Abstract
Objective. To evaluate the effectiveness of a home-based rehabilitation of balance using the Nintendo Wii Balance Board System (WBBS) in patients affected by multiple sclerosis (MS). Methods. In this 24-week, randomized, 2-period crossover pilot study, 36 patients having an objective balance disorder were randomly assigned in a 1:1 ratio to 2 counterbalanced arms. Group A started a 12-week period of home-based WBBS training followed by a 12-week period without any intervention; group B received the treatment in reverse order. As endpoints, we considered the mean difference (compared with baseline) in force platform measures (ie, the displacement of body center of pressure in 30 seconds), 4-step square test (FSST), 25-foot timed walking test (25-FWT), and 29-item MS Impact Scale (MSIS-29), as evaluated after 12 weeks and at the end of the 24-week study period. Results. The 2 groups did not differ in baseline characteristics. Repeated-measures analyses of variance showed significant time × treatment effects, indicating that WBBS was effective in ameliorating force platform measures (F = 4.608, P = .016), FSST (F = 3.745, P = .034), 25-FWT (F = 3.339, P = .048), and MSIS-29 (F = 4.282, P = .023). Five adverse events attributable to the WBSS training (knee or low back pain) were recorded, but only 1 patient had to retire from the study. Conclusion. A home-based WBBS training might potentially provide an effective, engaging, balance rehabilitation solution for people with MS. However, the risk of WBBS training-related injuries should be carefully balanced with benefits. Further studies, including cost-effectiveness analyses, are warranted to establish whether WBBS may be useful in the home setting.
Keywords
Introduction
Multiple sclerosis (MS) is the commonest cause of progressive neurological disability in young people. Lack of balance is among the most disabling symptoms of MS and it may affect about 75% of patients over the course of the disease. 1 Fatigue, muscle weakness, and spasticity further affect balance and postural control, thus predisposing MS patients to fall.2-4 All these impairments may also prevent patients from performing their daily life activities. 5
Recently, there has been a growing interest in the investigation and treatment of balance disorders in people with MS. Unfortunately, pharmacological approaches aimed at ameliorating imbalance in patients with MS are often inadequate. 6 It has been also reported that some drugs that are broadly used in a neurological setting may affect gait and balance. 7 Therefore, the majority of treatments rely heavily on rehabilitation, which is still considered the only way to improve function in MS. 8
New rehabilitative strategies regarding motor learning and plasticity are mainly focused on high-intensity, repetitive, and task-specific practice. 9 According to these principles, virtual reality and visual feedback training are being used to improve several deficits, such as impaired balance and walking, in both people with MS10-12 and other populations with neurological disorders.13,14
The Nintendo Wii software/hardware game package (Nintendo, Kyoto, Japan; http://www.nintendo.co.uk/index.html) has recently been included into the definition of visual feedback/virtual reality training, 15 and its use in the neurorehabilitation process is still growing.16-19 Some Wii videogames require a force platform (ie, the balance board) to be played. Once connected to a common home-TV, the Wii Balance Board System (WBBS) provides a constant visual feedback about accuracy of movement patterns by means of pressure sensors and wireless signals.
A WBBS-related improvement in balance has been reported in the elderly, 20 in people with acquired brain injuries, 17 and in Parkinson disease. 18 The WBBS has been recently proposed even for people with MS as a strategy to enhance physical activity behavior 21 and to improve balance and walking ability.22,23 Thus, WBBS could represent an alternative approach to engage in effective balance training. At the same time, it could allow patients to minimize MS-related barriers to rehabilitation, by means of home-based training programs. 21 However, so far there are no randomized controlled studies on people with MS investigating the efficacy and safety of WBBS in a home-based setting.
The hypothesis of the current study is that a home-based training using the WBBS might provide a safe, engaging, balance training solution for MS patients, particularly those still ambulatory, but at risk of falling. Hence, the main purpose of the present study was to evaluate the effectiveness of a home-based WBBS training protocol in ameliorating postural and balance control in a cohort of MS patients suffering from an objective balance disorder.
Methods
Participants
Patients affected by MS according to McDonald Revised Criteria, 24 and regularly attending the MS Centre of S. Andrea Hospital, were asked to participate at this 24-week, independent, randomized, controlled, 2-period crossover pilot study (Figure 1). The protocol was approved by the local ethics committee; each patient provided written informed consent before any study-related procedure.
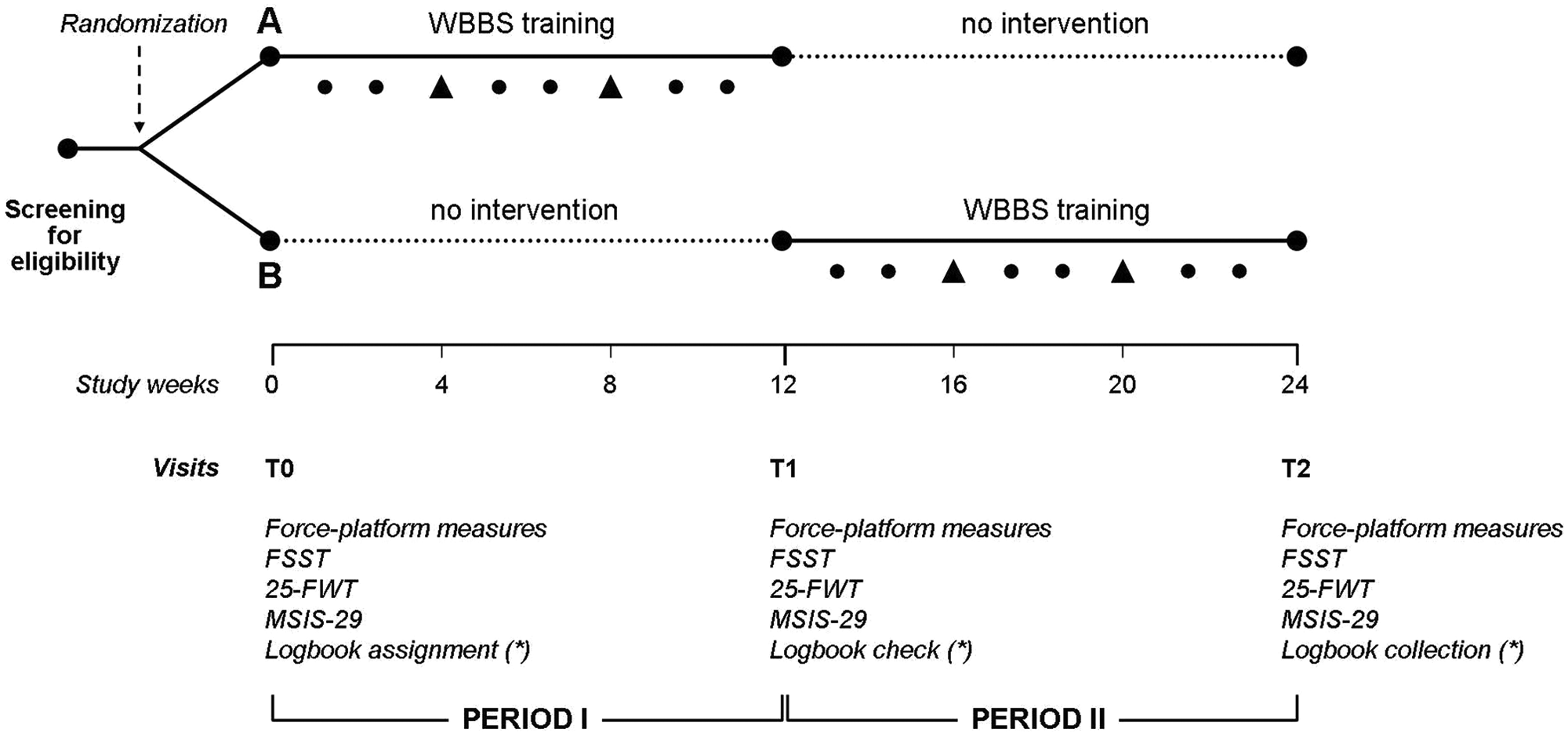
Study design Abbreviations: WBBS, Wii Balance Board System; FSST, 4-step square test; 25-FWT, 25-foot timed walking test; MSIS-29, 29-item Multiple Sclerosis Impact Scale.
Before study enrollment, 2 neurologists (LP and CP) screened patients for eligibility criteria, including a neurological examination by means of Neurostatus (www.neurostatus.net/scoring). Inclusion criteria were the following: an age of 18 to 50 years (inclusive); a relapsing–remitting or secondary progressive MS course; an Expanded Disability Status Scale (EDSS) score 25 equal or less than 5.5; ability to walk without resting for at least 100 meters; and presence of an objective balance disturbance (ie, impaired straight line walking, gait ataxia, or positive Romberg test). Exclusion criteria were the following: use of assistive device or foot ankle orthosis; relapses occurring over the previous 6 months; initiation of disease-modifying or symptomatic treatments, or any medication change occurring over the previous 3 months; seizures; severe blurred vision; concomitant otological or vestibular diseases (non-MS related); psychiatric disorders or severe cognitive impairment; and cardiovascular and respiratory disorders.
Patients enrolled in the trial also had to be willing to not change or start any medication for the entire study period, except for steroids required to treat MS exacerbations.
Study Design
Patients who met all eligibility criteria underwent study assessments and were randomly assigned in a 1:1 ratio to 2 counterbalanced arms by computer-generated random numbers. Randomization procedure was performed by an operator (LL) not involved in study measurements.
Group A started a 12-week period of home-based WBBS training (intervention period), followed by a further 12-week period without any intervention or specific training (observation period). Group B patients were given the treatment period in reverse order. Patients were further retested after the first 12-week period (T1) and finally at the end of the 24-week study period (T2). Thus, all patients underwent 12 weeks of WBBS training (active period) and a 12-week observation period.
A 2-period crossover study design 26 was adopted for the following reasons: (a) influence of confounding covariates is reduced because each subject serves as his/her own control, (b) a sample size smaller than a parallel design is required, (c) carryover effect is not expected in a chronic disabling condition such as MS, and (d) the impossibility of planning an appropriate washout period since the duration of the effect of WBSS training has not been investigated.
Intervention
During the 12-week active period, each patient was submitted to daily sessions (with the exception of the weekend) of home-based training with WBBS, each lasting 30 minutes. Patients were allowed to skip at maximum 1 session per week; therefore, we expected that each patient performed 48 sessions.
Training protocol consisted of repetitions of several games selected from the “Wii Fit Plus” package (http://www.wiifit.com/training/balance-games.html), according to the following rules. Each game started at basic level, and when a certain score was reached, patients were automatically transferred to a more advanced level by WBBS. Patients were then encouraged to play the next game if they had a level progress; otherwise, 10 minutes were allocated for each game. During the first 4 weeks of WBBS training, patients were allowed to play only “Zazen,” “Table Tilt,” and “Ski Slalom”; thereafter they could add remaining games. In the last 4 weeks of WBBS training, they were allowed to play games that they enjoyed the most. A detailed description of each game is reported in Table 1.
Description of Balance Games Included in the “Wii Fit Plus” Package and Used for the 12-Week WBBS Training.

Abbreviation: WBBS, Nintendo Wii Balance Board System.
The balance board contains 4 force sensors (located in each corner) that detect a subject’s center of balance and weight shifts. Users can interact naturally with the game by means of weight transferences on the balance board, thus constantly having a visual feedback through a sensory-enriched environment. All participants familiarized themselves with the exercises prior to starting the protocol. The device was connected to the patients’ home TV and the balance board was located at a distance ranging from 1 to 3 meters, according to TV display size. Home installation of WBBS, detailed explanations of training protocol, and supervision of the first complete training session were carried out by a trained physiotherapist (DF). Engagements with physiotherapists (DF and MRM) were scheduled every 4 weeks during the intervention period to supervise the correct execution of games and to monitor patients’ performance. Phone contacts were also scheduled every week during the intervention period to remind patients to complete the logbook and encourage them to perform the training.
Study Evaluations
The following outcome measures were collected at each scheduled visit (T0, T1, and T2) by 2 neurologists (LP and CG) unaware of the training order allocation: (a) force platform–based measures of static standing balance, 27 (b) the 4-step square test (FSST), 28 (c) the 25-Foot Walking Test (25-FWT), 29 and (d) the 29-item MS Impact Scale (MSIS-29). 30
Self-reported number of accidental falls (defined as an unexpected contact of any part of the body with the ground) that occurred in the 12-week period before randomization was also asked of each patient.
Force platform–based measures of static standing balance were collected with a monoaxial force platform (ProKin, Tecnobody, Bergamo, Italy; http://www.tecnobody.it) according to standardized procedures.27,31 The instant positions of the center of pressure (COP) on the ground were used to calculate the sum of displacements (millimeters) on the force-measuring platform (COP path), in open eye condition (mean of 3 consecutive trials). Force platform measures can overcome the main drawbacks of clinical balance examinations, such as variability in test performance (within and across different assessors), subjective nature of scoring system, and insensitivity to subtle changes. 32 Moreover, posturographic techniques have been reported as useful for documenting improvements in balance after rehabilitative intervention in MS setting.12,22,33
The FSST measures dynamic standing balance, and it was developed to identify elderly people suffering frequent falls. 28 The time (seconds) is registered while the subject rapidly steps over low obstacles in clockwise and counterclockwise directions (the first attempt is not timed).
The 25-FWT is a stop-watch measurement of time (seconds) to walk a 25-foot (7.6 m) distance (mean of 3 consecutive trials). 29 It has shown good reliability and validity in MS population as an individual component of the MS Functional Composite (MSFC) disability assessment.
The MSIS-29 is a 29-item self-administered questionnaire measuring physical and psychological impact of MS from the patient’s perspective. 30 It has been reported not only as valid and reliable, but it is also more sensitive than other quality of life (QoL) scales in detecting rehabilitation-induced changes. 34
All patients received a logbook describing the training protocol and other additional precautions. This logbook was also used to daily record the log of training (including time and type of game played) and the occurrence of accidental falls (as defined above) or any adverse event (defined as any untoward medical occurrence regardless of its causal relationship to the study intervention) during the 24-week study period. The logbook was given at baseline (T0), was checked at visit T1, and finally was returned to the study team at the end of the 24-week follow-up (T2).
Adverse events were graded as mild (minimal or no treatment required and no interference with daily living activities), moderate (may require treatment and cause some interference with functioning), severe (systemic drug or other treatment required, interruption of daily living activities), or life-threatening (immediate risk of death; http://ichgcp.net/12-adverse-event-ae). Patients were also encouraged to contact any component of the study team in case of adverse events, for any question regarding the study protocol or technical problems.
Endpoint Definition
The primary endpoint was the mean difference in static standing balance measures (ie, the COP path) at T1 and T2 visits, as compared with baseline (T0) evaluation.
Secondary endpoints were the mean differences in clinical scales (FSST and 25-FWT) and in self-administered questionnaire (MSIS-29) at T1 and T2 visits, as compared with to baseline (T0) evaluation.
Statistical Analysis
Given the exploratory nature of this pilot trial, no sample size analysis was performed.
Data are presented as mean (standard deviation) or median (range), as appropriate. Well-balancing of the 2 treatment groups after randomization were tested by using the Mann–Whitney U or the Fisher exact tests for continuous and categorical variables, respectively. Repeated-measures analyses of variance (RM-ANOVAs) with raw values at different time points (ie, T0, T1, and T2 visits) as the within-subjects factor and treatment group (A vs B) as the between-subjects factor were performed for each of the endpoints. A time × treatment interaction analysis was run to evaluate treatment effect on the aforementioned endpoints. Simple contrasts were conducted for each treatment group to determine the source of significant difference with respect to the baseline (T0). This analysis allowed us to determine the maintenance of treatment effect for group A and the reliability of study measures over time for group B. Finally, a comparison between number of patients free from accidental falls in the 12 weeks prior to study enrollment (self-reported at baseline) and number of patients free from accidental falls (prospectively collected) at the end of the 24-week study period was carried out by the McNemar–Bowker test.
P values less than .05 in either direction were considered as significant. Analyses were carried out by using the Statistical Package for Social Sciences, version 16.0 (IBM SPSS, Chicago, Illinois).
Results
From February to June 2011, a total of 45 patients were assessed for eligibility; out of these, 36 (25 women, 11 men) with a mean age of 36.2 (8.6) years, mean MS duration of 10.7 (5.8) years, and median (EDSS) of 3.5 (1.5-5.0) were randomized (18 were assigned to group A and 18 to group B). The 2 treatment groups were comparable in terms of baseline demographic and clinical characteristics (P values ≥.15 for all comparisons; Table 2).
Characteristics of Study Sample at Baseline. a

Abbreviations: SD, standard deviation; BMI, body mass index; EDSS, Expanded Disability Status Scale.
P values are ≥.15 for all the between-group comparisons.
After baseline evaluation, there were 2 patients who discontinued the study protocol: (a) a 39-year-old female patient with an EDSS score of 5.0 and assigned to group A withdrew the informed consent 2 weeks after baseline (T0) and (b) a 54-year-old female with an EDSS score of 5.0 and assigned to group B experienced a motor relapse after 5 weeks from baseline (T0), thus becoming unable to accomplish the study protocol. As a consequence, data of these 2 patients were excluded from the analyses (Figure 2). No other patient experienced relapses or EDSS worsening during the 24-week study period in both groups. There was no difference between the 2 treatment groups in the mean WBBS training time: 27.5 (17.1) hours for group A and 27.1 (15.9) for group B, corresponding to 137 (85) and 135 (79) minutes per week, respectively.
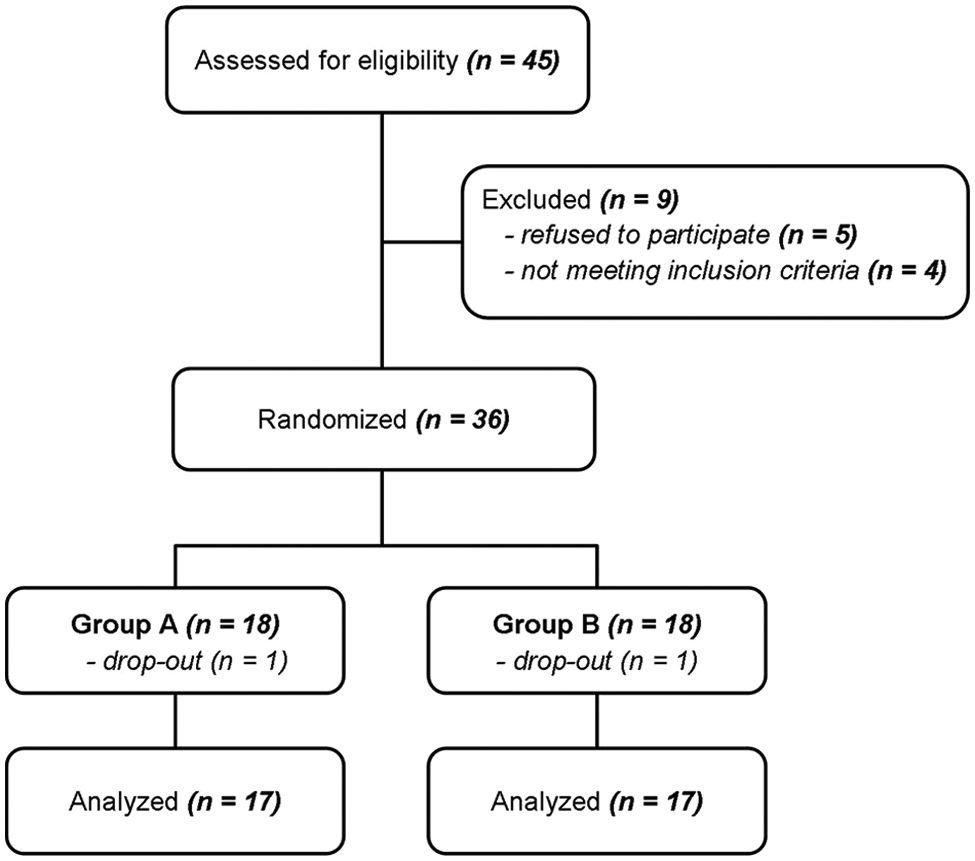
Study flowchart.
Table 3 shows the results of COP path, FSST, 25-FWT, and MSIS-29 for the 2 treatment groups at baseline (T0), after 12 weeks (T1), and after 24 weeks (T2), including their relative percentage changes, time effect, and time × treatment effect. We did not observe any time effect across groups for all endpoints, while there were significant time × treatment interaction effects in COP path (F = 4.608, P = .016), FSST (F = 3.745, P = .034), 25-FWT (F = 3.339, P = .048), and MSIS-29 (F = 4.282, P = .023). These findings indicate significant between-group differences over time favors WBBS training in static and dynamic balance, walking speed, and QoL (Figure 3).
Summary of Study Results.
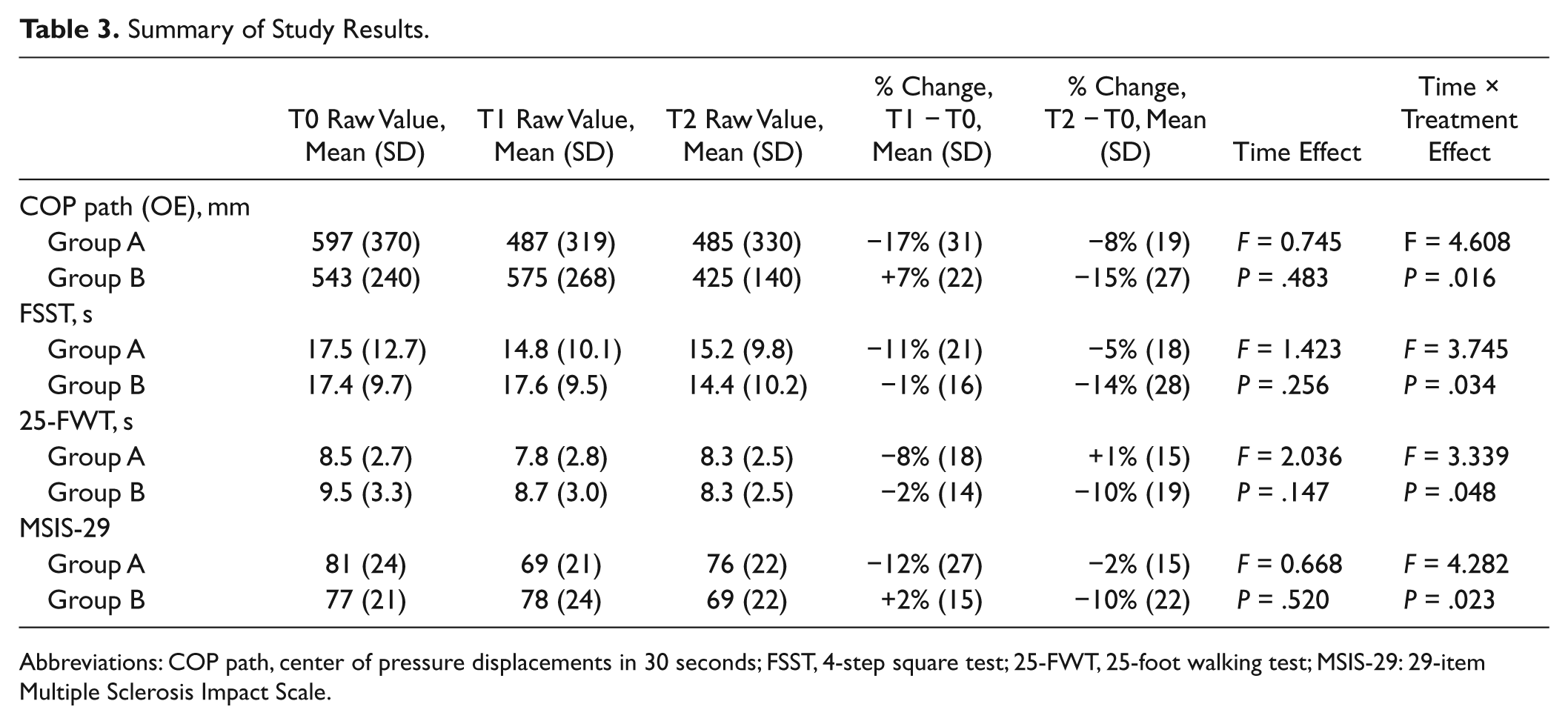
Abbreviations: COP path, center of pressure displacements in 30 seconds; FSST, 4-step square test; 25-FWT, 25-foot walking test; MSIS-29: 29-item Multiple Sclerosis Impact Scale.
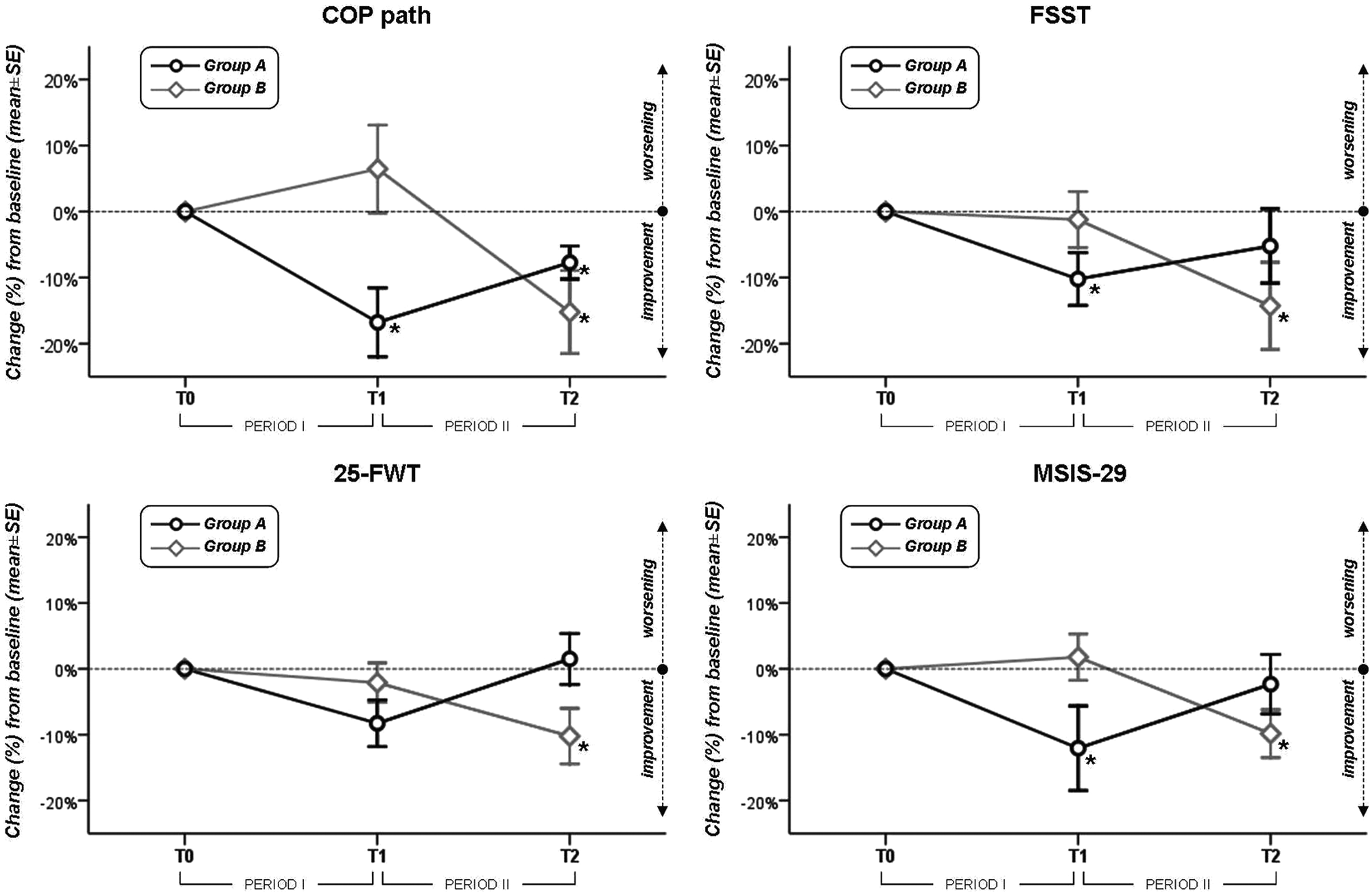
Percentage changes from baseline (T0) values as evaluated at 12-week (T1) and 24-week visits in force platform measures (center of pressure displacements in 30 seconds, COP path), 4-step square test (FSST), 25-foot walking test (25-FWT), and 29-item MS Impact Scale (MSIS-29) *P < .05 when compared with baseline (by RM-ANOVA—simple contrast analysis).
The simple contrast analyses also revealed that WBSS training was effective in all endpoints, regardless of the order of treatment. In group A, there was a significant improvement in COP path, FSST, and MSIS-29 at the end of intervention period and a trend toward a return to baseline values at the end of the observation period. A residual effect on primary endpoint was found in group A even at the end of the observation period.
In group B, all efficacy measures remained substantially unchanged during the observation period, while a significant improvement was observed after the intervention period. However, it is noteworthy that in group B the improvement in walking speed after WBBS training reached statistical significance (P < .05), whereas this did not happen in group A, probably due to the small sample size.
At the end of the 24-week study period, there was a proportion of nonfallers (n = 17, 50%) greater than that one (n = 12, 35%) relative to the 12 weeks before the study entry (P = .048 by the McNemar–Bowker test). No accidental falls were reported by patients while performing WBBS training.
During the 24-week study period, 24 (70%) patients reported at least 1 adverse event. Out of these, 5 graded as moderate (n = 2) and mild (n = 3) were considered as attributable to home-based WBBS training (Table 4). The majority of these WBBS training-related adverse events (4 out of 5) occurred after the introduction of the most challenging games. No extra contacts with the study team were required by patients, except for 1 patient in group B who needed technical assistance with WBBS after the device was set up.
Summary of the Adverse Events Related to the Training With Wii Balance Board System (WBSS).
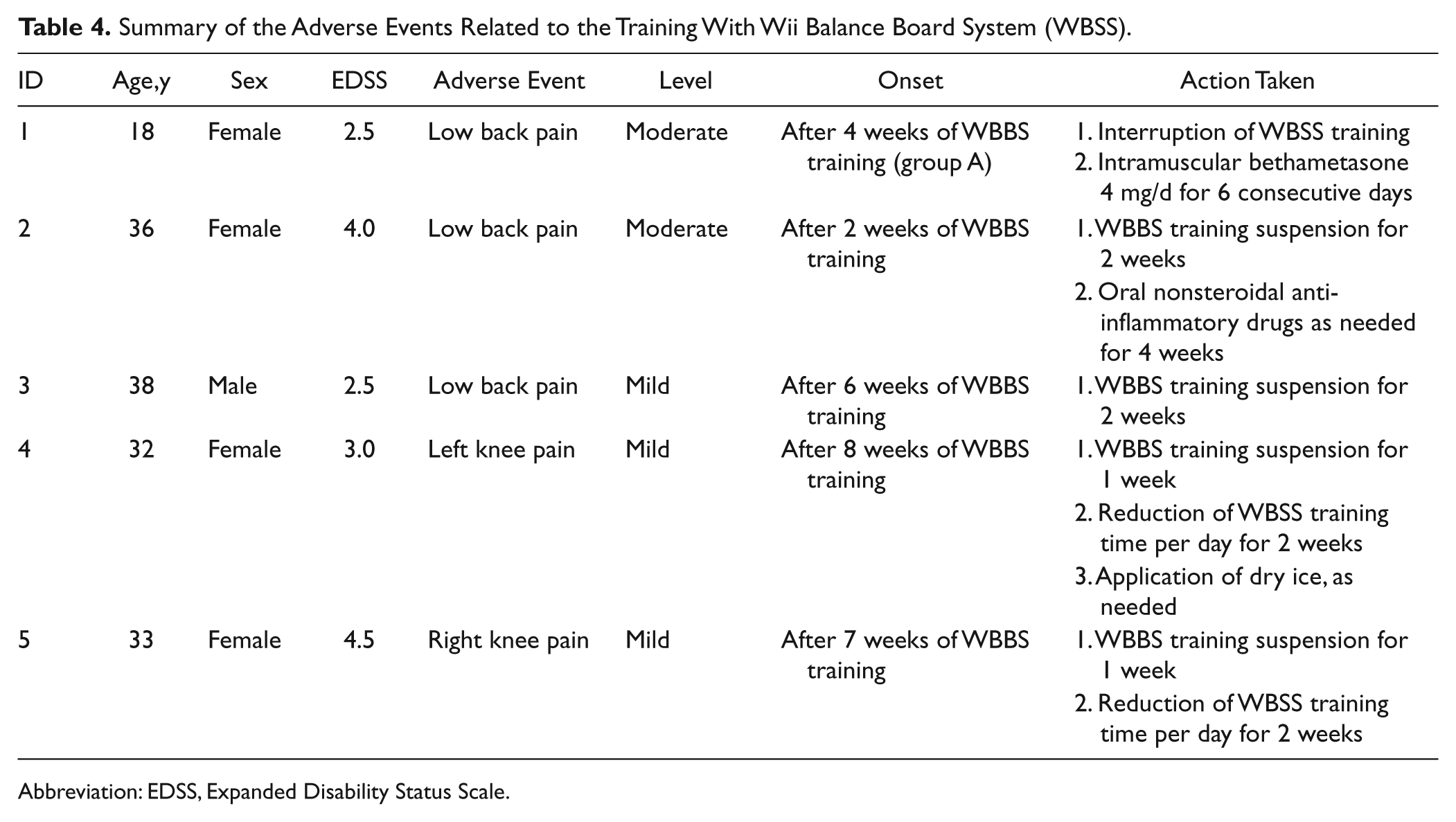
Abbreviation: EDSS, Expanded Disability Status Scale.
Discussion
The main finding of our study is that a 12-week WBBS training improved static and dynamic balance and reduced the impact of MS on patients’ QoL. The use of force platform–based measures, 27 which ensure linear, objective, and reproducible estimations of balance skills and risk of accidental falls, further strengthens our results.
A possible explanation for the benefits of the WBBS training is that it ameliorated the impairment of proprioceptive signal conduction by providing constant visual information about weight shifting on balance board using the visual display, thus enhancing the coupling between perception and action.12,35,36 In recent years, the specific retraining of sensory strategies has became an essential component of rehabilitative programs aimed at improving static and dynamic balance in MS. These interventions could reduce—at least theoretically—the risk of accidental falls while patients perform their daily living activities.12,37
An alternative hypothesis encompasses the role of mirror neurons. 38 During the WBBS training, patients see an avatar that mimics their movements while they are playing. Some authors suggest that watching one’s own movements, while executing an action, could facilitate motor relearning in neurorehabilitation.38,39 Task-oriented training and rehabilitation can lead to an enhancement of both function and structure of neural mechanisms, 40 but future studies are necessary to better examine this theoretical mechanism.
Last, improvement in standing balance observed after WBBS training might be even related to enhancement of lower limb strength.21,41
We found a 15% to 17% improvement in force platform measures after WBBS training. The COP path in open eye condition, which we adopted as the primary endpoint, has been recently suggested as reliable (95% concordance correlation coefficient), more sensitive (88% vs 37%), and accurate (75% vs 63%), but slightly less specific (67% vs 81%) than a common clinical test (ie, the Berg Balance Scale) in predicting accidental falls over a 3-month period. 27 Our findings are even more clinically relevant if we consider that the risk of accidental falls has been reported as increased by 8% for each 10-mm increase in COP path. 27
In our study, we also found an 11% to 14% improvement in the FSST. This clinical assessment has a sensitivity of 60% and a specificity of 75% in discriminating fallers and nonfallers among people with MS, if scoring above 16.9 seconds is used as a cutoff. 3 After WBBS training, both groups scored below this cutoff (see Table 3), thus suggesting its clinically relevant impact in terms of improved dynamic standing balance and reduced risk of falls.
We observed a slight but significant improvement (8% to 10%) in walking speed after WBBS training. This finding appears to be consistent with studies suggesting that an increase in more accurate postural control strategies leads to clinical improvement not only in balance skills but also in walking performance.12,21,22,37 The improvement in mobility, promoted by practicing high-intensity, repetitive weight shifting exercises, could be explained also by other mechanisms, such as muscle strength reinforcement,21,41 restoration of axial control and postural anticipatory strategies, 37 or simply enhancement of fitness level. 21 However, the threshold of 20% change, which is considered as clinically meaningful, 42 was reached only by about a quarter of patients after the intervention.
The 10% to 12% improvement in the MSIS-29 confirms previous assumptions that balance training can have a positive impact on patients’ QoL. 12 After WBBS training, the MSIS-29 scores of both groups were reduced, on average, by 9 or 10 points (see Table 3). This last finding is even more relevant when we consider that a minimal change score of 8 points in the MSIS-29 has been demonstrated as clinically significant. 43
Last, after WBSS training, the proportion of nonfallers was increased with respect to the 12-week period before randomization. However, this finding has to be interpreted cautiously because of the following: (a) retrospective self-report of falls is prone to recall bias, 3 (b) the time frame for optimal reporting of falls is unknown, 44 and (c) comparison was made between a self-reported measure and a prospectively collected measure.
In our study, we observed a maintained improvement only on the primary endpoint over the 12-week observation period post-intervention (Figure 3, group A), thus suggesting a stronger benefit of WBBS training on static balance, but not on dynamic balance, walking speed, and QoL. Yet this is not an unexpected result, if we consider that (a) WBBS training is specific for static standing balance rather than dynamic balance or gait and (b) a previous study demonstrated that benefits gained from rehabilitation are partly maintained until 6 months after the discharge, despite worsening neurological status. 45 On the other hand, there was a trend toward baseline values in group A after 12 weeks of not doing the WBBS. This could suggest the requirement for ongoing WBBS training to maintain benefit. Yet it is still unclear how long and how often people with MS need to exercise balance with WBBS.21-23
Although in our study only 1 patient had to retire from the study due because of to an adverse event related to WBBS training, 4 more patients reported the occurrence of knee or back pain (Table 4), graded as mild or moderate. This is not surprising, considering that injuries associated with specific videogames (the so-called Wii-itis or Nintendinitis) have been recently described even in healthy populations46-48 (see also http://www.nintendo.com/consumer/wiisafety.jsp). In our experience, games requiring body shifts in a fast motion, such as “Soccer Heading,” were more often related to occurrence of knee and back pain.
We scheduled engagements with physiotherapists every 4 weeks during WBBS training, thus leading to a better management of these symptoms. Although it has been reported that patients who have mild balance and mobility deficits can safely use WBBS in their homes, 21 no data are yet available on patients with more severe disability. Therefore, a home-based WBBS training is still applicable only to people with MS who are still ambulant (EDSS 5.5 or less).
Implications
The implications of our study may be significant for people with MS, who are most often neglected for rehabilitation care, especially in early stages of disease. Improvement in balance measures and mobility following the use of WBBS has been also reported in other MS populations,21-23 although one recent study suggests only moderate effect sizes not reaching statistical significance. 23
According to our findings, WBBS training seems to have a smaller effect than other balance rehabilitation approaches for MS, such as visuo-proprioceptive training, 12 torso-weighting, 33 sensory strategy retraining, 40 hippotherapy, 49 vestibular rehabilitation, 50 and resistance training. 51 However, the large standard deviations of percentage changes observed in both groups during the intervention period suggest that improvements deeply varied across patients. As a consequence, WBBS cannot yet be considered an alternative to standard rehabilitation.
Owing to the risk of adverse events and its relative effectiveness, we strongly suggest careful monitoring of patients during WBSS training, especially in the first stage and when more strenuous exercises are introduced. Thereafter, patients could be supervised less closely (monthly, for example), and just to verify their progresses over time. In this way, WBBS might potentially offer a cost-effectiveness at least equal (or even better, as previously reported 22 ) than standard care through the reduction in transport and staffing costs. Unfortunately, we did not carry out a cost-effectiveness analysis to support this statement.
Limitations
Limitations of the present study mainly concern the small sample size, absence of blindness, and the study design lacking a washout period.
Data regarding the occurrence of accidental falls prior to study enrollment were not reliably captured, therefore affecting a consistent comparison with respect to study period. Lower extremity strength was not measured; hence, we cannot assess whether muscle reinforcement may have played a role in determining the balance improvement. Moreover, although our computer-based measure of balance is accurate and reliable,27,31 it was evaluated only in the most simplistic condition (static condition, open eyes). Last, an evaluation of sitting balance performance is lacking, although WBBS training also included one game to be played in sitting stance.
Conclusion
A home-based WBBS training could be considered for MS patients with balance impairment, especially in those who have a mild to moderate disability level and are still employed. It would permit an inexpensive and pleasant homecare approach, also increasing the openness to rehabilitation. However, it could be used as part of a supervised physiotherapy-based exercise program, in order to minimize the risk of adverse events and training-related injuries. Further efforts are warranted to better estimate the risk–benefit ratio (adverse event rate vs efficacy) before the WBBS is used for clinical purposes.
Footnotes
Acknowledgements
We thank Mr Massimo Fortuna for his helpful contribution.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LP received consulting fees from Merck Serono and lecture fees from Biogen Idec and Teva. CP received consulting and lecture fees from Sanofi-Aventis, Biogen Idec, Bayer Schering, Merck Serono, and Novartis; he also received research funding from Sanofi-Aventis, Merck Serono, and Bayer Schering.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
