Abstract
Background
Low-frequency repetitive transcranial magnetic stimulation (rTMS) over the contralesional motor cortex (M1) has demonstrated beneficial effects on motor recovery, but evidence among patients with subacute stroke is lacking. We aimed to investigate whether 1-Hz rTMS over the contralesional M1 versus sham rTMS could improve arm function in patients with subacute ischemic stroke when combined with rehabilitative motor training.
Methods
In total, 77 patients who were within 90 days after their first-ever ischemic stroke were enrolled and randomly allocated to either real (n = 40) or sham rTMS (n = 37). We delivered 1-Hz 30-minute active or sham rTMS before each daily 30-minute occupational therapy sessions over a 2-week period. The primary endpoint was changes in the Box and Block Test (BBT) score immediately after the end of treatment (EOT). Secondary analyses assessed changes in Fugl-Meyer assessment, Finger Tapping Test (FTT), Brunnstrom stage, and grip strength.
Clinical Trial Registration
ClinialTrials.gov (NCT02082015).
Results
Changes in BBT immediately after the end of treatment did not differ significantly between the 2 groups (
Conclusion
The effects of real and sham rTMS did not differ significantly among patients within 3 months poststroke. The location of stroke lesions should be considered for future clinical trials.
Keywords
Introduction
Upper limb hemiparesis is one of the most common impairments after stroke and is associated with limited activities of daily living and restricted participation. 1 Therefore, adequate functional recovery of upper limbs is critical. Nevertheless, effective rehabilitation modalities are lacking, and novel therapeutic approaches that can be combined with conventional rehabilitation therapy are required. 2
Repetitive transcranial magnetic stimulation (rTMS) has been actively studied for upper limb recovery after stroke as a novel modality for neuromodulation. 3 Among various rTMS protocols, low-frequency rTMS over the contralesional primary motor cortex (M1) has been applied in attempts to normalize imbalanced interhemispheric inhibition (IHI) by suppressing the overinhibition of contralesional M1 toward ipsilesional M1. 4 Low-frequency rTMS is preferred to high-frequency rTMS due to its low risk of seizure and minimal effects on motor behavior in healthy corticospinal systems.5,6 Therefore, most rTMS studies on motor recovery after stroke have applied low-frequency (1 Hz) rTMS over the contralesional M1. 7 One meta-analysis has suggested that low-frequency rTMS may be superior to high-frequency rTMS for motor recovery after stroke. 8 However, the sample sizes in previous clinical trials using low-frequency rTMS were small, especially among patients with acute/subacute stroke. 9 Considering the optimal time window for neuroplasticity and recovery after stroke 10 and possible enhanced effects at acute/subacute stages compared to those at chronic stages, 7 sufficiently powered randomized controlled trials of low-frequency rTMS in patients with acute/subacute stroke are required. 11
Therefore, the purpose of this study was to assess whether applying low-frequency rTMS over the contralesional M1 combined with task-oriented upper limb rehabilitative therapy in patients with subacute stroke elicited greater motor recovery than that for upper limb rehabilitative therapy alone. We recruited patients with Brunnstrom hand stage rating of 3 to 5, who were capable of performing a hand motion from mass grasp with no finger extension to lateral or palmar prehension with voluntary finger extension in various ranges of motion, but were unable to perform the isolated individual finger movements, based on the potential predominant role of IHI in patients with mild motor impairments. 12 Our hypothesis was that low-frequency rTMS over the contralesional M1 would elicit greater upper limb motor recovery than would sham rTMS among patients with subacute stroke (≤90 days of stroke onset) with mild to moderate upper limb motor impairment.
Methods
Study Design and Randomization
This study used a randomized, sham-controlled, double-blind, parallel-group design to test the effects of rTMS on motor recovery. The trial was conducted at 3 university hospitals in South Korea with approvals from the Institutional Review Boards of the Seoul National University Bundang Hospital (No. E-1308/214-002, approved January 2014), Seoul National University Hospital (No. D-1508-171-701, approved October 2015), and Dongguk University Ilsan Hospital (No. 2013-01-075, approved September 2013). Eligible participants were randomly allocated (1:1) via sealed randomization envelopes to receive 1 Hz rTMS over the contralesional M1 (intervention group) or sham rTMS (control group). Real or sham rTMS was applied for 30 minutes per day over 10 days, and 30-minute task-based occupational therapy for upper limb recovery was provided within 10 minutes after the rTMS session (Figure 1). Throughout the 10 days of intervention, each patient received an additional daily 30-minute occupational therapy session and 2 sessions of physical therapy for gait recovery in addition to the protocol session (real or sham rTMS + 30-minute occupational therapy).
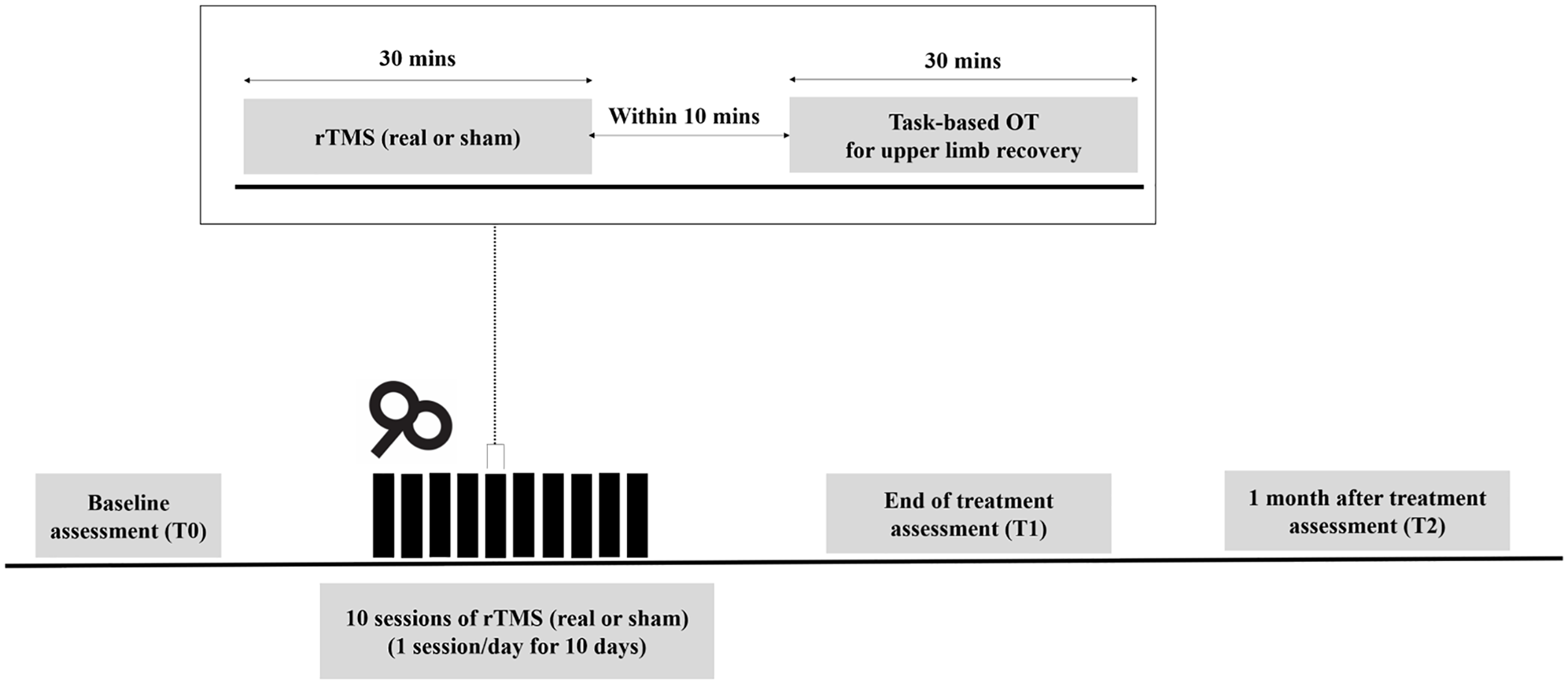
Experimental design. Patients received 30-minute daily real or sham repetitive transcranial magnetic stimulations (rTMS) over a period of 10 days. Task-based occupational therapy (OT) of 30 minutes for upper limb recovery was applied within 10 minutes after rTMS session. Assessments were performed prior to treatment (T0), immediately (T1), and 1 month (T2) after the last session of rTMS.
Protocol
Patients admitted to the department of rehabilitation medicine in 3 hospitals between June 2014 and January 2018 were enrolled (Clinical Trial Registry: NCT02082015). Patients were eligible for inclusion in the study if they displayed unilateral upper limb hemiparesis in Brunnstrom hand stage rating 13 of 3 to 5 within 90 days after first-ever ischemic stroke onset confirmed by magnetic resonance imaging (MRI) and were aged between 20 and 80 years. Exclusion criteria were hemorrhagic or recurrent stroke, previous history of traumatic brain injury, seizure or cerebrovascular surgery, need for intensive care due to severe complications of stroke, metallic materials in the body (eg, pacemakers, cochlear implants, aneurysm clips), pregnant or lactating women, those who were likely to become pregnant but did not agree to appropriate contraception during the trial, skin lesions around the contralesional M1 which interfered with rTMS, or those who could not regularly receive occupational or physical therapy. All participants received detailed information regarding the trial and provided their written consent. The research protocol was conducted in accordance with the regulatory standards of Good Clinical Practice and the Declaration of Helsinki.
Assessment of Study Outcomes
Primary and secondary outcomes were assessed by nontreating trained occupational therapists who were blind to the intervention assignment. Patients were assessed at baseline (T0), immediately after the end of treatment (EOT, T1), and 1 month after EOT (T2). Primary outcome was change in the Box and Block Test (BBT) results between baseline and immediately after EOT. Patients were instructed to move as many blocks as possible, one at a time, from one box to another for 60 seconds after a 15-second trial period. 14 The results of BBT were calculated by counting the number of blocks successfully moved. Although the motor domains of Fugl-Meyer Assessment (FMA) score is commonly used in clinical trials to investigate motor recovery, the responsiveness to the treatment in patients with stroke with mild upper limb hemiparesis is known to be limited due to its ceiling effect. 15 Given that participants were expected to move their hemiparetic hands partially, considering the selection criteria of subjects in this study, the low responsiveness and ceiling effect of FMA was a concern. Therefore, we selected the BBT as a primary outcome measure. BBT shows high interrater and test-retest reliability and correlates well with other commonly used outcome measures such as FMA or Actional Research Arm Test (ARAT). 16 BBT is also appropriate for evaluating dexterous function, the target for low-frequency rTMS in this study, and the ceiling effect is less of a concern.16,17
Secondary outcomes were changes in the following measures at each posttreatment time point (T1 and T2) compared to that at baseline (T0): BBT; upper extremity FMA score; Finger Tapping Test (FTT); hand grip, pinch grip, lateral prehension, and three jaw chuck strength; Brunnstrom hand and arm stage ratings 13 ; modified Ashworth Scale (MAS) in the elbow, wrist, and finger flexors; and Korean version of Modified Barthel Index (K-MBI). FTT was performed using the Digital Finger Tapping Test (Sybu Data). Patients were instructed to tap a square on an iPad screen with their index finger as fast as possible for 10 seconds, 3 times. The average number of taps of 3 trials was calculated for further analysis. Hand grip strength was measured using the Jamar hydraulic hand dynamometer (Sammons Preston), and the average of 3 trials was used for further analysis. Pinch grip, lateral prehension, and three jaw chuck strength were measured using the Jamar hydraulic pinch gauge (Sammons Preston), and the average of 3 trials was calculated. For pinch grip, patients were instructed to grasp the pinch meter with the tips of the thumb and index finger. For lateral prehension, the pinch meter was grasped between the pad of the thumb and radial side of the middle phalanx of the index finger. For three jaw chuck, patients positioned the pinch meter between the pads of the thumb, index, and long finger. Brunnstrom staging is a test to qualitatively assess motor recovery after stroke in 6 stages (from 1 [flaccid, no voluntary movement] to 6 [presence of isolated/coordinated joint movement]) based on muscle tone, synergistic pattern, and selective motor control. 13 Spasticity of the flexors in the upper limb was measured using the MAS. 18 All flexor muscles were examined from a fully flexed position within a tolerable range. Basic activities of daily living were measured using K-MBI with a score from 0 (totally dependent) to 100 (independent). 19 All adverse events were assessed during the intervention and follow-up. Expected adverse events included headache, pain in the face or neck, sensory disturbances in the hand, muscle spasm, nasal bleeding, fatigue, seizure, transient slowing or acceleration of thinking, and acute transient hypomania.
Assessment of Other Independent Variables
National Institutes of Health Stroke Scale was assessed at baseline to demonstrate stroke severity. 20 At 1-month follow-up after EOT, the dose of occupational therapy and physical therapy after EOT was investigated by asking the following questions: (1) How often did you receive physical or occupational therapy per week? (2) How many sessions of physical or occupational therapy did you receive per day? (3) How long was the session of physical or occupational therapy? Based on the answers to these questions, patients were categorized as receiving rehabilitation therapies or not, and we estimated the total hours of physical or occupational therapy after EOT. Patients’ MRIs were reviewed by the clinician. Stroke location was categorized as cortical, subcortical (corona radiata, internal capsule, or basal ganglia), cortical/subcortical, and/or brain stem.
rTMS Intervention
Patients were seated comfortably in a chair during the rTMS. The resting motor threshold (RMT) for the first dorsal interosseous muscle of the unaffected side was measured over the contralesional M1. RMT was defined as the minimum stimulation intensity needed to evoke a response of at least 50 µV in at least 5 of 10 consecutive stimulations. 21 In each session, contralesional M1 was stimulated through an 88-mm-diameter figure-of-eight coil powered by ALTMS (Remed, Korea), with a frequency of 1 Hz and intensity of 100% of the patient’s RMT measured in the contralesional M1 for 30 minutes, achieving 1800 stimuli per session. The stimulation intensity and duration were determined based on previous studies of low-frequency rTMS 9 and safety guideline. 6 For the sham rTMS, the coil was positioned perpendicular to the scalp on contralesional M1 with the same intensity and frequency as that in the real rTMS.
Task-Based Occupational Therapy
Task-based occupational therapy was provided and consisted of a standardized shaping training protocol, which was designed to improve the paretic hand function. The therapist applied the manageable tasks from a list of predefined tasks based on Wolf Motor Function Test 22 and Motor Activity Log 23 and provided the patient with explicit feedback according to each patient’s function. The level of difficulty of these activities was increased as the strength and coordination of the paretic hand improved.
Statistical Analysis
Sample size was calculated based on a previously published study that reported superior outcomes in BBT after real rTMS among patients with stroke. 24 To detect a difference of 3.6 in BBT with a standard deviation of 5.3 between the 2 groups (with β = .20 and α = .05), considering the 15% dropout rate, 42 participants in each group were required.
Continuous variables are presented as means ± standard deviations. Categorical variables are presented as frequencies (percentages). To compare the baseline characteristics between the 2 groups, Student’s
For the efficacy analysis, full analysis set (FAS), which included only patients who completed the intervention and primary outcome assessment immediately after EOT, was primarily used. Per-protocol (PP) analysis was additionally conducted for sensitivity analysis. For the primary outcome analysis, Student’s
Results
Participants
Of the 79 volunteers who provided consent to participate during the screening phase, 2 declined to participate before randomization and group allocation, and 77 patients in total were randomized to real rTMS (n = 40) or sham rTMS (n = 37). One participant in the real rTMS group did not receive the intervention due to consent withdrawal. Three in the real rTMS group were lost to follow-up due to consent withdrawal before EOT. At 1 month after EOT, one participant from each group withdrew their consent (Figure 2). In total, 42 participants in each group were the initial number of the enrollment plan, but the follow-up loss for primary outcome (4/77 = 5.2%) was less than the expected, yet the sample size required to achieve the anticipated statistical power (n = 35 in each group) was attained. Therefore, the clinical research organization and department in the Ministry of Food and Drug Safety that monitored this study decided to halt further enrollment following discussion with the principal investigators in each study site.
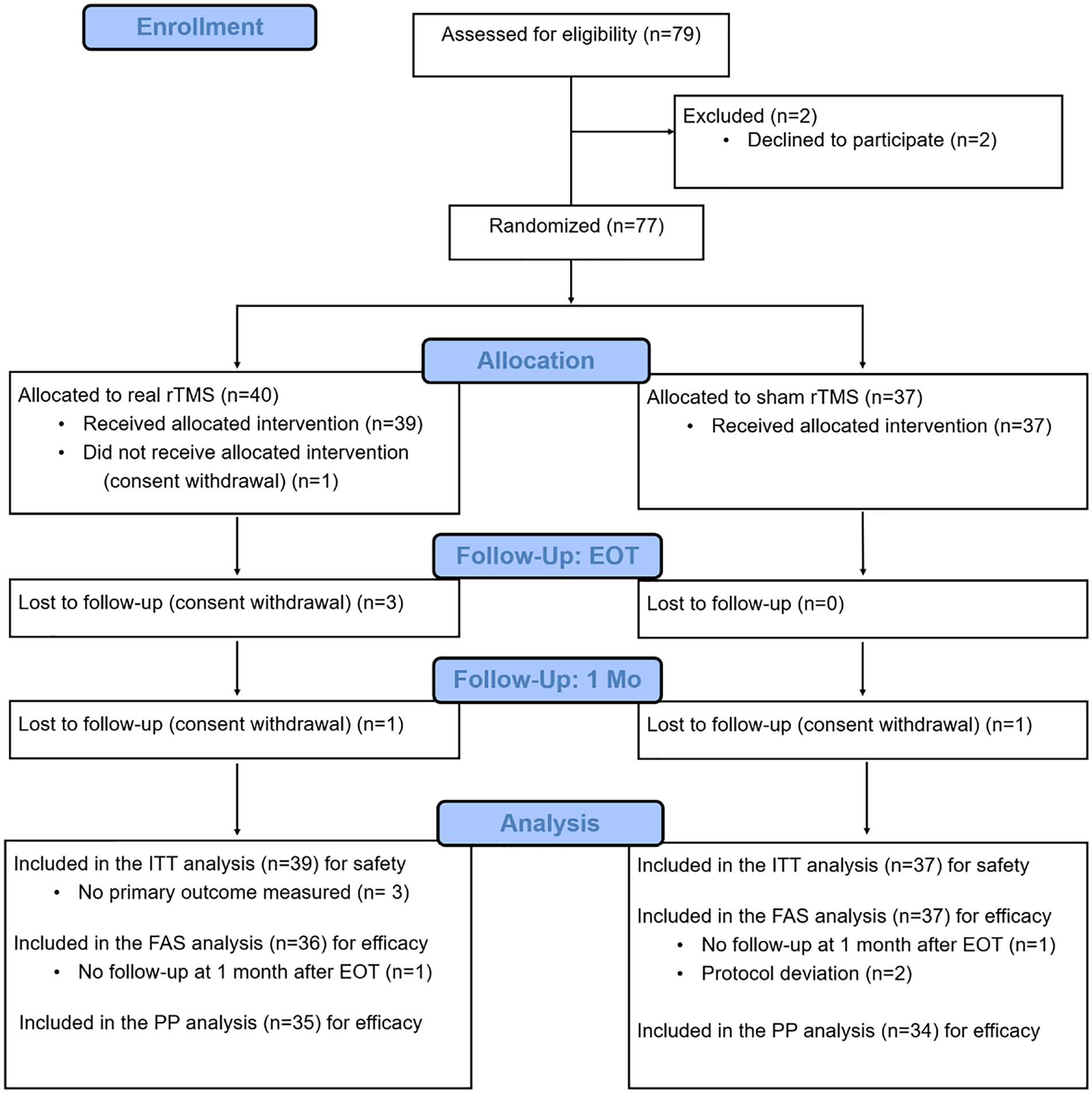
CONSORT flowchart with the number of participants in each phase. rTMS, repetitive transcranial magnetic stimulation; EOT, end of treatment; ITT, intention to treat; FAS, full analysis set.
Table 1 summarizes the baseline characteristics of the study participants. There were no significant differences between groups in baseline demographic, impaired side, stroke lesion location, initial stroke severity (revealed by National Institutes of Health Stroke Scale, upper extremity FMA, and B-stage), and other baseline functional measures (BBT, FTT, grip strength, and K-MBI), spasticity (MAS), RMT, or physical and occupational therapy time after discharge. The baseline characteristics of the PP set are presented in Supplementary Table 1.
Baseline Characteristics (n = 73).
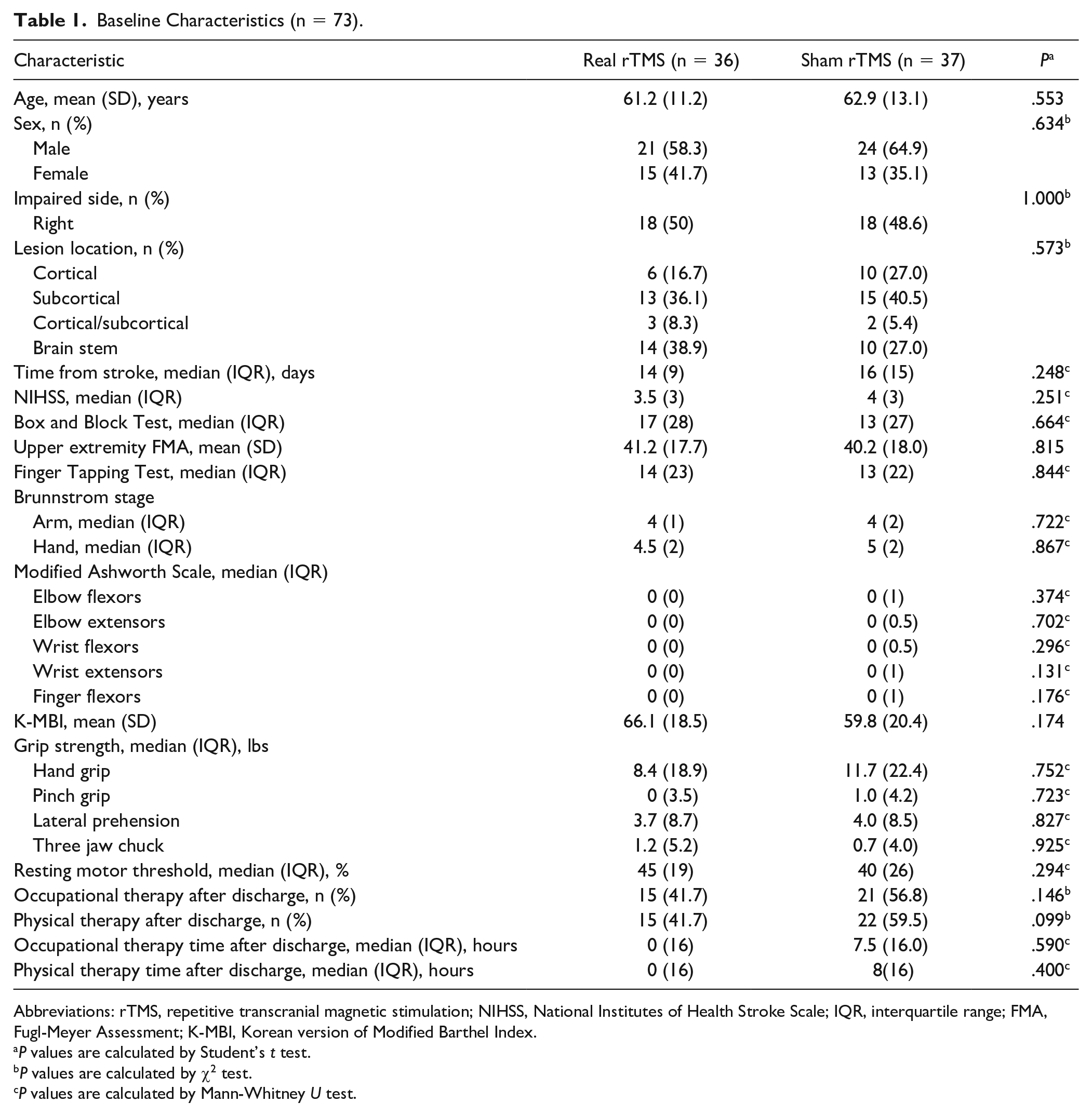
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; NIHSS, National Institutes of Health Stroke Scale; IQR, interquartile range; FMA, Fugl-Meyer Assessment; K-MBI, Korean version of Modified Barthel Index.
Primary and Secondary Outcomes
Changes in primary and secondary outcomes from baseline to EOT or 1 month after EOT in FAS are shown in Table 2. There were no significant differences in changes in any outcomes between real and sham rTMS. Equivalence test using TOST for primary outcome revealed that the effect of real rTMS was statistically equivalent with sham rTMS (
Changes in Outcome Scores from Baseline After Treatment (n = 36 in Real rTMS Group and n = 37 in Sham rTMS Group) a .
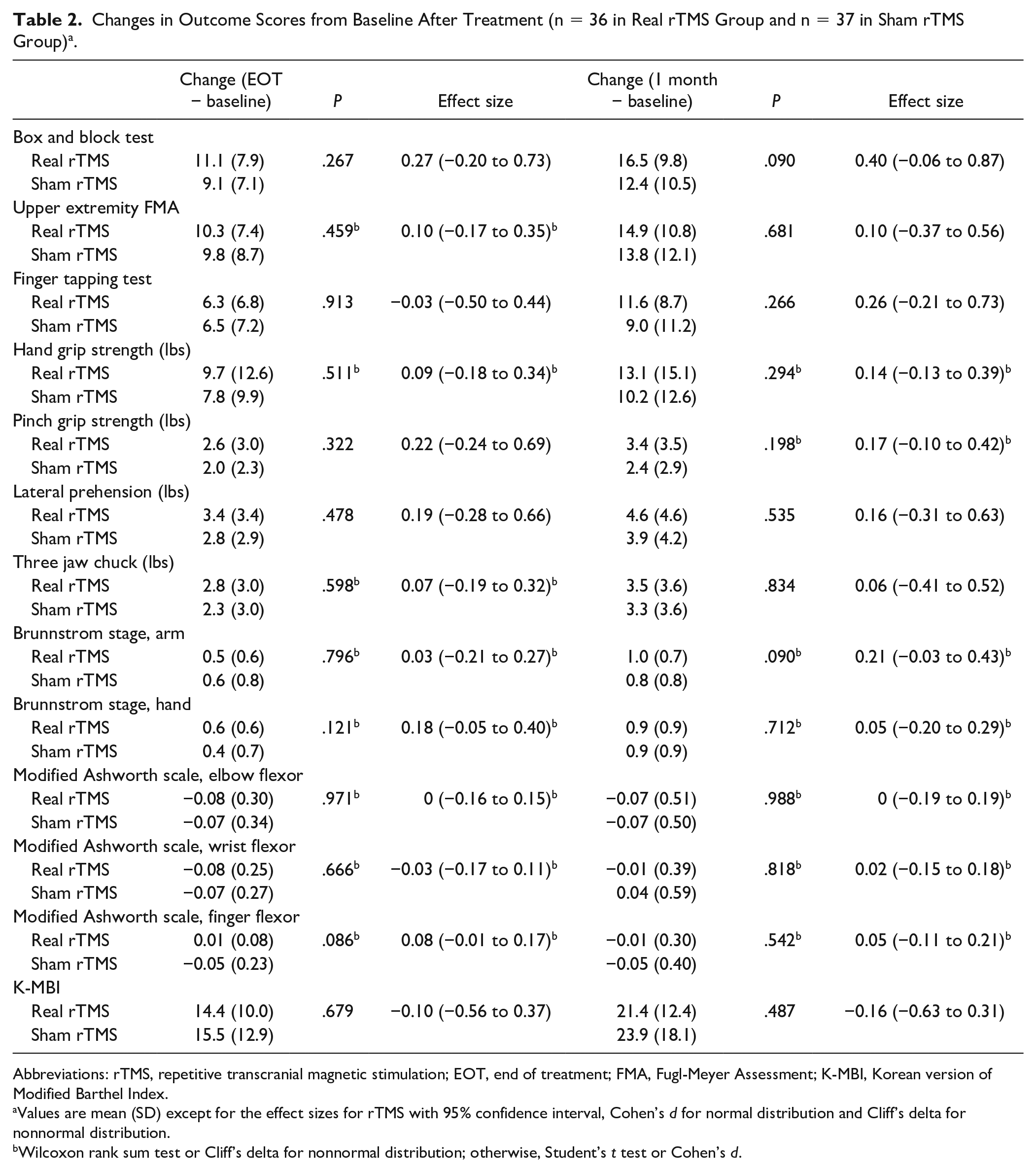
Abbreviations: rTMS, repetitive transcranial magnetic stimulation; EOT, end of treatment; FMA, Fugl-Meyer Assessment; K-MBI, Korean version of Modified Barthel Index.
Values are mean (SD) except for the effect sizes for rTMS with 95% confidence interval, Cohen’s
Wilcoxon rank sum test or Cliff’s delta for nonnormal distribution; otherwise, Student’s
Subgroup Analysis According to Stroke Lesion Location (Cortical Versus No Cortical Involvement)
Primary and secondary outcomes were additionally analyzed in 2 divided data sets according to the lesion location and any cortical involvement (cortical only or cortical/subcortical; real rTMS: n = 9; sham rTMS: n = 12) versus no cortical involvement (subcortical only or brainstem; real rTMS: n = 27; sham rTMS: n = 25). The representative results are presented in Figure 3. Detailed results are presented in Supplementary Tables 3 and 4. In outcomes that reflect fine motor function of the hand, improvements in the real rTMS group were noted only among the subgroup with no cortical involvement (Figure 3). At 1 month after EOT, BBT after real rTMS was improved compared to that after sham rTMS both in FAS (17.4 ± 9.8 real vs 10.9 ± 10.3 sham;
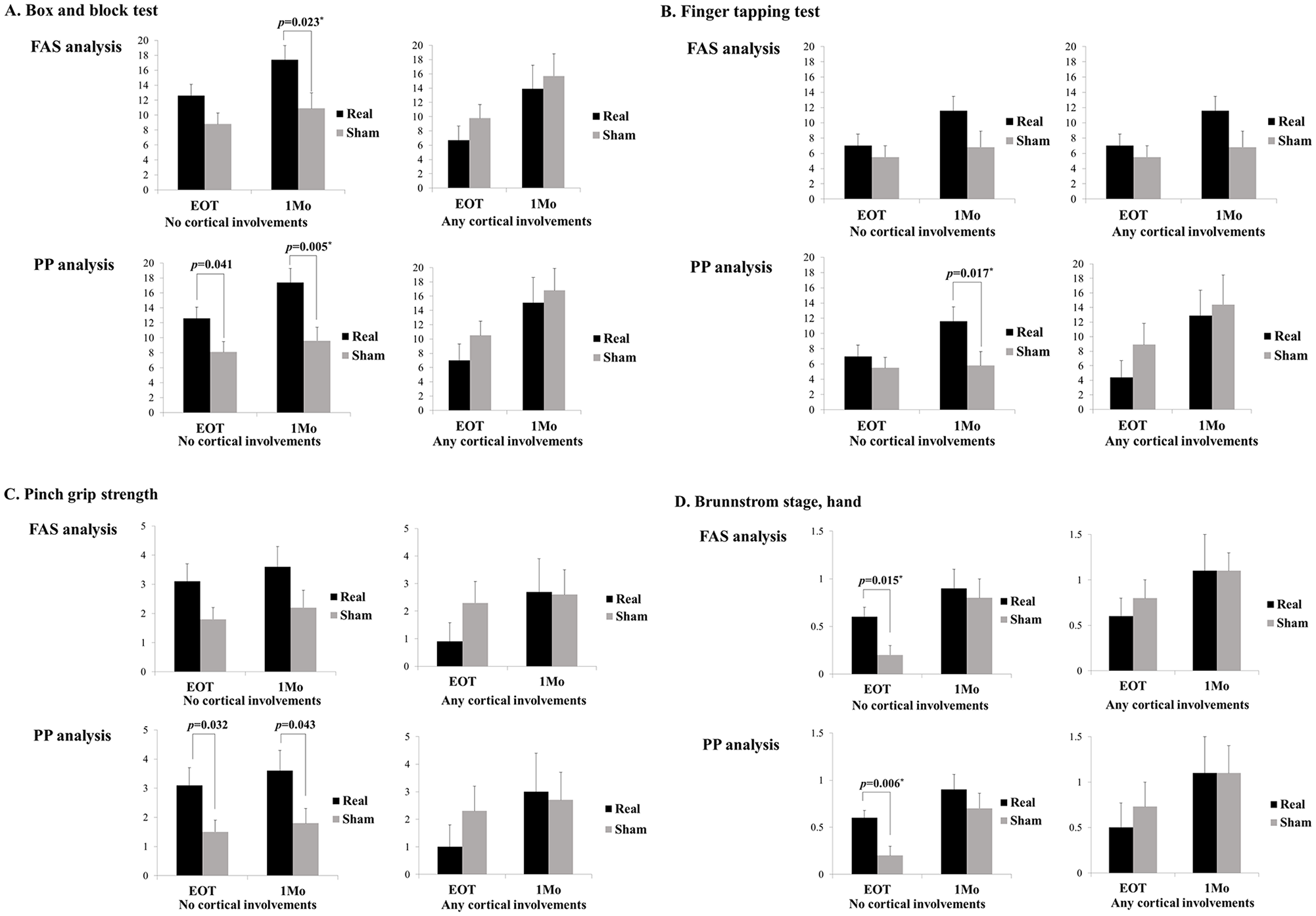
Subgroup analysis according to the stroke lesion location (any cortical versus no cortical involvement). (A) Box and block test. (B) Finger tapping test. (C) Pinch grip strength. (D) Brunnstrom stage, hand. Values are changes from baseline and presented as mean ± standard error.
Adverse Events
Of the safety population (n = 76), a total of 111 adverse events occurred in 48 participants and were more numerous among real rTMS (71 events in real rTMS [64%] vs 40 events in sham rTMS [36%];
Discussion
This study investigated the effects of low-frequency rTMS on motor recovery in patients within 3 months after ischemic stroke. In both study arms, significant improvements in upper limb function were observed, which may be observed in spontaneous recovery after stroke and associated with the effect of task-based occupational therapy, 26 but low-frequency rTMS combined with conventional rehabilitation therapy failed to appreciably enhance motor recovery in patients with subacute stroke.
The results of previous studies on low-frequency rTMS for motor recovery among patients with acute/subacute stroke are conflicting. In acute/subacute subcortical stroke, 3 studies reported possible beneficial effects of low-frequency rTMS on fine motor recovery or pinch force but had limitations of crossover design and small sample size.27-29 Among randomized controlled trials, several studies with small sample size revealed better motor recovery in patients who received low-frequency rTMS than in sham controls. Khedr et al reported superior motor improvements for low-frequency rTMS compared to that for high-frequency rTMS. 30 Three other randomized controlled trials demonstrated improvements in FMA.31-33 Two randomized controlled trials that included large numbers of patients with subacute stroke reported an improvement of upper limb function measured by FMA immediately after low-frequency rTMS.34,35 In contrast, 4 other studies reported no significant differences in the effects of low-frequency rTMS compared to sham control,36-39 although Lüdemann-Podubecká et al suggested a possible beneficial effect of low-frequency rTMS according to hemispheric dominance, 36 and Conforto et al reported that there were improvements in Jebsen-Taylor test scores and pinch force only among low-frequency rTMS participants but not in the sham control. 39 The results of this study weigh more on the negative results of low-frequency rTMS in patients with subacute stroke.
There are several possible reasons for the negative results of our study. First, IHI may contribute differently according to the stage of stroke (acute/subacute vs chronic). IHI was first introduced by studies to show the persistent premovement IHI from the contralesional to ipsilesional M1 using paired-pulse TMS in a small number of patients with chronic subcortical stroke.40,41 However, recent neurophysiological studies including patients with acute/subacute stroke did not support the role of IHI in motor recovery. One study of patients with subacute ischemic stroke using paired-pulse TMS demonstrated abnormally decreased IHI from the ipsilesional M1 toward contralesional M1 but normal IHI from the contralesional M1 to ipsilesional M1. 42 Stinear et al measured an ipsilateral silent period and reported that IHI was symmetrical and stable during the first 12 weeks after stroke. 43 Xu et al also reported that premovement IHI was stable during the acute and subacute period after stroke, and release of premovement IHI worsened as motor outcomes improved. 44 Therefore, IHI as a rationale for using low-frequency rTMS on the contralesional M1 in subacute stroke for upper limb recovery is not yet well elucidated, and further studies are required for clarification. Second, we oriented the rTMS coil vertically to the scalp for sham stimulation. Although the electrical field induced by this sham stimulation method is lower and less focal than that of real stimulation, 45 we could not exclude the possibility that this low electric field strength may have induced modulation of cortical neurons. 46 Third, when comparing our study with that of Zheng et al, 34 the largest study reporting the positive effects of low-frequency rTMS, the lower dose of rTMS intervention may have resulted in the negative results observed in the current study, considering the possible cumulative effects due to multiple sessions of rTMS. 47 The optimal dose of rTMS for motor recovery after stroke is not conclusive at this time and further study is required. Fourth, participants’ characteristics could affect the effect of low-frequency rTMS. In our study, we included only patients with ischemic stroke, but approximately 36% of the patients in the real rTMS group in Zheng et al’s study 34 had suffered hemorrhagic stroke. In a recent large randomized controlled trial including patients who experienced a stroke within 3 to 12 months of enrollment, low-frequency rTMS also produced improvements only for patients with hemorrhagic stroke. 48 The recovery pattern of hemorrhagic stroke may differ from that of ischemic stroke, 49 and stroke type should be considered in further clinical trials for rTMS. In addition, corticospinal tract integrity could affect the effect of low-frequency rTMS due to the differential role of IHI according to the degree of structural reserve, based on the bimodal balance recovery model. 12 In one study including 42 patients with subacute stroke in the low frequency rTMS group, positive results were observed to include only the patients with the presence of ipsilesional MEP. 35 Although we also included patients with mild motor impairment, there is the possibility that some participants with no ipsilesional MEP were included in our analyses, which may offset the beneficiary effects of low frequency rTMS.
In this study, most of the enrolled patients showed involvement of subcortical or brainstem structures (52/73, 71%; Table 1). In the subgroup analysis in this group (no cortical involvement), the BBT in the real rTMS groups was significantly more improved at 1-month follow-up, in both FAS and PP analysis (Figure 3). Brunnstrom hand stage ratings in the real rTMS group were also significantly improved at EOT. Changes in BBT and Brunnstrom hand stage ratings in the any cortical involvement group displayed a trend for less improvement in the real rTMS group, although this difference was not statistically significant. These results were observed as a results of the exploratory additional subgroup analysis that was not planned at the designing stage of this study, hence we cannot draw solid conclusions due to a high likelihood of these findings due to chance. 50 However, our results from the subgroup analysis are in consistent with those of previous studies indicating different effects of neuromodulation depending on the location of stroke lesions and suggest some consideration points for further study. In a study using bihemispheric tDCS with robot-assisted therapy, patients with subcortical stroke benefited more than those with cortical stroke. 51 Ameli et al demonstrated that high frequency rTMS on the ipsilesional M1 was effective only for patients with subcortical stroke but not in patients with additional cortical stroke. 52 Functional MRI studies have demonstrated that high-frequency rTMS decreased cortical activity in the contralesional M1 but induced widespread bilateral cortical activity in cortical stroke. 52 Several hypotheses may explain the superior effects of rTMS in cases of stroke without cortical involvement. First, the expected increase in cortical activity in the ipsilesional M1 by neuromodulation may not efficiently propagate to other nodes of the motor network for motor recovery due to structural or functional disconnection in stroke cases involving cortical structures.52,53 Second, greater suppression of GABA-ergic intracortical inhibition in cases of cortical stroke 54 and consecutive downregulation of GABAA receptors in both hemispheres 55 could be associated with reduced effects of rTMS in cortical stroke. Additionally, in patients with supratentorial stroke, recovery of interhemispheric balance may be rare and not related to motor recovery relative to that in patients with infratentorial stroke, and the former may use global reorganization of motor networks for recovery, which requires higher rewiring costs. 56
There are several limitations to this study. First, corticospinal tract integrity was not objectively measured using single-pulse TMS or brain imaging. The degree of corticospinal tract integrity is a strong predictor of motor recovery, 57 and the role of IHI in motor recovery may be less important in patients with severe corticospinal tract involvement, 12 which could be associated with the effects of low-frequency rTMS. Second, we did not measure the changes in cortical activation pattern or neurophysiological parameters using functional neuroimaging or paired-pulse TMS in all patients, although the main purpose of this study was to prove the clinical efficacy of low-frequency rTMS. Third, the possible differences in rTMS effect on motor recovery according to hemispheric dominance was not considered in this study. Lüdemann-Podubecká et al reported that low-frequency rTMS over the contralesional M1 improves the function of the affected hand only for cases in which stroke occurs in dominant hemisphere, but not in the nondominant hemisphere. 36 Fourth, most of the outcomes in this study were focused on assessing the hand function. Therefore, it is possible that the effects of rTMS on proximal arm recovery were not captured well. Fifth, medications which can promote motor recovery after stroke were not limited during this study. However, candidate drugs for motor recovery (eg, selective serotonin reuptake inhibitors, levodopa) did not result in significant effects in recent large clinical trials.58,59 Finally, the various rehabilitation modalities could not be exactly counterbalanced between the groups from EOT to 1-month follow-up, although we investigated the dose of physical and occupational therapies based on patients’ recall.
Conclusions
This study does not support the superiority of 2-week low-frequency rTMS on the contralesional M1 over sham rTMS for upper limb motor recovery when combined with task-oriented occupational therapy among patients within 3 months poststroke. Stroke lesion location may have to be considered in further clinical trials using low-frequency rTMS for motor recovery.
Supplemental Material
NNR_TAMAS_Supplementary_Tables_2ndRevision_final – Supplemental material for Low-Frequency Repetitive Transcranial Magnetic Stimulation Over Contralesional Motor Cortex for Motor Recovery in Subacute Ischemic Stroke: A Randomized Sham-Controlled Trial
Supplemental material, NNR_TAMAS_Supplementary_Tables_2ndRevision_final for Low-Frequency Repetitive Transcranial Magnetic Stimulation Over Contralesional Motor Cortex for Motor Recovery in Subacute Ischemic Stroke: A Randomized Sham-Controlled Trial by Won-Seok Kim, Bum Sun Kwon, Han Gil Seo, Jihong Park and Nam-Jong Paik in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors thank the Division of Statistics in Medical Research Collaborating Center at Seoul National University Bundang Hospital for statistical analyses.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Nam-Jong Paik is an unpaid medical advisor of the REMED Co since 2018 October after the completion of this clinical trial. The other authors have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number: HI15C0832). REMED, Korea, supported the repetitive transcranial magnetic stimulation machine in 3 hospitals.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
