Abstract
Background. High-intensity repetitive training is challenging to provide poststroke. Robotic approaches can facilitate such training by unweighting the limb and/or by improving trajectory control, but the extent to which these types of assistance are necessary is not known. Objective. The purpose of this study was to examine the extent to which robotic path assistance and/or weight support facilitate repetitive 3D movements in high functioning and low functioning subjects with poststroke arm motor impairment relative to healthy controls. Methods. Seven healthy controls and 18 subjects with chronic poststroke right-sided hemiparesis performed 300 repetitions of a 3D circle-drawing task using a 3D Cable-driven Arm Exoskeleton (CAREX) robot. Subjects performed 100 repetitions each with path assistance alone, weight support alone, and path assistance plus weight support in a random order over a single session. Kinematic data from the task were used to compute the normalized error and speed as well as the speed-error relationship. Results. Low functioning stroke subjects (Fugl-Meyer Scale score = 16.6 ± 6.5) showed the lowest error with path assistance plus weight support, whereas high functioning stroke subjects (Fugl-Meyer Scale score = 59.6 ± 6.8) moved faster with path assistance alone. When both speed and error were considered together, low functioning subjects significantly reduced their error and increased their speed but showed no difference across the robotic conditions. Conclusions. Robotic assistance can facilitate repetitive task performance in individuals with severe arm motor impairment, but path assistance provides little advantage over weight support alone. Future studies focusing on antigravity arm movement control are warranted poststroke.
Stroke is a leading cause of long-term disability in the United States, and hemiparesis is the most common motor impairment, which can have profound deleterious effects on functional mobility of the upper limb and significantly compromise quality of life. 1 It has been suggested that high-intensity repetitive training with close-to-normal movements can promote activity-dependent plasticity and improve motor skill.2-5 While it is possible to provide 300 movement repetitions in 1 hour for individuals with chronic upper limb hemiparesis, to approach the dosing in animal studies, 6 it is particularly challenging to provide this to individuals with significant poststroke weakness and spasticity. 7 As a result, the number of repetitions in typical practice settings tends to be extremely low. 8 Attempted movements of the upper limb against gravity in more impaired individuals may induce synergistic movements of several joints in a characteristic abnormal pattern.9-11 Repetitive training using such synergistic movements reinforces these abnormal patterns and could be detrimental over the long term. 12
Robotic devices have been designed for upper limb rehabilitation and provide an opportunity to explore fundamental biomechanical principles that can lead to better strategies to improve upper limb function. Robotic approaches can facilitate repetitive training by unweighting the limb and/or by improving trajectory control of the limb, but it is not known to what degree weight support and path assistance facilitate movement control, and if the extent of motor impairment leads to different responses to these aspects of robotic assistance. This is especially important as studies have shown that the response to treatment may differ substantially based on participant characteristics, type of treatment, intensity, and duration (for reviews, see Mehrholz et al13-16).
The purpose of this study was to examine the extent to which robotic weight support alone, path assistance alone, and path assistance plus weight support facilitate repetitive upper limb movement in patients with various degrees of poststroke arm motor impairment relative to healthy controls. A 3D circle-drawing task was chosen because it engages a wider range of upper limb muscles, better mimics real-life tasks, and provides the opportunity to examine the effect of abnormal motor synergies on function. 17 We chose a novel exoskeletal robot, the Cable Driven Upper Arm Exoskeleton (CAREX), which suspends the arm using a robot-controlled cable system, making it 10 times lighter and more versatile than other exoskeletal robotic rehabilitative devices.18-20 In a preliminary study, the CAREX was found to promote near-physiological muscle patterns, similar to those observed during movements without CAREX. 21 The CAREX can provide assist-as-needed force fields to keep the upper limb within the desired path of motion, and/or adjustable weight support to the arm to eliminate the effect of gravity, both of which can facilitate 3D task performance in individuals with poststroke hemiparesis. Furthermore, path assistance and weight support can be provided separately or may be combined. Performance on the 3D circle-drawing task can be quantified by examining the movement error from the target path, the speed of the movement, as well as the speed-error relationship.
We hypothesized that higher functioning individuals with lower arm motor impairment would show smaller movement error at higher preferred speeds compared with lower functioning subjects with higher arm motor impairment, on the 3D circle-drawing task. We also hypothesized that path assistance would facilitate trajectory control and task performance in higher functioning individuals with stroke, whereas both path assistance and weight support would be required in low functioning individuals. In addition, we sought to assess the relationship between measures of arm motor impairment and task performance metrics.
Materials and Methods
Subjects
Seven healthy control subjects and 18 subjects with chronic poststroke right-sided hemiparesis completed the study (Table 1). All subjects provided written informed consent approved by the institutional review board. The inclusion criteria were as follows: (1) radiologically verified stroke at least 4 months previously; (2) right-handed subjects with or without right hemiparesis (as robot is built for the right arm alone); (3) preserved passive range of motion at upper limb joints involved in the movement; and (4) spasticity <3 on the Modified Ashworth Scale. Subjects were excluded from this study if they had (1) previous injury to upper limb; (2) significant medical comorbidities; (3) abnormal involuntary movements that could endanger subject in robot; (4) fixed contractures; and (5) excessive spasticity or other condition that led to inability to comply with or complete the protocol.
Subject Demographics.
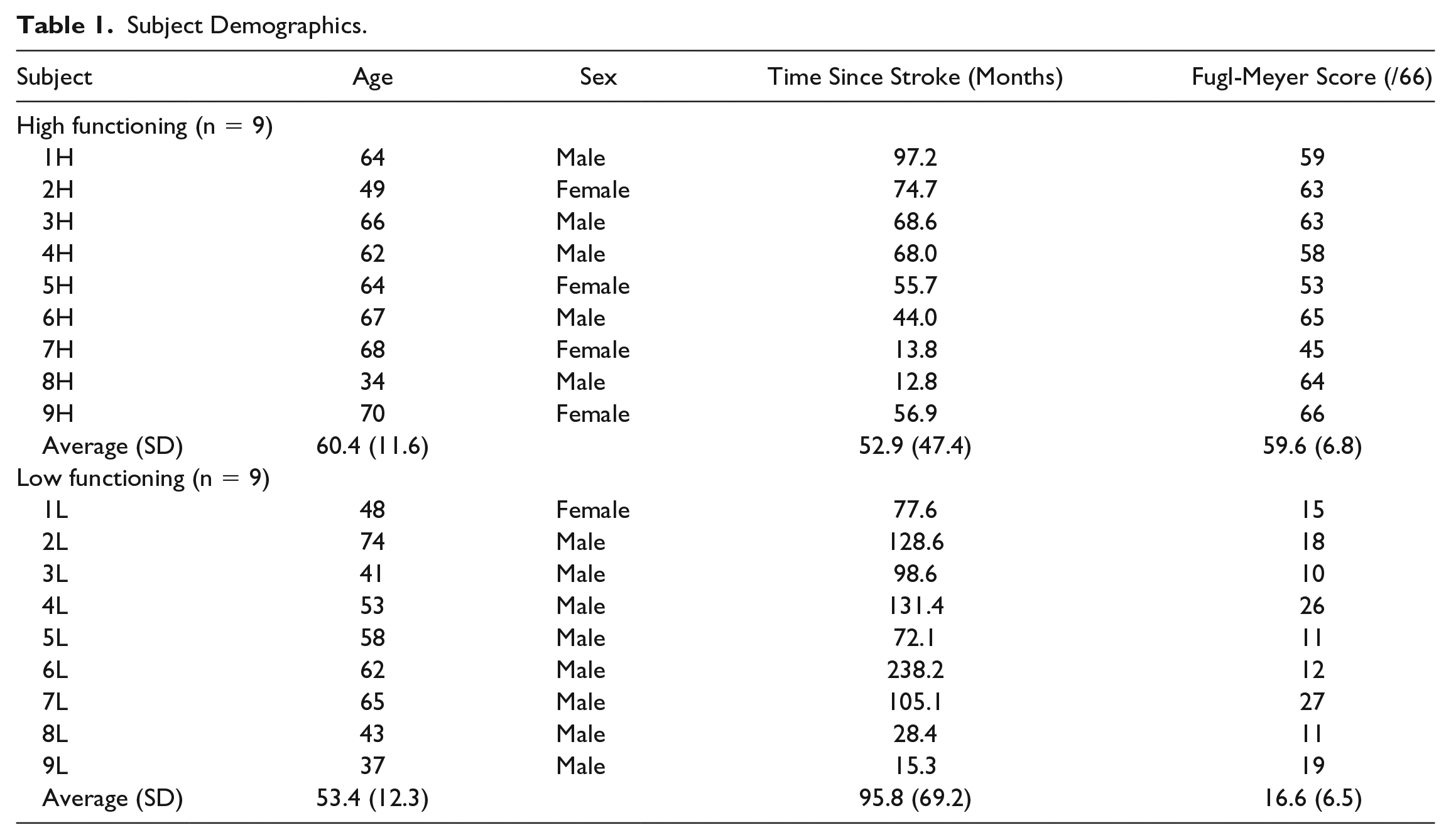
Apparatus
A 3D Cable-driven Arm Exoskeleton (CAREX) device was used in this study. The CAREX consists of 3 lightweight cuffs, one on the shoulder, one on the arm, and one on the forearm attached to motor-controlled cables, which facilitate engagement of the entire upper limb as shown in Figure 1A. The shoulder cuff is fixed to a chair on which the subject sits. Seven cables, routed through the shoulder cuff, provide weight support and/or path assistance to the arm during movement. Five cables are connected to the upper arm cuff, and the remaining 2 cables are routed through the upper arm cuff to the forearm cuff. The cables are driven by motors placed on a frame around the CAREX. As the cables are pulled using motors, torques are generated at both the shoulder and elbow joints. The CAREX can control all 3 degrees-of-freedom at the shoulder joint and 1 degree-of-freedom at the elbow joint (Figure 1B). A load cell (MLP50, Transducer Techniques, Temecula, CA) at the end of each cable measures the tension of the cable for automatic adjustment of cable-tension to prevent sagging of the cables. An inertial measurement unit (IMU) NV-100 (VectorNav Technologies, Dallas, TX), attached to the upper arm cuff, measures the shoulder angles.19,20 The upper arm cuff is connected to the forearm cuff by a hinge joint. A digital encoder (US Digital, Vancouver, WA) mounted on the hinge joint measures the elbow angle. The exoskeleton is controlled by a PXI controller (National Instruments, Austin, TX) to provide assist-as-needed force fields for path assistance and/or weight support.18,20
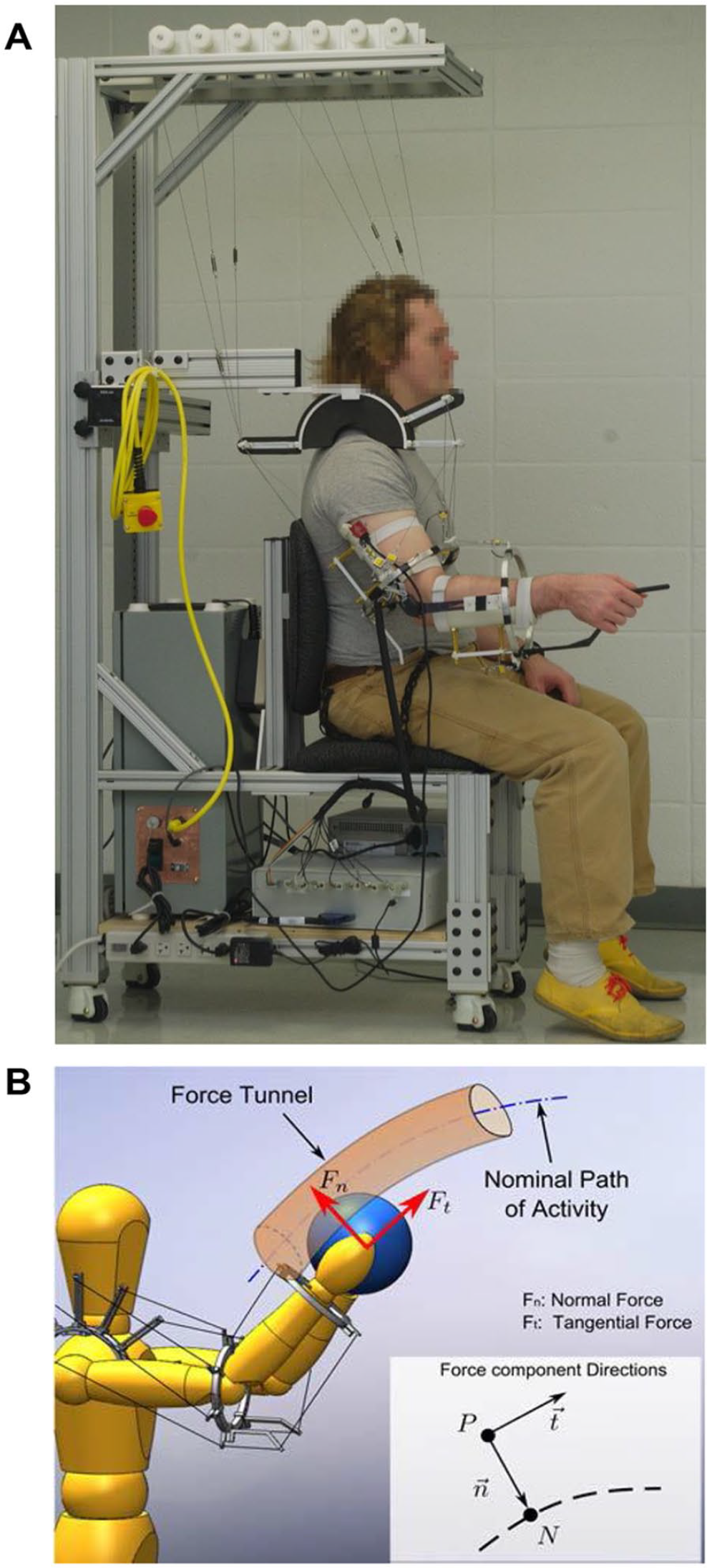
(A) Picture of CAREX used in the study. (B) Illustration of path assistance using CAREX created by a force tunnel that prevents the user’s hand from deviating from the nominal path.
Protocol
Subjects with stroke were assessed on clinical metrics which included measurement of upper limb motor impairment using the Fugl-Meyer Scale (FMS), 22 strength testing using the Manual Muscle Test (MMT), 23 active range of motion (AROM) measured by video motion analysis using Dartfish,24-26 and muscle stiffness or the resistance to passive joint movement measured using the Modified Ashworth Scale (MAS). 27
Circle-Drawing Task
The study was designed to test repetitive 3D task performance with the CAREX under 3 distinct conditions in both healthy subjects and in subjects with poststroke hemiparesis. For the experimental task, subjects were asked to complete a 3D circle-drawing task along a prescribed circular path with a constant diameter of 15 cm, at their preferred speed. Each subject was instructed to draw the circle through the 3 points of a triangle placed in front of their right arm (the template) as accurately as possible (Figure 2A). No feedback was provided to the subjects on their performance. We specifically chose the circle-drawing task to evaluate upper limb motor learning poststroke because it requires control of the arm in multiple directions, and the work-space covered by the circle is relevant to activities of daily living.17,28,29 In addition, the CAREX facilitated circle drawing in 3D space. Subjects sat on the CAREX chair and their torso was secured with a seat belt. The right arm was secured onto the upper arm and forearm cuffs of the CAREX. Each subject performed 3 sets of the circle-drawing task under 3 randomly assigned conditions: (a) path assistance alone (PA), (b) weight support alone (WS), and (c) path assistance and weight support combined (PA+WS). In the weight support conditions, the weight of the arm was estimated based on the height and weight of each subject. Subjects completed a total of 10 trials: 5 clockwise and 5 counterclockwise, each consisting of 10 circles. This amounted to drawing 100 circles for each condition, and a total of 300 circles during a single session, a training dose that was found to be necessary for motor learning in animal models and that is achievable in humans. 6 To reduce fatigue, rest breaks were provided between trials and between conditions. Kinematic data of the actual path drawn during each trial were acquired and processed using LabView (National Instruments, Austin, TX). Task performance was measured only with the robot under the various conditions. We did not test task performance without the robot as the low functioning patients could not perform the task without the robot. In addition, since the robot was used to measure performance, the measurements could not be made in a comparable manner without the robot.
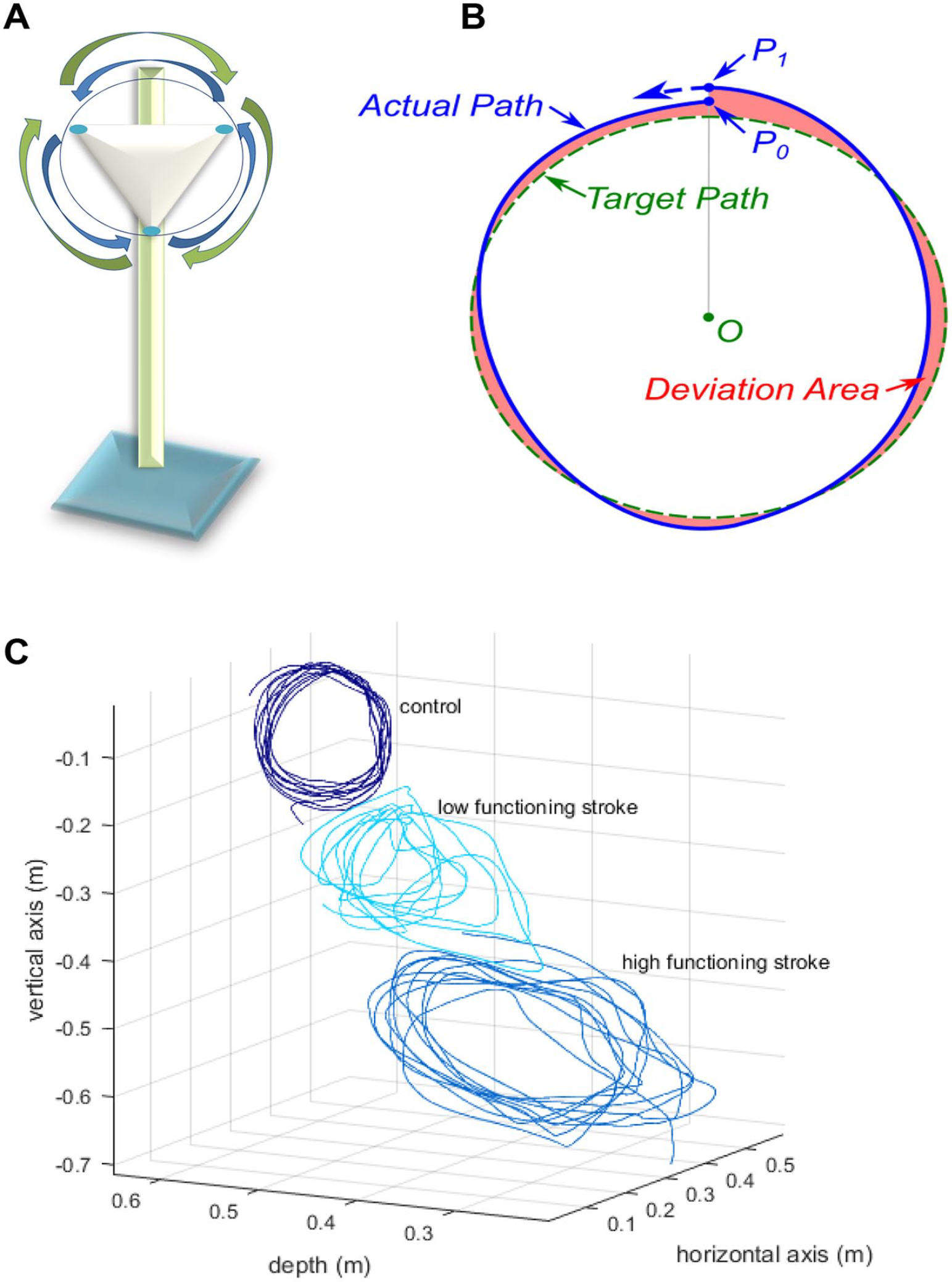
(A) Graphical representation of the circle-drawing apparatus with clockwise and counterclockwise arrows. (B) Illustration of the target path, actual path, and deviation area to compute the normalized error and speed metrics of task performance. (C) Representative circles drawn by a control subject, a low functioning stroke subject (Fugl-Meyer score = 19), and a high functioning stroke subject (Fugl-Meyer score = 59). The higher or lower location of the circle depends on the height of the subject.
Data Processing and Statistical Analysis
The 25 subjects were classified into 3 groups prospectively, as per the study design, according to their level of impairment—the control group, the high functioning group (FMS = 45-66), the low functioning group (FMS = 10-27). The cutoffs for the impairment groups are supported by prior studies. 30 Statistical analyses were conducted using RStudio (version 1.0.143). The significance level for all tests was set at 5%. We compared the demographic (age and time since stroke) and clinical variables (FMS, strength, active range of motion, and muscle stiffness) between the low functioning and high functioning groups using t tests.
Two main kinematic outcome measures were calculated from the circles drawn: the normalized error and speed. The green dashed line in Figure 2B represents the target path, that is, the prescribed circle. The subjects drew 10 circles continuously during each session to track the target path. This continuous path drawn by the subject was divided into 10 independent circles starting at the top of the circle, P0. The solid line represents one circle drawn by the subject, which started at P0 and ended at P1. This solid line is referred to as the actual path. The length of the actual path from P0 to P1 represents the trajectory of the actual path, drawn in time (t). The shaded area between the target path (dashed line) and the actual path (solid line) represents the deviation area of the actual path from the target path. The normalized error was calculated as the deviation area divided by the length of the actual path of the circle drawn. 1 The speed was calculated as the length of the actual path of the circle drawn divided by time it took to draw that circle.

Linear mixed effect models20,21 using package nlme (RStudio) were used to test the relationship between the normalized error/speed and the conditions, groups, and number of trials as shown in Equations (2) and (3). The relationship between the normalized error and the speed was assessed using Equation (4)
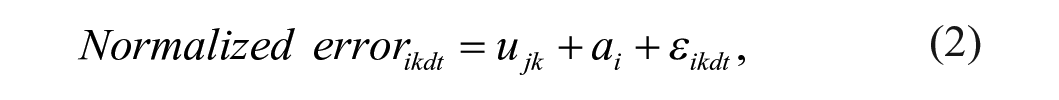
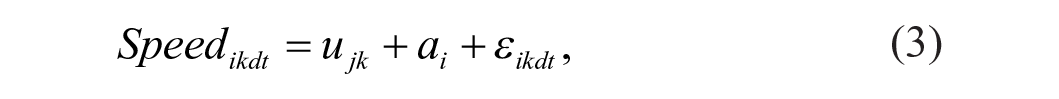
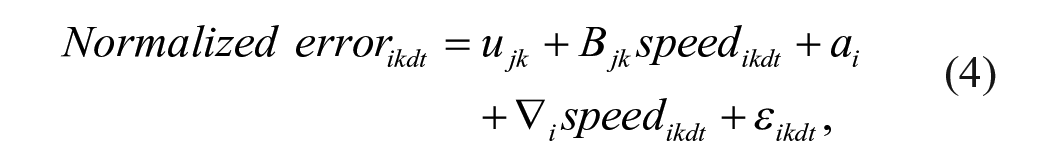
where speedikdt and normalized errorikdt denote the recorded speed and error values for the ith (i = 1, . . . , 25) subject under kth (k = 1, 2, 3; PA, WS, PA+WS) condition, t is the trial number and ε ikdt is the corresponding residual term, and d is direction (d = 1, clockwise and 2, counterclockwise). In Models (2), (3), and (4), ujk is the group and condition specific effect, j = 1, control; 2, high functioning; and 3, low functioning with respect to the outcome of interest. To control for the impact of individual variability, we modeled the subject specific intercept, ai, as a random effect with normal distribution ai ∼ N(0, σ a 2). In Model (4), Bjk is the slope coefficient associated with speed at condition k and for group j. Besides the random individual specific intercept ai, in this model we further considered an individual specific slope coefficient ∇ i for speed as a random effect with normal distribution ∇ I ~ N(0, σ ∇ 2). By incorporating random intercept ai and random slope ∇ i , we accounted for individual mean normalized error and speed (Models 2-4), and individual speed-error response patterns (Model 4). We did not find any trend over trials or direction hence these effects are not modelled in these analyses. Since the linear mixed effect model is estimated based on maximum likelihood, we conducted pair-wise comparisons using the likelihood ratio test (LRT) to assess the effect of group and condition.
Finally, to understand the relationship between movement impairment and performance on the circle-drawing task, we correlated the FMS scores and the active range of motion measurements with the mean normalized error and speed across all robotic conditions using Pearson’s correlation.
Results
The degree of motor impairment, as measured by the upper limb FMS scores, was significantly different between the high functioning (n = 9, FMS = 59.6 ± 6.8) and low functioning (n = 9, FMS = 16.6 ± 6.5) groups (P < .0001; Table 1). There was no significant difference in the age of the subjects in the 2 groups (P = .23). The time since stroke was longer in the low functioning group, but this was not statistically significant (P > .05). Clinical metrics, that is, active range of motion (AROM), muscle stiffness (MAS), and strength (MMT) at the shoulder, elbow, forearm, and wrist for the 2 groups are summarized in Table 2. Strength was significantly higher for all movements in the high functioning group compared to the low functioning group (P < .01). Active range of motion was also significantly higher for all movements in the high functioning group compared to the low functioning group (P < .05), except for elbow extension (which was more similar across the 2 groups), and radial and ulnar deviation (which had missing values in the low functioning group as the movement could not be performed by 8/9 subjects). In addition, the passive resistance to joint movement, as measured by the modified Ashworth score, was greater in the low functioning group compared to the high functioning group across all movements (P < .05) except for forearm supination and ulnar deviation, which approached significance (P < .1). Kinematic outcomes on the circle-drawing task performed with the CAREX, for a representative control subject, a low functioning, and a high functioning stroke subject are shown in Figure 2C. Note the clear differences in control of the circles drawn between the 3 subjects, reflective of their degree of motor impairment. Across all subjects, the FMS scores were positively correlated with speed (r = 0.48, P = .0003) and negatively correlated with normalized error (r = −0.48, P = .0006), indicating that the less impaired subjects with higher FMS scores moved faster and showed less error.
Clinical Assessments.
The mean normalized error of the circles drawn reflects the degree of control of the movement. As expected, we observe a clear group effect, the control group showed the smallest error and the low functioning group showed the largest error (mean normalized error ± SE: control = 0.067 ± 0.065, high functioning = 0.102 ± 0.057, low functioning = 0.346 ± 0.057; Figure 3A). Pair-wise group comparisons revealed that the error in the low functioning group was significantly different compared to the other 2 groups (LRT χ2(2) = 10.66, P = .0049). We found a significant interaction between group and CAREX training conditions (LRT χ2(5) = 17.22, P = .009). The low functioning group showed the most dramatic difference across the conditions with the smallest error when path assistance and weight support were combined compared with either path assistance alone (P = .026) or weight support alone (P = .030). Although the subjects were instructed to draw the circles at their preferred speed, the error in the trajectory of the circle may be correlated with speed. As expected, there was a significant group effect with the low functioning stroke group showing the lowest mean preferred speed (mean speed ± SE: control = 0.142 ± 0.019, high functioning = 0.13 ± 0.016, low functioning = 0.078 ± 0.016; LRT χ2(2) = 7.01, P = .030; Figure 3B). We found a significant interaction between group and condition (LRT χ2(5) = 31.32, P < .0001). Specifically, the high functioning group was faster with path assistance alone compared to either weight support alone or path assistance and weight support combined (P < .01).
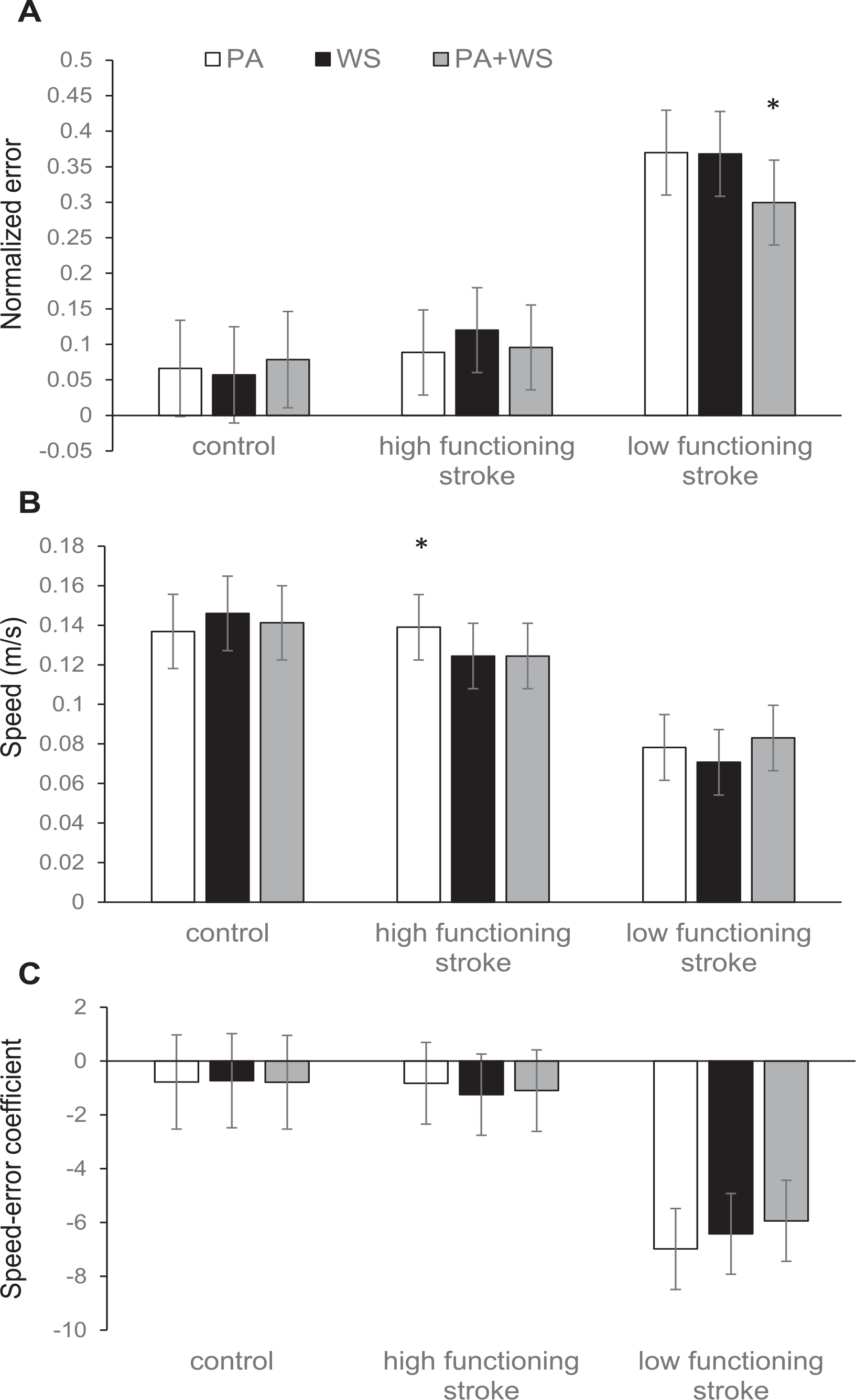
(A) Normalized error, (B) speed, and (C) speed-error coefficients with path assistance (PA), weight support (WS), and path assistance and weight support combined (PA+WS) with CAREX in the control, high functioning stroke, and low functioning stroke groups.
In Figure 3C, the speed and error are considered together and the estimated mean speed-error slope coefficients are plotted. Note that overall the coefficients are negative, suggesting that when speed increases the error tends to decrease across the 3 conditions with the CAREX. However, this negative correlation is not statistically significant for the control and high functioning groups. In the low functioning group, note the large negative speed-error slope coefficients, which suggests that increase in speed was associated with significantly reduced error on the 3D circle-drawing task. The error reduction associated with speed increase was largest with path assistance alone—the normalized error reduced by about 0.7 unit per 0.1 m/s speed increase (speed-error slope coefficient = −6.98 ± 1.5, P < .001), and second largest with weight support alone, at 0.64 unit per 0.1 m/s speed increase (speed-error slope coefficient = −6.42 ± 1.50, P < .001). With path assistance plus weight support, the speed-error reduction was the smallest at 0.59 unit per 0.1 m/s speed increase (speed-error slope coefficient = −5.93 ± 1.51, P < .001). However, it is important to note that there were no significant differences in the speed-error relationship with path assistance alone, weight support alone, and path assistance plus weight support, suggesting equivalence across these conditions in the low functioning group.
Most of the control subjects (except one subject; see Figure 4) performed the task at a narrow speed range (~0.1-0.15 m/s) and with the smallest error. The high functioning subjects with stroke were able to perform the task at comparable speeds to that of healthy controls, but with a wider range (~0.025-0.3 m/s), suggesting that their control of speed was not as consistent across trials and conditions as the control group. The normalized error was also slightly higher for the high functioning group compared to the control group. In contrast, many of the low functioning subjects performed the task at speeds slower than the other 2 groups (under 0.1 m/s), and with much higher error. However, it is important to note that the pattern we observe in Figure 4 can be confounded by between-subject variability in speed and error. For example, regardless of experimental conditions, the preferred speed and the extent to which the error is modified when speed changes may be different across individuals. The subject-specific slopes of the speed-error relationship across the 3 groups and 3 conditions are shown in Figure 5. These subject-specific speed-error responses explained a significant amount of the variance (LRT χ2(2) = 245.304, P < .0001), suggesting that individual subjects varied in their speed-error responses as represented by their slopes.
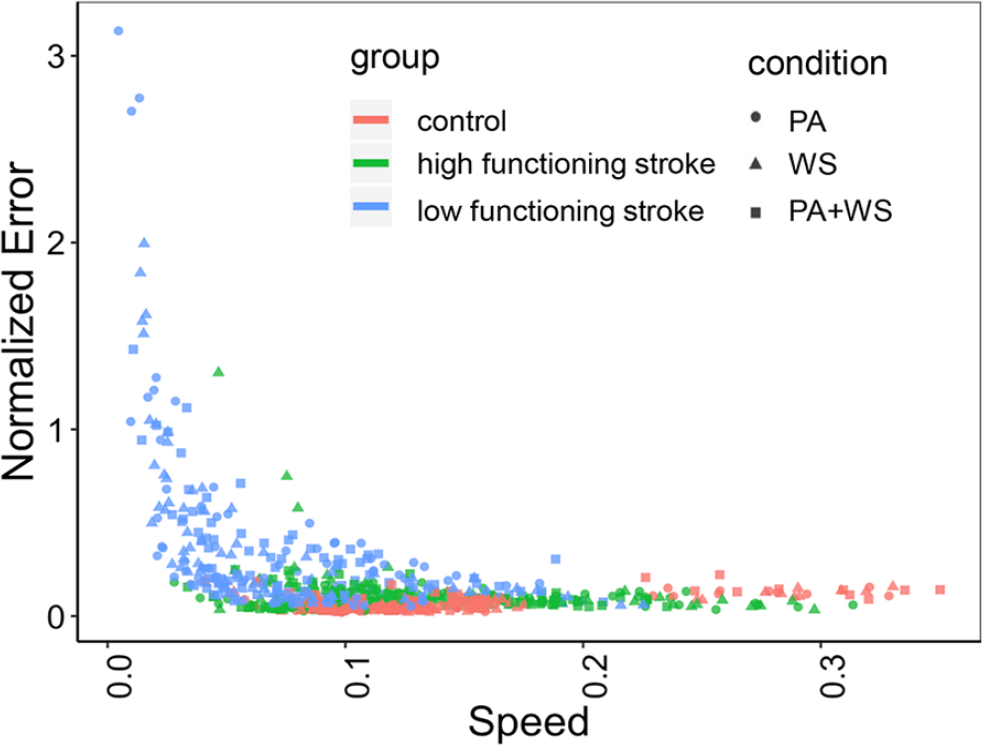
Speed-error relationship across all subjects and conditions.
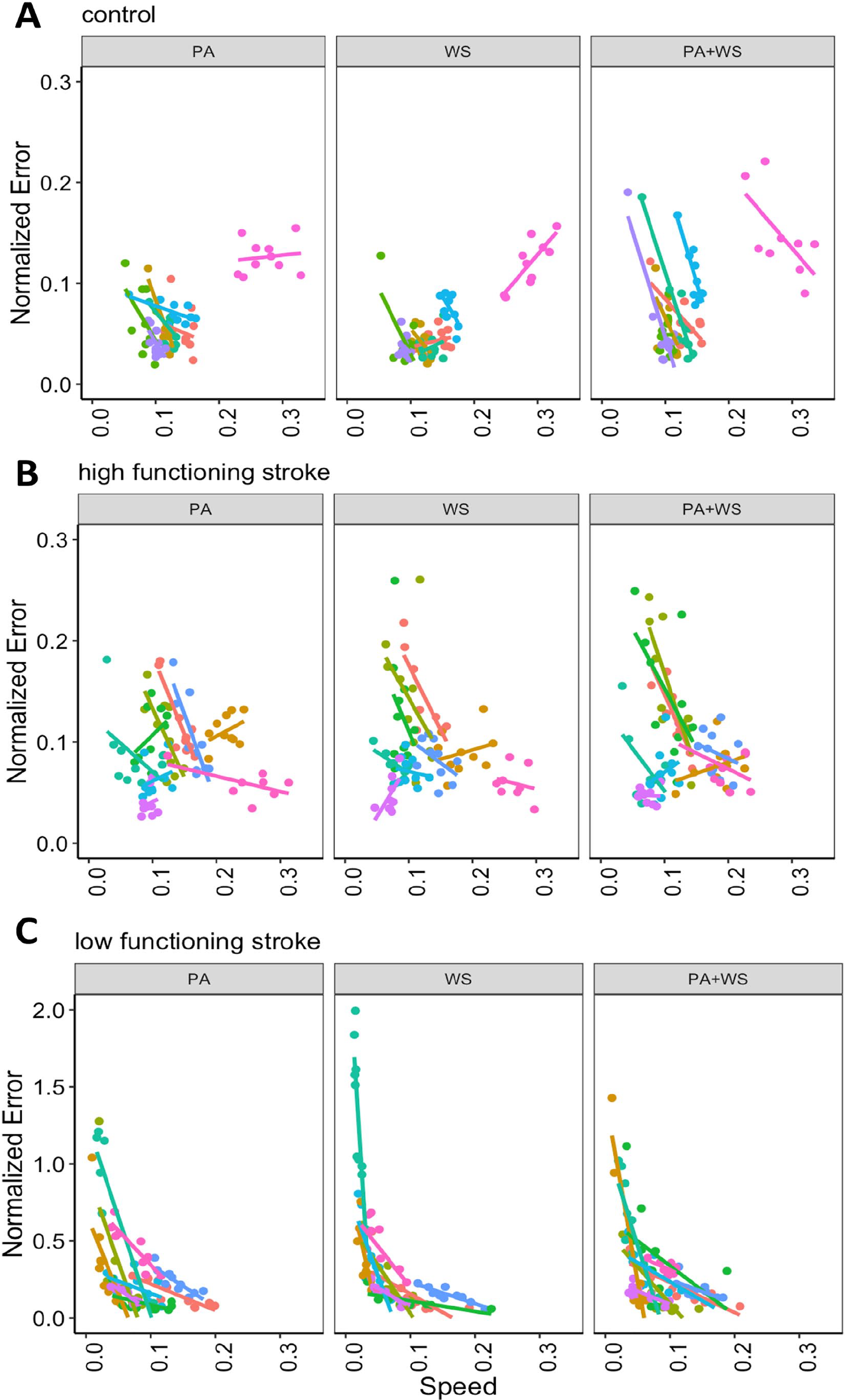
Speed-error relationship for individual subjects with path assistance (PA), weight support (WS), and path assistance and weight support combined (PA+WS) with CAREX in the (A) control, (B) high functioning stroke, and (C) low functioning stroke groups.
Finally, to understand the relationship between available movement and performance on the circle-drawing task, we correlated the active range of motion measurements with the mean normalized error and speed. We found positive correlations between shoulder flexion and mean speed in the low functioning group (Figure 6A; r = 0.73, P < .001), and forearm supination and mean speed in the low and high functioning groups (Figure 6B; low functioning, r = 0.86, P < .001; high functioning, r = 0.44, P = .04), suggesting that subjects with greater range of motion at these joints performed the task faster. Interestingly, shoulder extension showed a negative correlation with mean speed in both the low and high functioning groups (Figure 6C; low functioning, r = −0.81, P < .001; high functioning, r = −0.62, P < .001). Forearm pronation also showed negative correlation that approached significance in the low functioning group (Figure 6D; r = −0.49, P = .06).
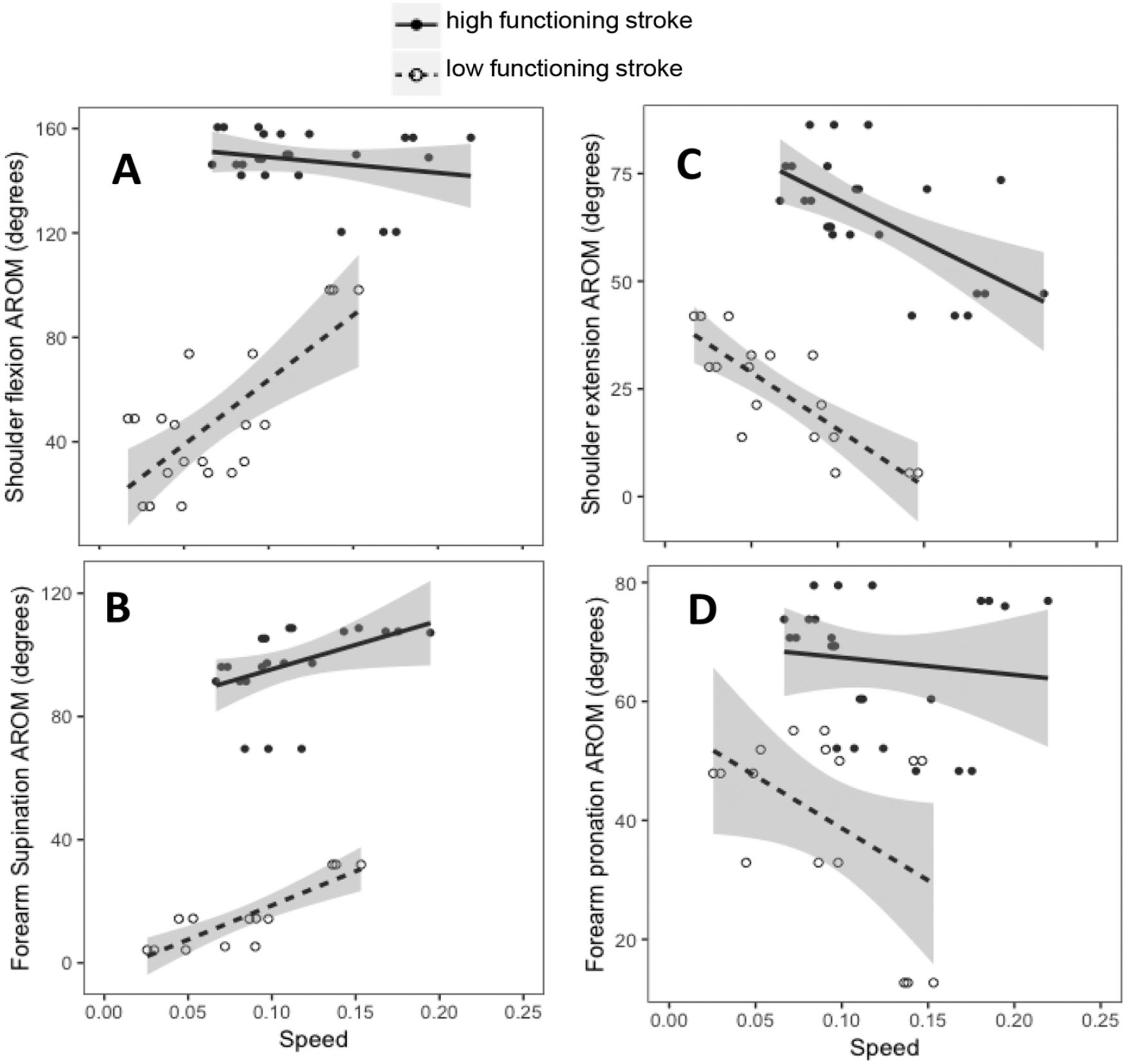
Relationship between speed and active range of motion in (A) shoulder flexion, (B) forearm supination, (C) shoulder extension, and (D) forearm pronation in the high functioning and low functioning stroke groups.
Discussion
In this study, we used a novel robotic CAREX to determine the role of robotic path assistance and weight support in facilitating repetitive 3D movements in individuals with poststroke hemiparesis. We hypothesized that higher functioning individuals would show smaller movement error at higher preferred speeds compared with lower functioning subjects on a 3D circle-drawing task. We also hypothesized that path assistance would facilitate trajectory control and task performance in higher functioning individuals with stroke, whereas both path assistance and weight support would be required in low functioning individuals. As expected, we found that the movement error was significantly greater, and the speed was significantly lower in the low functioning stroke group compared with the high functioning stroke group and controls. As hypothesized, the low functioning group showed the smallest error when path assistance and weight support were combined, whereas the high functioning group showed no difference in error but moved faster with path assistance alone compared to either weight support alone or path assistance and weight support combined. However, when both error and speed were considered together, the low functioning group showed a dramatic reduction in error and increase in speed across the trials, but weight support alone was as good as path assistance alone or path assistance and weight support combined. Interestingly, range of shoulder flexion and forearm supination positively affected movement speed, whereas shoulder extension and forearm pronation affected it negatively, particularly in the low functioning group.
Greater movement error and slower speeds in low functioning patients is consistent with their extent of motor impairment as represented by the Fugl-Meyer scores, as well as the degree of weakness and spasticity, as shown in Table 1. Path assistance and weight support combined significantly reduced the error in this group, consistent with the demonstrated effects in the literature of both haptic guidance and weight support in severe chronic stroke patients.11,31 Indeed, the patients in this group would not be able to perform 300 trials of the 3D task given the extent of their motor impairment 32 without robotic assistance. However, when both speed and error were taken into consideration, the error decrease and speed increase showed no significant difference with path assistance and/or weight support, suggesting that weight support alone may be adequate to facilitate task performance in this group.
When we examined the 100 trials for each condition, all 3 groups showed negative speed-error coefficients (Figure 3C), suggesting reduced error and increased speed, which was most marked in the low functioning subjects (see Figure 5). These results suggest that at higher speeds subjects showed less error perhaps due to better motor control. It has been shown that moving slowly is more difficult for humans as the control of movement is organized differently for movements at different speeds. 33 Furthermore, repetitive practice can lead to an improvement in both speed and error through various mechanisms.34,35 If repetitive practice led to the negative speed-error relationship, we might have seen a trial effect where earlier trials show slow speed and high error, whereas in later trials one may see faster speeds and lower errors. However, we did not find a statistically significant trial effect. This may be due to large heterogeneity across the subjects and between trials, which masked such an effect. Future studies may need to be designed to reveal the underlying mechanism of the observed speed-error relationship by examining the error when subjects are asked to perform the task at different speeds, and examining the potential carryover of the speed-error effect from one condition to the next.
Note that in this study, the circle diameter was constant, and subjects were not constrained to draw the circle within a specified amount of time or move their arm at any particular speed. Therefore, we did not expect a speed-accuracy tradeoff; the reduction in error and increase in speed resulted from each subject’s natural preference to move faster as a result of changes in motor control with practice. 36 Healthy controls showed little variability in task performance suggesting that the task was not difficult for them. However, one control subject who performed the task faster than the others to begin with and some high functioning stroke subjects showed a positive slope, that is, reduced error with increased speed (speed-accuracy tradeoff). 37 Interestingly, the speed-accuracy tradeoff was seen with weight support alone or path assistance alone but not when both weight support and path assistance were combined. None of the low functioning subjects showed a speed-accuracy tradeoff under the 3 conditions. These findings suggest that for some healthy and high functioning subjects, path assistance and weight support may make the task more difficult necessitating a change in control strategy that may lead to a speed-accuracy tradeoff. We propose that individual speed-error relationships with repetitive training may be able to provide information on practice strategies to facilitate meaningful changes in performance.
Partial weight support of the limb helps eliminate the effects of gravity on movement, which has been shown to reduce abnormal motor synergies in the upper limb after stroke and improve range of motion and function.38-40 For example, attempted reaching, which requires shoulder flexion and elbow extension, leads instead to involuntary shoulder abduction and extension or shoulder adduction and flexion, 41 accompanied by simultaneous elbow, wrist, and finger flexion in a synergistic pattern, making it very difficult if not impossible to reach normally. This abnormal pattern is responsible for reduced range of motion at multiple upper limb joints, limiting the workspace of paretic arm movements.42-46 Eliminating the effect of gravity by passively supporting the weight of the arm has been shown to lead to instant, albeit temporary, changes in the abnormal movement pattern and in functional ability.11,47-49 Our results concur with these findings.
Path assistance can improve trajectory control of the arm and may be provided by 2 distinct types of robotic devices. End-effector robotic devices control movement at the interface of the hand with the handle of the robotic device (eg, MIT Manus), but the movements at each joint cannot be independently controlled.50,51 In contrast, exoskeletal devices can control movements at each joint using motors (eg, Armin and MGA).52,53 The joints of the robotic exoskeleton are typically connected using rigid links, which adds inertia to the segments of the human arm making it heavier despite using strategies to reduce inertia of the exoskeleton such as by placing the motors away from the joints and driving the joints using cables and pulleys (eg, L-exos, CADEN-7, and MEDARM).54-56 The Cable Driven Upper Arm Exoskeleton, CAREX, used in this study, suspends the arm using a robot-controlled cable system, making it 10 times lighter and more versatile than other exoskeletal robotic rehabilitative devices.18-20 More sophisticated assistance can be provided by more expensive exoskeletal robots, which may however be less accessible to individuals who need them most. Our findings suggest that although there were interindividual differences across subjects, when overall task performance was considered, robotic path assistance for trajectory control provided little advantage over weight support alone. Ultimately, the goal of training is to restore movement and function without robotic guidance or support, and many new robotic approaches are being designed to provide just-enough support to individuals to facilitate task performance without promoting slacking behavior.57-60 Weight support is relatively less expensive to provide compared with robotic path assistance. 61 More important, the effects of weight support on arm movement control suggest that understanding the biomechanical principles underlying antigravity movements of the upper limb are important for overcoming poststroke synergy patterns and restoring movement poststroke. 62
We performed the study using a right-sided robot on individuals with right-sided hemiparesis from a left hemisphere stroke. However, all stroke subjects had adequate language and reasoning skills to understand the task and follow the instructions. We do not know if the results would be similar using a left-sided robot in individuals with left hemiparesis from a right hemisphere stroke, as interlimb differences have been found in the control of the right and left arm movements in healthy individuals as well as in subjects with stroke.63-65 Furthermore, available active range of motion is clearly important for task performance 66 and may be affected by side of the lesion. 67 The positive correlations between shoulder flexion and forearm supination with speed, but the negative correlations between shoulder extension and forearm pronation with speed, indicate how the extent and direction of joint motion enable or restrict the 3D circle-drawing task in this study. Restrictions in range of motion may need to be addressed prior to using robotic training strategies. This study was performed over a single session and we did not examine performance without the robot as the low functioning patients could not perform the task without robotic assistance, and the measurements could not be made in a comparable manner without the robot. Other studies have shown that repetitive task practice facilitated with a robot can lead to motor learning and generalized improvements in motor control without the robot, 68 although no benefits have been shown over dose-matched conventional therapy.69-71
Nevertheless, our findings show that low functioning subjects with stroke with severe arm motor impairment can perform 300 trials in 1 hour with the robot and show measurable changes in both speed and accuracy indicative of changes in motor control through practice. This is important as it is often difficult for low functioning individuals to engage in high-intensity repetitive task-practice due to the effort required and the onset and persistence of associated reactions during attempted movement, 72 and it is imperative that we find viable training options for this group. Our results suggest that robotic assistance for trajectory control provides little advantage over weight support alone over a single session of training. The next step is to determine the extent to which single session changes in performance are predictive of longer-term motor learning and changes in function.
Footnotes
Acknowledgements
The CAREX device was created by Dr Sunil Agrawal and Xin Jin at Columbia University and provided to Rusk Rehabilitation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Agrawal reports financial support to build the CAREX device from New York University School of Medicine; in addition, Dr Agrawal has a patent on Wearable cable-driven exoskeleton for functional arm training, 9,144,528, issued on September 29, 2015, not licensed. Dr Raghavan reports grants from National Institute of Health, grants from Foundation for PMR during the conduct of the study; in addition, Dr Raghavan has patents on rehabilitation devices for stroke and is cofounder and Scientific Consultant, Mirrored Motion Works, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by NIH R01HD071978 and the Rehabilitation Research Experience for Medical Students (RREMS) through the Association of Academic Physiatrist and the Foundation for PM&R.
