Abstract
Background. In preclinical stroke models, improvement in motor performance is associated with reorganization of cortical motor maps. However, the temporal relationship between performance gains and map plasticity is not clear. Objective. This study was designed to assess the effects of rehabilitative training on the temporal dynamics of behavioral and neurophysiological endpoints in a rat model of focal cortical infarct. Methods. Eight days after an ischemic infarct in primary motor cortex, adult rats received either rehabilitative training or were allowed to recover spontaneously. Motor performance and movement quality of the paretic forelimb was assessed on a skilled reach task. Intracortical microstimulation mapping procedures were conducted to assess the topography of spared forelimb representations either at the end of training (post-lesion day 18) or at the end of a 3-week follow-up period (post-lesion day 38). Results. Rats receiving rehabilitative training demonstrated more rapid improvement in motor performance and movement quality during the training period that persisted through the follow-up period. Motor maps in both groups were unusually small on post-lesion day 18. On post-lesion day 38, forelimb motor maps in the rehabilitative training group were significantly enlarged compared with the no-rehab group, and within the range of normal maps. Conclusions. Postinfarct rehabilitative training rapidly improves motor performance and movement quality after an ischemic infarct in motor cortex. However, training-induced motor improvements are not reflected in spared motor maps until substantially later, suggesting that early motor training after stroke can help shape the evolving poststroke neural network.
Introduction
A substantial body of research directed toward the resolution of motor impairments after stroke focuses on therapeutic interventions to optimize the expression of adaptive neural plasticity.1,2 Plasticity phenomena in spared tissue have been examined at many levels of analysis, from altered gene expression to structural reorganization of axonal pathways to reorganization of motor representations. Changes in motor map topography have been particularly relevant in translational studies since they can be examined in clinical populations with noninvasive imaging approaches 3 and can be addressed in more detail in preclinical studies using invasive approaches.4,5
Map expansion is thought to be related to functional restoration, as both develop in parallel during the early weeks to months following cortical injury. However, the temporal relationship of map plasticity and functional improvement is complex, as behavioral gains can plateau prior to expansion of motor maps. 6 Many early changes in motor maps may be independent of functional capabilities, presumably related to pathophysiological processes including diaschisis, edema, and hyperexcitability. Faced with a complex interplay of early pathophysiological events, spontaneous improvements in motor function, development of compensatory motor strategies, and initiation of regenerative processes, understanding the role of rehabilitative interventions in shaping neuroplastic events that ultimately will support recovered performance seems daunting.
To better understand how postinjury motor experience affects map plasticity and behavioral performance, a rat model of ischemic cortical injury was used. An ischemic infarct was directed at the forelimb representation within the primary motor cortex (caudal forelimb area; CFA). Motor maps were derived within the spared territory rostral to the infarct, including the rostral forelimb area (RFA), a motor field with many similarities to premotor cortex in primate species. 7 In different groups of rats, motor maps were derived either the day after a 10-day rehabilitative training period or after a 3-week follow-up period. Map plasticity was compared to improvements in motor performance and movement kinematics on a skilled reaching task. While a previous study in rats demonstrated reduced forelimb representations in the ipsilesional RFA after a traumatic injury to CFA, 8 this study represents the first examination of map changes in this area after an ischemic injury. It is also unique in its demonstration of how motor experience modulates map plasticity beyond the timeframe of rehabilitative training.
Materials and Methods
Subjects and Group Assignments
A total of 25 adult, male, Long-Evans hooded rats (Harlan; 4-5 month old, 300-400 g) was used in accordance with National Institutes of Health regulations, and approved by the Institutional Animal Care and Use Committee of the University of Kansas Medical Center. Rats were singly housed in a Plexiglas cage with ad libitum food and water on a 12 hour:12 hour light–dark cycle, with ambient temperature maintained at 68 to 71°F. Animals were randomly assigned to 1 of 4 post-lesion groups that varied by survival time (18 or 38 days) and postinjury behavioral experience (rehabilitative training, referred to as “rehab” group, or no rehabilitative training, referred to as “no-rehab” group). Thus, there were 4 groups: (a) rehab/short-term survival (n = 6), (b) rehab/long-term survival (n = 7), (c) no-rehab/short-term survival (n = 6), and (d) no-rehab/long-term survival (n = 6). Through post-lesion day (PLD) 18, rats in the 2 rehab groups received the same preoperative and postoperative experiences; rats in the 2 no-rehab groups also received the same experiences. Therefore, these groups were combined as rehab (n = 13) and no-rehab (n = 12) groups for analyses through PLD 18.
Pre-Infarct Behavioral Training and Motor Assessment
Single-Pellet Retrieval Task Training
This task has been used in numerous studies after focal cortical infarcts in rodents and nonhuman primates due to its high sensitivity and reliability. 9 High-resolution video recordings were made of training and assessment sessions for subsequent slow-motion and frame-by-frame analysis. Each animal was placed within a Plexiglas reaching box with a 1-cm-wide slot. Rats reached through the slot a distance of 2 cm to retrieve a single food pellet (45 mg, Bioserve) from a horizontal shelf. Forelimb preference was determined for each rat prior to training. A removable wall was inserted into the Plexiglas box to restrict the rat to reach only with the preferred forelimb (the forelimb used for more than 50% of the reaches). Pre-infarct training proceeded for 10 days (60 single-pellet trials/day).
Single-Pellet Retrieval Task Assessment
Pre-infarct motor performance was assessed on the day following the 10-day training period. The total number of successful retrievals and total number of reaching attempts (limb advances through reaching slot) were tallied based on 20 single-pellet trials. A successful retrieval required the rats to grasp and transport the food pellet to their mouth. Each trial ended with a successful retrieval or 5 unsuccessful attempts.
Postinfarct motor performance was assessed on the single-pellet retrieval task on PLD 7, 12, 17 and in the long-term survival groups, every 5 days through PLD 37. Performance was based on a 20-trial session. On assessment days that coincided with rehabilitative training days, assessment trials were conducted prior to rehabilitative training trials.
Kinematic Analysis
In the long-term survival groups, kinematics of forelimb use during the retrieval task were assessed using the Eshkol–Wachmann Movement Notation adapted by Whishaw and Pellis. 10 For each trial resulting in a successful retrieval, the quality of forelimb movements was analyzed. Specific movements consisted of pronation, grasp, supinate I (acquiring the pellet), supinate II (retrieving the pellet), and release.11,12 For each movement, a score of 0 was assigned if the movement was normal, 0.5 if the movement was abnormal, but present, or 1 if the movement was absent.
Foot-Fault Task
To assess forelimb performance during locomotion, a foot-fault task was conducted on the day prior to the lesion, PLD 7, 12, 17 and in the long-term survival groups, every 5 days through PLD 37. Each rat was placed onto an elevated grid (57 cm × 44 cm with 4 cm × 4 cm grid opening) and allowed to locomote freely for 3 minutes. A foot fault was defined as extension of the forepaw through the grid openings without any of the digits catching on a grid. The number of steps and the number of foot faults made with each forelimb were recorded. Performance was defined as the percentage of foot faults per step made with the forelimb contralateral to the lesion.
Postinfarct Rehabilitative Training
Rehabilitative training was initiated in the rehab groups on PLD 8. On PLD 8 to 12, a tray-reaching task was used. This task requires less precise reaching and grasping than the single-pellet retrieval task since rats retrieve pellets for 20 minutes from a tray filled with pellets. 13 On PLD 13 to 17, the single-pellet retrieval task was implemented (60 training trials per day). Rats assigned to the no-rehab groups had a similar number of food pellets available from the floor of the reaching box. Randomly selected videos of motor performance and movement kinematics were independently scored by separate examiners blind to the experimental condition as a reliability check.
Cortical Infarct Procedure
Anesthesia was induced with 3% isoflurane gas followed by ketamine (100 mg/kg, intraperitoneal) and xylazine (5 mg/kg, intramuscular). Additional doses of ketamine (20 mg/kg intramuscular) were used as needed. Six 0.7-mm diameter holes were drilled over the CFA contralateral to the dominant forelimb at anteroposterior +1.5, +0.5, and −0.5 mm and mediolateral +2.5 and +3.5 mm from bregma. 14 To induce cortical ischemia, 0.33 µL of endothelin-1 (ET-1; Bachem Laboratories, 0.3 mg/mL) was injected into each hole at a depth of 1.5 mm from the cortical surface, through a micropipette (160 µm o.d.) attached to a Hamilton syringe using a microsyringe injector (UltraMicro Pump III, World Precision Instruments). Appropriate postoperative care was provided under veterinary supervision.
Postinfarct Neurophysiological Assessment
Standard intracortical microstimulation (ICMS) techniques were used to derive forelimb movement maps in the cortex rostral to the ischemic lesion. 8 Rats in short-term and long-term survival groups underwent an ICMS mapping procedure on PLD 18 and 38, respectively. Rats were anesthetized with ketamine/xylazine and secured in a stereotaxic frame. Anesthesia was maintained throughout the procedure with bolus injections of ketamine (20 mg/kg, intramuscular) as needed to minimize spontaneous movements and toe pinch reflex. A craniectomy was performed over the frontal cortex. A digital image of the surface vasculature was obtained and imported into a graphics program to guide the placement of the microelectrode on a 250 µm grid pattern. A glass microelectrode (tapered to 15-20 µm o.d. with beveled tip; impedance = 500-700 kΩ) was filled with 3.5 M NaCl and connected to a constant-current stimulator (Model BSI-2, BAK Electronics) through a platinum wire. The electrode was lowered to 1700 µm (approximately Layer V) using a hydraulic microdrive (Model 650, David Kopf Instruments). The ICMS stimulus consisted of 13 cathodal pulses (200 µs each) delivered at 350 Hz. Movements and their threshold current levels (maximum = 80 µA) were recorded for each stimulation site. ICMS-evoked movements were defined via visual observation by an observer blind to the electrode placement. A second observer, blind to the experimental condition, verified the evoked movement. Distal forelimb movements were defined as visually observable movements of the wrist (extension, flexion, supination, pronation) and/or digits (extension, flexion). Proximal forelimb movements were defined as visually observable movements of the elbow (extension, flexion) and/or shoulder (extension, flexion). Movement representation maps were reconstructed from movement topography and areal extents measured with imaging software (NIH IMAGE vl.61). Rostral, caudal, medial, and lateral extents of forelimb representations were recorded based on the boundaries of reconstructed maps (rostral, caudal relative to bregma; medial, lateral relative to midline).
Histology
Immediately following the ICMS mapping procedure, rats were euthanized by an overdose of Buthanasia and perfused transcardially with normal saline followed by 4% paraformaldehyde in 0.1 M phosphate-buffered saline. Brains were postfixed in 20% glycerol, sectioned coronally (30 µm), and stained with cresyl violet. Lesion volume estimation was obtained by the difference of the cortical volume of the injured hemisphere subtracted from that of the intact hemisphere, 8 using the Cavalieri method in StereoInvestigator (Microbrightfield, Inc).
Statistical Analysis
Statistical analyses were performed using JMP v10.0 (SAS Institute, Inc, Cary, NC). Two-way ANOVAs were used to examine the effects of Group, Time, and Group × Time interactions on lesion volume, motor performance, kinematic scores, topography of forelimb motor maps, and ICMS current thresholds. Post hoc comparisons were performed with Tukey tests when appropriate (α = .05).
Results
Histological Results
The lesion extended through all cortical layers leaving the underlying white matter intact in all infarcted rats (Figure 1). No cortical damage was evident at the level of RFA in any of the cases. While the main effect of Group was not statistically significant (F1,21 = 3.9206; P = .0603), mean lesion volume in the rehab group was 36% larger than in the no-rehab group.
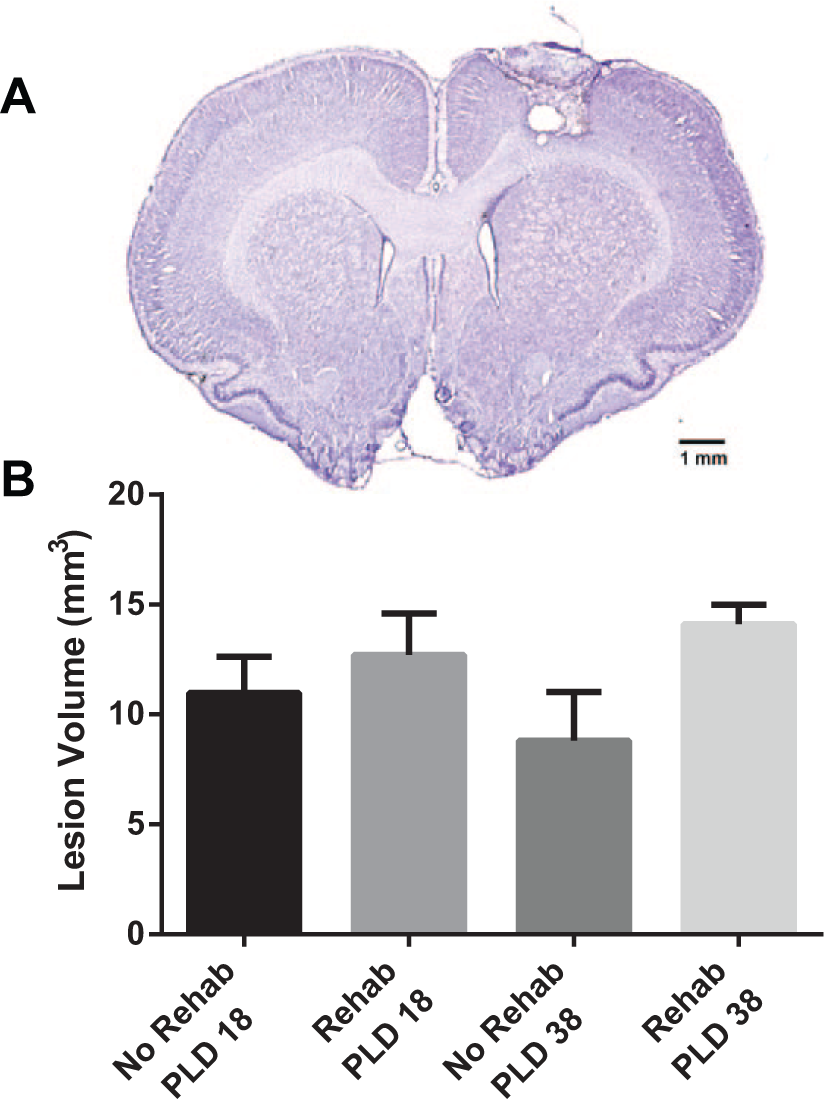
Histological results.
Effects of Rehabilitative Training on Motor Behavior
Behavioral results are described separately for 3 post-lesion phases (Figure 2): (a) Lesion phase (PLD −1 to PLD 7); (b) Rehabilitative training phase (single pellet retrieval training; PLD 12 to PLD 17); and (c) Follow-up phase (PLD 22 to 37).
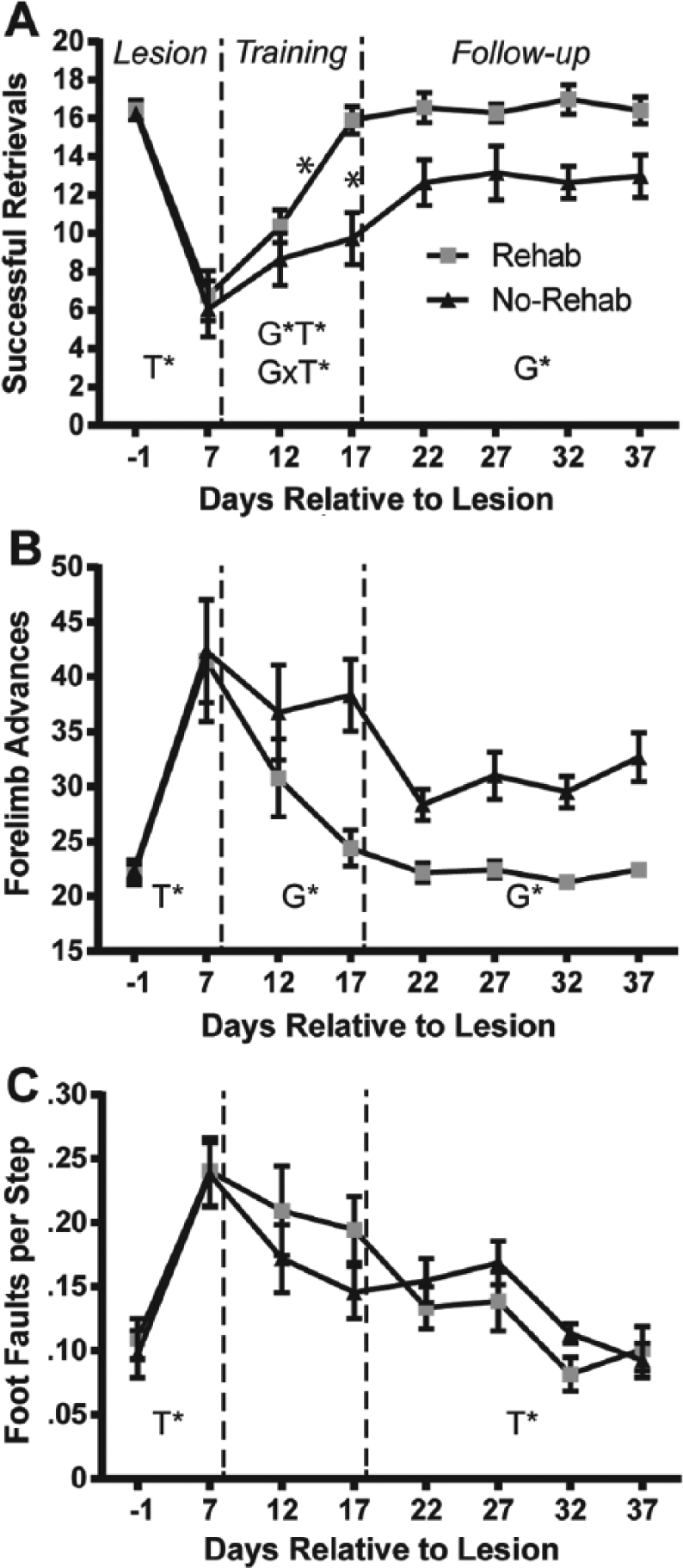
Motor performance results.
Successful Retrievals (Figure 2A)
Lesion phase
The analysis revealed a significant effect of Time (F1, 23 = 105.908, P < .0001), demonstrating that the lesion resulted in a deficit in both the rehab and no-rehab groups.
Rehabilitative training phase
There was a significant effect of Group (F1,23 = 8.465, P = .008), Time (F1,23 = 20.828, P = .0001), and Group × Time interaction (F1,23 = 9.428, P = .005). Post hoc tests revealed that the rehab group retrieved significantly more pellets on PLD 17, and also compared with both groups on PLD 12.
Follow-up phase
There was a significant effect of Group (Group F1,33 = 6.808, P = .024), demonstrating that the benefit of rehabilitative training persisted throughout the follow-up phase.
Reaching Attempts (Forelimb Advances; Figure 2B)
Lesion phase
Similar to the retrieval analysis, there was a significant effect of Time (F1,23 = 31.064, P < .0001), demonstrating a lesion effect in both groups.
Rehabilitative training phase
There was a significant effect of Group (F1,23 = 6.688, P = .017), indicating that the rehab group made fewer reaching attempts to retrieve pellets compared with the no-rehab group.
Follow-up phase
There was a significant effect of Group (F1,33 = 27.353, P = .0003), demonstrating that the benefit of rehabilitative training persisted after training was discontinued.
Foot Faults (Figure 2C)
Lesion phase
The ANOVA revealed a significant effect of Time (F1,23 = 43.195, P < .0001), demonstrating a deficit as a result of the infarct in both groups.
Rehabilitative training phase
There were no statistically significant effects.
Follow-up phase
There was a significant effect of Time (F3,33 = 3.945, P = .0163), demonstrating improvement in both groups on the foot fault task during the follow-up phase.
Movement Kinematics on Single-Pellet Retrieval Task
Lesion phase
There was a significant effect of Time (lesion effect) for supinate I (F1,10 = 21.922, P = .0009), supinate II (F1,10 = 44.705, P < .0001), and release (F1,10 = 26.406, P = .0004) (Figure 3). The effect of Time for grasp approached significance (F1,10 = 4.755, P = .054). There was no effect of Time for pronate, advance, or digit extend. Thus, the remaining analyses were limited to supinate I, supinate II, release, and grasp.
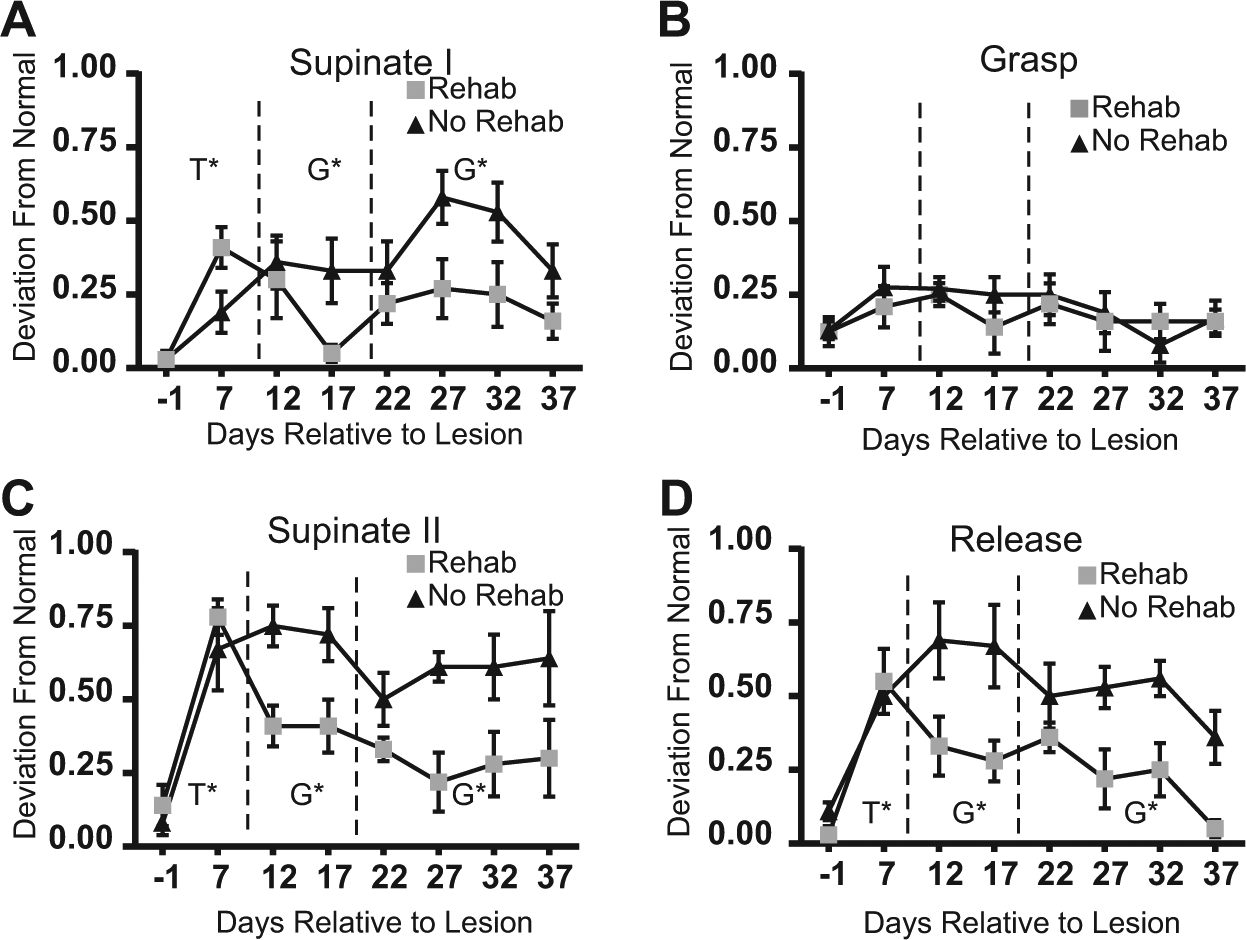
Kinematic results.
Rehabilitative training phase
The effects of Time on supinate I approached significance (F1,10 = 4.54, P = .059). Significant Group differences were found for supinate I (F1,10 = 6.47, P = .029), supinate II (F1,10 = 23.17, P = .0007), and release (F1,10 = 30.39, P = .0003), reflecting superior kinematic endpoints in the rehab group.
Follow-up phase
Group differences favoring the rehab group persisted for supinate I (F3,30 = 15.5, P = .0005), supinate II (F3,30 = 18.40, P = .0002), and release (F3,30 = 25.42, P < .0001). There was an effect of Time for release (F3,30 = 3.62, P = .024). Post hoc analysis comparing PLD22 to PLD37 revealed that both rehab and no-rehab groups showed significant improvement in release.
Effects of Rehabilitative Training on Motor Output Maps
Location of Forelimb Representations
ICMS maps were successfully obtained in 21 of 25 rats. Two rats died prior to the mapping procedure (long-term rehab group = 1, long-term no-rehab group = 1). In 2 additional rats (long-term rehab group = 1, long-term no-rehab group = 1), no evoked movements were observed from ICMS stimulation at the maximum current level (80 µA). Such outcomes are not uncommon in ICMS experiments and are typically attributable to improper anesthetic depth that cannot be corrected during the course of the procedure. 8 Subsequent analyses focused on the 21 rats with successful maps.
On both PLD 18 and PLD 38, the spared forelimb motor representations were largely contained within the typical RFA location (Figure 4). 8 In both rehab and no-rehab groups, neck, jaw, orofacial, and vibrissae movements were evoked on the medial, rostral, and lateral borders of the forelimb representation (as well as the caudal border in rats with more rostrally located forelimb maps). Because only the forelimb representation was mapped in its entirety, we did not examine these additional representations systematically.
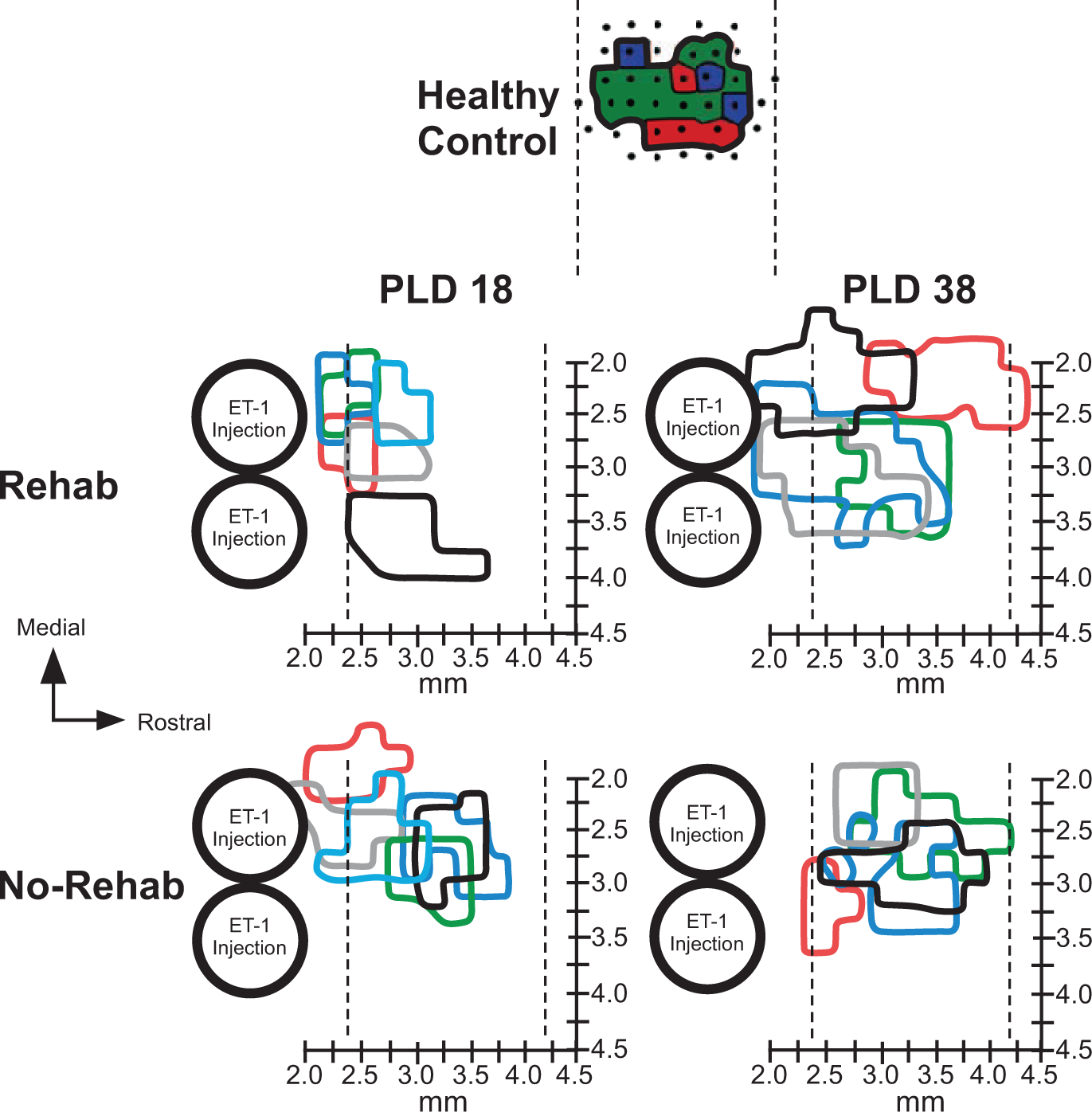
Forelimb movement representations rostral to infarct.
On PLD 18, in 2 rats in each group, movements could not be evoked rostral to the forelimb representation using the maximum current (80 µA). In 3 rats in each group, the forelimb representation extended further caudally to include the peri-infarct area immediately rostral to the infarct, where neck and orofacial representations typically are found.11-13 As this location shift was relatively small (<500 µm), we cannot rule out the roles of tissue cavitation (in the infarcted area), edema, or inflammation in such shifts. Thus, since it could not be determined that these forelimb representations were entirely within RFA, we conservatively refer to forelimb representations rostral to the lesion.
On PLD 38, in 3 rats in the rehab group, forelimb movements were evoked caudal to the typical RFA territory. There was a significant effect of Time in the rostral extent of forelimb movement maps (F3,20 = 5.7161, P = .0287), but no effect of Group nor Group × Time interaction. The map extended more rostrally in long-term versus short-term survival groups. There were no significant differences in medial or lateral extent of the maps.
Size of Forelimb Representations
On PLD 18, the total forelimb area rostral to the lesion in the rehab and no-rehab groups was 0.34 ± 0.06 mm2 (mean ± SEM) and 0.54 ± 0.05 mm2, respectively. This contrasts with the much larger RFA area in historical controls (0.95 ± 0.7 mm2).8,15 On PLD 38, the total RFA area was 1.08 ± 0.11 mm (14% larger than historical controls) and 0.60 ± 0.09 mm (37% smaller than historical controls) in the rehab and no-rehab groups, respectively.
There was a significant effect of Time in the total forelimb area rostral to the lesion (F3,20 = 23.4417, P = .0002) and a significant Group × Time interaction (F3,20 = 19.5069, P = .0004). Post hoc tests indicate that the forelimb area of the rehab/PLD 38 group was larger than any of the other 3 groups (P < .05, Tukey’s HSD; Figure 5A).
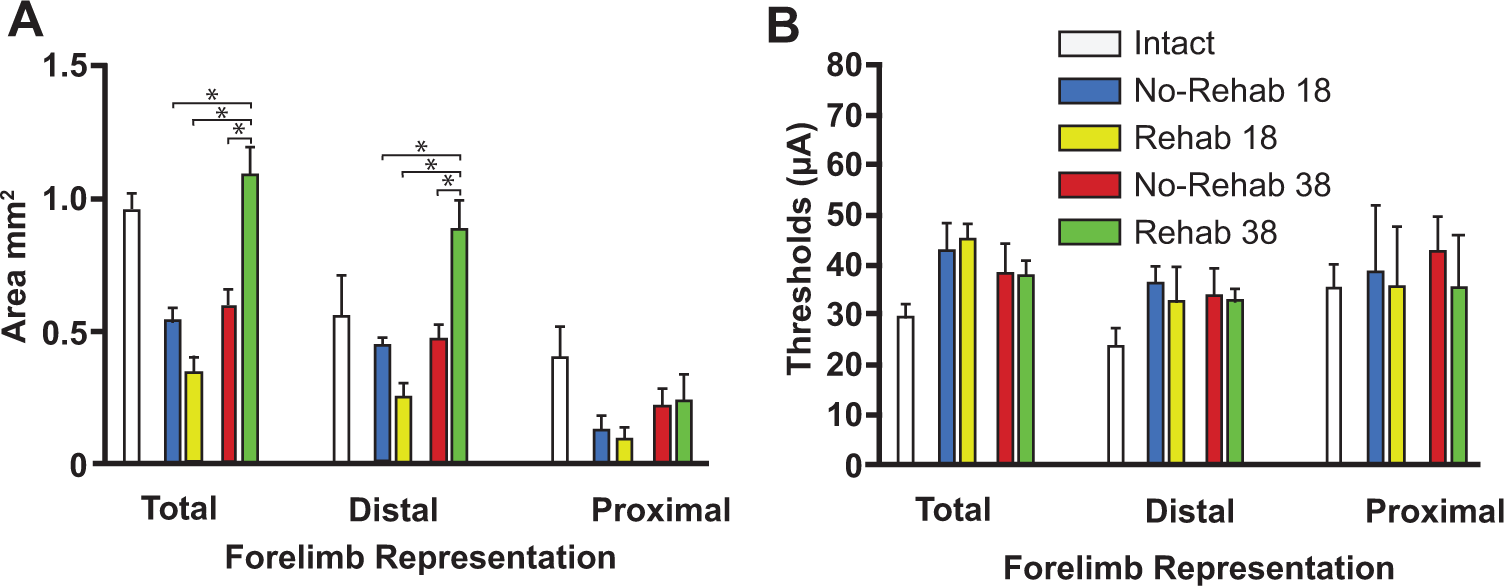
Motor map areas and thresholds.
Further analysis to differentiate distal versus proximal representations revealed that most of the change in the total forelimb area was due to changes in the distal area. While the effect of Group approached significance (F3,20 = 3.9535, P = .0631), there was a significant effect of Time (F3,20 = 16.6762, P = .0008) and a significant Group × Time interaction (F3,20 = 18.4697, P = .0005). Post hoc tests showed that the distal forelimb area was larger in the rehab/38 day group than any of the other 3 groups (P < .05). There were no significant effects for proximal forelimb area.
There were no significant linear correlations between map size and final performance or kinematic scores. The strongest relationship was between the distal forelimb area and release scores (PLD 38 rats; F = 2.5018, P = .1524).
ICMS Current Thresholds
There were no significant differences in the threshold currents required to evoke distal or proximal forelimb movements (Figure 5B).
Discussion
After a focal ischemic infarct in the rat’s primary motor cortex forelimb representation, rehabilitative training was effective in improving motor performance and movement quality on a single-pellet retrieval task. Motor scores returned to near-baseline performance by the end of the rehabilitative training phase, and gains were retained during a 3-week follow-up period. Spared forelimb motor maps were unusually small in both groups on PLD 18. Maps were significantly larger in the rehab group on PLD 38, tripling in size compared to PLD 18. These results demonstrate that rehabilitative training results in rapid improvements in motor performance and movement quality, and delayed expansions in spared motor maps.
Effects of Rehabilitative Training on Motor Performance and Movement Quality
The performance deficits and training-induced performance gains observed in this study are similar to those reported previously in rats after motor cortex lesions, though the severity of initial deficits and the time course of improvements are largely dependent on lesion size and location.16-19 Altered movement kinematic patterns during the single-pellet retrieval task also have been demonstrated frequently in rats after focal cortical lesions in motor cortex, especially in supination/pronation, aim, grasp, and release.11,16,17,20 Some impairments, such as grasp kinematics, are much more severe in rats with subcortical infarcts. 21 Since the rehabilitative training was focused on the retrieval task, requiring skilled use of the digits, it is not surprising that rehabilitative training did not affect performance on the foot-fault task. This suggests that training effects are task-specific and do not generalize to tasks that do not require skilled digit use.
It is typically assumed that motor performance gains are due to a combination of compensation (use of alternative kinematic patterns) and true recovery (return of baseline kinematic patterns).16,17,22-27 One of the key translational questions regarding poststroke rehabilitation is the following: Can rehabilitative interventions result in more extensive recovery of normal kinematic patterns, that is, movement quality? In the present study, rehabilitative training resulted in a sustained improvement in kinematics, in addition to greater and more rapid gains in functional performance. In a previous study after motor cortex lesions in rats, neither motor performance nor normalization of kinematic patterns was aided by poststroke practice. 16 Also, a recent study of chronic human stroke survivors undergoing a constraint-induced movement therapy intervention demonstrated improvement in functional outcomes, but no improvement in kinematic outcomes. 28 However, other studies have demonstrated that behavioral experience can alter not only the trajectory of motor recovery but can at least partially reduce compensatory movement patterns.17,18
It can be argued that performance gains by rats in the no-rehab group in the present study were largely compensatory. But the present results suggest that rehabilitative training provides benefits beyond simply improving compensatory skills and may promote true recovery. The results parallel those in a human stroke population in which motor performance improved over time, but those subjects with the greatest recovery of normal kinematic patterns had the most extensive functional improvement. 29
Temporary Disruption of Spared Motor Maps After Motor Cortex Lesions
The reduced size of forelimb representations on PLD 18 suggests that, at least for a few weeks after the infarct, the functional integrity of cortex rostral to the lesion (including RFA) is disrupted, perhaps by a diaschisis-like effect. Focal cortical infarcts produce hypometabolism throughout a large region of ipsilesional cortex, closely corresponding to areas with known corticocortical connections with the infarct core. 30 Thus, the functional integrity of neurons connected to the infarct is likely to be compromised. Since the CFA and RFA have dense reciprocal interconnections, neurons involved with motor control of the forelimb are most likely to be disrupted, allowing a competitive advantage for neurons more involved with motor control of more proximal (neck, orofacial, vibrissae) musculature. Similar hypotheses have been proposed to explain map changes after focal cortical impact injuries. 8 Such injuries result in widespread sublethal effects in the cortex ipsilateral to the damage. 31 It has long been known that the overlap of face/neck and forelimb representations in motor cortex provides a substrate for rapid map plasticity. Forelimb motor sites can convert to face/neck motor sites and vice versa within hours.32,33 If sublethal effects differentially compromise forelimb related neurons in RFA due to the dense reciprocal connections with CFA, then more proximal movement fields are likely to emerge. The present results suggest that this bias toward face/neck representations persists at least for a few weeks after the injury.
Temporal Relationship Between Cortical Plasticity and Behavioral Recovery
While forelimb motor maps were substantially smaller on PLD 18, motor performance had already plateaued. Thus, behavioral improvement preceded map expansion. Similar results were found in the supplementary motor area of monkeys undergoing spontaneous recovery after infarcts that damaged primary motor cortex and nearby premotor areas. 6 It was suggested that undetected changes in behavior, such as improvement in kinematic patterns, may have occurred during later stages and that late motor map reorganization may reflect improved movement quality.
Part of the rationale for conducting the present study was to determine whether late motor map changes reflect late improvements in kinematic patterns. However, as with motor performance measures, kinematic endpoints had already improved by the end of the rehabilitative training period. As there continued to be improvements in release during the follow-up period, it is still possible that some of the late map expansion was due to refinement in movement quality. It is possible that the smaller PLD 38 forelimb maps in the no-rehab group reflect the continued use of maladaptive compensatory movement strategies, sometimes called “learned-bad use.” 34 Such compensation may have dampened both motor performance gains and forelimb map expansion.
The lack of an effect of rehabilitative training on motor maps on PLD 18 contrasts with the large number of neuroimaging studies in stroke survivors demonstrating rapid structural and functional changes after rehabilitative interventions. 35 However, human studies correlating the effects of interventions on neuroimaging endpoints are typically done in a more chronic state. The present results suggest that as clinical trials are conducted at earlier time points after stroke, neuroimaging results may not be entirely predictable from results in chronic populations. Changes in functional maps may only be observable at later time points.
There is considerable evidence that cortical injury initiates a plethora of presumably regenerative neurophysiologic and neuroanatomic events in the peri-infarct and remote tissue that last for at least a few weeks.27,36 A time-dependent expression of both neuronal growth-promoting and growth-inhibiting genes occurs within the first week after stroke. Altered gene expression profiles are found in the peri-infarct tissue
37
and in neurons in the RFA that project to the infarcted zone.
14
Other early changes include synaptogenesis, axonal sprouting, dendritic arborization, and dendritic spine remodeling.38-44 Peri-infarct neuronal excitability changes occur rapidly and are mediated by extrasynaptic GABAA (γ-aminobutyric acid type A) receptors.45-47 Pharmacologic blockade of tonic GABAergic transmission during this early stage can rapidly improve recovery in mice after stroke.
47
Also, decreased excitation in the peri-infarct cortex is mediated by altered NMDA (N-methyl-
In conclusion, during the first few weeks after injury, 2 competing processes are at play: (a) Diaschisis results in sublethal, presumably reversible functional disruption of local and connected neurons; less affected neurons in these same regions gain a competitive advantage, resulting in altered map topography. (b) Regenerative processes are set into motion and are modifiable via behavioral or pharmacological interventions. Thus, rehabilitative training during the first few weeks postinjury results in rapid behavioral gains, but such gains typically are not expressed in motor output maps due to the ongoing diaschisis. However, cortical reorganization continues long after behavioral recovery has stabilized. It is likely that rehabilitative training guides the eventual neuroanatomical and neurophysiological changes that will persist in chronic stages, much like motor training in healthy rats induces synaptogenesis that is reflected later. 50 The implication of these results for clinical stroke rehabilitation is that the process of postinjury neural plasticity can be guided in a powerful way by the type and quality of early postinjury motor experience.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by NIH R37 NS030853 to RJN.
