Abstract
Background. Mirror therapy (MT) is a promising therapeutic approach in stroke patients with severe hand paresis. Objective. The ipsilateral (contralesional) primary sensorimotor cortex (SMC) and the mirror neuron system have been suggested to play decisive roles in the MT network. The present study investigated its underlying neural plasticity. Methods. Two groups of healthy participants (n = 13 in each group) performed standardized fine motor tasks moving pegs and marbles (20 min/d for 4 days) with their right hand with either a mirror (mirror training group, MG) or a nonreflective board (control training group, CG) positioned orthogonally in front of them. The number of items moved by each hand was tested after each training session. Functional MRI (fMRI) was acquired before and after the training procedure to investigate the mirror training (MTr)-specific network by the analysis of the factors Time and Group. Results. The hand performance test of the trained right hand did not differ between the 2 groups. The untrained left hand improved significantly more in the MG compared with the CG. fMRI analysis of action observation and imitation of grasping tasks demonstrated MTr-specific activation changes within the right dorsal and left ventral premotor cortex as well as in the left SMC (SMCleft). Analysis of functional and effective connectivity showed a MTr-specific increase of functional coupling between each premotor region and the left supplementary motor area, which in turn showed an increased functional interaction with the ipsilateral SMCleft. Conclusions. MTr remodels the motor system by functionally connecting hand movement to the ipsilateral SMC. On a system level, it leads to interference of the neural circuit related to motor programming and observation of the trained hand with the illusionary movement of the untrained hand.
Introduction
Effective stroke management is important as the treatment of stroke consumes a significant amount of resources. 1 Functional recovery can occur even in the chronic stage of stroke with plastic changes induced in the affected primary motor cortex (M1) by intense motor training of the paretic hand.2,3 Additional commitment of noninvasive stimulation over the affected M1 by repetitive transcranial magnetic stimulation (TMS) 4 or by transcranial direct current stimulation 5 promotes functional recovery in association with plastic changes within the affected M1. However, the effectiveness of such strategies is limited to patients with mild to moderate hand paresis. Thus, an effective therapy regime for patients with severe hand paresis or loss of hand function is still lacking.
Mirror therapy (MT) is a promising therapeutic approach in stroke patients, even in those with complete loss of hand function. By using a mirror that is positioned orthogonally in front of the patient, the reflection of the right arm in the mirror provides an illusion that the left arm is being moved. MT was first described by Ramachandran and colleagues 6 in patients with phantom pain after limb amputation and has been suggested to improve hand functional deficit in stroke patients. 7 Ramachandran and Altschuler 8 suggested that the ipsilateral (contralesional) corticospinal tracts from the M1 play a role while training with a mirror, 8 but a recent functional MRI (fMRI) study reported an activation shift toward the ipsilesional sensorimotor cortex (SMC) after MT in chronic stroke patients in contrast with a control group. 9 It has also been suggested that the mirror neuron system (MNS) plays an important role while observing one’s hand in the mirror and training via action observation (AO) and imitation (IMI).8,10
The underlying neural plasticity of MT is currently unknown. Therefore, in the present study, fMRI was used to address this issue. We focused on training-induced plasticity by training with a mirror rather than the direct effect of observing one’s hand in the mirror, as was investigated with the use of functional imaging11,12 or TMS.13-15
Methods
Participants
A total of 30 healthy volunteers were recruited for the study. All of them provided written informed consent. This study was approved by the local ethics committee. All participants were right handed according to the Edinburgh Handedness Inventory.
Experimental Procedure
Healthy participants were studied in order to analyze a healthy neural network—therefore, the term mirror training (MTr) is used rather than mirror therapy.
Healthy participants were divided into a control training group (CG) and a mirror training group (MG). In each group, 2 participants were excluded because of head movement during fMRI. Therefore, in the final analysis, n = 13 participants were included in each group (mean age in the CG was 23.9 years, and there were 7 women; mean age in the MG was 25.5 years, with 6 women). All participants trained with their right hand for 20 minutes daily over 4 days. Five training skills, each for 2 minutes, were practiced. These skills were repeated after a rest for 3 minutes. The skills included picking up a peg from a 9-hole peg test board, placing it on the desk in a predefined position, and then returning the peg to the board. In another task, participants used a teaspoon to move marbles from one bowl to another. The training skills were standardized by definition of their procedures. Participants were encouraged to execute the skills quickly and to remain focused on the task while an instructor sat beside them.
The MG practiced the training skills with a mirror positioned orthogonally in front of the participants, and they continually looked in the mirror. A board of the same dimension was positioned in place of the mirror in the CG (Figure 1). During training, both the MG and the CG could not see their untrained left hand; they did not move their left hand while training with their right hand. Both hands were tested on the first day of testing (baseline), each day after the daily training session and a day after the 4-day training period (post; this test was termed the hand performance test). The same skills used during the training session were tested (hand performance tests lasted 10 minutes and included 5 trained skills for 2 minutes each). For the hand performance tests, the mirror in the MG and the board in the CG were removed. The number of successful trials was counted for each skill during the 2-minute testing period—for example, the number of marbles moved from one bowl to another. The number of successful trials for each of the 5 skills was averaged for each day and for each hand.
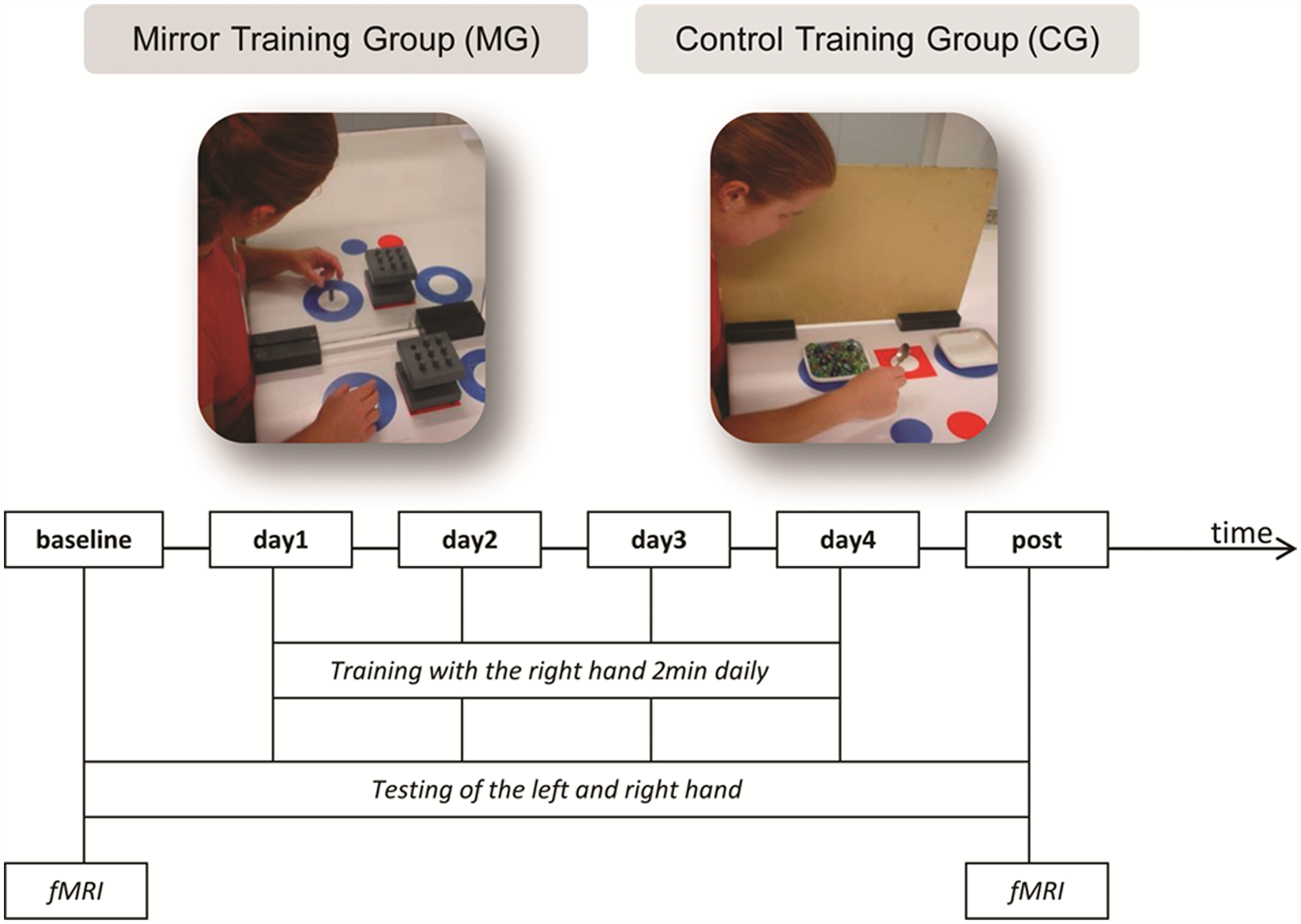
Schematic of the study design: the MG practiced training skills with a mirror, whereas the CG used a board. Both groups trained for 20 minutes daily with their right hand for 4 days (day 1, day 2, day 3, and day 4). The untrained left hand and the trained right hand were tested before starting the training period (baseline), the day after day 4 (post), and after daily training sessions. The tests included the same skills that were practiced daily. Imaging data were acquired at baseline and posttraining. Abbreviations: MG, mirror training group; CG, control training group; fMRI, functional MRI.
The data were analyzed using a between-group (MG vs CG) and within-group (baseline, day1, . . . , post) analysis with analysis of variacne (ANOVA; threshold for significance set at .01, Bonferroni correction for multiple comparisons). To analyze whether the MG and the CG were different at baseline, which might explain any difference in the increase of hand performance test curves independent of the training effect, a 2-sample t test was carried out between the CG and the MG for time point baseline; the threshold for significance was set at P < .05.
MRI Acquisition
MRI was performed in a 3-T whole-body MRI system (TIM-TRIO, Siemens, Erlangen, Germany) equipped with a standard head coil. fMRI was acquired before training (baseline) and a day after training (post). For fMRI, contiguous multislice echoplanar images (TE = 60 ms) were obtained in the axial orientation. A total of 30 slices (3-mm thickness) were acquired every 2.49 s (voxel size 3 × 3 × 3 mm3). For T1-weighted MRI, 160 sagittal slices (1 × 1 × 1 mm3 voxel size; TE = 3.93 ms) were acquired.
The following conditions were applied in a block design: during REST, participants fixated on a cross in the middle of the screen. For AO, videos of a hand grasping objects were presented in one block with the right hand and in a further block with the left hand. Each block lasted 25 s, with 5 different objects being grasped for 5 s. For IMI, participants first observed a hand grasping an object and were requested to imitate the observed movement with their right hand in 1 block and with their left hand in another block. Participants performed the grasping movement 5 times in each block. Objects were fixed on Velcro tape, which was fastened around the hip before MRI acquisition. The signal to begin to grasp from a predefined position was delivered by an acoustic stimulus via headphones. This signal was controlled by a PC running “Presentation” Software (Neurobehavioral-Systems, Inc, Albany, California) synchronized to the scanner. Before scanning, participants were trained to perform this movement accurately in the MR environment. Motor responses were controlled via inspection within the scanner and by bipolar surface EMGs (from both flexor digitorum superficialis and extensor digitorum communis, using subtraction of the averaged recorded gradient signal; the high-pass filter was 70 Hz). To ensure that IMI with the right and left hand were comparable across both investigation time points and did not change with practice, the EMG data were compared over the duration of the active condition and for the peak-to-peak amplitude during the active blocks on each investigation day. This comparison was performed offline. If a participant’s EMG activity differed between both investigation time points, they were excluded. Each IMI and AO block lasted 25 s. Each active block was repeated 4 times and alternated in a pseudorandomized manner with rest. Thus, for 16 active blocks and 16 rest blocks, the time for the fMRI acquisition for 1 participant was approximately 14 minutes. Visual stimuli were projected on a screen positioned in front of the bore of the scanner with a horizontal field of view of 19°. Participants viewed the visual stimuli through prism glasses.
fMRI Data Processing
fMRI data processing was performed with SPM8 (Welcome Department of Cognitive Neurology, London, UK) running under Matlab7a (Mathworks, Sherborn, Massachusetts). The first 4 images of each run were discarded to allow for equilibration of longitudinal magnetization. Calculation of online motion and distortion-corrected fMRI volumes was performed at the scanner. Resulting volumes were spatially normalized to a symmetric template based on the Montreal Neurological Institute reference brain using the normalization parameters estimated during segmentation of the T1-anatomical scan. Normalized fMRI images were then smoothed with an isotropic 12-mm full width at half maximum Gaussian kernel.
fMRI Statistical Analysis
A general linear model based on a model of the time course and the hemodynamic response function was used. Stimuli onsets were convolved with a canonical hemodynamic response function, as implemented in SPM8. Low-frequency components of fMRI time series were removed by high-pass filtering. In the second level analyses, volunteers were treated as a random effect, and the contrast images of the conditions of interest were entered into a 2-sample t test.
We were interested in the contrast of time (baseline vs post and vice versa) in the MG for AO and IMI. We also verified the analysis of interaction of AO and IMI between Groups × Hands × Time (the threshold was set at P < .05 corrected across the whole brain, familywise error).
Functional connectivity (FC) was used to evaluate the functional interaction between different regions. 16 FC was evaluated by a correlation analysis between the extracted time series from a region of interest (ROI). 16 Individual ROIs for each participant were defined according to the results of the group analysis (MG vs CG). Significant activity was identified in SMCleft, the right dorsal premotor cortex (dPMCright), left ventral premotor cortex (vPMCleft), and the left inferior parietal lobule (IPLleft), respectively. Next, time series were extracted and were included as a regressor to participants’ fMRI images for baseline and for postacquisition times. A differential contrast between post versus baseline was calculated via a 2-sample t test between MG and CG (MG, post vs baseline, vs CG, post vs baseline) for each extracted time series (corrected P < .05, familywise error).
Dynamic causal modeling is used to estimate and make inferences about the coupling among brain areas by using the procedure of Bayesian model selection. 17 This procedure depends on the model evidence, P(ym), which is given by the posterior probability of observing the data y given a particular model m integrated over the model parameters. The best model (models are represented in Figure 2) is the one with the highest log-evidence: ln[p(ym)]. Different models were conceived based on different connectivities between areas: SMCleft, dPMCright, IPLleft, vPMCleft, and left supplementary motor area (SMAleft). We extracted the BOLD time series from these 5 ROIs of 5-mm radius.
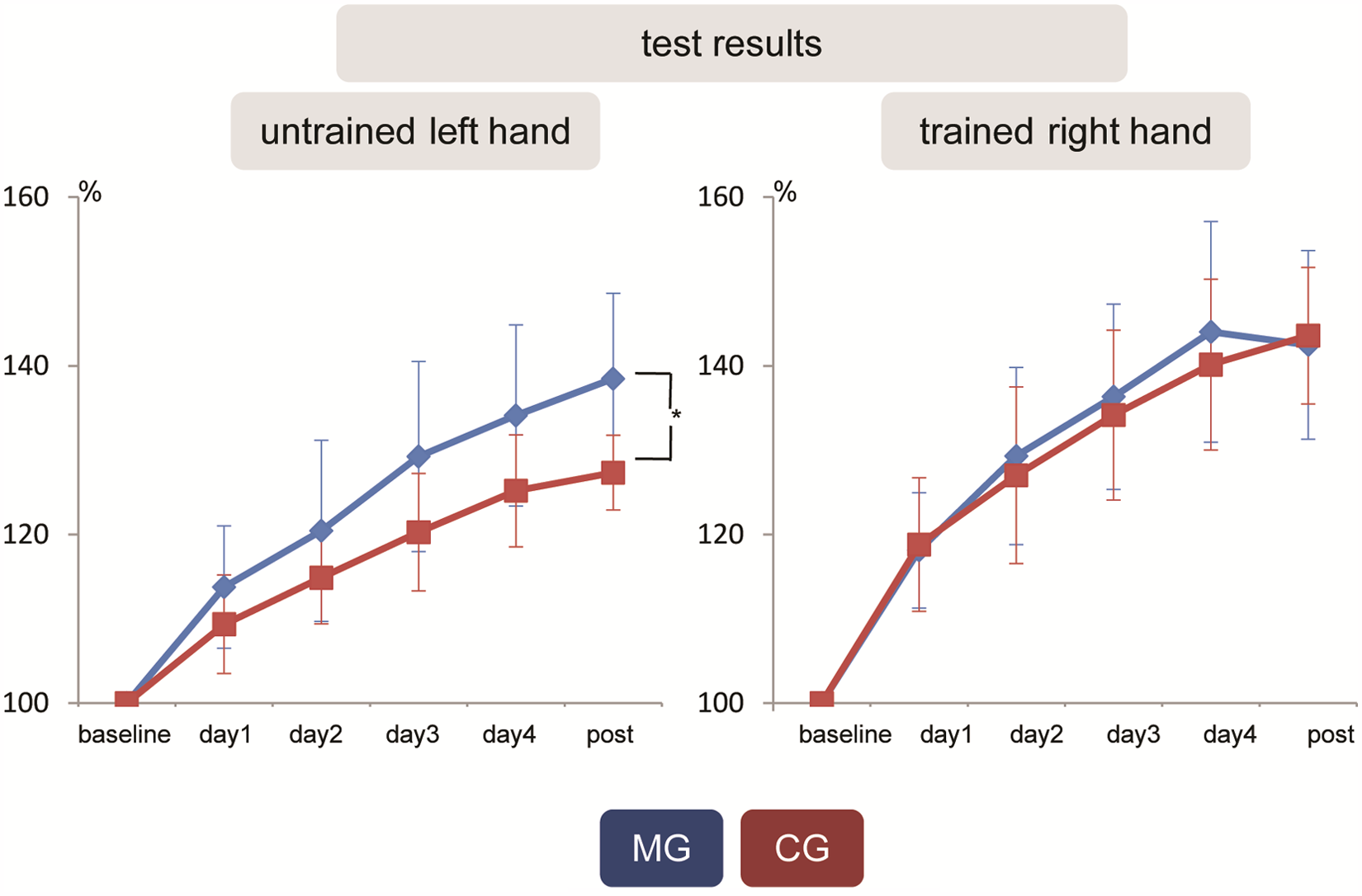
Hand performance test for the trained right and untrained left hand (with standard deviations). Hand performance tests of the trained right hand were not different between the MG and the CG. Hand performance test of the untrained left hand increased with the duration of training in both groups and showed a significantly greater increase in the MG compared with the CG at post (day 1, P < .4; day 2, P < .2; day 3, P < .2; day 4, P < .1; and post, P < .001). The curves represent the average of the numbers from 5 tasks normalized to the average of the baseline (expressed in percentage); that is, Xday(n+1)% = (Xday(n+1):Xbaseline) × 100% (X = average number from 5 skills at time point dayn). Abbreviations: MG, mirror training group; CG, control training group.
Using FC and dynamic causal modeling, we found that we cannot rule out the participation of other areas in information processing between ROIs. Our models were chosen to represent biologically plausible models. Alternative models could have been tested for a higher log-evidence. Anatomical description is based on the probabilistic cytoarchitectonic maps as implemented in the SPM Anatomy toolbox (http://www.fz-juelich.de/inm/inm-1/DE/Forschung/_docs/SPMAnantomyToolbox/SPMAnantomyToolbox_node.html). 18
Results
Hand Performance Test
At baseline, hand performance testing between the MG and the CG were not different for the right and left hands. For the trained right hand, no significant difference was observed between the MG and the CG on any day. For the untrained left hand, hand performance increased with the duration of training in both groups (within-group analysis: baseline vs day 1, baseline vs day 2, baseline vs day 3, baseline vs day 4, and baseline vs post in both groups in all tests; P < .0001). There was a significantly greater increase for the MG in comparison with the CG at post (between-group analysis: for day 1, the comparison between the groups for the left hand was P < .4; day 2, P < .2; day 3, P < .2; day 4, P < .1; and post, P < .001; Figure 3).
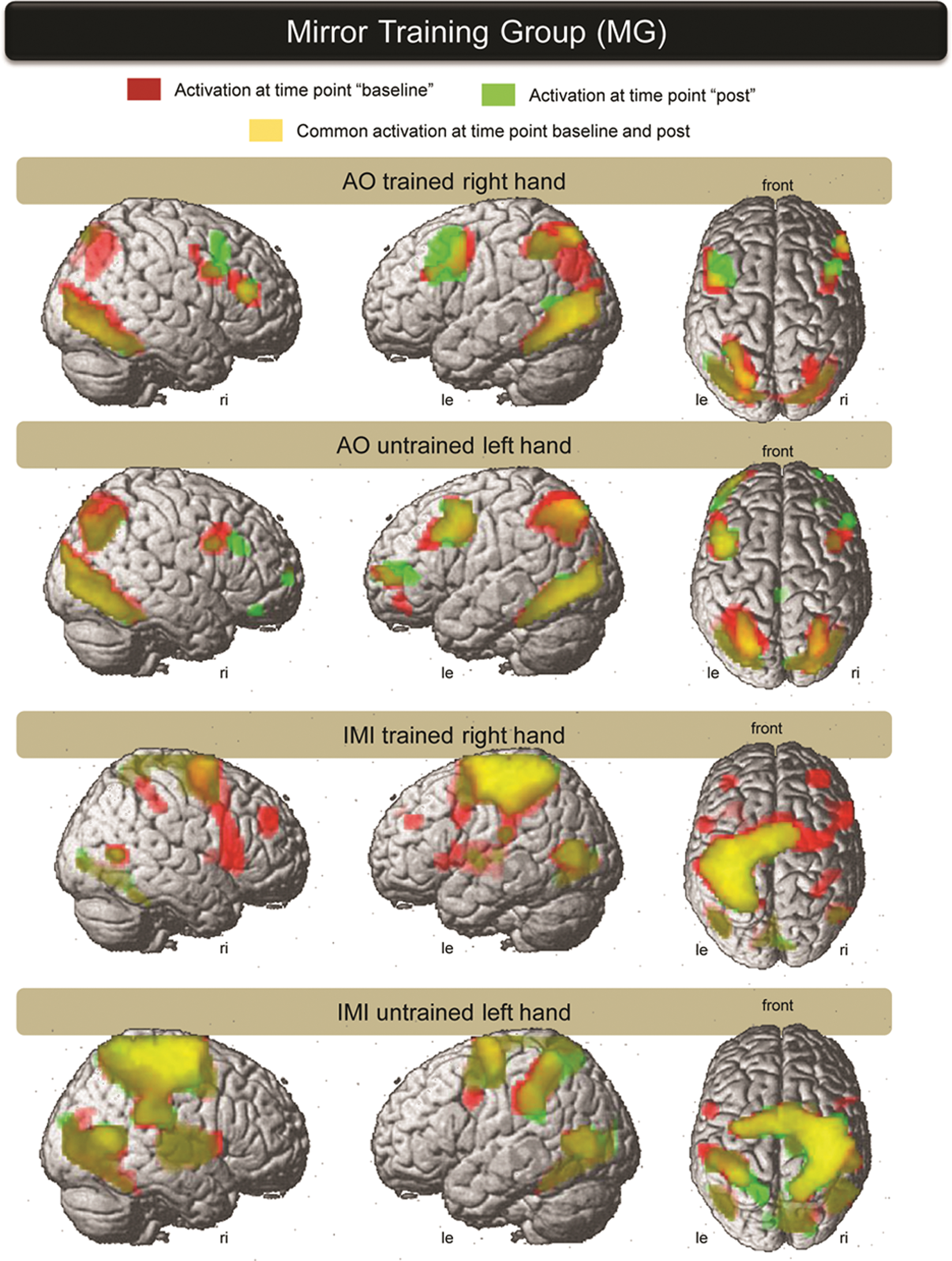
Group activation of the MG during AO and IMI (of the trained right and untrained left hand) are overlaid from both investigation time points. Baseline activation is presented in red and the activation of post is overlaid in green. The common activation is presented in yellow (the threshold of P value was set at P < .05 corrected across the whole brain). The picture on the left represents the right hemisphere, the middle picture is a view of the left hemisphere, and the picture on the right side is a view from above. AO (of the trained-right and untrained-left hand) showed an activation of a network involving the temporal cortex, inferior parietal cortex, premotor cortex, and frontal areas on both hemispheres. The primary SMC is omitted. IMI of both hands showed a widespread activation of the temporal cortex, the superior and inferior parietal cortex, premotor cortex, and frontal areas. The IMI of the trained right hand showed SMC activation on the left side and IMI of the untrained left hand showed right SMC activation. Abbreviations: AO, action observation; IMI, imitation.
fMRI Results
Group activation of MG at time points baseline and post versus rest
AO of the trained right hand at baseline activated the ventral temporal cortex, visual cortex, IPL, vPMC, dPMC, and Broadmann area (BA) 44 and BA45 of both hemispheres (Figure 4). At post, a similar network was activated with higher activation within both premotor cortices. The AO of the untrained left hand at baseline and post showed comparable activation with the AO of the trained right hand.
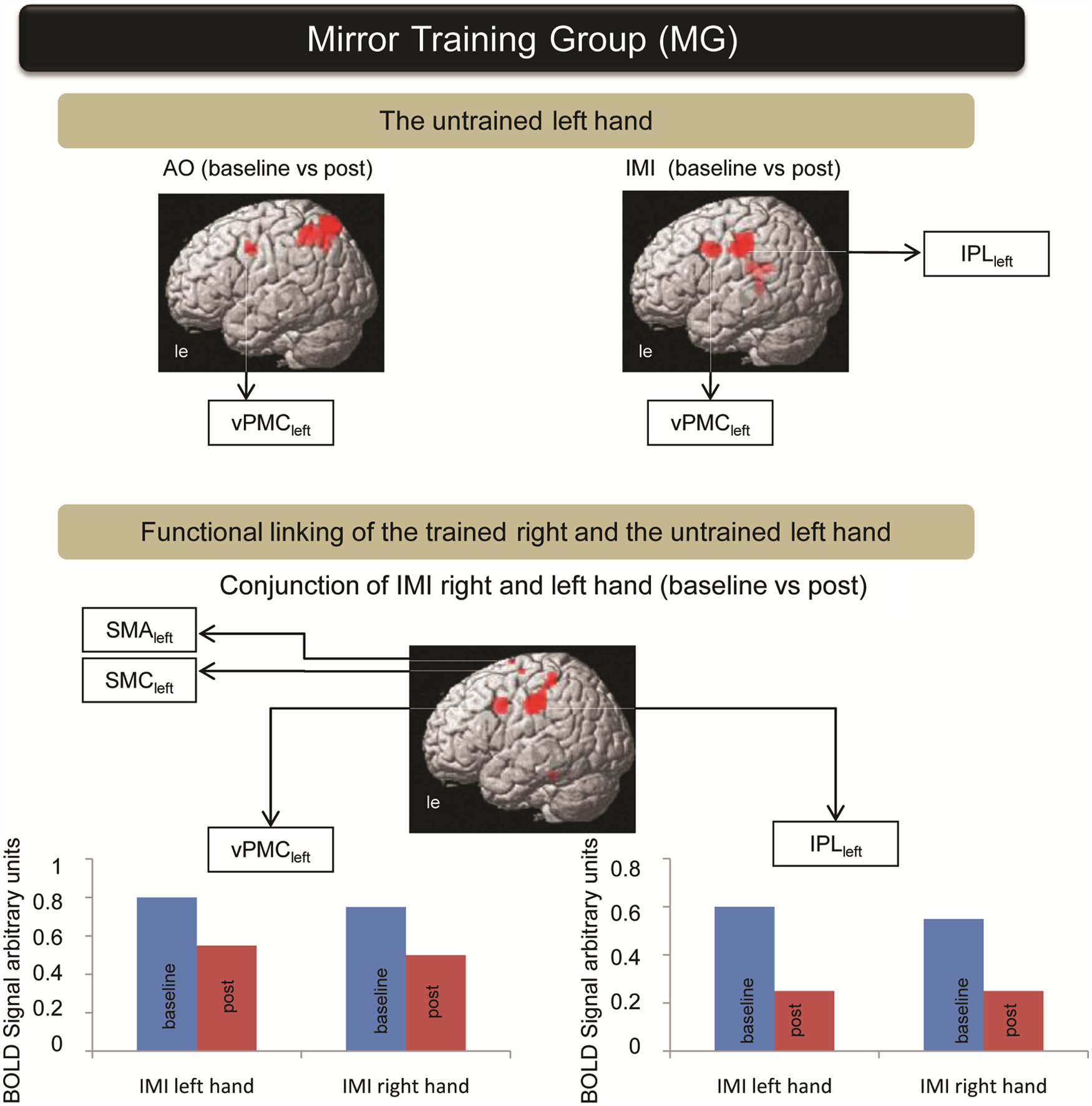
The mirror training group (contrast of time): AO of the untrained left hand (baseline vs post) showed pronounced involvement of the vPMCleft and left posterior parietal cortex. Untrained left hand IMI (IMI baseline vs post) exhibited the involvement of 2 ROIs within the vPMCleft and IPLleft. Conjunction analysis of the baseline versus post difference for the IMI untrained left hand and IMI trained right hand showed a common activation change within the IPLleft, vPMCleft, SMAleft, and SMCleft. Their activation demonstrated functional linking of the untrained left to the trained right hand. Plot bars represent fMRI activation level for different time points and hands. Abbreviations: AO, action observation; IMI, imitation; ROI, region of interest; vPMCleft, left ventral premotor cortex; IPLleft, left inferior parietal lobule; SMCleft, left primary sensorimotor cortex; SMAleft, left supplementary motor area; fMRI, functional MRI.
The IMI of the trained right hand and IMI of the untrained left hand at baseline activated a network of the posterior temporal cortex, superior parietal cortex and IPL, vPMC and dPMC, and BA44 and frontal regions of both hemispheres. The side of SMC activation was dependent on the side of hand movement. At post, the PMC showed an apparent focusing of activation during IMI of the trained right hand.
It must be considered that overlay of 2 activation maps per se does not provide information about the differences between these 2 overlays. A differential contrast (obtained by subtraction of one map from another map) must be analyzed.
Activation changes for MG
AO and repetitive movement were elements of the training session for the MG. Participants continually observed their own (right) hand in the mirror during repetitive training, providing the illusion that their left hand was being moved. During AO of the untrained left hand, activation within the left posterior parietal cortex and vPMCleft decreased (AO of the untrained left hand baseline vs post). During the IMI paradigm for the untrained left hand, activation also decreased within the vPMCleft and IPLleft (IMI of the untrained left hand baseline vs post). Conjunction analysis of the baseline versus post difference for the IMI of the untrained left hand and IMI of the trained right hand revealed a common activation change within the IPLleft, vPMCleft, the SMAleft, and left SMC (SMCleft). This analysis emphasizes the functional linking effect of the mirror regarding real movement of the right hand and the illusion that the left hand is being moved (Figure 5).
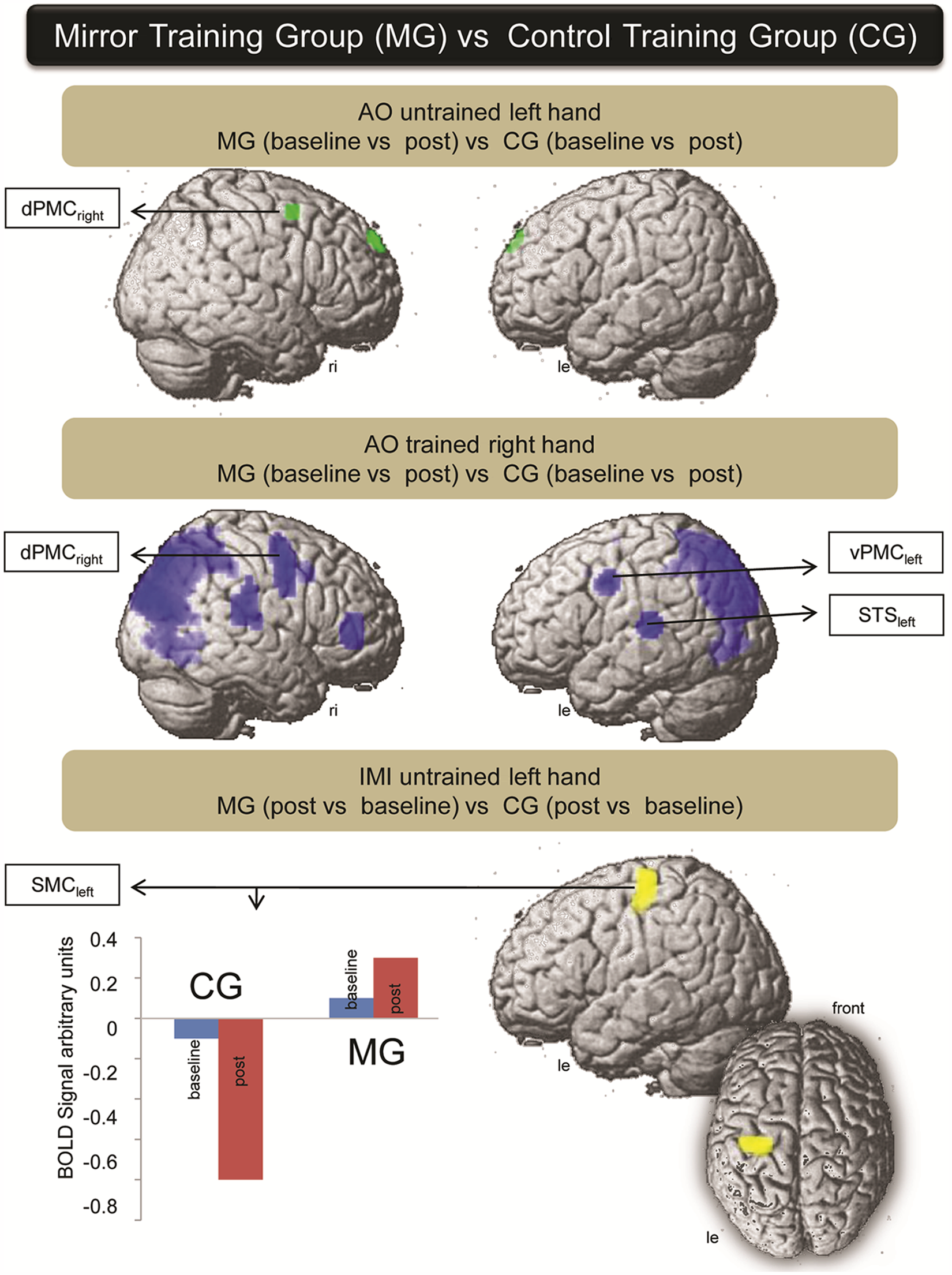
Contrast difference between the MG and the CG: AO of the untrained left hand between MG (baseline vs post) versus CG (baseline vs post) showed activation changes within the dPMCright. AO of the trained right hand between MG (baseline vs post) versus CG (baseline vs post) showed activation changes within the dPMCright, vPMCleft, left and right superior temporal gyrus, and superior parietal lobule and right IPL. IMI of the untrained left hand between MG (post vs baseline) versus CG (post vs baseline) showed an activation increase within the ipsilateral SMCleft (view from left side and from above). Plot bars represent activation level at different time points for both groups: by moving the left hand, ipsilateral SMCleft activation increased in the MG, whereas the activation within the CG decreased further. Abbreviations: AO, action observation; IMI, imitation; dPMCright, right dorsal premotor cortex; vPMCleft, left ventral premotor cortex; SMCleft, left primary sensorimotor cortex.
Contrast between the MG and CG by time and hands
AO of the trained right hand between MG versus CG revealed, among other regions, activation changes within the right dPMCright, vPMCleft, left and right superior temporal gyrus and superior parietal lobule, and IPLright (AO of the trained right hand between MG [baseline vs post] vs CG [baseline vs post]; for Montreal Neurological Institute [MNI] coordinates of the activated regions, see Table 1). AO of the untrained left hand also demonstrated changes within the dPMCright. IMI of the trained right hand between MG, for baseline versus post, versus CG, for baseline versus post, and between MG, for post versus baseline, versus CG, for post versus baseline, showed no activation change. IMI of the untrained left hand between MG (post vs baseline) versus CG (post vs baseline) showed an activation increase within the ipsilateral SMCleft. Thus, the following regions were relevant in the MG in comparison with the CG: dPMCright, IPLleft, vPMCleft, and SMCleft (Figure 6).
Coordinates of the Interaction AO Right Hand, MG Versus the CG (Group × Time) a
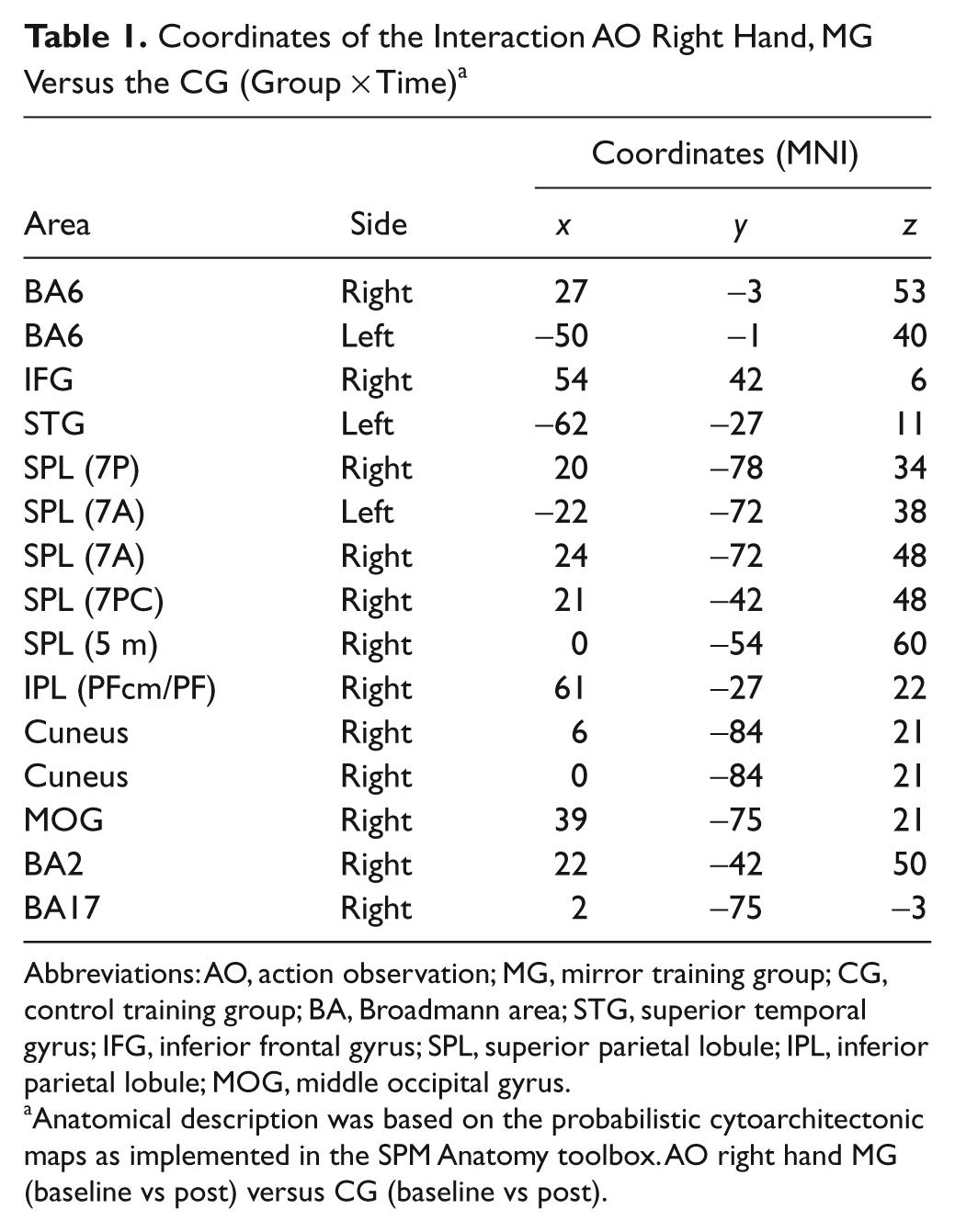
Abbreviations: AO, action observation; MG, mirror training group; CG, control training group; BA, Broadmann area; STG, superior temporal gyrus; IFG, inferior frontal gyrus; SPL, superior parietal lobule; IPL, inferior parietal lobule; MOG, middle occipital gyrus.
Anatomical description was based on the probabilistic cytoarchitectonic maps as implemented in the SPM Anatomy toolbox. AO right hand MG (baseline vs post) versus CG (baseline vs post).
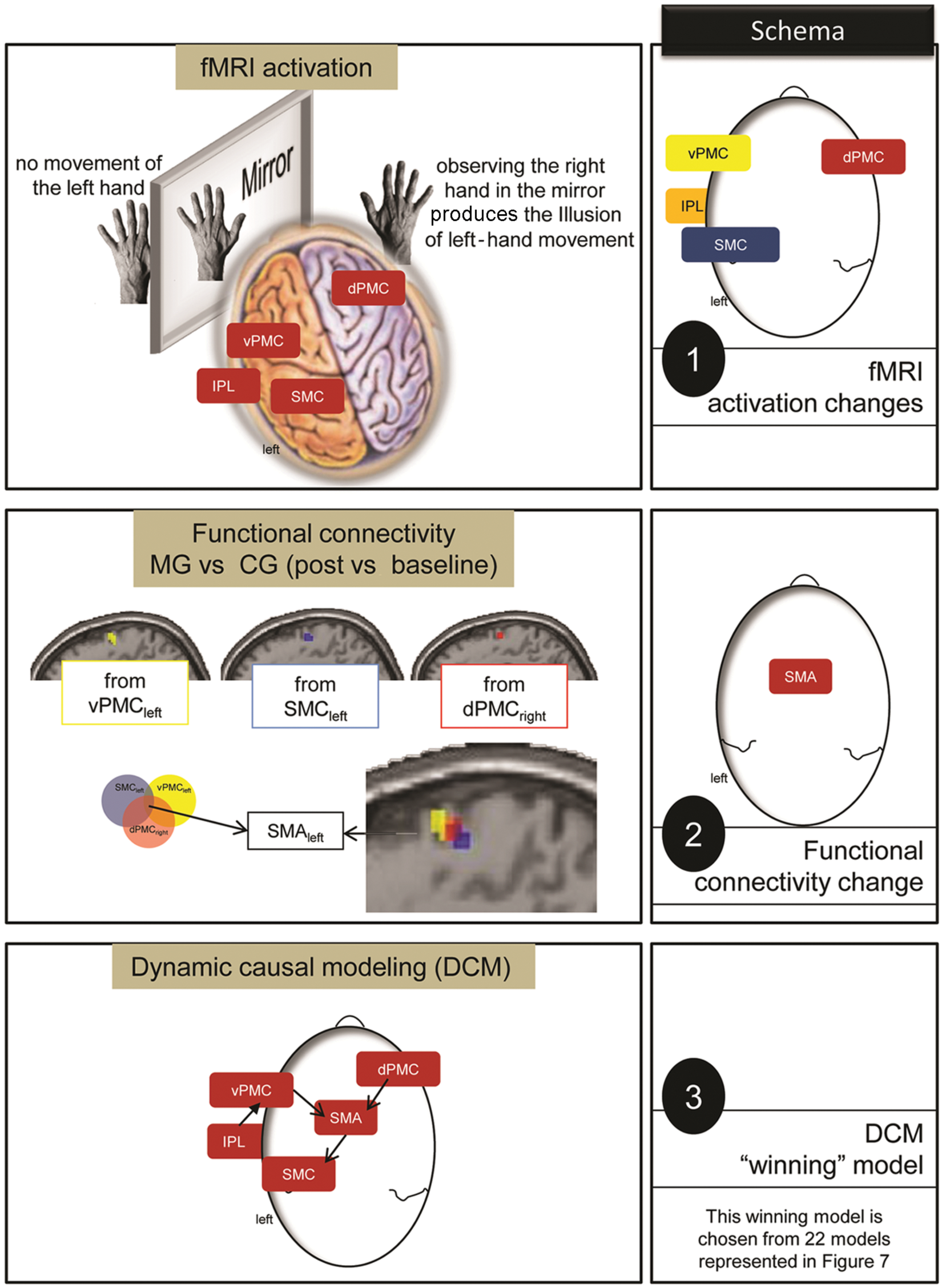
Contrast of fMRI paradigms (AO and IMI) between Groups × Hands × Time revealed an activation difference within the dPMCright, vPMCleft, IPLleft, and SMCleft (schema 1). To analyze the network interaction among the dPMCright, vPMCleft, IPLleft, and SMCleft, the FC was evaluated between MG versus CG (post vs baseline). This analysis revealed a common connectivity of the dPMCright, vPMCleft, and SMCleft with the SMAleft. Consequently, this region interacted with the dPMCright, vPMCleft, and SMCleft in MG versus CG (schema 2). Therefore, SMAleft had to be considered in further network analyses. Schema 3 shows the winning model: it demonstrates interaction between dPMCright and vPMCleft with the SMAleft, which has access to the SMCleft (and the IPLleft interacted with the vPMCleft). Abbreviations: fMRI, functional MRI; AO, action observation; IMI, imitation; ROI, region of interest; dPMCright, right dorsal premotor cortex; vPMCleft, left ventral premotor cortex; IPLleft, left inferior parietal lobule; SMCleft, left primary sensorimotor cortex; SMAleft, left supplementary motor area; MG, mirror training group; CG, control training group.
Functional connectivity
The FC of the dPMCright, vPMCleft, and SMCleft revealed a superior FC in a common region (SMAleft) in the MG over the CG. This area was shared in all FC analyses from the dPMCright, vPMCleft, and SMCleft. Consequently, the dPMCright, vPMCleft, and SMCleft showed a pronounced functional interaction with the SMAleft after MTr. These regions were subsequently added to further networking analyses, particularly because FC cannot determine the direction of interaction (Figure 7).
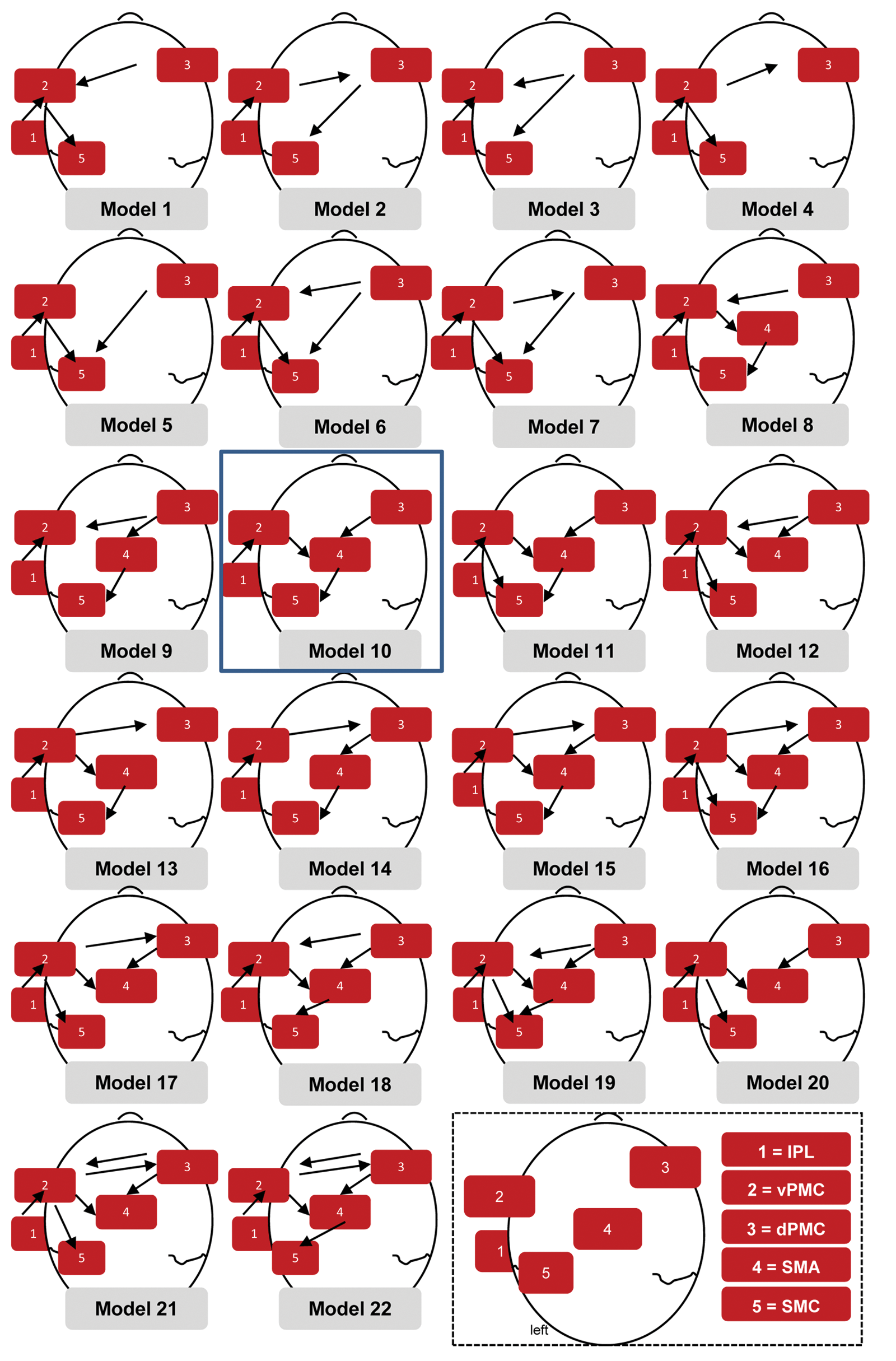
Different models are presented based on different interaction hypotheses among these areas (dPMCright, vPMCleft, IPLleft, SMCleft, and SMAleft). These models were compared by DCM. The “winning” model was the model number 10. Abbreviations: dPMCright, right dorsal premotor cortex; vPMCleft, left ventral premotor cortex; IPLleft, left inferior parietal lobule; SMCleft, left primary sensorimotor cortex; SMAleft, left supplementary motor area; DCM, dynamic causal modeling.
Dynamic causal modeling
Comparing different models (presented in Figure 2), the log-evidence of model 10 had the highest value. In this model, the dPMCright and vPMCleft interacted with the SMAleft, which in turn had access to the SMCleft (and the IPLleft interacted with the vPMCleft).
Discussion
Intensive training of stroke patients with mild to moderate hand paresis effectively promotes hand function recovery (19) in association with plastic changes in the affected SMC (2-3). In patients with severe hand paresis after stroke, effective output from M1 limits its participation in leading to improved function with rehabilitation.20,21 Consequently, the undamaged motor network must support any recovery process. Thus, finding a way to access this undamaged network (eg, the contralesional M1) via a specific therapeutic approach is of paramount importance. MT has been suggested to be an appropriate therapy method to recruit the contralesional M1 8 to promote functional recovery. Although specific evidence is currently lacking, this notion is supported by data from recent clinical studies emphasizing the role of MT in patients with severe hand paresis.22,23
In the present study, healthy participants who trained on simple motor tasks with their right hand with and without a mirror were compared. Performance, measured by the number of pegs and marbles moved, of the trained right hand increased comparatively in both groups. This emphasizes that difficulty and cognitive demand (eg, spatial transformation) of the selected motor skills are comparable in both training groups. Performance of the left, untrained hand increased in CG as well as in MG. It is known that practice of reading Braille improves the performance of the trained and the untrained hand.24,25 This effect on the untrained hand may be based on interhemispheric transfer of information. The neural basis of the interhemispheric transfer is still unclear. It was suggested that during training sessions, a simultaneous passive training of the untrained hand appears (motor overflow). The callosal access concept favors the retrieval of information of the trained hemisphere through transcallosal fibers. 26 But the MG showed a significantly stronger increase in hand performance of the untrained left hand in comparison with the CG.
We detected MTr-specific activation changes within the left ipsilateral SMC by moving the untrained left hand. This finding supports the role for the ipsilateral SMC in MT. The mirror serves as the connecting element by functionally linking the untrained left to the trained right hand.
In a recent fMRI study, an activation shift toward the affected SMC was described in 9 chronic stroke patients after 6 weeks of home-based MT. 9 This shift was contrasted with the lesser shift of a control group that had bimanual exercise. It can be assumed that the lower activation shift of the control group was based on the reported increase of SMC activation within the unaffected SMC, as a consequence of bimanual therapy. Furthermore, along with hand function improvement after MT, active hand movement during fMRI needs to be constant in both performance and effort 27 to allow distinguishing between activation changes induced by neuroplasticity and those induced by accelerated hand movement.
TMS studies have focused on the excitability of M1 corresponding to the hand behind the mirror13-15 (transferring to the current study, only the excitability change over the right M1 was evaluated). It is noteworthy that the excitability of M1 corresponding to the hand behind the mirror was comparable when participants moved their ipsilateral hand, irrespective of whether they observed the moving hand in the mirror.14,15 This finding is comparable with the current activation change within SMCright, where no different activation change was evident between MG and CG.
Using functional and effective connectivity, we decoded the functional information processing to the ipsilateral SMCleft. Both premotor regions on both hemispheres (vPMCleft and dPMCright) increased their FC with the SMAleft. The SMAleft also showed an increase of functional coupling to the SMCleft. In other words, the output function to the left hand was linked to the ipsilateral SMCleft, and its related information was processed within the lateral (vPMCleft and dPMCright) and medial premotor regions (SMAleft). Executing a movement while simultaneously observing an incongruent movement interferes with the performance of the executed movement. 28 It is assumed that movement execution of the paretic hand is improved through interference with the observation of the smooth movement of the unaffected hand in a mirror (giving the illusion of a smooth movement of the paretic hand). Thus, the illusionary visual feedback of the trained right hand is synchronized with initially small or erratic discharges in the brain, which gradually gain significance with ongoing training. Simultaneously, there is a learning effect through the training of the right hand. These signals from the right hand may “gate” motor commands for the untrained left hand. Therefore, representation of the trained motor skills of the illusionary untrained left hand is coded within the contralateral dPMCright. Information processing between the dPMCright and SMAleft increased through MTr. The SMA is known to be involved in bimanual coordination tasks.29-31 Therefore, we suggest that the SMAleft is related to an illusory bimanual movement (a real right hand movement and an illusory left hand movement) and its information conveyed from the right and left premotor regions.
Two visual areas, the superior temporal and superior occipital gyrus, were active when comparing fMRI activation of participants who tapped with the right hand in the mirror versus participants who made the same movement but without a mirror. 12 These regions may play a dominant role in transforming visual information to the motor network responsible for MTr-induced plasticity.
In MTr, the MNS is activated during observation of one’s hand in the mirror (AO) and by repetitive training of motor skills in the mirror. 32 It has been reported that training through AO and execution activates the vPMCleft, bilateral dPMC, and SMA. 33 The vPMC was found to relate to specific individual effectors, and the dPMC was suggested to be responsible for spatial positions for each effector. 33 However, when novel observed actions are learned by imitation, this new motor skill is incorporated into the learner’s own motor repertoire. Consequently, observation of this known action (embodied action) activates MNS-related regions, probably by motor simulation (eg, Cross et al 34 ). Activation within premotor regions decreased after MTr, supporting the notion of learning. 35
Both features of the MNS (AO and imitation) have been used to improve hand paresis in stroke patients, termed video therapy (VT). Patients initially observe a video (AO) and then imitate the observed action. 36 Their activation within the bilateral PMC, bilateral superior temporal gyrus, and SMA is similar to the pattern of activation within the tertiary and secondary motor regions presented here. Thus, both VT and MT functionally activate similar networks but also differ. VT is feasible only in stroke patients with mild to moderate hand paresis. In contrast, MT is particularly beneficial in stroke patients with no or minimal hand function.22,23 Furthermore, whereas VT activates the ipsilesional SMC, MT activates the contralesional (ipsilateral) SMC.
Conclusion
The mirror is the element functionally linking both hands to one (ipsilateral) SMC. Its relevant information is conveyed by networks related to the illusion of bimanual hand movement and to representation of motor skills by the specific training of AO and repetitive hand movement.
Footnotes
Acknowledgements
We are grateful to all the individuals who participated in this study. We thank H.-J. Mast, A. Vuck, J. Wanschura, and M. Tepper for their support during data acquisition.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was partially funded by the European Commission under the 7th Framework Program “Health,” Collaborative Project “Plasticise” (223524).
