Abstract
Background. Upper-limb (UL) dysfunction is experienced by up to 75% of patients poststroke. The greatest potential for functional improvement is in the first month. Following reperfusion, evidence indicates that neuroplasticity is the mechanism that supports this recovery. Objective. This preliminary study hypothesized increased activation of putative motor areas in those receiving intensive, task-specific UL training in the first month poststroke compared with those receiving standard care. Methods. This was a single-blinded, longitudinal, randomized controlled trial in adult patients with an acute, first-ever ischemic stroke; 23 participants were randomized to standard care (n = 12) or an additional 30 hours of task-specific UL training in the first month poststroke beginning week 1. Patients were assessed at 1 week, 1 month, and 3 months poststroke. The primary outcome was change in brain activation as measured by functional magnetic resonance imaging. Results. When compared with the standard-care group, the intensive-training group had increased brain activation in the anterior cingulate and ipsilesional supplementary motor areas and a greater reduction in the extent of activation (P = .02) in the contralesional cerebellum. Intensive training was associated with a smaller deviation from mean recovery at 1 month (Pr>F0 = 0.017) and 3 months (Pr>F = 0.006), indicating more consistent and predictable improvement in motor outcomes. Conclusion. Early, more-intensive, UL training was associated with greater changes in activation in putative motor (supplementary motor area and cerebellum) and attention (anterior cingulate) regions, providing support for the role of these regions and functions in early recovery poststroke.
Introduction
A stroke results in permanent damage to the brain, but most patients make some recovery. In the first few hours postevent, this relates to early reperfusion, but following this, most of the recovery is related to brain reorganization (neuroplasticity).1-3 Nonetheless, up to one-third of patients need ongoing assistance, with upper-limb (UL) dysfunction being a major contributor to this disability. 4 Evidence indicates that earlier, more-intensive, and/or more-challenging training can improve functional outcomes5-8 and influence brain activation, which may be a marker of neuroplasticity.3,9
Techniques such as functional magnetic resonance imaging (fMRI) can identify changes in regional cerebral blood flow and, in turn, brain activation, during a motor task. 10 A recent quantitative meta-analysis 11 found that better motor performance (not specifically linked to motor training) was associated with greater activation likelihood in the ipsilesional primary motor cortex, supplementary motor area, contralesional premotor cortex, and cerebellum and that the pattern of activation varies with time poststroke. Most imaging studies investigating UL motor recovery in ischemic stroke patients have recruited participants at least several months after the event, as indicated by a systematic review published in 2008. 3 In 2010, Buma et al 12 reviewed 19 studies that recruited between 2 and 14 participants; it included a study by Ward et al 13 who recruited 2 participants within 2 weeks poststroke and, when comparing their findings to 7 healthy volunteers, found bilateral activity after recent motor strokes. Buma et al 12 found a correlation between favorable UL outcomes and changes in activity in motor and nonmotor areas in the first few months poststroke; however, none of these studies specifically investigated the impact of motor retraining. The only study that has recruited stroke patients in the first 2 weeks postevent and investigated the impact of repetitive, task-specific UL training, which, in this instance, was part of the standard care provided by a Comprehensive Stroke Unit, was reported by Askim et al. 14 When compared with healthy controls, the authors found bilateral reorganization in the primary sensorimotor areas in the first month. Interpretation of neural outcomes associated with specific motor training early poststroke may be particularly influenced by the presence of spontaneous recovery and/or spontaneous changes in neural activation, including maladaptive plasticity.1,15 -19 Under these circumstances, a randomized controlled trial (RCT) design is critical. Although increasing evidence supports the association between poststroke recovery and changes in task-related activation, the patterns of brain activation underlying recovery early poststroke and in response to specific interventions requires systematic and controlled investigation. 20
To date, no study has used a RCT design to compare changes in brain activation in response to different intensities of UL motor training in the first month poststroke. 21 Clinical guidelines recommend training of motor functions at this time. 22 Moreover, intensity of training is a critical component of successful training and is associated with learning-dependent plasticity. 23 In this preliminary study, we investigated the impact of different intensities of task-specific UL training on brain activity and UL recovery in a cohort of adult, first-ischemic stroke patients with an affected UL. We hypothesized that there would be increased activation of putative motor areas, specifically in the ipsilesional primary sensorimotor, premotor, supplementary motor, and anterior cingulate areas and in the contralesional cerebellum in those receiving intensive, task-specific UL training when compared with those receiving standard care.
Methods
This prospective, single-blinded, RCT recruited stroke patients admitted to 2 acute stroke units. The study received ethical approval from the region’s human research ethics committees, and informed consent was obtained from all participants.
Participants
Participants were recruited within 1 week of being diagnosed with a first-ever, ischemic stroke resulting in UL motor impairment. Patients were identified as potential participants by their admitting neurologists. All stroke unit neurologists were informed of the study’s inclusion/exclusion criteria, but a decision to inform the researchers of a potential participant was at the discretion of the treating physician. Therefore, some patients may not have been referred because they were screened as ineligible by the treating physician. Patients were excluded from the study if they were left-handed, scored more than 16 on the UL component of the Motor Assessment Scale (UL-MAS), 24 were unable to correctly respond to a 2-step command (NIHSS, 25 item 7a), were fearful of confined spaces, and/or had a condition incompatible with MRI. In all, 24 patients who fulfilled the eligibility criteria consented to participate in the study, with one subsequently withdrawing before randomization because of claustrophobia. All 23 participants remained in the study for the 3-month duration (Figure 1).
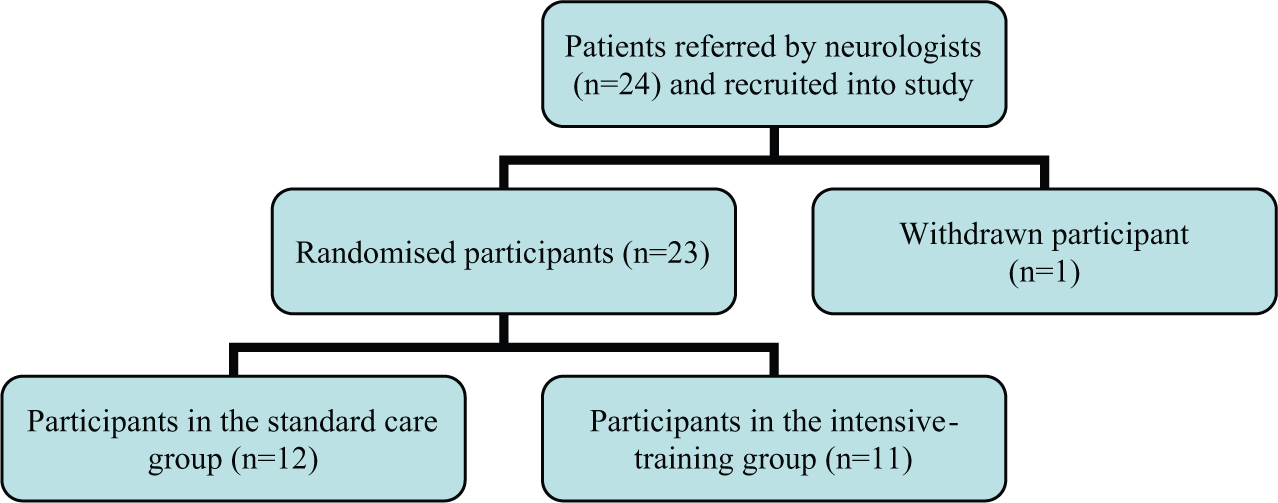
CONSORT diagram of patients recruited to the study.
Study Design
Participants were randomized 1:1 to either the standard-care group or the intensive-training group, who received an additional 30 hours of UL training that was task specific. Randomization was stratified for hemispheric side of lesion. The randomization protocol was established prior to the onset of recruitment and was independently administered by an associate researcher not otherwise involved in this study. Before the study started recruiting participants, 2 sets of envelopes were collated, one for each side of the lesion. Each envelope contained a listing of either the standard-care or intensive-training group, and all envelopes were then sealed. The 2 sets remained separate, and both were shuffled and placed in 2 locked containers that remained with the independent administrator throughout the duration of the study. As each participant was recruited into the study, the next envelope was opened by the independent administrator who notified the researchers. All assessors were blinded to randomization but not to the study’s objectives.
All participants received standard care. On average, this consisted of a mean 6 occasions of service, for a mean 31.5 minutes of occupational therapy and physiotherapy, during a mean 11.5 days as an inpatient of an acute hospital. 26 The intensive-training group received an additional 30 hours of task-specific UL training during the first month poststroke, applied 2 hours a day, 5 days a week, for 3 weeks. It was prescribed by the “treating” therapist and applied by the therapists or by a therapy assistant supervised by the prescribing therapists. The intervention for both groups was based on the motor relearning principles using repetitive, task-specific training. 27 The only between-group difference was the intervention group receiving more face-to-face therapy than the standard-care group. Irrespective of groups, intervention was individualized with patients starting at different levels and progressing at different rates. Therapy was designed to challenge at a level appropriate to the motor capacity of the individual. The finger tapping task was not included as part of the task-specific training. It was only rehearsed in the moments prior to each participant’s imaging sequence. All participants were discharged during the 3-week intervention period, but for those in the intensive-training group, the additional task-specific UL training continued irrespective of discharge destination.
Clinical and imaging measures were collected at 3 time points poststroke: 1 week (baseline), 1 month (postintervention), and 3 months (follow-up). The baseline assessments were acquired before randomization. The clinical assessments were conducted by therapists with more than 2 years of neurological experience. To ensure consistency, therapists were trained in administering the assessments using scripted protocols to maximize reproducibility and consistency. All demographic data were extracted from the patients’ medical records. Infarct volumes were measured using commercial software MIStar (Apollo Medical Imaging Technology, Melbourne, Victoria, Australia) at the baseline imaging time point using an automated threshold-based technique. 28 All data were encoded and either password protected or stored under lock and key.
Study Procedures and Outcomes
The primary outcome was change in brain activation as measured using fMRI, an in vivo, noninvasive, neuroimaging technique that is based on blood oxygen–level dependence (BOLD; dependent variable). 20 Task-related functional imaging sequences were acquired during an alternating finger-tapping and rest paradigm. This was presented in a blocked design comprising 30 s of rest followed by 30 s of finger tapping. The rest-tap cycles of 60 s acquired over 20 consecutive whole-brain echo-planar images (EPIs) were repeated 4 times in alternate order for each UL over 2 consecutive imaging sequences. Therefore, for each participant, an anatomical image and 160 EPIs (80 tap/rest/left; 80 tap/rest/right images) were acquired for each of the 3 poststroke imaging sessions. The imaging technicians instructed participants via headphones as to when to start and stop tapping their finger. During the finger-tapping cycles, observational records were kept of the number of finger taps performed per cycle and any extraneous participant movement. During all the imaging sequences, participants lay in a supine position with their eyes open. Both ULs were supported in splints designed to limit movement of the wrist and nontapping fingers and thumb.
The secondary outcomes were UL motor function and the impact of stroke. These were measured using the UL-MAS and the modified Rankin Scale 29 (mRS), respectively. The UL-MAS4,24 is scored from 0 to 18, with a higher score indicating better UL function. The study protocol followed specified instructions, 24 and scoring was nonhierarchical, as recommended.30,31 If a patient achieves a score of 18, it means that they have the dexterity and speed to write legibly and are able to participate in most activities that involve the UL. In terms of severity, Rehme et al 18 define this as mild motor impairment if the UL-MAS score is 14 to 18 points, moderate if it is 3 to 13, and severe if the score is 1 to 2. The UL-MAS has been validated in stroke cohorts and has high interrater and intrarater reliability. 32 The mRS 33 is scored 0 to 6, with a higher score indicating a higher degree of disability.
Image Data Acquisition and Processing
MR images were acquired for each participant at 1 week, 1 month, and 3 months poststroke using a 1.5 T Magnetom Siemens Avanto (Erlangen, Germany) MRI scanner. Anatomical images of each participant’s brain were acquired using a high-resolution, T1-weighted, spin-echo imaging sequence. The echo delay time was 4.94 ms, the repetition time was 11 ms, and the flip angle was 15°; the matrix was 256 × 256; and the field of view was 256 mm. This provided 176 sagittal slices at 1 mm3 resolution. Images were acquired using an EPI sequence. The echo delay time was 50 ms, the repetition time was 3080 ms, the matrix was 64 × 64, and the field of view was 220 mm. This provided 36 axial images at a resolution of 3.4 × 3.4 × 3 mm, with a 0.8-mm interslice gap.
All imaging data were preprocessed and analyzed using Statistical Parametric Mapping (SPM) 34 version 5 and Matlab 35 version R2011b software packages. For each of the 3 sessions, 1 T1 and 160 EPIs were acquired in Dicom format and later converted to Analyse format for processing in SPM5. For each session, the EPIs were acquired in 2 imaging blocks of 80 images, where the participants undertook either left or right hand rest-tap cycles of 30 s and where the order of left versus right finger tapping was counterbalanced across participants.
To standardize EPI data across participants, images for participants with right hemispheric lesions (n = 12) were reversed, so that for all participants, the “left” hemisphere represented the lesioned hemisphere and the “right” hemisphere represented the contralesional hemisphere. To facilitate analyses across participants, all the T1 images and EPIs for each participant were transformed into standardized Montréal Neurological Institute (MNI) stereotaxic space. To achieve this, the 3 T1 images for each imaging session were realigned and coregistered, and a mean T1 image was derived and transformed into MNI space using a symmetrically modified version of the original SPM/MNI 305 MRI template (“avg305T1.nii”) created using left-right symmetrical tissue classification priors according to the procedure used by Didelot et al. 36
The EPIs were then realigned and coregistered to the first functional image acquired in the first session (1 week) and an across-sessions mean-EPI was derived. This individual across-sessions mean EPI was then coregistered and normalized to the symmetrical mean-T1 image, and the resulting normalization parameters were applied to each individual’s EPI data over all 3 imaging sessions. The integrity of the EPI data was then assessed using the TSDiffAna 37 procedure to identify any within-session changes in mean EPI intensity between successive EPIs greater than 1 standard deviation. No such artifacts were identified, nor was any within-session movement identified that was greater than 3 mm for both shifts and rotations in the 3 image planes (x, y, and z; supplement A). To account for the variance or artifact associated with the realignment of EPIs within and across sessions, the realignment parameters (x, y, and z shifts and rotations) for each session were included as regressors in the subsequent SPM5 first-level general linear model analyses, as described below.
Image Data Analysis
A within-subject or first-level analysis was carried out for each individual participant’s normalized EPI using canonical hemodynamic response functions in SPM to convolve the EPI series data to the intensive-training study design. For each participant, 3 SPM t-contrast images were created to compare ipsilesional finger tap versus rest, contralesional finger tap versus rest, and both finger taps versus rest (combined ipsilesional and contralesional) for each of the 3 sessions for which imaging data were collected. An additional 3 equivalent contrasts were also created across all 3 sessions (supplement C). This resulted in 12 SPM t-contrasts per participant.
Data Analysis: Differences in Brain Activation
To investigate between-group results, the 12 SPM t-contrast images for each participant’s first-level, fixed-effects contrast images (threshold P < .001 uncorrected) were submitted to second-level, random-effects analyses using only the stroke-affected UL contrasts for the conditions of: intensive-training versus standard care. The a priori P value was set at <.001 (uncorrected), and a correction was applied using a family-wise error cluster threshold set at P < .05. Full factorial analysis identified the clusters of voxel-based activity combined across all imaging sessions (effect of group) and at each of the 3 time points (effect of time). Flexible factorial analysis identified the interaction effect of Group × Time. The voxel size was set at 3 × 3 × 3 mm3. Locations of significant activation were interpreted using the Anatomy Toolbox in SPM 2005. 38 To correct for participant heterogeneity, a small volume correction was applied using a template created using the method of Eickhoff et al 38 and the MarsBaR software package. The template included the bilateral Brodmann areas 1, 2, 3, 4, and 6 (includes the premotor cortex, the supplementary motor area, and primary sensorimotor cortex) and the cingulate area and cerebellum.
To investigate region-based activity in putative motor regions, a post hoc analysis was conducted to identify changes at specific time points. The a priori selection of regions was based on the sensorimotor areas identified in previous research.3,12,21 Extent of activation data was used to analyze the percentage of active voxels at 1 week (baseline), and over time in the 1-month and 3-month poststroke time periods. Regions were defined using the cytoarchitectural probability atlas from the Anatomy Toolbox in SPM (version 17). 38 The STATA 39 version 11.0 software package was used to investigate differences at baseline and the change relative to baseline in the first month and in the first 3 months.
Data Analysis: Differences in UL Function
Between-group differences were investigated using the UL-MAS and mRS data and the STATA 39 version 11.0 software package. The t-test (confidence interval [CI] = 95%) was used to investigate between-group differences in the mean age, disability (mRS), and frequency of finger tapping at 1 week. General linear mixed models, with a random effect for each participant, were used to determine if there were differences between the standard-care and intensive-training groups between 1 week and 3 months poststroke. The models included the independent variables of time and group and an interaction term between time and group. The P value of the interaction term was used to determine significance. The interaction term was dropped from the models to investigate change across time points within the whole cohort. Differences in the variation between groups were investigated using the UL-MAS data and Levene’s 40 robust test to compare the standard deviations in the standard-care group and the intensive-training group.
Results
The study recruited 23 participants. Of these, 12 were randomized to the standard-care group and had a mean age of 69.3 years, and 11 were randomized to the intensive-training group and had a mean age of 61.7 years (Table 1). At 1 week poststroke, there was no significant between-group difference in age (t = 1.57; P = .13; CI = 95%), disability (mRS; t = 0.07; P = .93; CI = 95%), UL function (UL-MAS; t = −0.98; P = .34; CI = 95%), or frequency of finger tapping (t = 0.56; P = .58; CI = 95%; Table 2). All participants were admitted to a stroke unit and remained in the hospital for a mean of 8.3 days; 7 participants (30.4%) were discharged directly home, 15 (65.2%) received inpatient rehabilitation, and 15 received outpatient rehabilitation.
Participants (n = 23) Group, Stroke Subtype, Gender, and Age. a
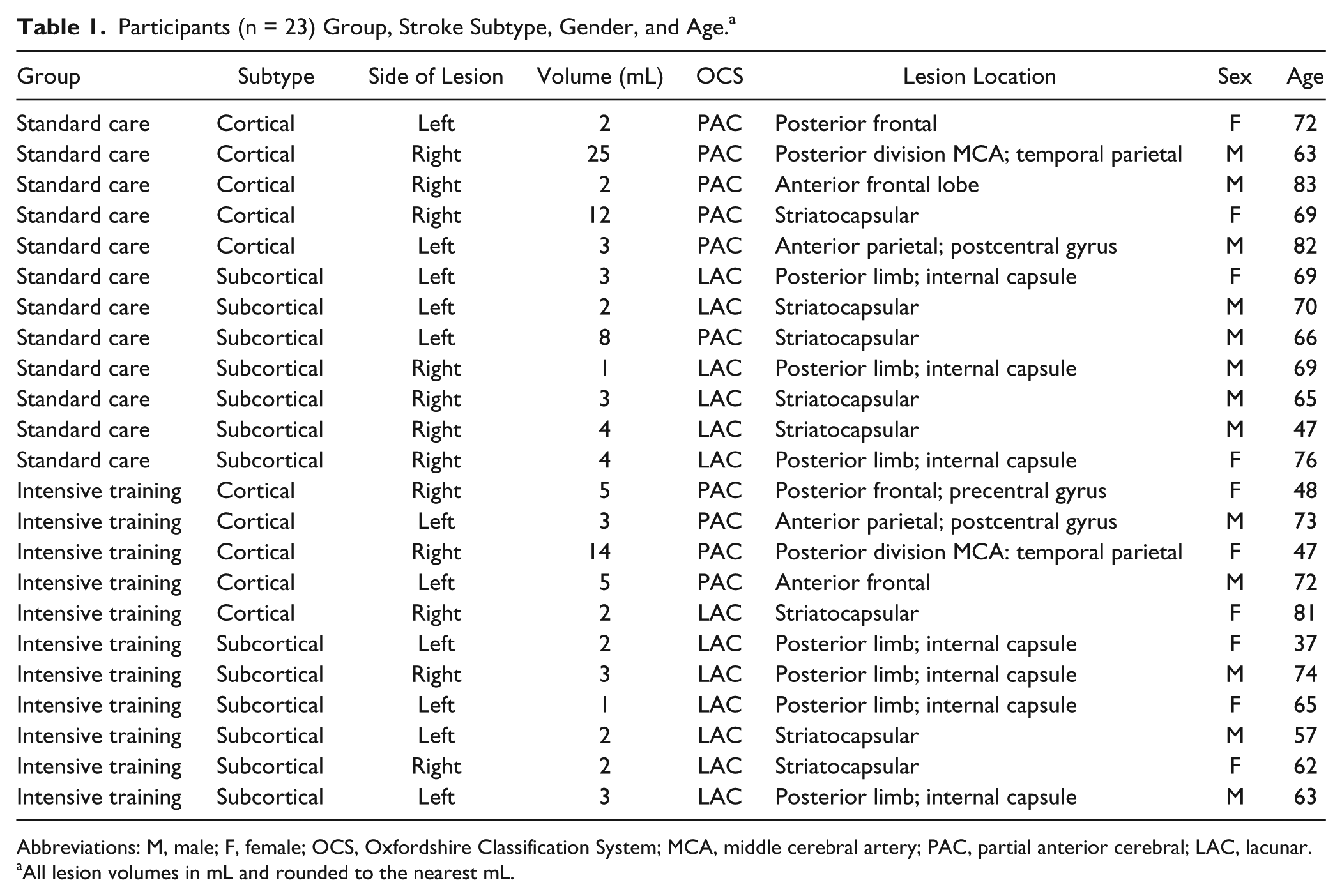
Abbreviations: M, male; F, female; OCS, Oxfordshire Classification System; MCA, middle cerebral artery; PAC, partial anterior cerebral; LAC, lacunar.
All lesion volumes in mL and rounded to the nearest mL.
Demographic and Baseline Characteristics of Participants.
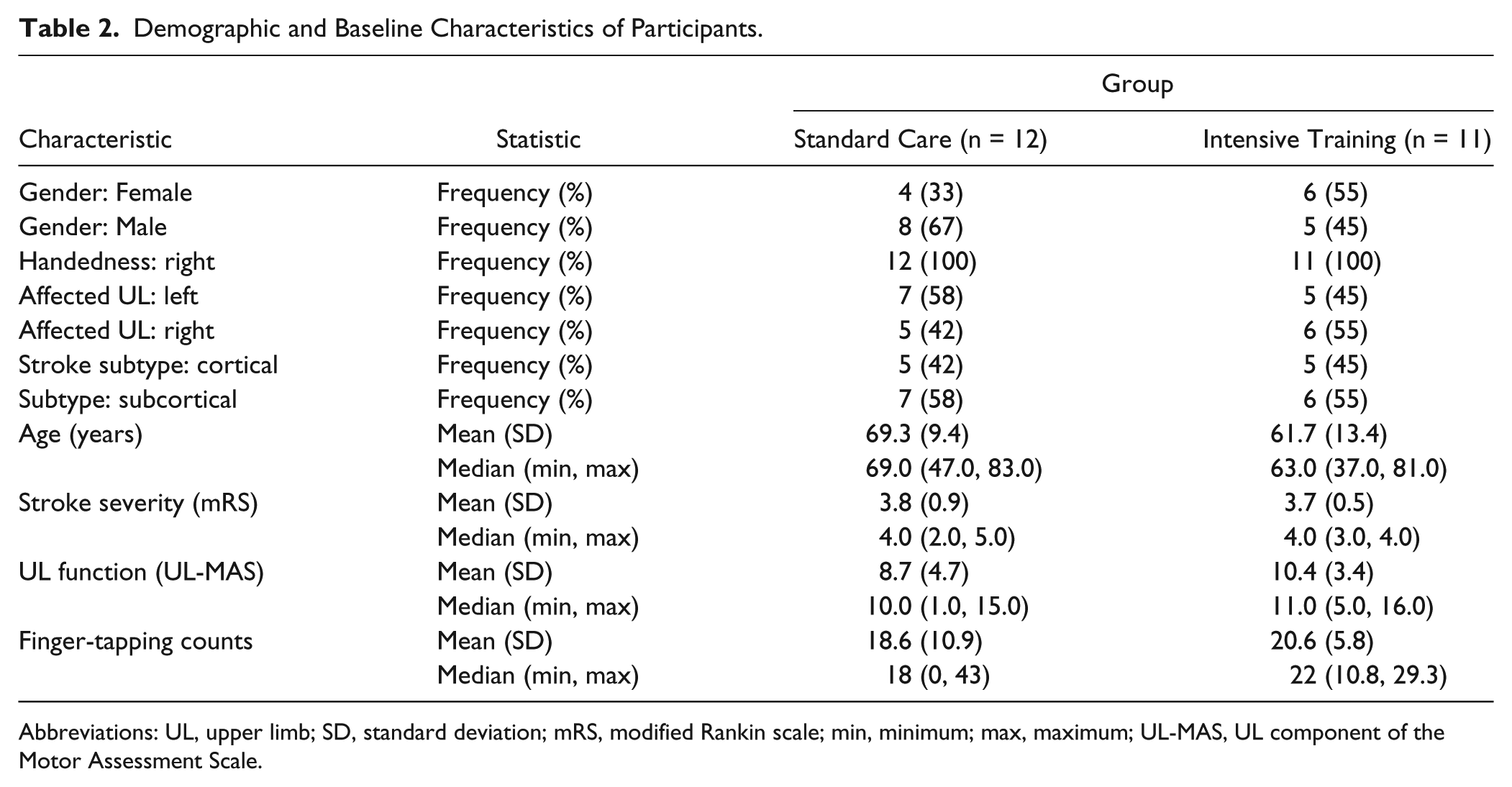
Abbreviations: UL, upper limb; SD, standard deviation; mRS, modified Rankin scale; min, minimum; max, maximum; UL-MAS, UL component of the Motor Assessment Scale.
Differences in Brain Activation
Clusters of brain activation were associated with the affected UL in the ipsilesional hemisphere across all 3 time points. When compared with the standard-care group, the intensive-training group had significant clusters of greater activity in the ipsilesional anterior cingulate and supplementary motor areas across these time points. Figure 2 reports results from the affected UL, finger-tapping versus rest, across all 3 time points combined. The voxel-wise cluster-forming significance threshold is P < .001, and the family-wise error cluster threshold is P < .05. This analysis showed a positive effect of group, but no significant result was found to be associated with either time, or Group × Time. At 3 months poststroke, the intensive-training group demonstrated trends toward greater activity in the ipsilesional anterior cingulate and supplementary motor areas (Table 3).
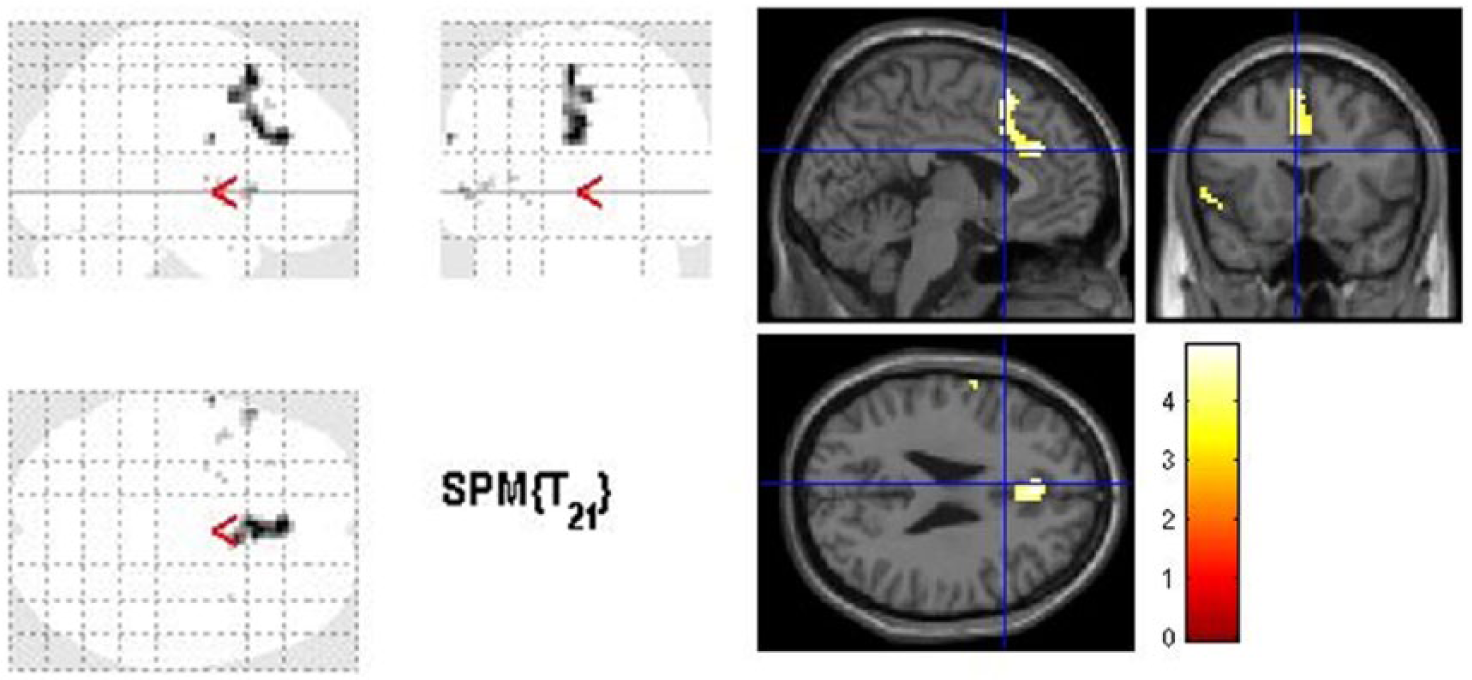
Sagittal, coronal, and axial slices showing clusters of brain activation in the ipsilesional anterior cingulate and supplementary motor areas that are significantly greater in the intensive-training group when compared with the standard-care group. The contrasts analyzed were those reporting the affected finger tapping versus rest, across all 3 sessions. The SPM t-image is displayed at a voxel-wise significance threshold of P < .001 uncorrected and a family-wise error cluster threshold at P < .05. The data have not been smoothed.
Brain Activation Associated With the Affected Upper Limb Following a Small-Volume Correction. a
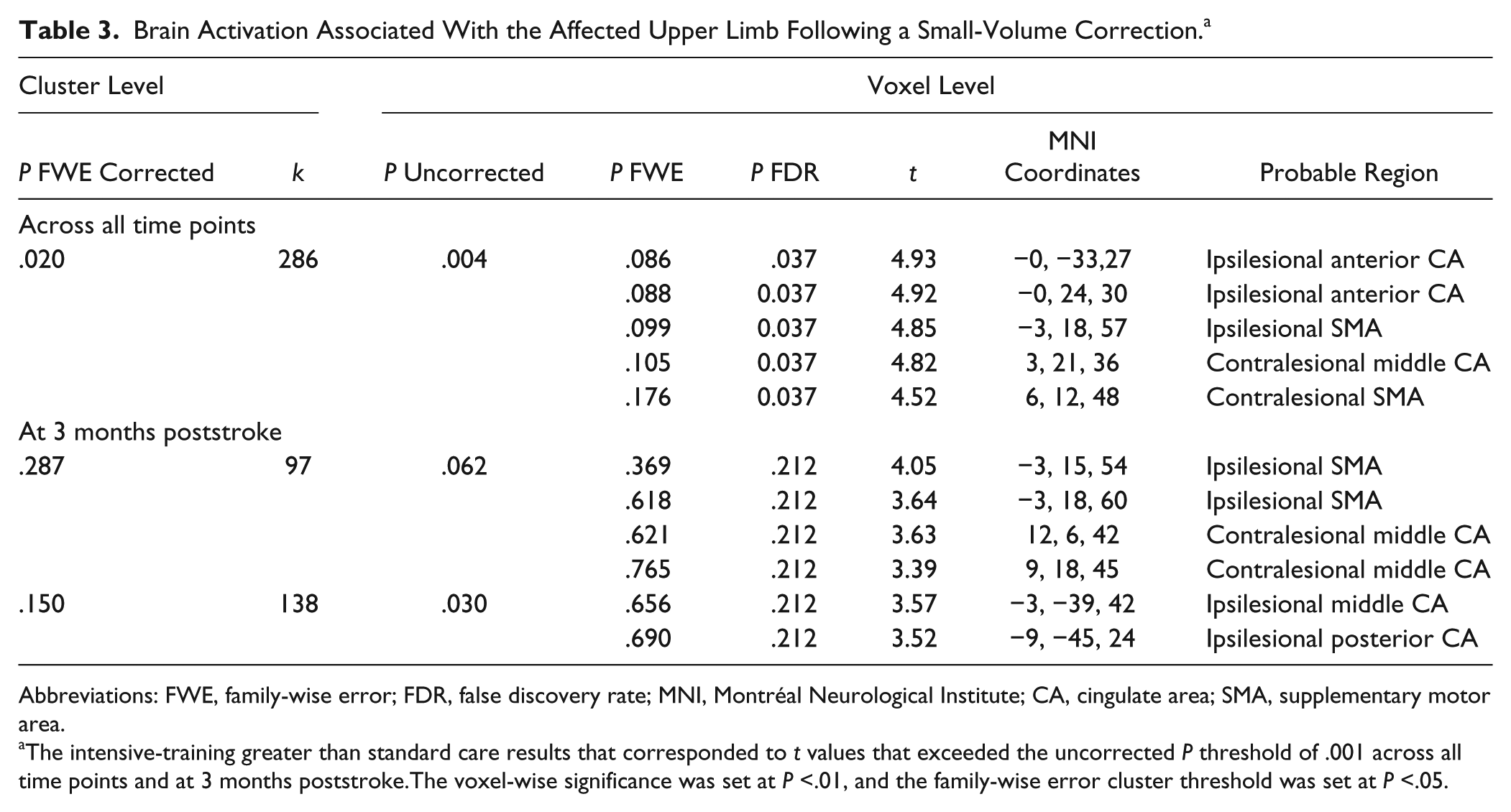
Abbreviations: FWE, family-wise error; FDR, false discovery rate; MNI, Montréal Neurological Institute; CA, cingulate area; SMA, supplementary motor area.
The intensive-training greater than standard care results that corresponded to t values that exceeded the uncorrected P threshold of .001 across all time points and at 3 months poststroke.The voxel-wise significance was set at P <.01, and the family-wise error cluster threshold was set at P <.05.
Region-Based Activity in Putative Motor Regions
No differences were found at baseline when comparing the extent of activation between the intensive-training and the standard-care group in the bilateral supplementary motor area, the primary sensorimotor area, the cingulate area, or the cerebellum. When investigating change over time relative to baseline, the extent of activation in the intensive-training group reduced significantly more in the contralesional cerebellum (rank sum P = .02) in the first 3 months poststroke (Table 4) when compared with the standard-care group.
Change in Percentage of Active Voxels in Defined Regions of Interest at 1 Month and 3 Months Poststroke Relative to Baseline: Differences Between the Standard-Care and Intensive-Training Groups. a
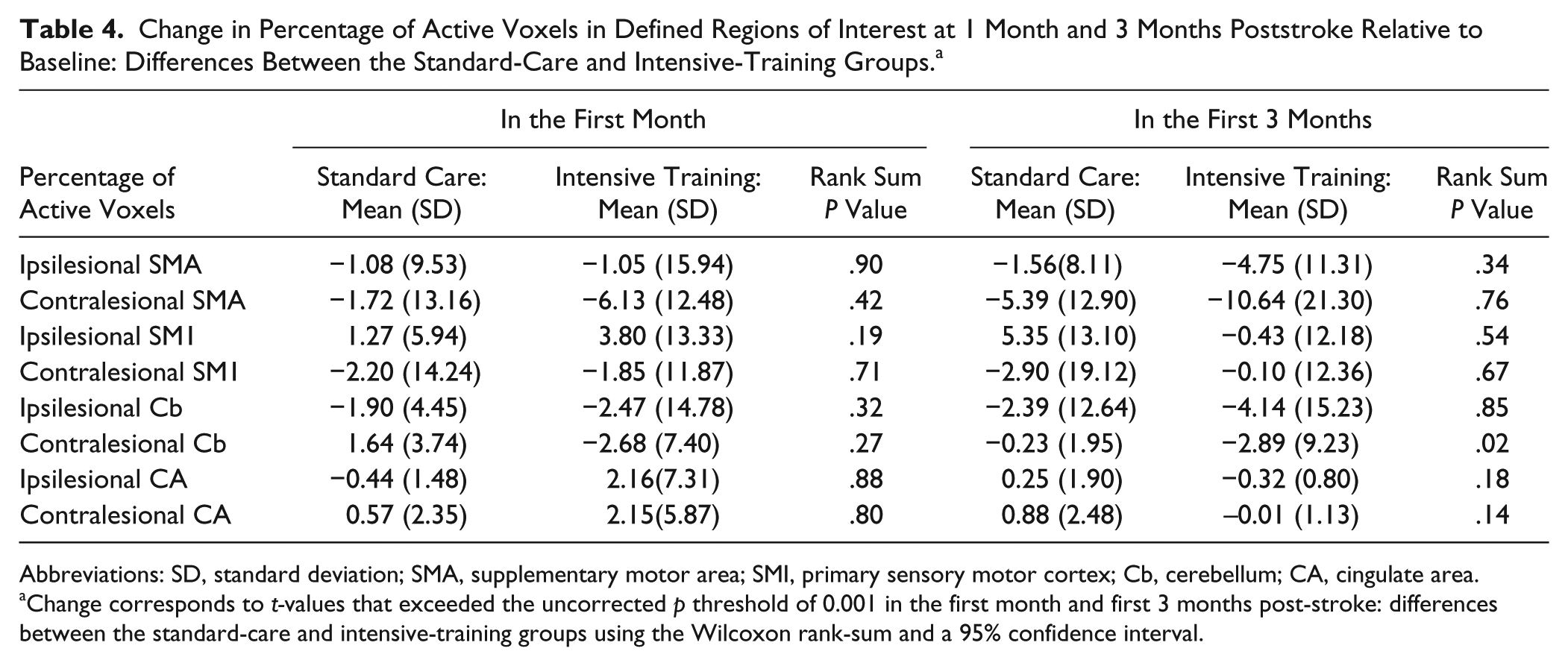
Abbreviations: SD, standard deviation; SMA, supplementary motor area; SMI, primary sensory motor cortex; Cb, cerebellum; CA, cingulate area.
Change corresponds to t-values that exceeded the uncorrected p threshold of 0.001 in the first month and first 3 months post-stroke: differences between the standard-care and intensive-training groups using the Wilcoxon rank-sum and a 95% confidence interval.
Differences in UL Function
In the first 3 months, all participants showed significant improvement in the motor function of their affected UL and in their degree of disability: UL-MAS, P < .0001; mRS, P < .0002 (supplement B). Additionally, the level of recovery achieved at 1 month poststroke was maintained at 3 months. There were no significant between-group differences in change over time in the clinical outcome scores. Although there were no significant differences in mean recovery of UL function between the 2 groups, the intensive-training group had a smaller deviation from mean recovery at 1 month (Pr>F0 = 0.017) and 3 months (Pr>F = 0.006), indicating more consistent and predictable improvement in motor outcomes.
In summary, across the first 3 months poststroke, when compared with those in the standard-care group, the intensive-training group had increased activity in the ipsilesional anterior cingulate and supplementary motor areas, and they experienced a greater reduction in the percentage of active voxels in the contralesional cerebellum. All participants experienced significant improvement in their degree of disability and in the motor function of their affected UL, but the UL improvement in those recruited to the intensive-training group was more consistent.
Discussion
This preliminary study is the first to show that more intensive rehabilitation (task-specific UL training) in the first month poststroke is associated with increased brain activation in putative motor and attention areas. Intensive UL training also led to more consistent recovery of clinical UL function. This study is original in its use of RCT methodology and involvement of an acute stroke sample, tested at 1 week and trained within the first month poststroke. The findings suggest that increased activation of ipsilesional supplementary motor and anterior cingulate areas with more intensive training poststroke may contribute to more consistent recovery of the affected UL after stroke across individuals. Increased activation of these regions has been associated with early motor learning and attention, 11 consistent with the early intensive motor training provided. Involvement of these areas is also consistent with learning-dependent plasticity.3,12 This study is also the first to report a potential association between more intensive UL training in the first month poststroke and more predictable and consistent motor recovery. It demonstrated that this UL training regimen is effective, safe, and clinically feasible. Finally, this study found that changes in brain activation were evident at the end of the 3-month follow-up period poststroke.
Differences in Brain Activation Patterns
Previous studies have shown an association between improved clinical recovery and brain activation but not in a randomized trial.3,12 This is the first RCT of more intensive, early UL therapy in stroke patients that used fMRI measures of brain activation as an outcome alongside clinical measures. The finding that increased brain activation in motor-related and attention areas was associated with recovery of UL function in those that received increased intensity of training is clinically relevant. There is increasing agreement that the intensity of intervention in the first month poststroke influences outcomes,2,41 and this is also the time when most stroke patients are engaged in rehabilitation. We argue that the increased intensity allows increased opportunity for attentive, task-specific training of motor function and that this process would be expected to include brain regions involved with motor learning and attention—for example, supplementary motor areas and cingulate regions,11,18 consistent with our findings and with learning-dependent plasticity. 20 However, the awareness that the activation seen may be unrelated to recovery is supported by the fact that there seems to be no relationship between functional outcome and increased activity in the brain, except that the group receiving the additional hours of therapy performed finger tapping more times, with less effort, and more consistently than the standard-care group.
The Significance of Region-Based Activation
Evidence increasingly identifies the ipsilesional supplementary motor area as important to UL recovery in the first 3 months poststroke 18 and, to a lesser degree, the ipsilesional anterior cingulate area.9,42,43 The current study’s findings support the theory that stroke recovery may be associated with the recruitment of spared sensorimotor3,12,44 and attention45-47 regions. However, the authors acknowledge that other areas of the brain may also be making a functionally significant contribution but that this activation may be present in both groups or may be subthreshold. No single pattern of neuroplastic change is observed during recovery; rather, they are distinctly different in different regions and seem to depend on deficits caused by an initial lesion as well as training interventions.20,48
The finding of an association between activation of the ipsilesional anterior cingulate area and more intensive, task-specific UL training is clinically significant. It suggests that this rehabilitation paradigm may require patients to pay more attention to the task.46,49 The anterior cingulate area has been shown to be associated with effortful tasks, error detection, attention, and learning. 45 The potential involvement of the cerebellum may highlight the interconnectedness between the cortex and cerebellum—a phenomenon yet to be fully understood.46,50 Evidence indicates an increased reliance on the cerebellum with transfer of motor learning.51,52 The fact that no significant differences were found in areas where differences were anticipated—for example, the primary sensorimotor cortex—is noteworthy because the location of most of the lesions in this study were in a sensorimotor region or in directly connected subcortical regions. To find that the only between-group differences were in the ipsilesional supplementary motor area and the ipsilesional anterior cingulate area, a region linked with effortful tasks and early learning45-47,53,54 is important and may be linked with the additional motor learning involved at this early time poststroke. Furthermore, evidence of significant changes in activity in the cerebellar and anterior cingulate regions provide a basis for alternative hypotheses related to stroke recovery; that is, that the differences between the intensive-training and standard-care groups may reflect differences in the effort, error checking, and attention that patients apply when undertaking motor tasks and/or that these regions are differentially accessed during the process of intensive motor training. Further investigation is warranted.
Intensive UL Training in the First Month
This study found that all participants experienced improvement in the first month poststroke, which was maintained at 3 months. It also demonstrated that patients in the first month tolerated an additional 30 hours of UL training and that this dose was effective, safe, clinically feasible, and associated with more consistent improvement in motor function. Thus, this study provides evidence to support current recommendations in clinical practice.22,55 Because this study was undertaken in the first month poststroke, it was anticipated that both groups would experience recovery because of the combination of natural history and poststroke rehabilitation. 2 However, the finding that more-intensive, task-specific UL training resulted in more consistent, and predictable, improvement in UL function is novel and clinically important. Theories of learning indicate that performance becomes more consistent and predictable when well learnt.56,57 It is possible that the additional training provided to those in the experimental group led to more stable improved performance, not only in individuals but in the group as a whole. Even though there is consensus that patients with stroke should receive early, more-intensive training,6,7,58,59 questions remain about how much additional intervention is required to ensure more consistent outcomes; for example, Horn et al 8 found an association between more-intensive, early UL intervention and higher scores in functional independence, whereas Rodgers et al 6 found no differences. However, the current study was unable to show that increasing intensity improved the overall magnitude of motor recovery. This is most likely because of low participant numbers and a known ceiling effect in the UL-MAS. 31 To find an association between more predictable recovery and intensive UL training suggests that testing for between-group variance may be a more sensitive outcome than purely measuring mean between-group changes only.
Neurobiological Change Is Evident After Physical Recovery
The finding of persistent increases in brain activation in the intensive-training group at 3 months, beyond the first period when most of the clinical recovery occurred, is noteworthy. Although increased activation at 1 month paralleled clinical recovery, the persistence of increased activation is highly likely to reflect cortical reorganization. Additionally, it appears that such plasticity may have contributed to improved clinical UL function. These findings suggest that it may be possible to use fMRI as a measure of plasticity following stroke, and if this were the case, it may be a useful biological measure of rehabilitation interventions.
Strengths and Limitations
This study’s strengths lie in its RCT design, its recruitment of patients during their acute admission poststroke, and when compared with other fMRI studies, the relatively high (n = 23) participant numbers. It also recruited similar numbers of participants with left and right hemispheric stroke and cortical and subcortical stroke. However, the sample size is still limited and likely hindered our ability to find between-group differences with the traditional standard clinical outcomes. This was further challenged by the ceiling effect in the UL-MAS. Both issues should be taken into consideration in future UL research that recruits participants with mild to moderate stroke. We were also not able to specify the number of stroke patients in the acute stroke unit who did and did not meet criteria, thus affecting generalizability. We did not include a no-intervention arm for ethical reasons. Because the finger-tapping task was not controlled for, it was not possible to identify whether the increased activity observed in the anterior cingulate area was a by-product that arose from finger-tapping performance or an increased effort during the motor task in the scanner and may be unrelated to recovery. It was also not possible to test for possible between-group differences in the way finger tapping was performed by patients. Nonetheless, the increased activity was differentially observed in the group receiving increased intensity of UL training, despite similar levels of impairment and recovery across groups. Because most participants will demonstrate early recovery, more-sensitive measures of between-group variances in improvement, as used in the current study, may need to be considered as outcomes.
Conclusion
This is the first RCT of first-ischemic-stroke patients with an affected UL that compared early, intensive, task-specific UL training with standard care. The study found that those who received intensive training had more activation in the anterior cingulate and the supplementary motor areas and experienced more predictable and consistent UL recovery. Our findings highlight the additional involvement of secondary motor and attention regions associated with more intensive involvement of motor training early poststroke. Involvement of these areas is consistent with learning-based plasticity. Increasing the dose of UL training by an additional 30 hours in the first month poststroke was clinically safe and feasible. These findings have considerable translational value because they contribute to the knowledge supporting an evidence-based approach to UL recovery poststroke and provide direct evidence that more-intensive rehabilitation contributes to neuroplasticity. In particular, we provide novel evidence of neural plastic changes and brain regions associated with intensive motor rehabilitation within the period of assumed spontaneous recovery early poststroke.
Footnotes
Acknowledgements
The authors would like to acknowledge the participation of stroke survivors in the study and the support of the John Hunter hospital’s Acute Stroke Research team and Imaging Department.
Authors’ Note
The authors have no disclosures of any commercial interest relevant to the subject of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Heart Foundation (Grant Number GO189945 to MWP and IJH); the National Stroke Foundation (Grant Number PP08S 4082 to IJH); the Hunter Medical Research Institute (Grant Number G0187851 to IJH); and an Australian Research Council Future Fellowship (number FT0992299 to LMC and FT0991128 to MWP) and the Victorian Government’s Operational Infrastructure Support Program (to LMC). The funding sources had no role in conduct of the study or writing of the report.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
