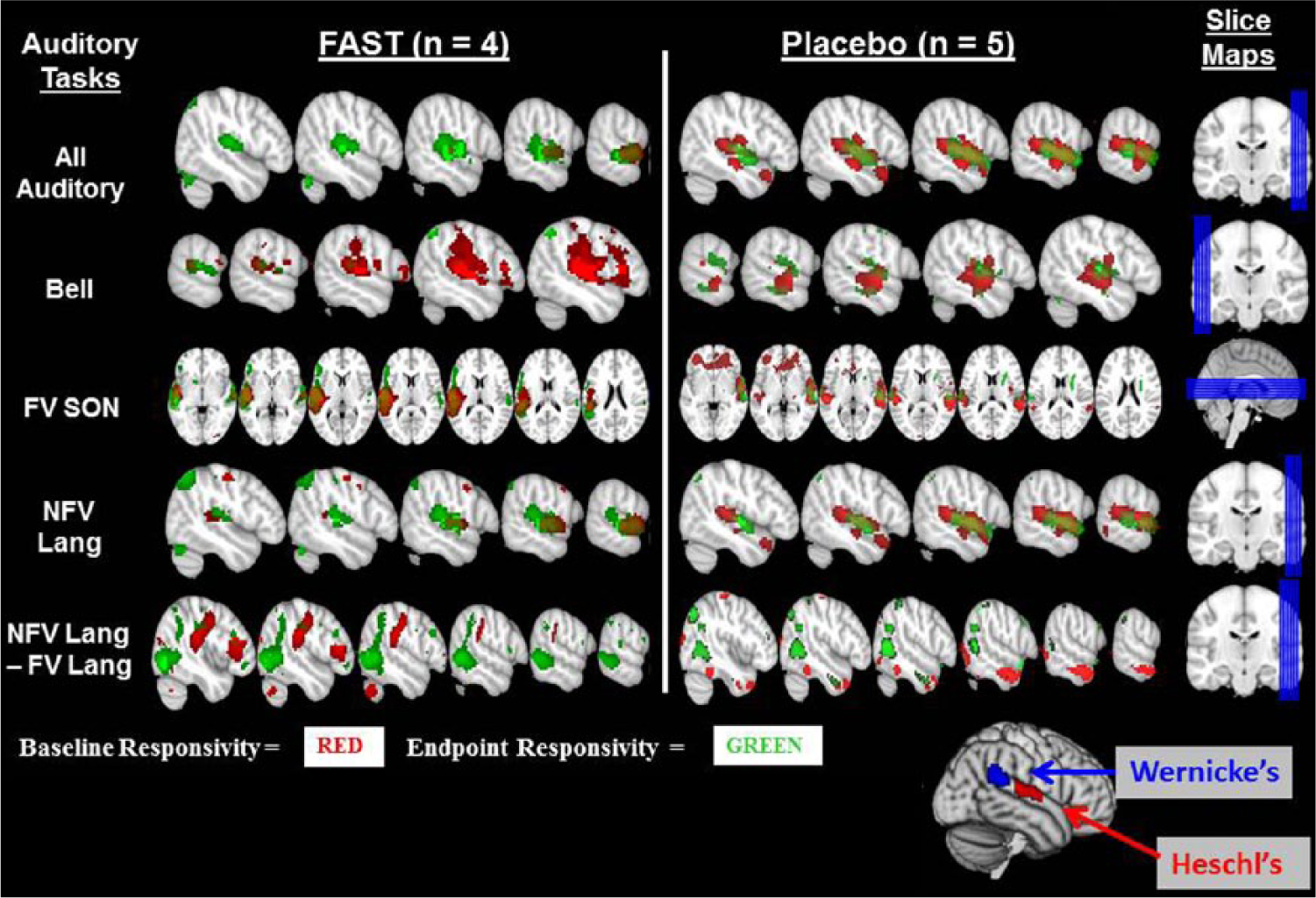
Responsivity to fMRI auditory testing paradigm.
Functional MRI Results Within TBI Subjects Relative to Healthy Controls
The within-subject analyses indicate that the majority of TBI patients in both groups (75% to 100%) had FACs that significantly differed from the average healthy group’s FACs. The Placebo group (6% to 16%) had twice the number of FACs that significantly differed from healthy controls compared to the FAST group (0% to 7%) and about twice the number of ROIs (17% to 44%) that significantly differed from healthy control ROI FACs (for elaboration of findings see Supplement Section IIg).
Discussion
For persons with DOC after TBI, the FAST protocol is significantly related to more CNC gains. The observed effect size for CNC measures indicates that you would need to treat 2 persons with the FAST for 4 weeks, rather than placebo, in order to find 1 additional patient who would improve more than 2 CNC units. This finding is supported by CNC mixed effects analyses, descriptive clinical findings, and changes in neural activation in response to specified auditory stimuli. Findings from nonrandomized and noncontrolled studies suggest that sensory stimulation might enhance arousal to and awareness of vocal, 51 acoustic, 11 and familiar stimuli and furthermore that arousal and awareness might be enhanced when stimulation is provided in short and frequent increments. 52 Our findings, derived from a placebo controlled RCT, demonstrate that sensory stimulation using familiar vocal and linguistic stimuli provided in 10-minute increments 4 times per day is related to improved arousal and awareness.
We found that the observed treatment effect, based on CNC measures, is significantly different between groups, and when based on the DOCS-25 measures it is not. Absence of significant differences in DOCS measures (both over time and between groups) may reflect that DOCS measures are based on best elicited responses whereas CNC scores reflect consistency of responses. Consistency translates to more arousal and awareness manifested as more ability to follow auditory commands, visually track, localize to sounds, vocalize, and respond to pain and touch.
We tested the hypothesis that study participants demonstrating neurobehavioral gains will also demonstrate neural activation changes that resemble the neural activation of healthy persons. Our finding that the majority of FAST patients had fewer FACs differing significantly from healthy persons suggests that the FAST protocol supports adaptive neurobehavioral recovery. Evidence is emerging to indicate that this patient population, when provided with common treatments such as neuropharmacologic therapies, 53 do make previously undetected neurobehavioral gains. The translation of these statistically significant gains to clinical impact is challenging, in part, because the minimally clinically important differences for the neurobehavioral tests are not yet available. 28 It is also challenging because the mechanisms underlying these neurobehavioral gains have yet to be determined. The observed FAST effect, as measured by the CNC (d = 1.88), and the evidence of adaptive neural activation, together suggest that gains in arousal and awareness may be important for supporting adaptive recovery. While this study suggests that the FAST protocol may be important to support adaptive recovery, we do not know what the patients in VS and MCS are doing with the sensory input. This raises the question of whether or not it is possible to have increased neural activation in logical brain regions given specified stimuli and not engage key neural networks? That is, do treatments related to improved arousal and awareness such as the FAST enable recovery of awareness by “exercising” key networks (eg, language, attention, default mode) or by engaging these networks via exercising of residual neural integrity within key networks?
The finding that the majority of FAST patients had fewer FACs significantly differing from healthy persons suggests that the FAST protocol supports adaptive neurobehavioral recovery and that the neurosciences literature regarding the responsiveness of the healthy brain can be leveraged to advance our understanding of the functional neural activation findings related to the FAST protocol. The reported fMRI findings regarding neural activation in response to auditory stimuli salient to this study, interpreted within the context of the neurosciences literature provides insights into the mechanisms of plasticity that could be contributing to the improved arousal and awareness. These inferences are made in the next paragraphs to identify avenues for future research.
The FAST group had significantly increased whole brain activation in response to a familiar voice calling a patient’s name aloud, which is consistent with existing evidence that calling a healthy person’s name aloud captures that person’s attention and primes their brain to attend to incoming stimuli.13-16 Together, this evidence suggests that the FAST protocol supported recovery by priming the brain to receive information that subsequently enhanced covert awareness of incoming information.
The FAST group had significantly increased activation within Heschl’s and Wernicke’s regions in response to a nonfamiliar voice reading the SCATBI story aloud. Since the only difference between the FAST and Placebo groups is that the FAST group was provided with familiar stories told by familiar voices, this finding is consistent with evidence (from healthy persons and persons in states of DOC) that familiar voices enhance likelihood of eliciting brain responses to novel auditory information included in the fMRI auditory paradigm.13-16,43,44 This suggests that improved arousal and awareness could be due to repetitive exposure to familiar voices or familiar stories. These findings also correspond with the plasticity principles of saliency and specificity specifying that the nature of the neural changes (increased activation within language regions) is dictated by the nature of the training experience, 54 which is repetitive exposure to familiar voices and familiar stories. The theoretical basis for the FAST is based, in part, on applying the principles of saliency and specificity. 8
The FAST group had reduced activation in response to a nonvocal sound (bell) within Prefrontal and Heschl’s regions. This reduced response is likely due to habituation to a nonfamiliar sound that lacks saliency to the patient.
Our findings regarding changes in neural activation together with evidence from the literature suggests that repetitive exposure to familiar voices and familiar stories contributed to improved arousal and awareness, in part, because of a priming effect and because of the saliency and specificity of the FAST. The effect of brain priming on subsequent processing of stimuli needs more direct examination with this patient population. Future research could, for example, measure the patient’s activation in response to their name being called aloud immediately followed by nonvocal versus vocal stimuli. Future research is also needed to directly inform us about the injured brain’s response to less salient and less specific stimuli. Research comparing neural activation in response to familiar stories versus nonfamiliar stories read aloud by familiar versus nonfamiliar voices would, for example, inform us about how salient and how specific sensory stimuli needs to be to yield a therapeutic effect. Studies examining responses to linguistic versus nonlinguistic acoustic stimuli would also inform us about the acoustic parameter(s) driving the brain’s responses. Additional avenues for future research also include examinations of different doses, maintenance of effects, and effects when the FAST is coupled with other treatments and interventions. Given restrictive eligibility criteria, enrollment was lower than planned and a study limitation is achieved sample size. Future research on a larger sample is needed and should also represent subacute and chronic recovery stages to determine FAST effects by time postinjury.
Conclusions
We conclude that persons remaining in states of DOC for 29 to 170 days after TBI and receiving FAST for 4 weeks have more clinically meaningful CNC gains compared to a group not receiving any structured sensory stimulation. Our results suggest that improved arousal and awareness may be due to the FAST protocol priming the brain to be more responsive to salient stimuli. Clinicians should consider providing the FAST protocol to support patient engagement in neurorehabilitation. Findings indicate that clinicians should consider FAST as a neurorehabilitation intervention though additional research is needed to confirm findings.
Footnotes
Acknowledgements
The authors thank Ms Bessie Weiss, the nurses, and staff within Northwestern University’s clinical research unit who demonstrated a dedication to excellence that makes clinical trial research possible. We also recognize Dr Elliott Roth, Northwestern University Feinberg School of Medicine, who supported our research efforts.
Authors’ Note
Study Statisticians: Dulal Bhaumik, Weihan Zhao, and Domenic Reda. Study Psychometrician: Trudy Mallinson.
Clinical Trial Registry: NCT00557076; The Efficacy of Familiar Voice Stimulation During Coma Recovery; http://www.clinicaltrials.gov/ct2/show/NCT00557076?term=NCT00557076&rank=1.
The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the US Department of Veterans Affairs or the US government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Veterans Affairs, Office of Research and Development, Rehabilitation Research and Development Merit Grant # B4591R and career development transition award #B4949N. Northwestern University’s Clinical and Translation Sciences Institute, which is supported by the National Center for Research Resources, National Institutes of Health (Grant UL1RR025741). The Nick Kot Charity for traumatic brain injury (![]() ).
).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
