Abstract
Background. Several pilot studies have evoked interest in robot-assisted therapy (RAT) in children with cerebral palsy (CP). Objective. To assess the effectiveness of RAT in children with CP through a single-blind randomized controlled trial. Patients and Methods. Sixteen children with CP were randomized into 2 groups. Eight children performed 5 conventional therapy sessions per week over 8 weeks (control group). Eight children completed 3 conventional therapy sessions and 2 robot-assisted sessions per week over 8 weeks (robotic group). For both groups, each therapy session lasted 45 minutes. Throughout each RAT session, the patient attempted to reach several targets consecutively with the REAPlan. The REAPlan is a distal effector robot that allows for displacements of the upper limb in the horizontal plane. A blinded assessment was performed before and after the intervention with respect to the International Classification of Functioning framework: body structure and function (upper limb kinematics, Box and Block test, Quality of Upper Extremity Skills Test, strength, and spasticity), activities (Abilhand-Kids, Pediatric Evaluation of Disability Inventory), and participation (Life Habits). Results. During each RAT session, patients performed 744 movements on average with the REAPlan. Among the variables assessed, the smoothness of movement (P < .01) and manual dexterity assessed by the Box and Block test (P = .04) improved significantly more in the robotic group than in the control group. Conclusions. This single-blind randomized controlled trial provides the first evidence that RAT is effective in children with CP. Future studies should investigate the long-term effects of this therapy.
Keywords
Introduction
Cerebral palsy (CP) is a neurodevelopmental disease related to nonprogressive cerebral abnormalities that occur before birth or early in life, and it affects 2 to 3 children out of every 1000. 1 Children with CP children have hemiplegia, quadriplegia, or diplegia, which could be associated with abnormal sensibility, motor control, strength, and tonus (ie, spasticity) of the upper limb.2,3 These impairments may restrict the functional capacity and participation in activities of daily living (ADL) of a child with CP.4,5
Recent recommendations state that intensive rehabilitation is necessary for improving motor function in children with CP.4,5 These recommendations, based on motor learning theories, suggest that repetitive, goal-directed, assist-as-needed movements that are associated with sensory feedback and an attractive environment are likely to promote reorganization of the neuronal networks (ie, neuroplasticity) and motor development after brain injuries.4,6,7
Robot-assisted therapy (RAT) of the upper limb has the potential to satisfy these recommendations in children with CP.8-12 RAT is conducted using robotic devices that enable the patients to perform specific upper limb movements. 12 The main interest in using robots is to allow the patients to achieve a large amount of movement in a limited time. For instance, children with CP were able to perform 640 movements during 60-minute RAT sessions. 8 Additionally, the attractive human–machine interface has the capacity to motivate the child to perform his or her therapy. 12 This visual interface can be adapted to be kid-friendly through playful games, such as car races,8,10,11 or to perform exercises that mimic ADL, such as reaching for a cup. 10 Moreover, robotic devices allow the patient to receive visual, auditory, or sensory feedbacks.8,11 Finally, the haptic interaction of the robot gives performance-based assistance to the patients.13,14 This assistance can enhance the neuronal plasticity by enabling the patients to initiate and accomplish movements as actively as possible. 13
RAT efficacy has been studied in stroke patients. 15 A recent meta-analysis concluded that RAT could improve upper limb structure and function and the ADL of these patients. Some pilot studies have described the feasibility and interest in using this therapy in children with CP.8,10,11,16,17 However, there are no currently published randomized controlled trials, and a recent review has noted that such studies are needed to confirm the usefulness of RAT in childrent with CP. 4
Pilot studies have investigated RAT efficacy in place of, but not combined with, conventional therapy (CT).8,10,11,16,17 In everyday life, the combination of RAT, involving substantial movement, and CT could enable the therapist to reallocate his or her time and energy to transferring the benefits of these repetitive movements (for instance, motor control improvement in stroke patients) 15 to ADL and patient social integration.
According to these considerations, the purpose of this study was to assess the effectiveness of RAT combined with CT compared to conventional therapy alone in children with CP. This comparison was performed in a single-blind randomized controlled trial. The assessment protocol was in accordance with the 3 International Classification of Functioning (ICF) domains.
Patients and Methods
The ethics board of our Faculty of Medicine approved this study. All parents freely accepted the participation of their children in the study and provided written informed consent.
Patient Selection
Sixteen patients were recruited from a school for children with physical disabilities (Institut Royal de l’Accueil du Handicap Moteur, Brussels, Belgium). This sample size was dependent on the recruitment possibilities in the school. MG and AR enrolled the children. The patients’ characteristics are described in Table 1. The inclusion criteria were a history of CP, a maximum age of 18 years, the ability to understand simple instructions, and moderate to severe impairments of the upper limbs, corresponding to a Manual Ability Classification System (MACS) score greater than 1. The exclusion criteria were epileptic patients and upper limb therapeutic intervention within the previous 6 months, such as a Botulinum toxin injection or neuro-orthopedic surgery. The patients were equally randomized into 2 groups (1:1): a Robotic group and a Control group. A stratified randomization assigned participants to their groups after the first evaluation using a computer-generated random number. The same persons (MG and AR) generated each allocation sequence. The stratification classified the subjects according to their upper limb manual capacity, as assessed by the MACS score (moderate disability, MACS range = 2-3; and severe disability, MACS range = 4-5). The trial was registered at ClinicalTrials.gov, number NCT01700153.
Characteristics of the Subjects Included in the Robotic and Control Groups and Results of the Baseline Measurement Comparison Between the Groups and the Learning Effects of the Upper Limb Kinematics in All Children.
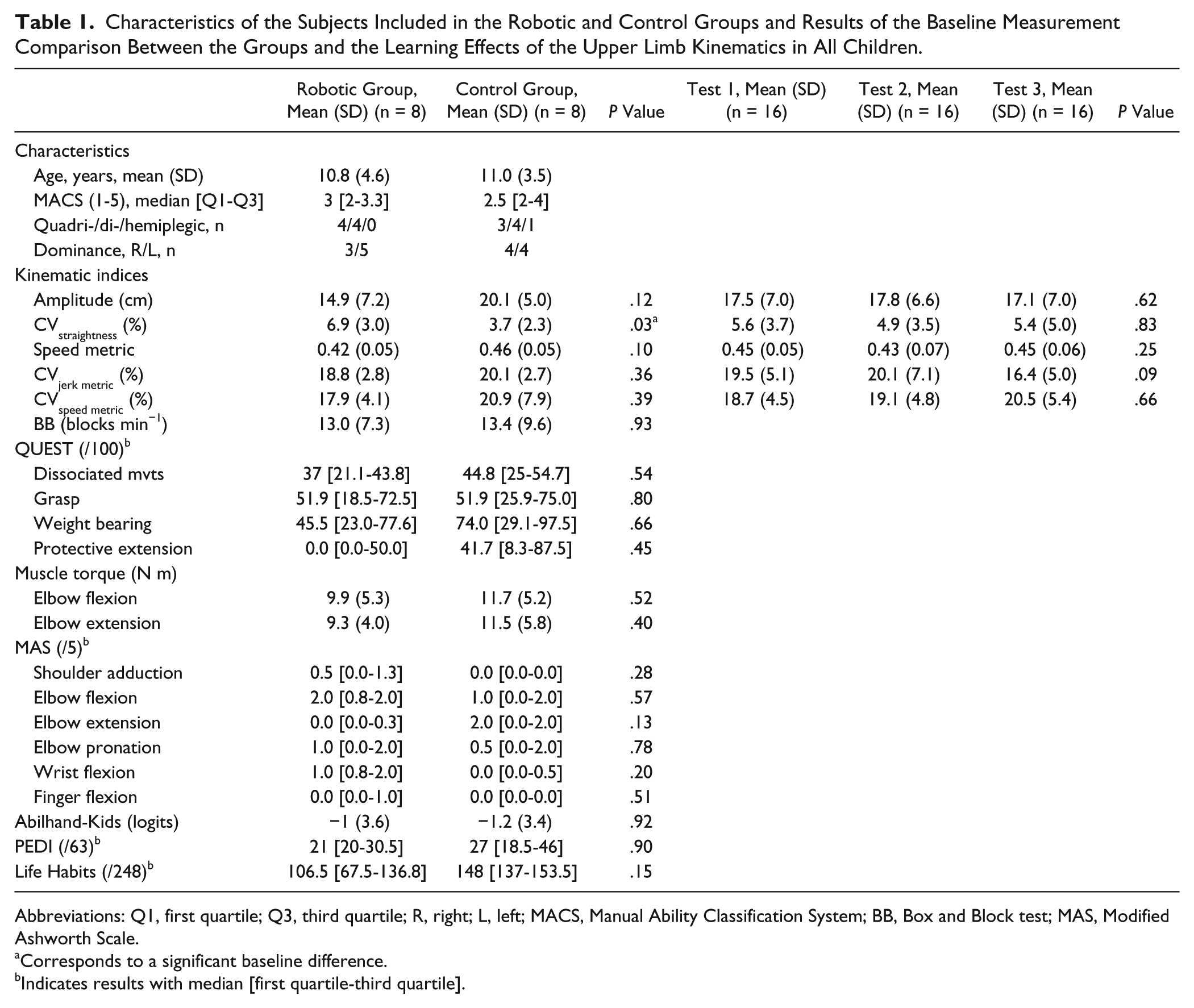
Abbreviations: Q1, first quartile; Q3, third quartile; R, right; L, left; MACS, Manual Ability Classification System; BB, Box and Block test; MAS, Modified Ashworth Scale.
Corresponds to a significant baseline difference.
Indicates results with median [first quartile-third quartile].
REAPlan
The robot used in this study was a robot research prototype named REAPlan, which is illustrated in Figure 1A. 14 REAPlan is an end-effector robot than can move the patient’s upper limb in a horizontal plane via a handle that the patient can grasp or to which he or she may be attached by an orthosis if his or her hand is too weak.

(A) View of REAPlan. (B) Zoomed-in view of the visual interface during a session of robotic-assisted therapy.
REAPlan is fitted with force and position sensors (acquisition frequency = 100 Hz), allowing for control of the lateral (
The patient has to perform the movement along a reference trajectory. This reference trajectory corresponds to the ideal path that the patient must follow to perform the exercise. (
The size of the workspace was adapted to the child’s morphology, within a square that was 0.8 m long on each side. Indeed, this workspace was as large as possible to stimulate the children, with regard to their arm lengths, to perform the largest movements with the robot. A screen and a speaker were installed in the robot to give visual and auditory feedback for performance.
Interventions
Both groups (Robotic and Control) received 5 sessions of therapy per week over the course of 8 weeks (40 sessions in all). Each session lasted 45 minutes. For the Control group, all the sessions were CT. The Robotic group received 2 RAT sessions and 3 CT sessions per week.
The children underwent their CT sessions with their regular physiotherapists and occupational therapists. The physiotherapists practiced neurodevelopmental therapy, and occupational therapists specifically focused on the ADL. The therapists maintained their standard protocols and adapted the rehabilitation to match each child’s needs.
All RAT sessions were supervised by the same physiotherapist (MG), who is experienced with the use of robot. RAT sessions consisted of many duplicate exercises. Each exercise consisted of 160 consecutive movements toward a specific target, as suggested by Fasoli et al, 8 with the REAPlan robotic device (for illustration, see Figure 1B). A force field helped the children reach the targets (see the REAPlan section). The reaching of each target consecutively resulted in audio feedback, the deletion of the target, and the appearance of a new target on the screen. This new target was randomly placed on the visual screen at a distance of 10 cm from the last one. These targets were enlarged for children with visual impairments. For half of the exercises, the target was motionless as long as the patient did not reach it. For the other half, the target was dynamic, moving a distance of 1 cm vertically or horizontally every 0.5 seconds.
The amount of movement, adapted to each patient, was as high as possible to stimulate improvements but was also adapted to the child’s tiredness. Each child could have an optional rest between exercises and during each exercise (of approximately 1 minute). Finally, the RAT sessions were in the form of video games. An avatar (the cursor that the child had to move) and a cartoon animal (the target that the child had to reach) were integrated into an appropriate landscape (Figure 1B). The cartoon animal and its corresponding landscape were changed each week. Finally, at the end of each exercise, a personalized feedback was posted on the visual interface to congratulate the child and give him his time score for achieving the 160 targets.
ICF Assessment
All the children were assessed before and after the intervention through a protocol that took into account the 3 domains of the ICF.
The primary outcome was upper limb kinematics. Gilliaux et al18,19 provided a standardized protocol to quantitatively assess active movements of the upper limb in stroke patients, including several kinematic indices. The short version of this protocol (5 indices) was used for this study. This protocol consisted of performing 4 different tasks (Free Amplitude, Target, Square, and Circle), as described below.
For the Free Amplitude task, the subject had to reach straight out in front of them as far as they could and brought the arm back to the starting position. For the Target task, the subject made movements in the most precise and direct manner toward a specific target placed at a distance of 10 cm in front of the subject. After performing this task, the robot brought the subject’s arm back to the starting position. For the Square and Circle tasks, the subject had to draw 2 geometrical shapes: a square of 6 cm side and a circle of 4 cm radius. These shapes were performed clockwise with the right upper limb, and counterclockwise with the left one. These tasks were performed with REAPlan, without any assistance (ie, no interaction forces) and at spontaneous speeds.
For the Free Amplitude task, the computed indices were the amplitude and the coefficient of variation (CV) of the straightness. For the Target task, the speed metric index was calculated. For the Square and Circle tasks, the CVjerk metric and CVspeed metric indices, respectively, were computed. Each index of this short protocol was computed for each of the 10 cycles of movement and was then averaged.
The kinematic assessment started after a 10-minute training phase to limit learning bias. For the acquisition phase, the order of tasks was randomly assigned. Each task was performed 10 consecutive times, during which the end-effector position was recorded (acquisition frequency = 100 Hz). The rest period between tasks was 1 minute. This kinematic assessment was performed 3 times within the 2 weeks preceding the intervention to evaluate a possible learning effect of the protocol and then once after the intervention. The same blinded physiotherapist (DD) performed each kinematic assessment.
For the secondary outcomes of the body structure and function domain of the ICF, the assessment included the Box and Block test (BB) 20 ; the 4 subscales (dissociated movements, grasps, weight bearing, and protective extension) of the Quality of Upper Extremity Skills Test (QUEST)21,22; the Modified Ashworth Scale (6 muscular groups were tested: shoulder adductors, elbow flexors and extensors, pronators, wrist and finger flexors) 23 ; and the strength of 2 muscular groups (elbow flexors and extensors), assessed with a hand-held dynamometer (Microfet2TM, Orsay, France). 24 For the calculation of muscle torque, the result obtained with the dynamometer was multiplied by the distance measured between the lateral epicondyle and radial styloide. 25 All these assessment tools were reliable and valid for the studied population 26 and were used by the same blinded occupational therapist (DH).
The secondary outcomes of the activity and participation domains of the ICF correspond to 3 questionnaires. For the activity domain, the French versions of the Abilhand-Kids 27 and Pediatric Evaluation of Disability Inventory (PEDI) 28 questionnaires were filled out by each child’s therapist. For the participation domain, the French version of Life Habits was completed by each child’s parent. 29
Statistics
Statistical tests were performed using SigmaStat 3.5 software (WPCubed GmbH, Munich, Germany). For tests with parametric measures, the normal distribution and equality of variance were verified for all comparisons. For each test, the significance level was .05.
For each parametric and nonparametric measure, a 1-way analysis of variance (ANOVA) or a Mann–Whitney test was performed to verify the parity of the baseline results between groups. To verify the learning effect of the primary outcome, a 1-way repeated ANOVA was performed for each kinematic index on the 3 measures computed before the intervention.
For each parametric variable, a 2-way repeated ANOVA was performed to analyze the interaction between the time (before intervention vs after intervention) and groups (Control group vs Robotic group). For each significant interaction, a Bonferroni-adjusted post hoc (Holm Sidak) test was used to analyze the differences in the change between groups. For each nonparametric variable, a Mann–Whitney test and a Wilcoxon test were performed to analyze the treatment effects between groups and within each group, respectively.
Results
The recruitment and baseline assessments were performed in September 2012, and the interventions were started in October 2012. The final interventions were completed in December 2012. The final assessments were performed between 1 and 7 days following the final rehabilitation session. The flowchart of this study is illustrated in Figure 2.
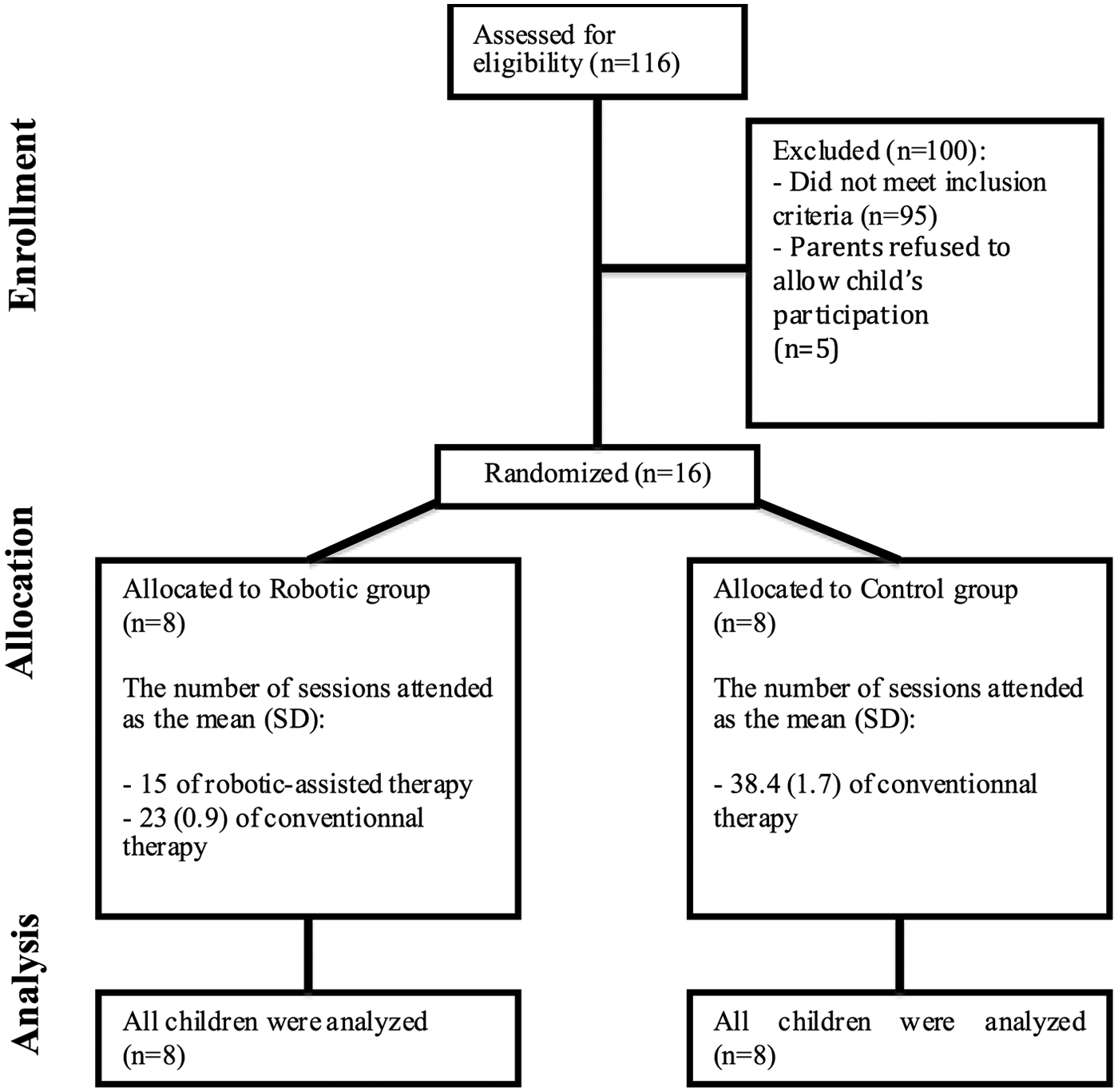
Flow diagram of the participants through each stage of the study (ie, Enrollment, Allocation, and Analysis).
All 16 patients completed the study. During each RAT session, the patients performed 744 (224) (mean [SD]) movements on average with the REAPlan. For the Robotic group, the children performed 15 (0) sessions of RAT and 23 (0.9) sessions of CT. For the Control group, the children received 38.4 (1.7) sessions of CT. No adverse events were reported. All results are presented in Tables 1, 2, and 3 and are illustrated in Figure 3.
Two-Way ANOVA Results for Parametric Variables Comparing the Treatment Effects Between the Robotic and Control Groups.
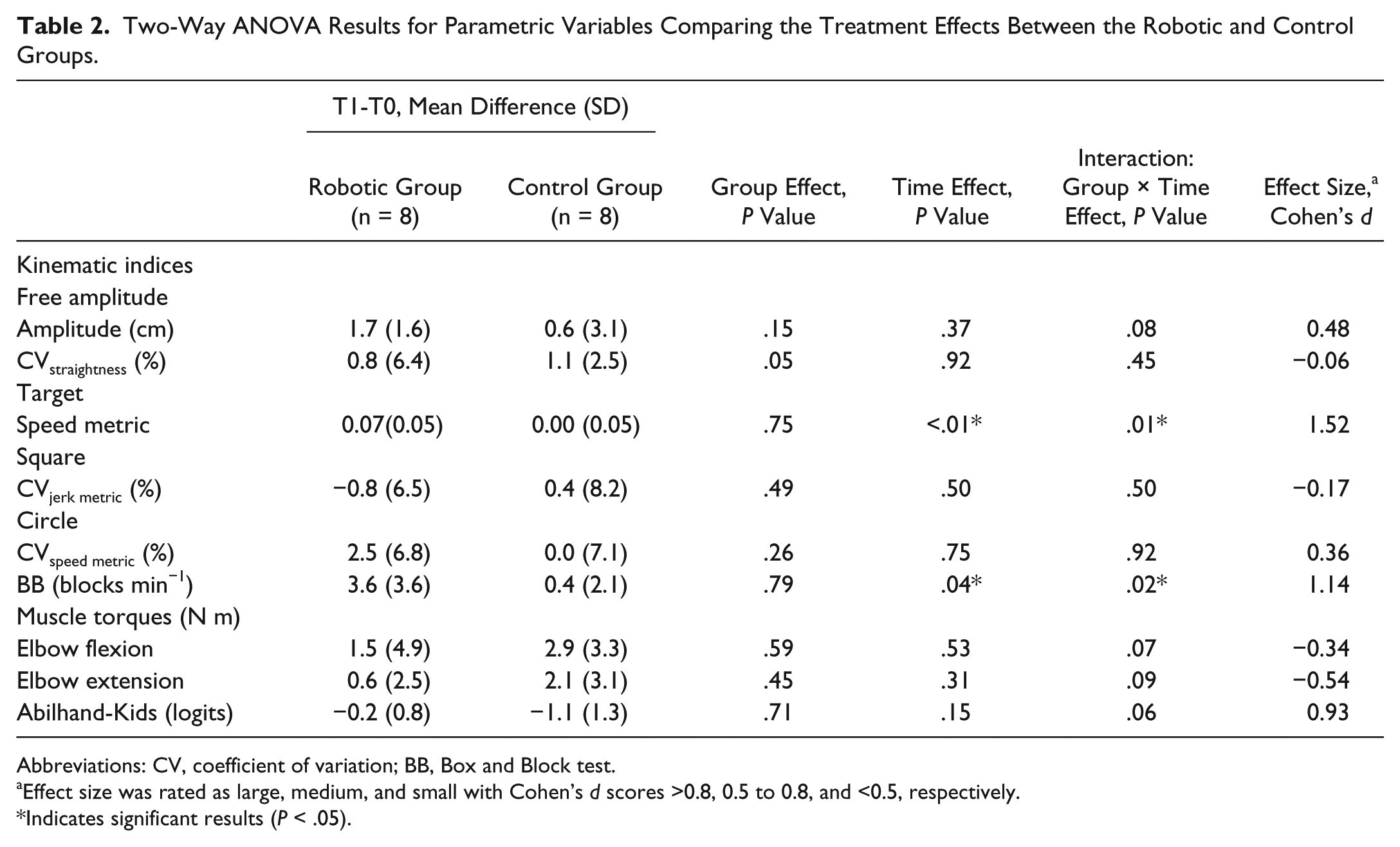
Abbreviations: CV, coefficient of variation; BB, Box and Block test.
Effect size was rated as large, medium, and small with Cohen’s d scores >0.8, 0.5 to 0.8, and <0.5, respectively.
Indicates significant results (P < .05).
Results of the Mann–Whitney and Wilcoxon Tests for Nonparametric Variables Comparing the Treatment Effects Between the Robotic and Control Groups and Within Each Group.
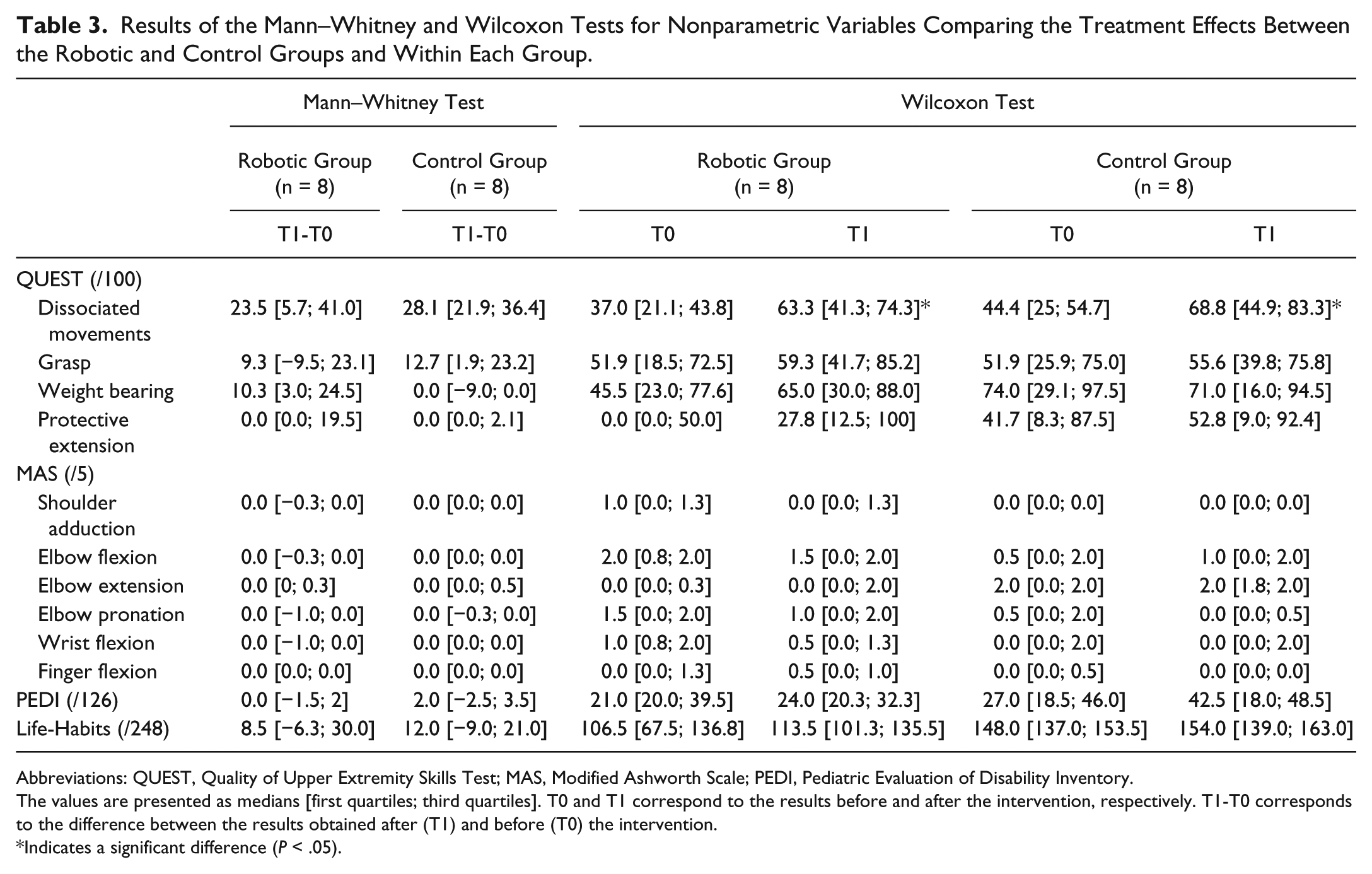
Abbreviations: QUEST, Quality of Upper Extremity Skills Test; MAS, Modified Ashworth Scale; PEDI, Pediatric Evaluation of Disability Inventory.
The values are presented as medians [first quartiles; third quartiles]. T0 and T1 correspond to the results before and after the intervention, respectively. T1-T0 corresponds to the difference between the results obtained after (T1) and before (T0) the intervention.
Indicates a significant difference (P < .05).
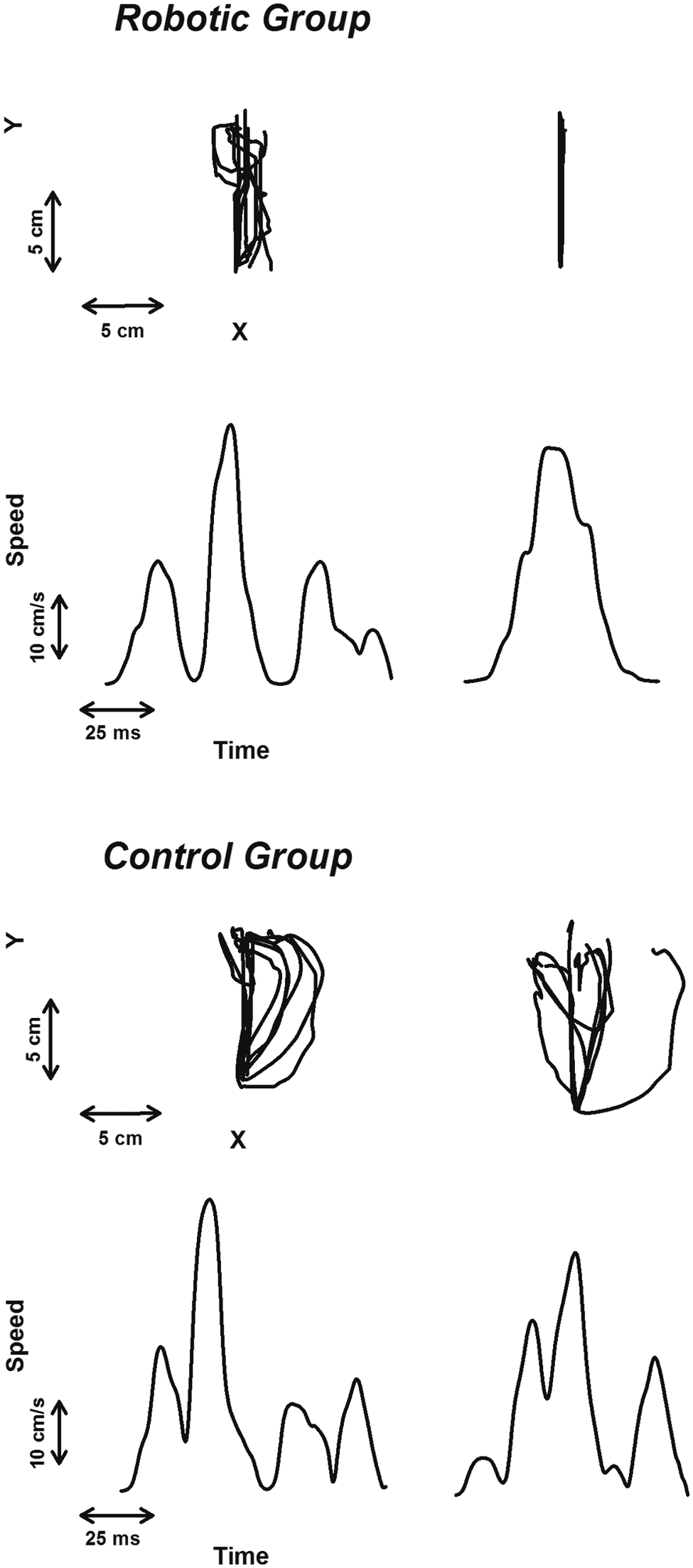
For the target task in each group (Robotic and Control), illustration of the typical traces computed for one child before (left graphs) and after (right graphs) treatment.
Similarity Between Groups at Baseline and Learning Effect
Before the interventions, the results of the kinematic indices were similar between groups, except for the CVstraightness of the Free Amplitude task (P = .03; Table 1). There was no learning effect for the primary outcome (Table 1). Indeed, for all merged tasks, the kinematic indices were similar within the 3 measures computed before the intervention (P > .09). For this reason, the average of these 3 kinematic measures was considered to be the baseline results.
Effect of Therapy
For the body structure and function domain, an interaction between the time and groups revealed that the smoothness in discrete and unidirectional upper limb movements only improved in children who received RAT (P < .01; Table 2). Indeed, for the Target task, the speed metric index increased from 0.42 (0.05) to 0.49 (0.03) in the Robotic group, but this index score did not change in the Control group (0.46 [0.05] to 0.46 [0.06]). For the 3 other tasks (Free Amplitude, Square, and Circle), the kinematics indices did not change after intervention (P > .05).
An interaction between time and groups showed that the manual dexterity of the upper limb improved significantly more in children who received RAT than children who only received CT (P = .04; Table 2). Indeed, the BB score improved from 13.0 (7.3) to 16.6 (9.9) blocks/min in the Robotic group, while this score increased only slightly from 13.4 (9.6) to 13.8 (9.7) in the Control group.
The capacity to perform analytical movements of the upper limb similarly improved in both groups (P < .05; Table 3). Indeed, the scores of the dissociated movements subscale of the QUEST significantly increased for the Robotic (median increased from 37.0 to 63.3/100) and Control (median increased from 44.4 to 68.8) groups (P < .04). However, these improvements were not different between the groups (P = .87; Table 3).
There was no significant effect of treatment for the other scales and for the 3 questionnaires assessing the activity and participation domains (P > .06; Tables 2 and 3).
Discussion
The aim of this study was to compare the effect of conventional therapy (CT) to the combination of RAT and CT in children with CP in a single-blind randomized controlled protocol. This comparison took into account the 3 domains of the ICF. 30
Body Structure and Function Domain
Some upper limb kinematic indices, assessed by the robot REAPlan, and manual dexterity, assessed by the Box and Block test, had significantly more improvement after RAT and CT than after CT alone. Both results suggest a motor learning effect. 7 Indeed, the RAT of this study consisted of repetitive discrete movements (reaching targets), and the observed improvements were specifically related to discrete movements (Target Task of the kinematic assessment and BB).
The assessment protocol of this study followed current recommendations. Indeed, kinematics was chosen as a primary outcome to quantitatively and objectively assess upper limb movements, 31 avoiding the disadvantages (eg, nonparametrical statistics) of qualitative, subjective, and ordinal scales.31,32 After that, this protocol was established to be easily reproduced in clinical routines. Because a robotic device, such as the REAplan, has the potential to rehabilitate and assess patients, 19 we suggest that combining both abilities in one tool is more advantageous in clinical routines than adding other kinematic assessment tools, such as an expensive optoelectronic system.
However, one can argue that the kinematic improvement observed in the Robotic group could be related to the child’s learning of the specific robot tasks. Even so, the 3 kinematic assessments performed before the intervention did not show any leaning effect. More important, the Robotic group transferred the improvement to a more functional task (BB) that was not directly related to robot therapy. Indeed, improvement in the BB test showed a high effect size (Cohen’s d = 1.1), which suggests that RAT can significantly influence gross manual ability in children. However, although minimal detectable change in CP is unknown, the change measured (3.6 blocks/min) is below the known minimum for stroke patients (6 block/min) 33 and cannot therefore be assumed to represent a meaningful improvement. These results are consistent with an observational study that has shown kinematic improvements after RAT in children with CP. 34 Finally, randomized controlled trials assessing RAT efficacy in stroke patients have also shown improvements in kinematics and manual dexterity, as assessed by a robot35,36 and the BB, 37 respectively.
The dissociated movements of the QUEST showed improvements in both groups, but no difference between groups. Previous observational studies identified significant improvements on this same subscale after RAT.8,34 The present results illustrate the necessity of a control group in clinical trials to avoid misinterpretation of the results. 38 The improvement observed in both groups could be explained by the fact that this study started after the summer holidays (lasting 2 months), during which most children did not have rehabilitation. This observation suggests that CT, with or without RAT, could preserve the children’s capacity to dissociate upper limb movements.
Activity and Participation Domains
The improvements of impairments after only 8 weeks of RAT seem promising. However, these improvements did not translate into improved ADL. This result is disappointing because the patients yearn to improve their capacity for ADL as well as their social integration. These results can be explained by the following hypotheses. First, the various exercises were designed to stimulate the patients to repeat discrete reaching movements. However, ADL involves discrete reaching movements (eg, pushing on a light switch) along with rhythmic reaching movements (eg, washing the upper body) and grasping movements (eg, open a bag chips) (for these examples, see Abilhand-Kids 27 ). Further studies should expand the exercises to enable the patients to repeat a wide variety of movements (eg, rhythmic, discrete, with or without hand implication), as suggested by Krebs et al. 34 Moreover, these exercises could be in the form of ADL 10 or serious games. 39 Second, the activity and participation assessments were presented in the form of questionnaires completed by parents and therapists.27-29 Because the parents and therapists were not blinded, their judgment could have been altered. To increase the responsiveness of this activity assessment, future studies should also use tools to assess the child’s performance in ADL, such as the Melbourne Assessment of Unilateral Upper Limb Function 40 or the Assisting Hand Assessment. 41
Limitations and Perspectives
This study lays the groundwork for future research on the use of RAT in CP.
Even though the sample size was sufficient to show significant results, it was too small to generalize the results to the clinical setting and to determine the subgroups of children who will be more responsive to RAT. Finally, the sample size did not allow the stratification to take other factors, such as the patients’ ages, into account. This bias was limited because the mean age of each group was similar (Table 1). Then, further multicenter trials should be planned to (a) confirm these results with a larger sample and variety of settings 38 ; (b) add other stratification factors, such as the patients’ age, since learning capacities and video games playing experience are not the same among young children and adolescents; and (c) establish the correlations between the improvements and the children’s characteristics (eg, impairment severity, age, etc).
In this study, the proportion of robotic sessions (2/5) was limited by the feasibility of performing the study at school, and the number of RAT sessions (n = 15) was chosen in agreement with previous studies.8,11,13 However, we still do not know whether more intensive use of the robot or the use of RAT over a longer period of time would yield better results. Additionally, we do not know whether similar results could be obtained with reduced use of the robot. This issue could be addressed through a study conducted over a longer period of time with a regular evaluation of the evolution of the patients’ function instead of only at the beginning and after therapy.
This study assessed the effect of RAT directly after therapy but not a few months later for logistical reasons. There was no long-term follow-up; therefore, the results do not provide any indication that benefits are maintained or of the necessity to repeat RAT regularly or to use it continuously. This limitation could be addressed by evaluating the evolution of improvements over time, as Krebs et al 34 have shown for kinematics.
Finally, Sakzewski et al 5 are interested in combining botulinum toxin with other upper limb therapies. Future studies should evaluate the effectiveness of the combination of RAT with botulinum toxin injection in the upper limb. This combination showed promising results in a pilot study and could maximize the improvements to upper limb impairments and activities after RAT. 42
Conclusions
In conclusion, this study is the first single-blind randomized controlled trial to assess the efficacy of RAT in children with CP. This therapy improved upper limb kinematics and manual dexterity but did not improve functional activities and social participation. Further studies should confirm these preliminary results on larger populations and assess if RAT could lead to more functional improvements in the long term. The REAPlan robotic device provides an intensive, goal-oriented, and assist-as-needed therapy associated with motivational and performance feedbacks. Robotic devices offer children fun and intensive rehabilitation that a human therapist cannot provide. These robots can be easily integrated as a relevant complement to therapy in the clinical setting.
Footnotes
Acknowledgements
The authors would like to thank Catherine Hereng, Catherine Gillain (Institut Royal de l’Accueil du Handicap Moteur, Brussels, Belgium), Martin Vanderwegen, and the Van Haesendonck Company for their collaboration, along with all the children who participated in this study. The authors would also like to thank the Mushroom studio (Belgium) for the graphical conception of the video game integrated in the robot.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Saint-Luc Foundation (Brussels) supported all phases of this study.
