Abstract
Background. Stretching is a widely accepted standard-of-care therapy following spinal cord injury (SCI) that has not been systematically studied in animal models. Objective. To investigate the influence of a daily stretch-based physical therapy program on locomotor recovery in adult rats with moderate T9 contusive SCI. Methods. A randomized treatment and control study of stretching in an animal model of acute SCI. Moderate SCIs were delivered with the NYU Impactor. Daily stretching (30 min/day, 5 days/wk for 8 weeks) was provided by a team of animal handlers. Hindlimb function was assessed using the BBB Open Field Locomotor Scale and kinematically. Passive range-of-motion for each joint was determined weekly using a goniometer. Results. Declines in hindlimb function during overground stepping were observed for the first 4 weeks for stretched animals. BBB scores improved weeks 5 to 10 but remained below the control group. Stretched animals had significant deficits in knee passive range of motion starting at week 4 and for the duration of the study. Kinematic assessment showed decreased joint excursion during stepping that partially recovered beginning at week 5. Conclusion. Stretch-based therapy significantly impaired functional recovery in adult rats with a moderate contusive SCI at T10. The negative impact on function was greatest acutely but persisted even after the stretching ceased at 8 weeks postinjury.
Introduction
Widely accepted standard-of-care manual therapies for spinal cord injury (SCI) patients have developed over time with the goal of preventing further sensorimotor impairment, promoting functional recovery acutely and improving quality of life for chronic patients. 1 Guidelines for manual therapies cite promising preclinical data from animal studies, case studies, anecdotal evidence, and historical references as the rationale for prescribing these treatments. 2 Significant health care resources, in the form of manually applied stretching and range-of-motion (ROM) 3 treatments, are allocated in an effort to maintain joint mobility acutely after SCI, or to manage contractures for the chronically injured. However, as demonstrated by Harvey and colleagues, when a commonly employed 30 min/day regimen of stretch therapy in patients with chronic injuries is subjected to the standards of a randomized controlled trial, the results show no benefit for SCI patients or are inconclusive.2,4,5 Furthermore, the timing, dosage, or intensity of stretch needed to maintain or increase joint mobility is unclear.2,6,7
Loss of soft-tissue extensibility has been the focus of efforts to maintain function and to allow for optimal neurological plasticity. As outlined in Ben and Harvey, 8 numerous animal studies have shown a positive effect of stretch on muscle remodeling and extensibility, sarcomere length, collagen arrangement, and increased force production.9,10 However, these findings may not reflect any practical effects on patients because the majority of human stretch studies involve extensibility outcomes that are measured immediately on completion of the stretch, and it is likely that an initial positive effect of the stretch is not sustained beyond the short term. Contracture management for those with chronic SCI will require strategies that maintain extensibility over the long term.2,11
As a whole, laboratory animal care for experimental SCI studies does not include traditional stretch or ROM interventions; however, the practice simply may not be reported. Case studies have reported on the use of physical interventions to increase mobility after canine SCI; stretching in the form of traction, 12 passive ROM, isometric stretch, and locomotor training 13 have been used. Any improvements reported in these studies were not attributed solely to the physical interventions employed. Hypothesis-driven studies on the effect of stretch and/or ROM interventions, both with and without concomitant activity-based rehabilitation, on functional locomotor recovery after SCI in animal models are needed.
In a recent study focused on wheelchair immobilization after acute SCI, we found that hindlimb stretching negatively influenced the normal course of recovery in spinal cord injured but not immobilized control animals following mild thoracic contusions. 14 Animals that received the stretch protocol were unable to achieve consistent forelimb–hindlimb coordination (Basso, Beattie, and Bresnahan Open Field Locomotor Scale [BBB] scores ≤ 14), a significant deficit when compared to a non- stretch group of control animals (BBB score ≤ 18). Furthermore, the stretch protocol had no influence on the prevalence of ankle contractures in animals that were wheelchair immobilized overnight during the same 8-week period. These data were surprising to us, so the current study was designed to assess the effect of stretching on the recovery of coordinated stepping in animals with a moderate thoracic contusive injury. We hypothesize that a hindlimb stretch therapy, applied acutely after contusion injury, will disrupt central pattern generation and delay or reduce functional locomotor recovery. Our data suggest that locomotor recovery is significantly altered by only 30 minutes of daily hindlimb stretch in rats with lower thoracic moderate contusion SCI.
Materials and Methods
Spinal Cord Injury and Experimental Design
Nineteen young adult female Sprague-Dawley rats (190-215 g) were used in this study. All the procedures were approved by the University of Louisville Institutional Animal Care and Use Committee. Animals were randomized into 2 experimental groups: Acute Stretch (SR), n = 7, or controls, n = 12. Each animal was anesthetized with pentobarbital (55 mg/kg IP) and given prophylactic antibiotics (Gentamicin sulfate 15 mg/kg SC) prior to surgery. A single-level laminectomy was performed at the T9 vertebrae before receiving a moderate contusion injury (12.5 g cm) performed at the T10 spinal cord level using the NYU Impactor (W. Young, Rutgers University, NJ) as previously described. 15 All animals received Buprenorphine (0.1 mg/kg, SC) twice a day for 3 days. After recovery, all animals were housed socially, 2 per cage, for the duration of the study. All animals were housed in the same room with a 12-hour light/dark cycle and received daily postoperative care, including manual bladder expression as needed. Beginning at postinjury day 4 (Monday), the Acute SR group received a hindlimb stretch protocol administered each weekday morning (Monday to Friday), for 8 weeks. Assessments and daily care were continued for another 5 weeks. At 10 weeks, a subset of the control animals (chronic stretch group, n = 6) were chosen to undergo the daily stretch protocol for 6 days, Monday through Friday and the following Monday. All control animals, including the chronic stretch group (n = 12), were handled daily throughout the study.
Daily Stretch Protocol
Animals were wrapped in terry cloth hand-towels so that their hindlimbs were exposed. The animals were held securely, in a supine position, and it was common to observe them resting calmly or grooming during stretching. Stretch protocols were administered in a group setting. A moderator kept time and monitored finger placement to ensure that each stretch maneuver isolated a single muscle group. The handler to rat pairings were randomized throughout the study. The stretch protocol lasted ~30 minutes: 1 full minute of static stretch-and-hold of each major hindlimb muscle group in the order shown in Figure 1A-F bilaterally, for 2 sets. Thus, each major muscle group received 2 full minutes of static stretch-and-hold daily.
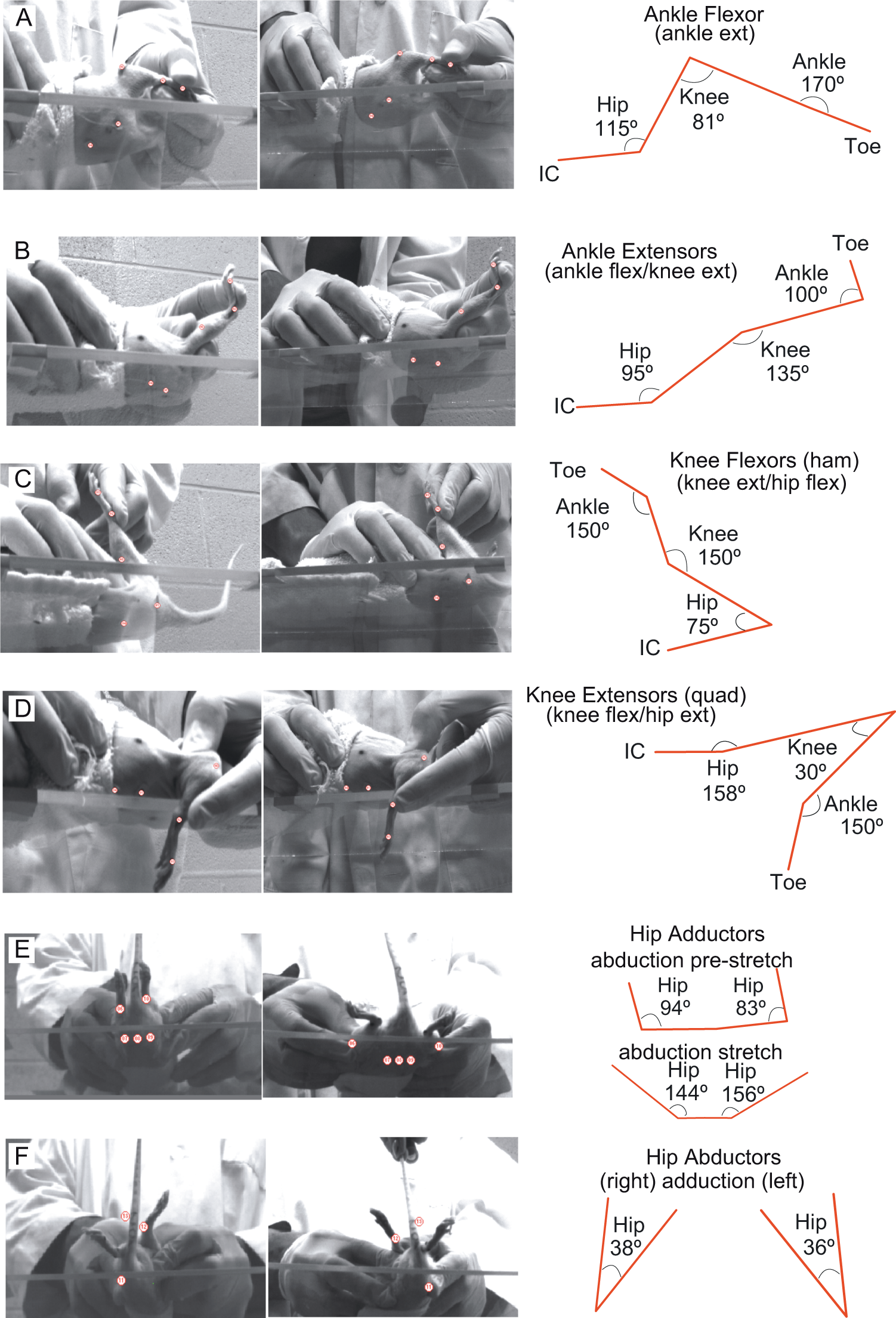
Kinematic representation of hindlimb stretch positions.
The intensity or force applied during each stretch was monitored carefully by observing the joint angles achieved. There was a soft end-feel for all stretch positions except ankle extension, which had a firm capsular end-feel 1 as assessed by coauthor Darryn Atkinson, PT. All animals had some degree of reflexive and/or volitional response to stretch, which we recorded as the “Immediate Stretch Responses” (ISRs). The ISRs occurred during the static hold and/or immediately on release of the hold. Observed ISRs were recorded for each stretch position during every stretching session for each animal (Supplementary Figures 3 & 4 and Table 2).
Behavioral Assessments
Behavioral assessments were performed by individuals blinded to the experimental groups. Overground stepping was assessed using the BBB Open Field Locomotor Scale as previously described. 16 BBB testing began at 4 days postinjury, twice each Monday, am (before the stretch protocol) and pm (3 hours after the stretch protocol) and on Friday afternoons (after 5 consecutive days of daily stretch) for 8 weeks. Monday pm BBB testing sessions were not done on weeks 9 to 13. Overground stepping was also assessed kinematically at weeks 3 and 13 (Supplementary Figure 1). Swimming was assessed using the Louisville Swim Scale (LSS) at week 2 and every other week thereafter. Finally, passive ROM of the hip, knee, and ankle were assessed preinjury and every other week postinjury (Supplementary Table 1).
Ventral view videos recorded during shallow water walking (SWW) were processed to calculate 3 different indices of coordination: the central pattern index (CPI), the regularity index (RI), and the plantar stepping index (PSI). 17 CPI was calculated as the number of correctly patterned step cycles (dorsal and plantar) divided by the total number of cycles (dorsal and plantar regardless of ability to achieve plantar paw placement. The RI, originally described by Hamers et al 18 and Koopmans et al, 19 was calculated as the number of correctly patterned planter step cycles/total cycles (dorsal and plantar) and gives an indication of interlimb coordination of plantar only stepping. The PSI is calculated as the number of hindlimb plantar steps divided by forelimb plantar steps and indicates how consistently the animal is able to achieve plantar hindlimb stepping.
Magnetically Evoked Potentials From Tail Stimulation
Magnetically evoked muscle responses (MER) were assessed at 3.5, 6.5, and 9.5 weeks. Each animal was positioned and secured on a piece of wood using a cloth stockinette as previously described. 15 A figure eight magnetic transducer coil placed at the base of the tail was used to directly stimulate afferent nerves resulting in electromyographic responses (EMG, motor output) in hindlimb muscles. Three stimulus amplitudes were used (60%, 70%, and 80% of maximal output) as previously described. 20 The transducer coil produces a 4.6 T magnetic field with a ~1 cm deep by ~1 cm wide peak magnetic field. The transducer coil was positioned with care to precisely angle the coil relative to the tail to avoid direct stimulation of the hindlimb muscles and/or the spinal cord. EMGs were recorded bilaterally from the lateral gastrocnemius muscles using 26-G needle electrodes connected to AI 405 head stages and a CyberAmp 380 (Axon Instruments). Responses were analyzed for onset latency, peak-to-peak amplitude, and recovery to baseline.
Histological Processing
Animals were euthanized with excess anesthetic (Pentobarbital, 110 mg/mL IP) at 13 weeks postinjury. The lateral and medial gastrocnemius and tibialis anterior muscles were blunt dissected, weighed, and flash frozen. Animals were then perfused transcardially with 4% paraformaldehyde and calcium-free tyrodes. Spinal cords were harvested and prepared for analysis of spared white matter as previously described. 21 Briefly, adjacent slide sets containing every fifth section were stained with eriochrome cyanin and cresyl violet, thus allowing the total area of white matter, healthy gray matter, unhealthy gray matter, cavity, and total area to be identified in micrographs and traced using a Wacom Intuos (Vancouver, WA) drawing tablet. Traced images were opened in ImageJ (NIH) for the area calculation of each tissue region.
Statistical Analysis
All data are presented as group means with standard deviations (±SD). Outcome measures were analyzed using repeated-measures analysis of variance (ANOVA) with groups as a factor, and were followed by Tukey’s HSD and Bonferroni post hoc t tests where appropriate. Group differences were considered statistically significant for P values of at least ≤ .05. The binomial proportions test was used were appropriate, and no correction to the proportions was required. The nonparametric Wilcoxon signed-rank test was used in the case of ROM measurements in which standard deviations were ±0. Pearson correlations of at least P ≤ .05 were used to determine the relationships of the ISRs and BBB Scores.
Results
Behavioral Assessments: Stepping
All BBB scores are shown in Figure 2A and Monday/Friday pm only scores are shown in Figure 2B. Beginning on the second Monday of the experiment (week 2), we observed an acute loss of locomotor function following a single stretching session and hindlimb function improved from Friday afternoons to Monday mornings, when no stretching was administered (Figure 2A). Thus, the BBB scores present as a “saw-tooth” pattern with substantial variability because animals were not equally vulnerable to locomotor loss after a single stretching session on Monday mornings. We plotted the drop in BBB scores for every Monday on a per handler basis and could find no handler–response relationship (data not shown). Overall, the BBB scores increased gradually from week 5 to week 9, while still exhibiting transient drops after each Monday morning stretching session (Figure 2A and B). Representative 3-dimensional kinematic analysis of overground stepping is available in Supplementary Figure 1.
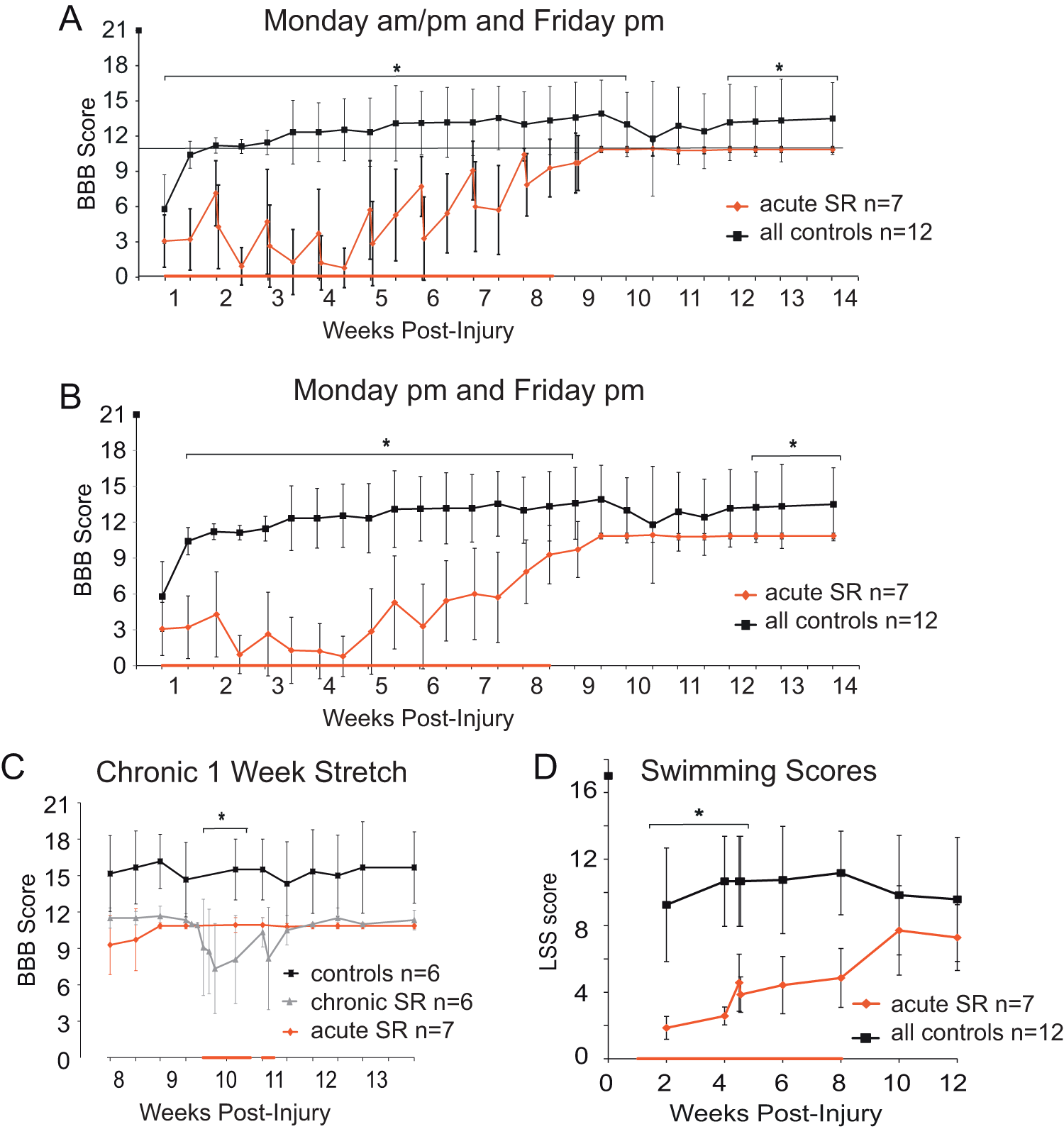
BBB and LSS scores daily stretch.
At 10 weeks we chose a subset of the original control animals to stretch for 6 days (stretch group, n = 6, gray trace, Figure 2C). We separated the data from these animals beginning at week 8 to show that the chronic stretch animals had lower BBB scores than the remaining control animals (black trace, n = 6). Control animals with BBBs > 16 would not tolerate stretching and were excluded from the chronic stretch group (Figure 2C). We performed daily BBB assessments on the chronic SR animals (bold red bar on the x-axis) and compared daily BBB scores within the chronic SR group rather than among groups. Similar to the acutely stretched animals, the chronic SR group had a “saw tooth” BBB profile of loss of function. Importantly, after only 2 days of hindlimb stretch these animals were no longer capable of weight support, even at stance (BBB < 9). As anticipated, recovery of BBB scores occurred after the chronic daily stretch ended (Figure 2C).
Behavioral Assessments: Swimming, ROM, ISRs
In the current study, swimming was used as an assessment of locomotion that does not require the ability to support body weight. Swimming was assessed using the LSS as previously described 22 beginning at week 2 and every other week thereafter. Animals in the current study scored within the 0 to 5 range indicating poor swimming (little or no hindlimb movement, high dependence on their forelimbs for forward motion, severe trunk instability and/or body angle), or in the intermediate swimming range, 6 to 11, (retained dependency on forelimbs for forward movement and with varying degrees of hindlimb movement, trunk instability, and/or body angle).
Daily SR negatively influenced swimming ability over weeks 1 to 4, the same period that showed the greatest loss of overground function (Figure 2D). The SR group remained dependent on their forelimbs for forward movement with only occasional hindlimb kicking, severe trunk instability, and poor body angle as hallmarks of their swimming during weeks 1 to 4 (LSS ~ 3). Similar to the BBB profile, LSS scores increased in weeks 4 to 8 and reached a plateau for weeks 10 to 12. We tested the animals for swimming ability on Monday (am and pm) at week 4.5 to determine if a transient loss of function occurs for swimming as in overground locomotion. At that time only a single point drop in swimming ability was evident (Figure 2D). As a group, the non-SR animals were intermediate swimmers with more hindlimb kicking, greater trunk stability, and better body angle (LSS ~ 11) throughout the study. Overall, the LSS scores reveal that the SR protocol appears to negatively influence the recovery of swimming in a manner similar to that of overground stepping.
Passive range-of-motion (pROM) assessments and ISRs also support the overall loss and gain profile of locomotor recovery for SR animals shown in Figure 2B and C. This change in recovery over the 4 to 6 week range, postinjury, occurred even though the intervention of daily stretch was still ongoing (Supplementary Figures 2-4 and Tables 1 and 2).
Interlimb Coordination and Plantar Stepping
Gait analysis in shallow water allows for assessment of plantar stepping and footfall pattern when partial weight support is provided by buoyancy. 17 Group data from postinjury weeks 3 (during stretch) and 13 (5 weeks poststretch) were compared for CPI, PSI, and RI. Significant group differences are evident for all 3 indices at week 3; SR animals were not capable of stepping and had minimal movement of hindlimb joints with no weight support (Figure 3A-C). By 13 weeks the ability to produce the correct footfall pattern (CPI) had improved for the SR group and was not different from the non-SR group (Figure 3A). Similarly, the SR animals showed improved ratio for plantar paw placement of the hindlimbs (PSI) with respect to the forelimbs(Figure 3B). However, the RI remained significantly lower for the SR group at the 13-week time point (Figure 3C). The large standard deviation for the RI values indicates that at least some of these animals struggled to combine plantar stepping with a correct pattern, even though the plantar stepping ratio alone and pattern formation alone were high (~80%) at week 13.
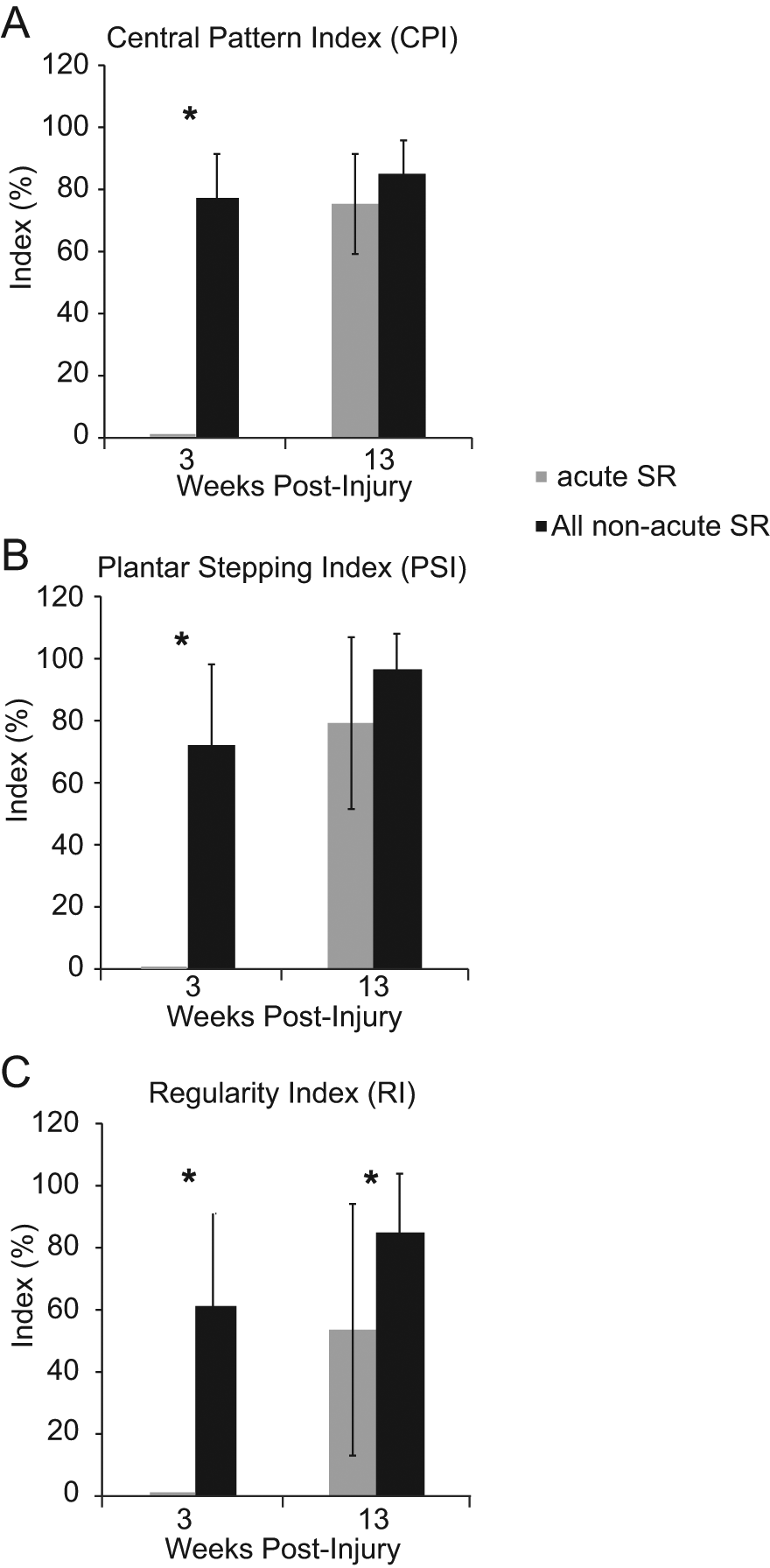
CPI, PSI, and RI Measures of Shallow Water Walking. (A-C) shallow water walking analysis at week 3 indicated that SR animals were dragging their hindlimbs, with no swing or stance phases due to lack of weight support. (A and C) By 13 weeks, the CPI and RI indices of gait had recovered for SR animals. (C) However, the RI shows that significant deficits still exist if the correct pattern is dependent on plantar steps only. (B) Importantly, the ratio of plantar stepping by the hindlimbs (compared to the forelimbs) was delayed but recovered by 13 weeks, indicating that the SR animals were able to weight support and achieved proper plantar paw placement in a few weeks without the daily stretch protocol. Abbreviations: CPI, central pattern index; PSI, plantar stepping index; RI, regularity index.
Magnetically Evoked Hindlimb Muscle Responses
Magnetic stimulation at the base of the tail evokes a short-latency (5-6 ms) muscle response, presumably via a monosynaptic reflex, indicating the state of excitability of motor neurons innervating ankle extensors (Figure 4). A trend toward decreased amplitude for the SR group exists at weeks 3.5 and 9.5, but do not reach significance (Figure 4A and C). Response amplitudes were significantly lower for the SR group only at week 6.5 (Figure 4B). Furthermore, at week 6.5, the response amplitude for the SR group is positively correlated with BBB scores from Monday afternoons (Figure 4D) suggesting that motor neuron excitability was recovering. This recovery is consistent with the profile of BBB scores (~weeks 5-8) even though the stretch protocol was still being administered daily.
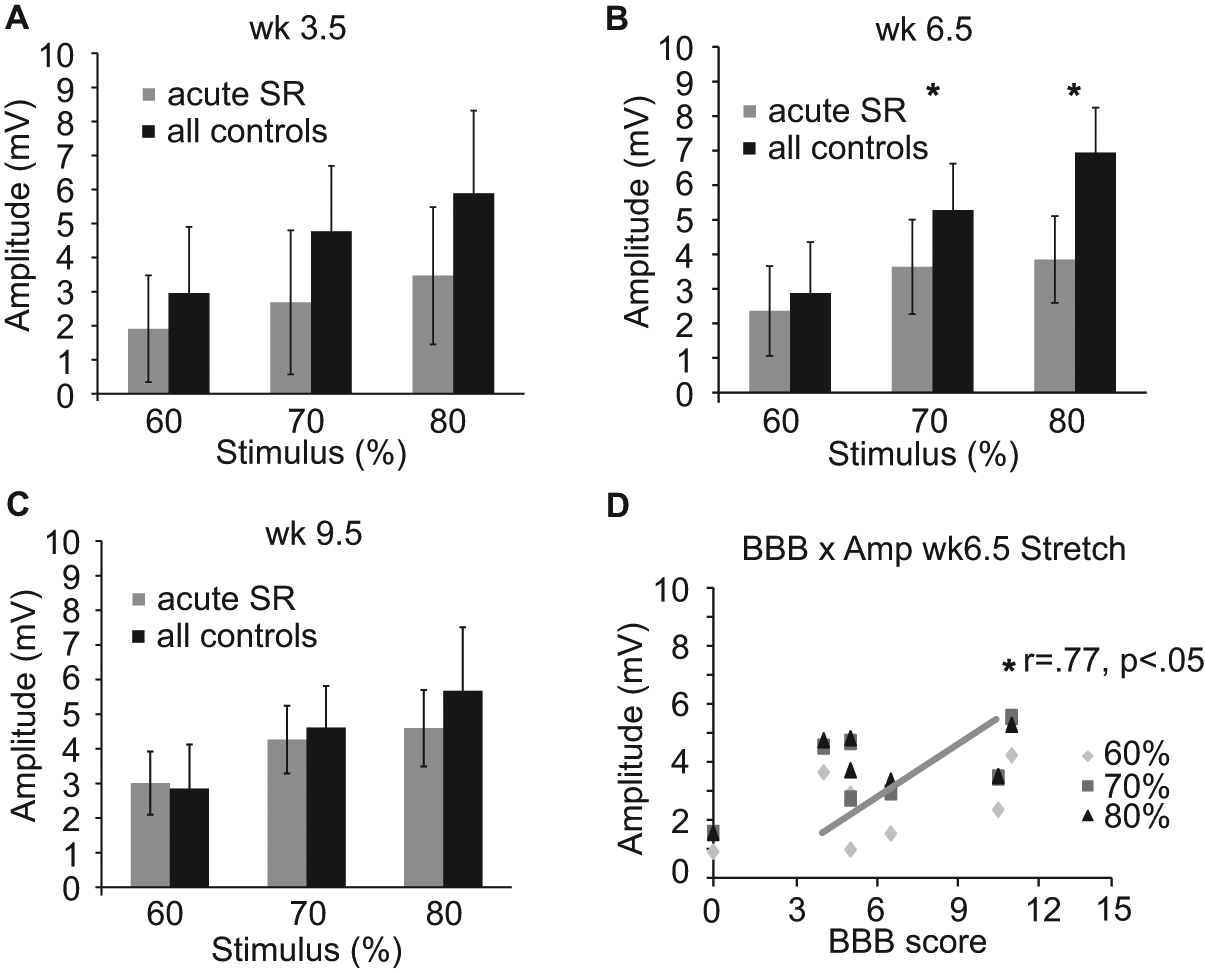
Magnetically evoked muscle responses daily stretch.
Muscle Weight and Histological Assessment of Epicenter
Hindlimb muscles were fresh dissected and weighed immediately. Unexpectedly, ankle extensor and flexor muscle groups were significantly lighter in SR animals as compared to non-SR controls (Table 1). Body weights were not significantly different between groups throughout the study, and there were no group differences in spared white matter at the injury epicenter (Table 1).
Muscle Weight and Histology a .
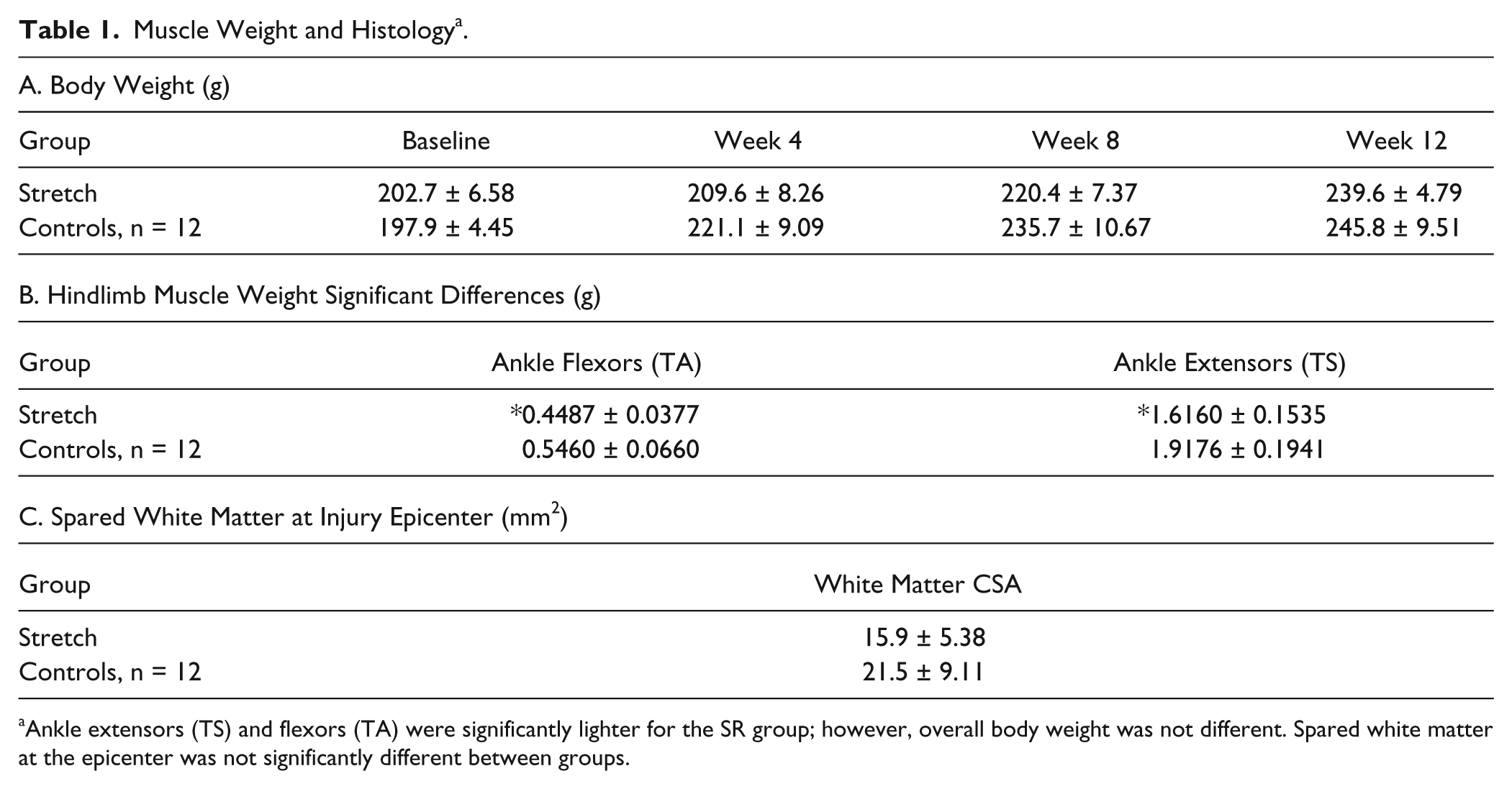
Ankle extensors (TS) and flexors (TA) were significantly lighter for the SR group; however, overall body weight was not different. Spared white matter at the epicenter was not significantly different between groups.
Discussion
The loss of locomotor function due to hindlimb stretching after contusive thoracic SCI has now been demonstrated for very mild 14 and moderate injury severities (present study). The stretching protocol was modeled on practices used in clinical settings of SCI management. However, the majority of our handlers were not trained therapists, and we discuss the implications of possible overstretching in the Supplemental Materials. Our outcomes suggest that repeated stretch, traditionally used to maintain muscle length and joint ROM after SCI, may delay and limit long-term functional locomotor recovery in an animal model that consistently shows substantial recovery by 6 weeks. When stretching began within 4 days of contusive SCI and occurred daily for 8 weeks, locomotor function was severely diminished through the first 4 weeks, and then recovered to suboptimal levels even though daily stretching was continued through weeks 5 to 8. Significant locomotor deficits persisted after the daily stretching ceased in weeks 9 to 13. When a separate group of animals underwent only 6 days of stretching that began in the chronic phase, at 10 weeks, there was a significant but transient decline in BBB scores that did not affect locomotion in the following weeks.
Loss and Gain of Locomotor Function
The profile of locomotor function over time in the current study is similar to that of 8 weeks of hindlimb immobilization in animals that were only mildly contused. 14 This congruent loss and gain of locomotor function is suggestive of a common mechanism for the 2 interventions, both of which model aspects of standard-of-care in SCI patients. We began our interventions acutely postinjury when animals just begin to move spontaneously in their cages using only their forelimbs for propulsion. Regardless of whether signs of inflammation and peripheral tissue pathology were 14 or were not (current study) outwardly visible, our models can be expected to induce some inflammation and to activate nociceptors. Sensitization of spinal cord circuitry resulting from noxious afferent input is NMDA and Substance P dependent, 23 and excessive stimulation by Substance P has been shown by Baumbauer and colleagues24,25 to diminish plasticity and induce a deficit in a model of spinal instrumental learning after transection. Furthermore, the key proinflammatory cytokine tumor necrosis factor-α (TNF-α) has also been implicated in spinal cord dysfunction. Direct application of TNF-α decreases, almost to extinction, compound action potentials recorded from isolated spinal cord white matter 26 and is both necessary and sufficient for long-term inhibition of instrumental learning. 27 Whether or not TNF-α, other inflammatory mediators, nociceptor activation, or Substance P are associated with the observed decline in locomotor function, it appears that stretching reduces the excitability of hindlimb motoneurons. We used a surrogate for the H-reflex to assess motoneuron responses to magnetic stimulation of afferents in the base of the tail at weeks 3.5, 6.5, and 9.5. SR animals had lower response amplitudes at all time points tested, but due to the inherent variability of repeated EMG measures, these were statistically significant only at the week 6.5 time point. Importantly, we found a significant correlation between BBB scores and evoked reflex amplitude (Figure 4D).
We also determined that the drop in locomotor function was not limited to stepping. Using the LSS, 22 we found that SR animals had significantly lower LSS scores at weeks 2 and 4 postinjury (Figure 2). This observation argues that stretch-induced declines in locomotor function are not specific to overground stepping and are likely to involve a generalized influence on spinal cord circuitry. It has been known for many years that muscle-spindle afferents (Ia) provide convergent excitatory input onto inhibitory interneurons,3,28 which, in-turn, are thought to play important roles during normal locomotion. The influence of therapeutic muscle stretch on spinal cord circuitry has been investigated very little. However, it is known that both ballistic and static stretch of triceps surae muscle group induces a significant decrease in soleus motoneuron excitability as assessed using the Hoffman Reflex. 29 How long this kind of inhibition lasts, how it might influence locomotor pattern generation, or if it is present or augmented after an incomplete SCI is currently unknown.
Clinical Implications and Conclusion
Stretching of the lower limbs is regarded as necessary for patients in the subacute and chronic phases following SCI, when potentially debilitating spasticity is developing and threatens to dramatically reduced the quality of life and interfere with rehabilitation efforts. 30 Our data suggest that any and all daily afferent input may contribute to or detract from spinal cord function/excitability and optimal recovery. The interaction of retained descending input with any and all afferent input will, in toto, “conduct the plasticity” to result in adaptive and/or maladpative functional changes (J. Wrathall, personal communication). In order to optimize functional recovery, combined therapies might focus on maintaining the potential for adaptive plasticity, retraining components of recovery that are not intrinsic to the locomotor circuitry, and minimizing exposure to afferent input that does not contribute to functional recovery. Wheelchair immobilization and hindlimb stretching 14 both appear to render animals unable to maintain intrinsic locomotor function or to involve their hindlimbs during day-to-day in-cage activity. Similarly, animals that are stand trained lose the ability to step, 31 possibly due to the barrage of extensor load and fusimotor proprioceptive input that is contrary to the patterned and timed afferent input required to optimize locomotion. Thus, task-specific gain of function coupled with loss of function in other intrinsic nontrained behaviors may imply that there are certain activities that need to be avoided in order to improve a given function, or “be careful what you train for.” 32 These findings support the notion that there may be a limited behavioral repertoire that can be retrained depending on the amount and location of spared tissue 32 and that this repertoire may decrease over time postinjury. 17
Despite the lack of primary research on the influence of stretching on spinal cord circuitry, we found a brief discussion in a primary physical therapy textbook indicating that facilitated, “hands-on,” therapies such as stretch and ROM can be detrimental to the recovery of SCI patients with some retained motor control below the level of injury.1(p372) If this notion is common knowledge for physical therapists, yet widely employed for SCI patients in rehabilitation facilities, it would behoove us to continue animal studies to refine and reassess the application of stretch as treatment only for patients that truly need it. In addition to the obvious detriments imposed by hindlimb stretch described herein, the most relevant implication of our study is that clinical assessments of motor function may be substantially influenced by stretch-based therapies and thus not provide an accurate view of neurological function. This is of vital importance because American Spinal Injury Association scores, Functional Independence Measure, and other neurological assessments are not only prognostic indicators but can also dictate both the course of treatment and the expected level of recovery. Increased communication, observation, and collaboration among basic science researchers, clinicians, and physical therapists are necessary to optimize treatment for SCI patients. 33 We posit that our data are not surprising to many physical therapists and that the presentation of such variation of locomotor function may have already been described but not systematically studied.
Footnotes
Acknowledgements
The authors wish to thank KSCIRC’s outstanding Core personnel: Darlene Burke, Johnny Morehouse, Christine Yarberry, and Kim Cash.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Defense (CDMRP SC110169), the Kentucky Spinal Cord and Head Injury Research Trust and the NIH (NIH/NIGMS P30 GM103507, R01 NS052292 & R56 NS052292).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
