Abstract
Introduction
Multiple sclerosis (MS) is the most common progressive neurological disease 1 and the main cause of nontraumatic neurological disability 2 in young adults. Individuals with MS frequently experience impairments in walking and balance. Approximately 85% of individuals with MS report gait disturbance as their main complaint, 3 and within 15 years of onset of MS, up to 50% of individuals require assistance with walking. 4 Balance impairments contribute to unsteady gait and increased fall risk. 5
Slowed walking speed has been linked with impairments in strength and sensation,6-8 somatosensory integration, 9 and postural control in static standing.10,11 Individuals with reduced strength demonstrated slower walking speeds, regardless of the subtype of MS. 8 Additionally, vibratory sensation deficits in the feet predict static standing balance in MS, even in those with short symptom duration. 12 Reduction in vibration sensation in the lower extremities has also been linked with increased postural sway and reduced postural stability. 13 Previous work has implicated slowed spinal somatosensory conduction 14 and cerebellar atrophy,15,16 rather than weakness in MS imbalance; however, impaired strength has been linked with reduced walking speed and greater sway in static conditions. 17 Interestingly, lower-extremity strength can compensate for somatosensory loss in some individuals with MS, 8 suggesting that multiple factors influence walking and balance.
Measures of dynamic balance that involve self-generated perturbations represent an important consideration in the determination of balance impairment, but the relationship with walking speed is not clear. Few studies18,19 have evaluated the relationship of dynamic standing balance to walking, and none have evaluated this in combination with common impairments such as sensory loss, weakness, and static balance. This is particularly relevant in individuals with MS, where impairments typically occur in combinations, emphasizing the complexity of their functional limitations. 20 A comprehensive evaluation of the role of dynamic standing balance on gait velocity in combination with common impairments will allow us to develop appropriate quantitative behavioral outcome measures. This information may improve our understanding of what deficits play the greatest role in increasing fall risk in individuals with MS.
The objective of this study was to determine the association of measures of strength, sensation, dynamic balance, and static balance with walking velocity in individuals with MS. We hypothesized that dynamic balance measures would be more important contributors to walking velocity than static measures.
Methods
Participants
A total of 52 participants with clinically definite MS, as defined by the 2005 McDonald criteria, 21 who had volunteered for an ongoing longitudinal parent study at the MS Center at Johns Hopkins Medical Institutions between 2005 and 2009 were recruited for this study. We used a single time point for individuals who met our study criteria. Participants were included if they had received a clinical diagnosis of MS and were capable of standing without an assistive device. All participants demonstrated full understanding of the study and study-related tests. Participants were excluded if they had experienced an MS relapse within 3 months of testing or reported a history of peripheral neuropathy or any other orthopedic, neurological, or cognitive condition that might interfere with study procedures.
For comparison, we tested age-matched healthy adults to generate norms for strength (n = 61) and balance measures (n = 44; Table 1). All participants gave written informed consent prior to participation, and the institutional review boards at Johns Hopkins Medical Institutes and Kennedy Krieger Institute approved all procedures.
Participant Characteristics. a
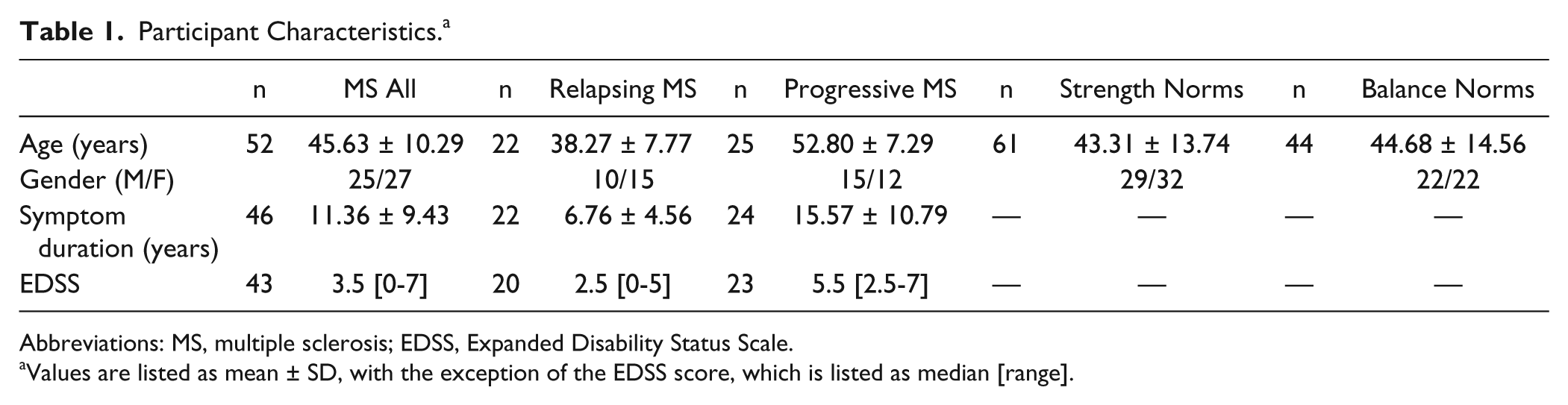
Abbreviations: MS, multiple sclerosis; EDSS, Expanded Disability Status Scale.
Values are listed as mean ± SD, with the exception of the EDSS score, which is listed as median [range].
Functional and Quantitative Impairment Measures
Participants completed all measures within a single session.
Vibration Sensation
Vibration sensation thresholds (vibration units) for the right and left great toes were quantified using the Vibratron II device (Physitemp, Huron, NJ). For this test, each person was required to determine which of 2 rods was vibrating using a 2-alternative forced choice procedure. 22 This is a valid and objective measure of sensation in MS. 6 More- and less-sensitive sides were determined, and the values for the more-sensitive side (ie, better vibration sensation) were used for analysis.
Strength Measures
Maximal voluntary contraction was quantified by hand-held dynamometry via break testing. Quantitative strength testing is valid and objective in MS. 6 The averages of 2 maximal efforts of hip flexion, hip extension, and ankle dorsiflexion bilaterally were evaluated using a Microfet2 hand-held dynamometer (Hoggan Health Industries, West Jordan, UT). The weaker and stronger sides for each measure were determined using an average of the 2 trials for each muscle for each individual. The strong muscle groups were used in the analyses because we hypothesized that they would provide the greatest propulsive force driving fast walking speed.23-25
Posturography
All measures of balance were evaluated using a Kistler 9281 force plate (Kistler Instrumente, Winterthur, Switzerland) at a sampling rate of 1000 Hz. Position data were low-pass filtered at 10 Hz. Participants were positioned on the force plate such that the force plate’s x-direction corresponded to medial-lateral (ML) body sway movement and the y-direction corresponded to anterior-posterior (AP) sway. Static balance was assessed under 2 conditions: eyes open, feet apart (EOFA) and eyes open, feet together (EOFT). Participants were asked to stand as still as possible during each condition for two 20-s trials. Static posturography is a reliable measure of static balance in persons with MS. 26 The distance between the first metatarsal heads was measured in the EOFA condition, and this distance was used to ensure uniform foot position in both dynamic conditions. Data were normalized to foot spread (ML trials) and foot length (AP trials) following previous methods from our laboratory. 27
Dynamic balance was assessed under 2 conditions: voluntary ML sway and voluntary AP sway. Participants were instructed to shift their weight as far as possible in either the ML or AP directions while keeping their feet flat on the ground. Participants were instructed to utilize ankle movements rather than hip movements to complete these movements, but there were no instructions given regarding speed of movement. Two 20-s trials were recorded for each dynamic condition following the methods from our laboratory.27,28
For analysis of the static and dynamic balance conditions, sway amplitude was calculated. Sway amplitude is a measure of the average distance the center of pressure deviates from the mean center of pressure, which is assumed to be the center of stability for each trial. Sway amplitude was the mean of this series of distances. For static balance trials, sway amplitude was calcualted with the following formula:
Gait Analysis
Gait kinematics were measured with an Optotrak 3020 Motion Capture System (Northern Digital Inc, Waterloo, ON, Canada). 3D coordinates were obtained from infrared light-emitting markers placed bilaterally on the foot (fifth metatarsal head), ankle (lateral malleolus), knee (lateral joint space), hip (greater trochanter), pelvis (iliac crest), and shoulder (acromion process). Wearing their usual comfortable shoes, participants were asked to walk for 3 to 5 trials along the 10-foot laboratory walkway as quickly and safely as possible. Walk velocity was computed using a custom-made MATLAB (The MathWorks Inc, Natick, MA) program. Individuals who regularly used an ankle-foot orthosis or an assistive device for safety were permitted to use it during all walking trials.
Statistical Analysis
Statistical analyses were performed using Statistica software (version 6.1; StatSoft, Inc, Tulsa, OK). Normality of the data distribution was checked using the skewness and kurtosis test and the Shapiro-Wilk test. In conditions where the assumptions of normality were not satisfied, nonparametric statistics were used. Outliers were determined for each condition using box-and-whisker plots. Mann-Whitney tests were used to compare individuals with MS to age-matched norms and to compare subtypes of MS (relapsing vs progressive) to norms and to each other. Corrections for multiple comparisons were not performed, following the methods of Spain et al, 11 because these groups were known to be different, and corrections would exaggerate the type II error. Spearman correlation coefficients were utilized to assess associations between walking velocity and strength, sensation, static balance, and dynamic balance measures in individuals with MS. A forward stepwise multiple regression analysis was then performed to describe the association between gait velocity (dependent variable) and those independent variables with an R value of 0.3 or greater (indicating a fair to moderate or greater correlation) 29 on Spearman’s correlation. P values <.05 were considered statistically significant.
Results
A total of 52 individuals with MS participated in this study (age [mean ± SD] = 45.63 ± 10.29 years; Expanded Disability Status Scale score, median [range] = 3.5 [0-7]). Participants with MS were subgrouped by diagnosis (relapsing-remitting MS vs progressive MS; Table 1). Quantitative measures for hip flexion, hip extension, and ankle dorsiflexion muscle strength were significantly weaker in individuals with MS when compared with age-matched norms (P < .001; Table 2).
Strength and Static and Dynamic Balance Measures. a
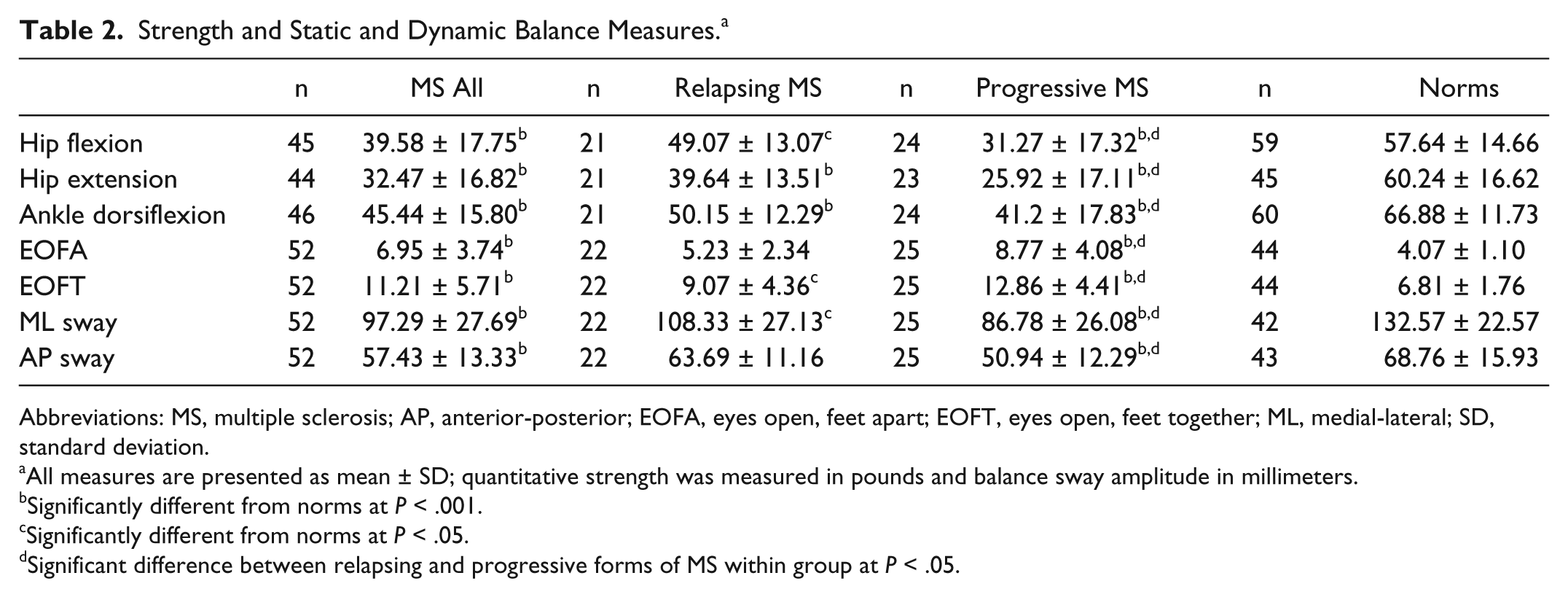
Abbreviations: MS, multiple sclerosis; AP, anterior-posterior; EOFA, eyes open, feet apart; EOFT, eyes open, feet together; ML, medial-lateral; SD, standard deviation.
All measures are presented as mean ± SD; quantitative strength was measured in pounds and balance sway amplitude in millimeters.
Significantly different from norms at P < .001.
Significantly different from norms at P < .05.
Significant difference between relapsing and progressive forms of MS within group at P < .05.
All individuals with MS, regardless of disease subtype, showed significantly impaired static and dynamic balance as compared with age-matched norms (P < .001 for all conditions; Table 2). Individuals with MS moved significantly more than norms in the static balance condition (P < .001). Figure 1 shows the results from the curve fitting; R2 values ranged from 0.64 to 0.97. During dynamic balance conditions, individuals with MS moved significantly less (P < .001) than norms.
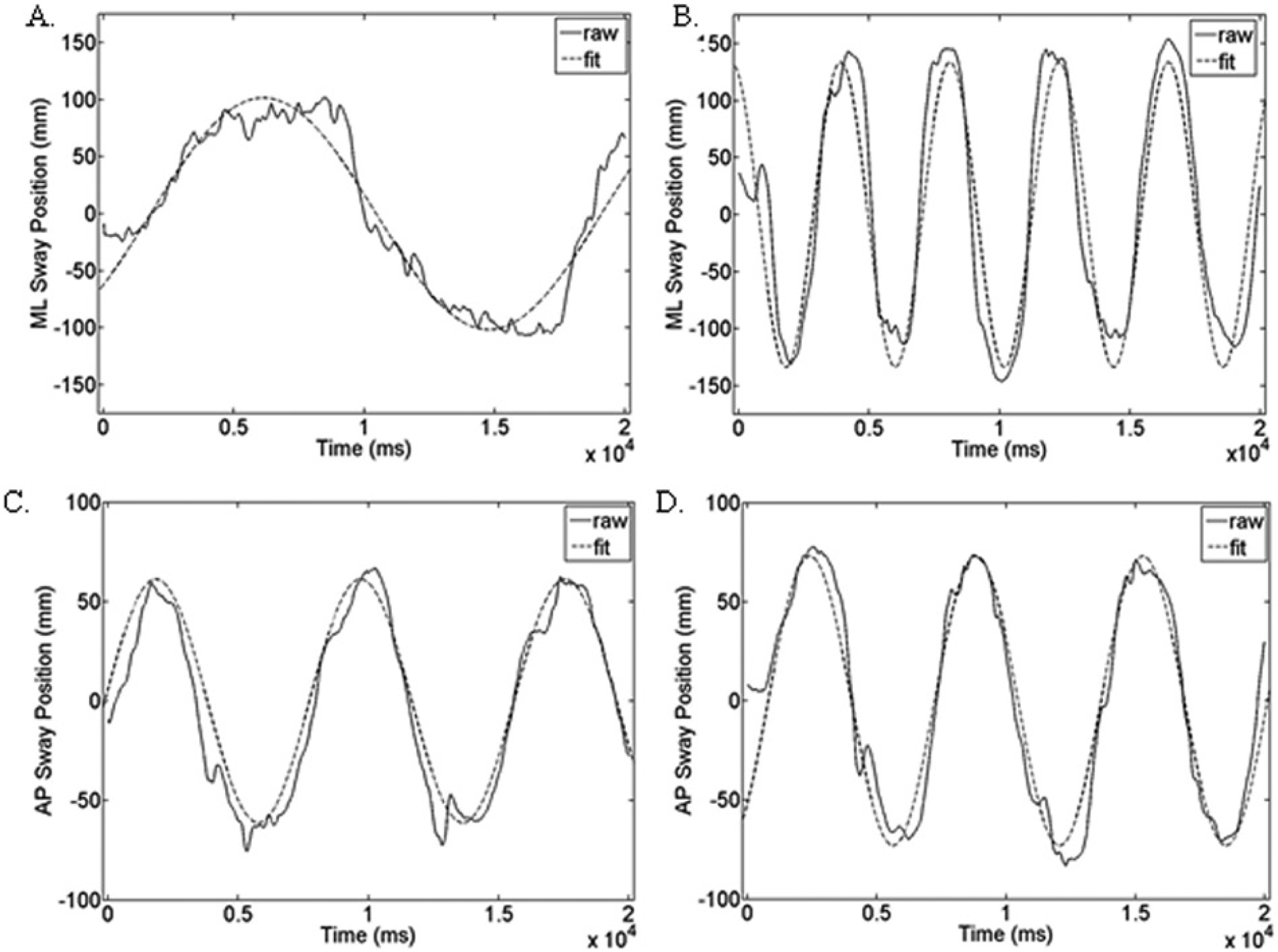
Postural sway plots of a representative individual with MS and a control. A. ML sway for individual with MS: R2 = 0.924, amplitude = 101.7 mm. B. ML sway for an age-matched norm: R2 = 0.867, amplitude = 133.8 mm. C. AP sway for individual with MS: R2 = 0.927, amplitude = 61.2 mm. D. AP sway for an age-matched norm: R2 = 0.862, amplitude = 73.0 mm. Each plot shows the raw frequency data from a single participant as well as the fitted data from a linear sinusoidal model.
Fast walking velocity for all individuals with MS averaged 1.37 ± 0.61 m/s, with those in the relapsing subtype walking faster (1.67 ± 0.45 m/s) than those in the progressive subtype (1.01 ± 0.57 m/s). Individuals with MS walked considerably slower than the published norms for fast walking velocity in adults 40 to 49 years old, 30 which is 2.46 m/s for men and 2.12 m/s for women.
Individuals in the relapsing subtype exhibited better sensation (3.30 ± 2.32 vibration units for the less sensitive toe and 2.61 ± 1.97 vibration units for the more sensitive toe) than those in the progressive subtype (6.94 ± 4.09 vibration units for the less sensitive toe and 5.21 ± 3.04 vibration units for the more sensitive toe). Individuals with MS demonstrated poorer vibration sensation values than the normalized reference values for individuals 36 to 50 years old and 51 to 65 years old taken from the Vibratron package insert: 2.56 and 2.89 vibration units, respectively. Indeed, values greater than 4.64 and 5.25 mark severe dysfunction in these 2 groups. 31
To examine which factors related to walking performance in MS, a correlation analysis was performed. In individuals with MS, there was a strong association (>0.7) between hip extension strength and walking speed as well as moderate relationships (0.50-0.69) between ML amplitude, AP amplitude, hip flexion strength, and ankle dorsiflexion strength and walking speed (Table 3). Interestingly, the association between the dynamic measures of balance and walking velocity were much stronger than the measures of static balance.
Correlations Between Fast Walking Velocity and Measures of Strength, Balance, and Sensation.
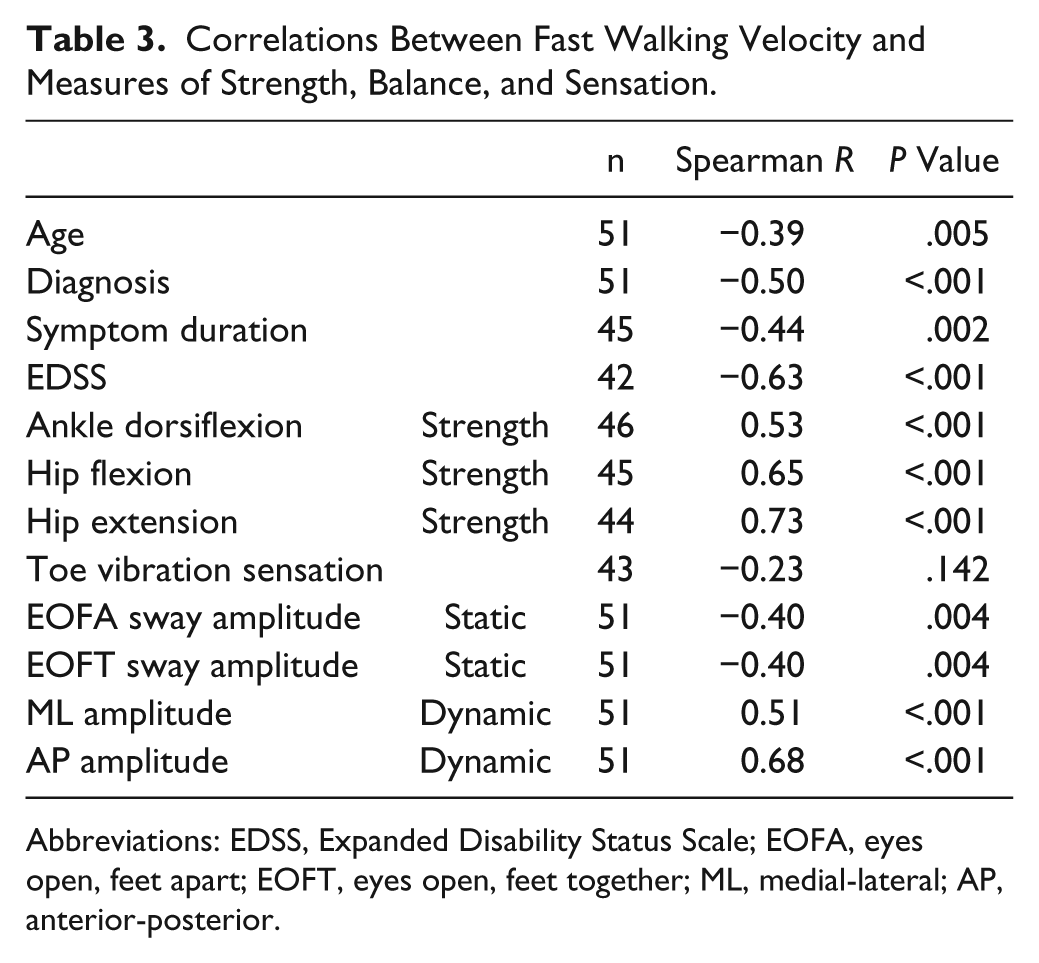
Abbreviations: EDSS, Expanded Disability Status Scale; EOFA, eyes open, feet apart; EOFT, eyes open, feet together; ML, medial-lateral; AP, anterior-posterior.
To specify the unique contribution of each predictor to walk velocity, we show the adjusted R2 and partial correlation coefficients (ρr) from the regression analysis (Table 4). AP sway amplitude was the most significant contributor to walking speed (ρr = 0.71; P < .001), with hip extension strength also strongly contributing to walking velocity (ρr = 0.54; P < .001). EOFT, a static balance measure, was also significantly associated with walking velocity (ρr = −0.41; P = .01) but to a lesser degree than AP dynamic sway and hip extension strength. With these th3ree measures as the strongest factors, the model explained greater than 70% of the variance (adjusted R2 = 0.747) for walking velocity (P < .001).
Forward Stepwise Multiple Regression Show That Measures of Strength and Static and Dynamic Balance Predict Walking Speed in MS.
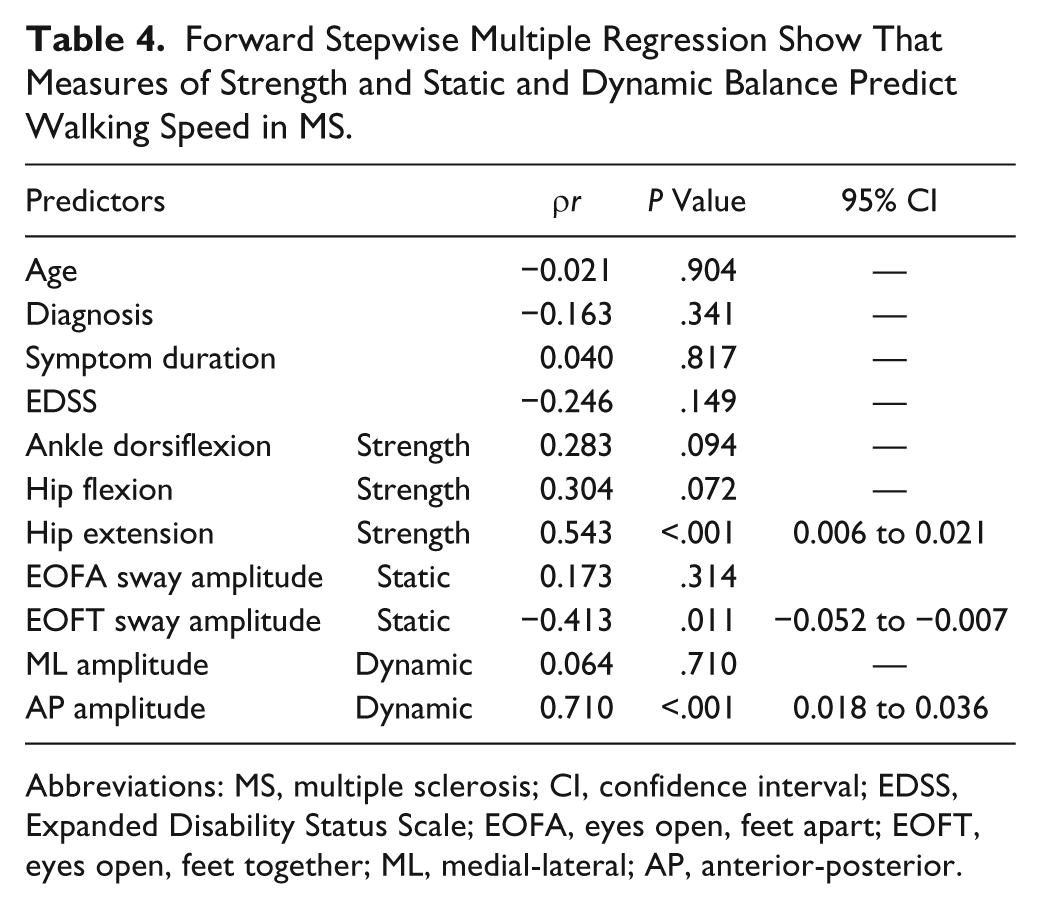
Abbreviations: MS, multiple sclerosis; CI, confidence interval; EDSS, Expanded Disability Status Scale; EOFA, eyes open, feet apart; EOFT, eyes open, feet together; ML, medial-lateral; AP, anterior-posterior.
Discussion
Our data show that measures of dynamic balance that capture self-generated perturbations contribute significantly to walking speed in MS. The combination of hip extension strength, EOFT, and AP sway amplitude explain more than 70% of the variance in walking speed. Vibration sensation, EOFA sway amplitude, and ML sway amplitude did not significantly contribute to the model. Interestingly, hip flexion and ankle dorsiflexion were also not significant contributors to the model despite their strong relationship to walking speed. This suggests that hip extension strength provides independent information separate from other strength measures. Our data support the hypothesis that dynamic balance measures (AP sway amplitude) are more strongly related to walking speed than static measures (EOFT; Figure 2). We posit that self-generated dynamic balance measures, particularly AP sway amplitude, may contribute more to walking speed because these movements mimic the displacement required during gait initiation to a greater degree than static measures.
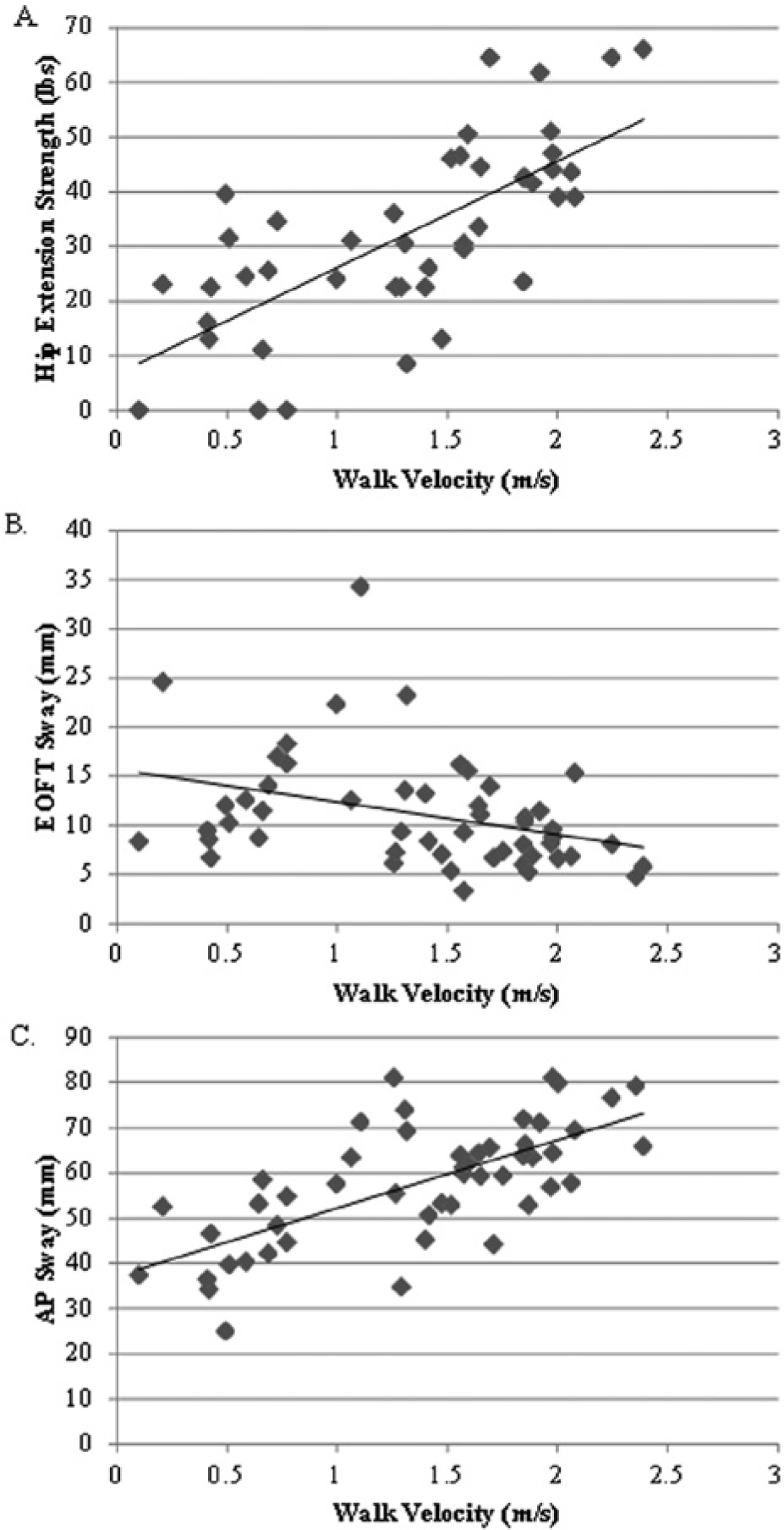
Scatterplots showing values for each individual for the 3 significant contributors to walking velocity. A. Hip extension strength: ρr = 0.54, P < .001. B. EOFT static sway amplitude: ρr = −0.41, P = .01. C. AP dynamic sway amplitude: ρr = 0.71, P < .001.
We know from previous studies that individuals with MS demonstrate increased postural sway in both ML and AP directions in static standing,9,32 delayed responses to postural perturbations, 14 and decreased ability to move toward their limits of stability. 9 However, clinical balance tests may not accurately identify balance impairment in MS. Spain et al 11 report that static postural deficits existed even when no differences were present on the Timed Up and Go or Timed 25-foot Walk. In contrast, reports exist where both high-functioning individuals with MS 33 and those with severe disability 34 have normal sway in quiet stance. In both cases, balance impairments only emerged with dynamic balance testing.35,36 Thus, the usefulness of static posturography may be limited, and balance assessment in MS may be augmented by dynamic posturography measures that more closely resemble the dynamic nature of walking.
The findings from this study show that individuals with MS lean less and stay further from their limit of stability, and this may be a limitation when initiating gait. 18 In our model, one measure of static balance, EOFT, also contributed significantly to walking speed. Deficits in static balance have been linked with reduced walking speed 37 as well as fall risk, particularly in the EOFT condition. 38 Additionally, reduction in walking speed has been linked with decreased muscle strength, and lower peak torque values in the hamstrings and quadriceps were associated with greater sway area in static standing. 17 Interestingly, sensation as measured by the Vibratron II device, was not a significant contributor to fast walking speed in our closed, laboratory environment. Perhaps sensory integrity is more critical for static balance and may play a larger role when assessing gait mechanics in open, unpredictable environments.
Although this study demonstrates that dynamic balance has important implications for understanding gait velocity, it may also play a role in understanding fall risk and fall prediction. Fall risk in MS has been linked to reduced balance control and quality of gait 39 as well as neurological impairments that include weakness, sensory dysfunction, and poor motor planning strategies. 40 Deficits in forward limits of stability, or leaning, along with lower-extremity weakness have also been associated with recurrent fallers in MS.41,42 Kasser et al 42 also demonstrated that reduced limits of stability on voluntary movement during dynamic posturography accurately identified frequent fallers in a sample of women with MS. Slips and falls frequently occur in the AP (forward-backward) direction; thus, AP sway may be more relevant to fall risk than ML sway. 43 Dynamic posturography in the elderly has been linked to fall risk 38 and may have better sensitivity than clinical measures of balance such as the Timed Up and Go test. Interestingly, Kalron et al 44 show data suggesting that falls are also related to the inability to allocate attention to balance, particularly in a situation requiring dual tasking (performing 2 tasks simultaneously). Impairment in the ability to dual task has been well documented in MS. 45 Therefore, measures of dynamic balance may be more relevant to functional rehabilitation because they are associated with a greater challenge, 46 requiring more attention to balance than static balance tasks. Combining dynamic balance measures with dual tasks may be useful in both fall prediction models and intervention strategies and requires further study.
Limitations
There are several limitations in this study. We examined dynamic balance cross-sectionally and are thus unable to make predictions about the effect of balance on the slowing of walking speed over time. To address this, we plan to use the baseline values from this study in a longitudinal study. We used dynamometry for strength testing, which can be variable between testers; to limit variability in technique, 1experienced tester (KMZ) trained all testers and monitored their technique. Furthermore, we strength tested only 3 muscle groups; however, the quadriceps, hamstrings, and ankle dorsiflexors are common sites of weakness in MS and account for both proximal and distal impairments that contribute to walking ability. 6 Ankle plantarflexors were not included because testing with dynamometry has been noted to be influenced by examiner strength and is not valid in this muscle group. 47 There are factors such as spasticity and proprioceptive loss that could contribute to walking speed. Such factors are more subjective and less quantitative and were, therefore, not assessed. Finally, we measured dynamic balance using a force plate. This provides a more objective and quantitative measure of balance important to understanding impairments; however, this equipment is not always readily clinically accessible. A clinical measure of dynamic leaning balance has been developed 48 and recently validated in MS 37 ; however, its use in clinical practice is not yet widespread. Given the results of this study, it is clear that dynamic balance should be incorporated into clinical testing. Therefore, further testing of measures that quantify dynamic balance is an important future direction.
Conclusion
Measures of dynamic balance that capture self-generated perturbations are important to consider when assessing and quantifying walking performance in MS. Overall, our model shows that dynamic balance, static balance, and strength are important factors to test when evaluating walking speed in individuals with MS. Dynamic balance assessment alone may uncover balance impairments that were not noted in static balance or during clinical testing. Regular assessment of dynamic balance may lead to a better understanding of walking mechanisms, identification of how the human body compensates for balance or strength deficits, and development of more targeted evaluation and intervention strategies to assess and improve walking and prevent falls. Future studies should explore the role of dual tasks in dynamic balance testing and examine additional measures of dynamic balance, including tasks that incorporate weight transfer.
Footnotes
Acknowledgements
The authors would like to thankfully acknowledge Joe Wang, Amy Bastian, Danny Reich, and all the participants in our study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH/NICHD K01 Award [HD049476-04].
