Abstract
Background. Action observation has been suggested as a possible gateway to retraining arm motor function post stroke. However, it is unclear if the neuronal response to action observation is affected by stroke and if it changes during the course of recovery. Objective. To examine longitudinal changes in neuronal activity in a group of patients with subacute stroke when observing and executing a bimanual movement task. Methods. Eighteen patients were examined twice using 3-T functional magnetic resonance imaging; 1 to 2 weeks and 3 months post stroke symptom onset. Eighteen control participants were examined once. Image time series were analyzed (SPM8) and correlated with clinical motor function scores. Results. During action observation and execution, an overlap of neuronal activation was observed in the superior and inferior parietal lobe, precentral gyrus, insula, and inferior temporal gyrus in both control participants and patients (P < .05; false discovery rate corrected). The neuronal response in the observation task increased from 1 to 2 weeks to 3 months after stroke. Most activated clusters were observed in the inferior temporal gyrus, the thalamus and movement-related areas, such as the premotor, supplementary and motor cortex (BA4, BA6). Increased activation of cerebellum and premotor area correlated with improved arm motor function. Most patients had regained full movement ability. Conclusions. Plastic changes in neurons responding to action observation and action execution occurred in accordance with clinical recovery. The involvement of motor areas when observing actions early and later after stroke may constitute a possible access to the motor system.
Introduction
The reorganization of motor areas after stroke has been examined in numerous imaging studies, focusing primarily on possible correlations between task execution and recovery of motor function, for example, reviews by Johansen-Berg 1 and Rehme et al. 2 Results from behavioral and neurophysiological studies have suggested that mere observation of movement facilitates motor memory function and motor performance both in healthy individuals and patients with stroke.3-5 The potential mutual relationship between task execution and task observation is commonly referred to as mirror activity. More specifically, neurons that are activated both when a task is executed and when a similar task is observed are referred to as mirror neurons, and were first reported in macaques more than 2 decades ago. 6
The existence of mirror neurons in humans, however, remains controversial; for example, the review by Turella et al. 7 One point of discussion is why neurons with mirror properties are seemingly more widespread in the human brain in contrast to the spatially well-defined regions originally reported in macaques. 8 It has been pointed out that large differences in choice of methodology such as the lack of inclusion of both an action observation and action execution task, combined with large differences in task selection can possibly account for many of these discrepancies. 9 Neurons responding to the observation of hand actions have been reported in the premotor and supplementary motor cortex, the inferior frontal gyrus, the inferior parietal cortex and in the posterior middle temporal gyrus in healthy adults. 10
There is a considerable interest in exploiting mirror neurons to improve motor function after stroke. Initial studies applying the observation of motor activities as a treatment approach have shown promising results, indicating that action observation could be a possible avenue to retraining motor function after stroke.3-5 However, little is known about reorganization processes in neurons with mirror activity after stroke. With focus on rehabilitation, it is important to investigate if similar neuronal responses exist in patients with lesions affecting the motor network. In the current study, functional magnetic resonance imaging (fMRI) based on blood oxygenation level–dependent contrast was used to investigate the neuronal activation when observing and executing a bimanual motor task in a group of patients with subacute stroke.
The main objective of this study was to identify for the first time a possible action observation/action execution matching system in patients with stroke during the course of recovery. An overlap of neurons involved in execution and observation in both patients and healthy control participants was expected, and that overlapping clusters of neurons would include brain regions that are typically associated with the mirror neuron system, such as premotor cortex, inferior frontal gyrus and inferior parietal cortex.
Materials and Methods
Participants
Twenty patients with stroke as confirmed by routine diagnostic scanning were included, see Table 1. All patients underwent standard physical, radiological (computed tomography/MRI) and neuropsychological examination when admitted to the stroke unit. Inclusion criteria were adults (>18 years), time since stroke symptom onset <15 days, impaired upper extremity motor function (<0.5 NHPT [Nine-Hole Peg Test], see below), 11 but some preserved visible active flexion and extension of the fingers, and ability to stand and transfer. Exclusion criteria were no prior history of stroke, pronounced neglect, visual impairment interfering with the study protocol or major cognitive problems (<21 on the Mini-Mental State Examination). The patients were scanned twice, the first time within 2 weeks (mean 8.9 ± 4.1 days) and the second time 3 months (mean 89.3 ± 8.3 days) after stroke onset. All but one patient, who participated in additional arm training, received comprehensive standard rehabilitation.
Patient Characteristics and Outcome Scores.
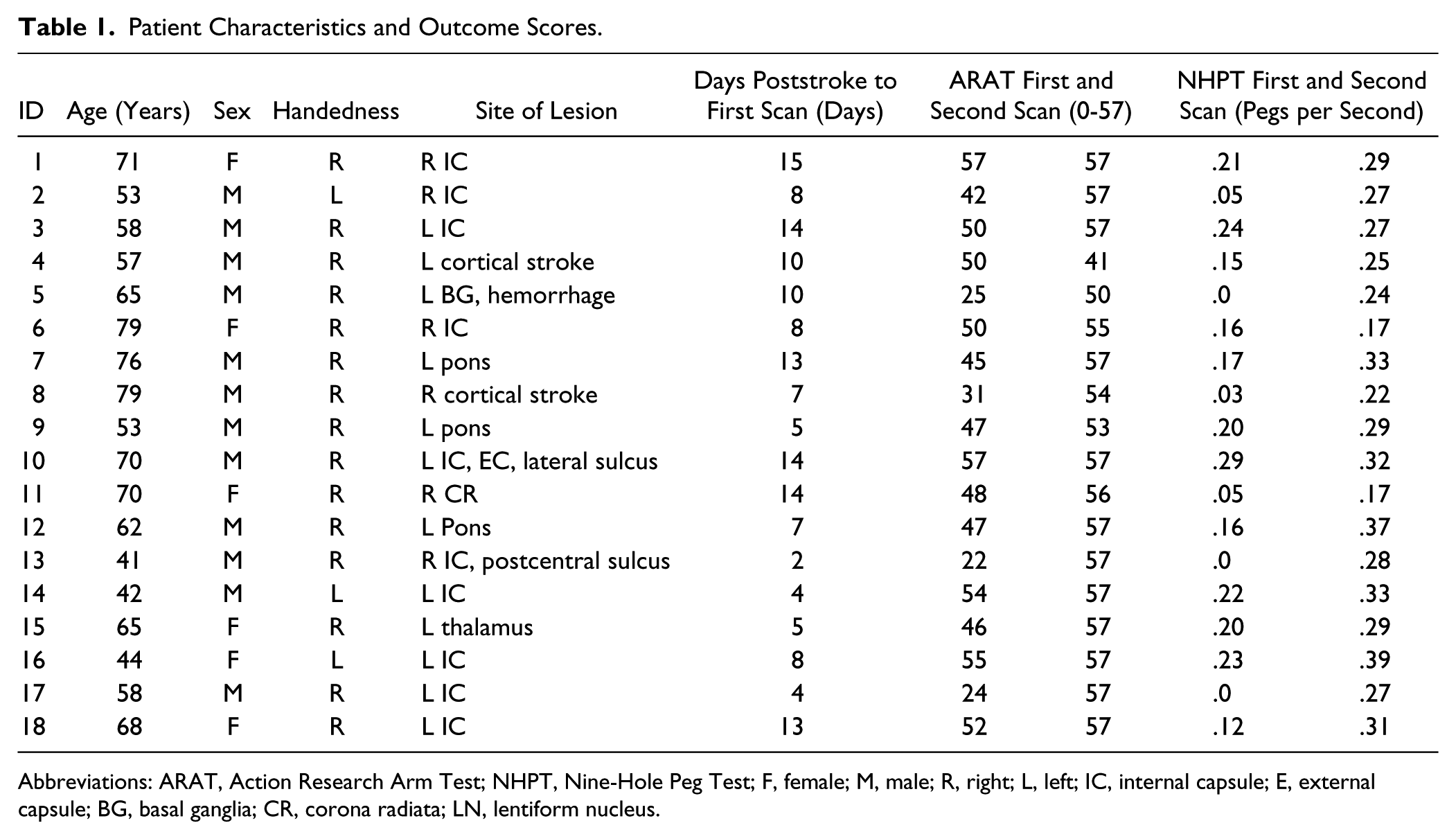
Abbreviations: ARAT, Action Research Arm Test; NHPT, Nine-Hole Peg Test; F, female; M, male; R, right; L, left; IC, internal capsule; E, external capsule; BG, basal ganglia; CR, corona radiata; LN, lentiform nucleus.
Twenty age- and sex-matched control participants, with no history of neurological disease, mean age 60.6 ± 11.7 years were scanned once. The control participants received a small financial compensation for participation covering travel costs. The study was approved by the regional ethic committee and informed written consent was obtained from all participants according to the Declaration of Helsinki.
Two patients were excluded from the subsequent analysis because they did not participate in the follow-up scanning. The remaining patient group included patients with subcortical and cortical stroke lesions (n =18, 60.7 ± 11.6 years old, 6 females, 3 left-handed, details in Table 1 and Figure S1 in Supplementary Material). Data from 2 neurologically healthy persons were excluded from the analysis, because they did not comply with the task instructions, leaving 18 in the control group, 60.6 ± 11.9 years old, 7 females, 1 left-handed).
Clinical Assessments of Motor Function
Patients underwent arm motor assessment on both test occasions with the Action Research Arm Test (ARAT) 12 and the NHPT. 13 The ARAT comprises 19 items for measuring different aspects of arm motor function, with a total score ranging from 0 to a maximum of 57 for best motor function. The NHPT is used to assess fine motor skills, and is measured as the time it takes for a patient to place and subsequently remove 9 pegs in holes. Arm motor function change scores from the first to the second assessment are expressed as pegs per second. ARAT and NHPT scores were examined by related-samples Wilcoxon signed rank test using SPSS 19.0 (IBM SPSS, Armonk, NY).
Functional Magnetic Resonance Imaging Tasks
The functional imaging consisted of 2 tasks; first to execute a repeated, self-paced bimanual twisting of a cylindrical device, and then to observe a video clip showing the same device being twisted (Supplementary Figure S2). An fMRI block-design was used in both tasks where 2 rest blocks (each 30 seconds) were interleaved by one active block (30 seconds). The rest condition in the execution task was holding the device without twisting it, while the rest condition in the observation task was to watch a still image of the device being held at rest. In both tasks, a pair consisting of one rest block and one active block was repeated 4 times.
All participants were thoroughly instructed before entering the scanner. A “start” and “stop” sign indicated when the participant was to start the twisting of the device and when to rest. The cylindrical device was placed in and removed from the hand of the participant for this task. Compliance with the instructions was ensured by visual inspection throughout the exam.
Functional Magnetic Resonance Imaging Acquisition
Imaging was performed using a 3T Signa Excite (General Electric, Milwaukee, WI) employing an identical protocol for the acute phase exam and the follow-up exam. T1-weighted anatomical images (3D FSPGR, 180 slices, 1 mm3 voxel resolution, echo time [TE]/repetition time [TR] = 3.2/8.0 ms, flip angle = 11°) were acquired first to anatomically position the slices in the fMRI acquisitions parallel to the anterior and posterior commissure.
For the fMRI acquisitions, a gradient recalled echo EPI (echo planar imaging) sequence (TE/TR = 25/2000 ms, flip angle = 90°, 120 volumes/task, 35 slices/volume, voxel size 3 × 3 × 4 mm3) was sensitized to the blood oxygenation level–dependent contrast in time. Stimulus presentations were triggered in time by the MRI scanner and presented to the participants through dedicated MRI compatible goggles with correction for eyesight (Stimulus presentation software and hardware solutions NordicNeuroLab Inc, Bergen, Norway).
Functional Magnetic Resonance Imaging Analysis
The analysis was carried out using the Statistical Parametric Mapping software package (SPM8, Wellcome Department of Cognitive Neurology, London, UK) using Matlab 7.7 (Mathworks Inc, Natick, MA). The first 5 EPI image volumes in each task were discarded to account for signal intensity variations because of lack of steady state signal. The remaining image volumes in each time series were realigned to the first functional image volume in the series to correct for head movement.
In order to perform group analysis across participants, it was necessary to normalize (3 mm3) all image data to a stereotaxic space using the provided EPI template in the SPM8 software. Lesion sizes were determined based on clinical diffusion-weighted imaging (b = 1000 s/mm2, Supplementary Figure S1). Because larger lesions may substantially deteriorate the performance of automated registration algorithms and because large lesions may cause artificial activation signals over a large spatial area, 3 additional patients (IDs 4, 5, and 13; Table 1) were excluded from further analysis. All normalized image volumes were visually inspected to assess potential problems with normalization and then smoothed (Gaussian kernel with full-width at half-maximum of 8 mm) to meet the requirements of the subsequent statistical modeling. A high-pass filter (128 seconds) was applied to remove unwanted physiological influences such as cardiorespiratory cycles.
To perform a best fit of the measured fMRI time series using a general linear model, a boxcar function defining the timing of the paradigm was convolved with a canonical hemodynamic response function. Areas of statistically significant differences in signal intensity were determined using t-statistics on a voxel basis. In each individual, the resulting statistical maps were contrasted in all cases to the respective “rest” condition before being used in a second level analysis. Because lesions in the current patients were localized in either hemisphere, patients with lesions in the right hemisphere were mirrored to produce a common “affected hemisphere” (left hemisphere), and a corresponding seemingly “unaffected hemisphere” (right hemisphere).
For the second level analysis, a full factorial design was used with 2 tasks, action observation (AO) and action execution (BIL), resulting in statistical maps corresponding to patient group at 2 weeks poststroke (AO1 and BIL1), patient group 3 months poststroke (AO2 and BIL2), and baseline control group (AO and BIL), respectively. In all baseline activations, a whole-brain significance threshold of P < .05 false discovery corrected for multiple comparisons was used. A one-way within-subjects analysis of variance was performed repeatedly for the action observation and action execution task and also, it was investigated whether changes in neuronal activation in the action observation task correlated with improvements in motor function (ARAT score). For this, a multiple regression analysis was performed using the normalized ARAT score as covariate [(ARAT2 – ARAT1)/(ARAT1 + ARAT2)]. Five regions of interest were particularly investigated using small volume analysis to further explore changes in time; the cerebellum, the thalamus, the premotor cortex (BA6), the inferior parietal cortex and pars opercularis (BA44). A conjunction analysis between action observation and action execution was performed to better identify neuronal clusters that were active in both tasks (“overlap”).
Finally, to investigate differences in laterality of the neuronal activation, a region of interest (ROI) analysis was performed. A total of 9 ROIs were extracted based on the mounting literature on brain regions described to have mirror neuron properties,9,10 that is, the cerebellum, the pre- and postcentral gyrus, the supplementary motor cortex, the superior and inferior part of the parietal cortex, 2 regions in the frontal lobe, using IBASPM. 14 A laterality index (LI) based on the number of activated voxels in the “affected” hemisphere and “unaffected hemisphere” was estimated for each ROI [(Affected hemisphere – Unaffected hemisphere)/(Affected hemisphere + Unaffected hemisphere)]. A cut of LI = 0.5 was chosen to be reported, with LI = 0 indicating symmetric patterns of activation and LI = 1 indicating asymmetric patterns of activation across hemispheres.
Statistically significant voxel coordinates were transformed from MNI (Montreal Neurological Institute) to Talairach space 15 by nonlinear transformation (using the “icbm2tal” transform, http://brainmap.org/icbm2tal). 16 The peak voxel coordinates, maximum of 3 peaks reported per cluster, were labeled using the Talairach Daemon software (http://www.talairach.org/daemon.html). 17 The resulting activation maps were visualized using the rendering tool in SPM8 or the MRIcron software (Version 4. http://www.mricron.com).
Results
Clinical Assessment of Motor Function
Most patients (n = 16) significantly improved arm motor function from first to second exam, obtaining a score in the ARAT test of more than 50 points, see Table 1. One patient’s motor function worsened on ARAT because of shoulder pain. Almost two thirds (n = 11) of the patients recovered well enough to achieve the top score of 57 points at the second assessment, and one had already achieved 57 at first exam. Similarly, all experienced a significant improvement on the NHPT test (P < .05). Scores of functional assessments are presented in Table 1.
Neuronal Activation When Observing the Bimanual Task
Concomitant to the improvement of arm motor function the spatial extent of activation increased after 3 months in patients (Figure 1, Table 2, and Supplementary Figure S3).
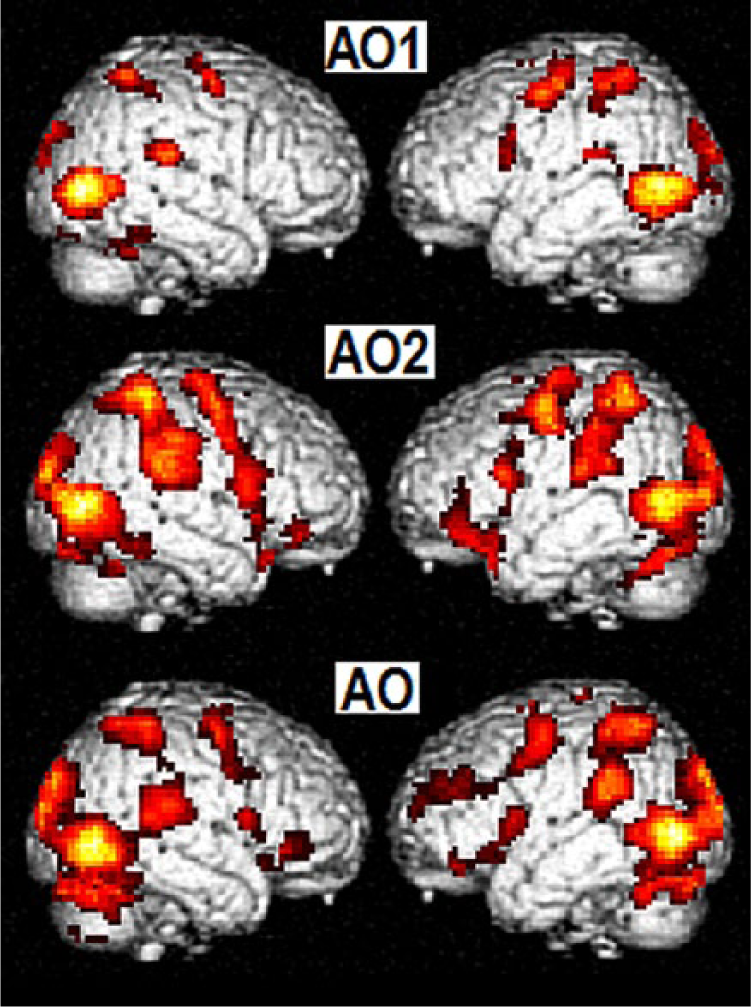
Observation of bimanual movement in patient group—2 weeks poststroke (top row, AO1) and 3 months poststroke (middle row, AO2)—and control group (bottom row, AO). Neuronal activation mapped on to a standard rendering template: right hemisphere (left column), left hemisphere (right column) at statistical threshold P < .05, false discovery rate corrected, including only clusters with spatial extend of 20 voxels. The neuronal activation in the observation task is reduced after 2 weeks compared with 3 months poststroke and control group.
Within-Patients Change in Neuronal Activation During Observation (AO1, AO2) and Performance (BIL1, BIL2) of Bimanual Movement Across Time. a
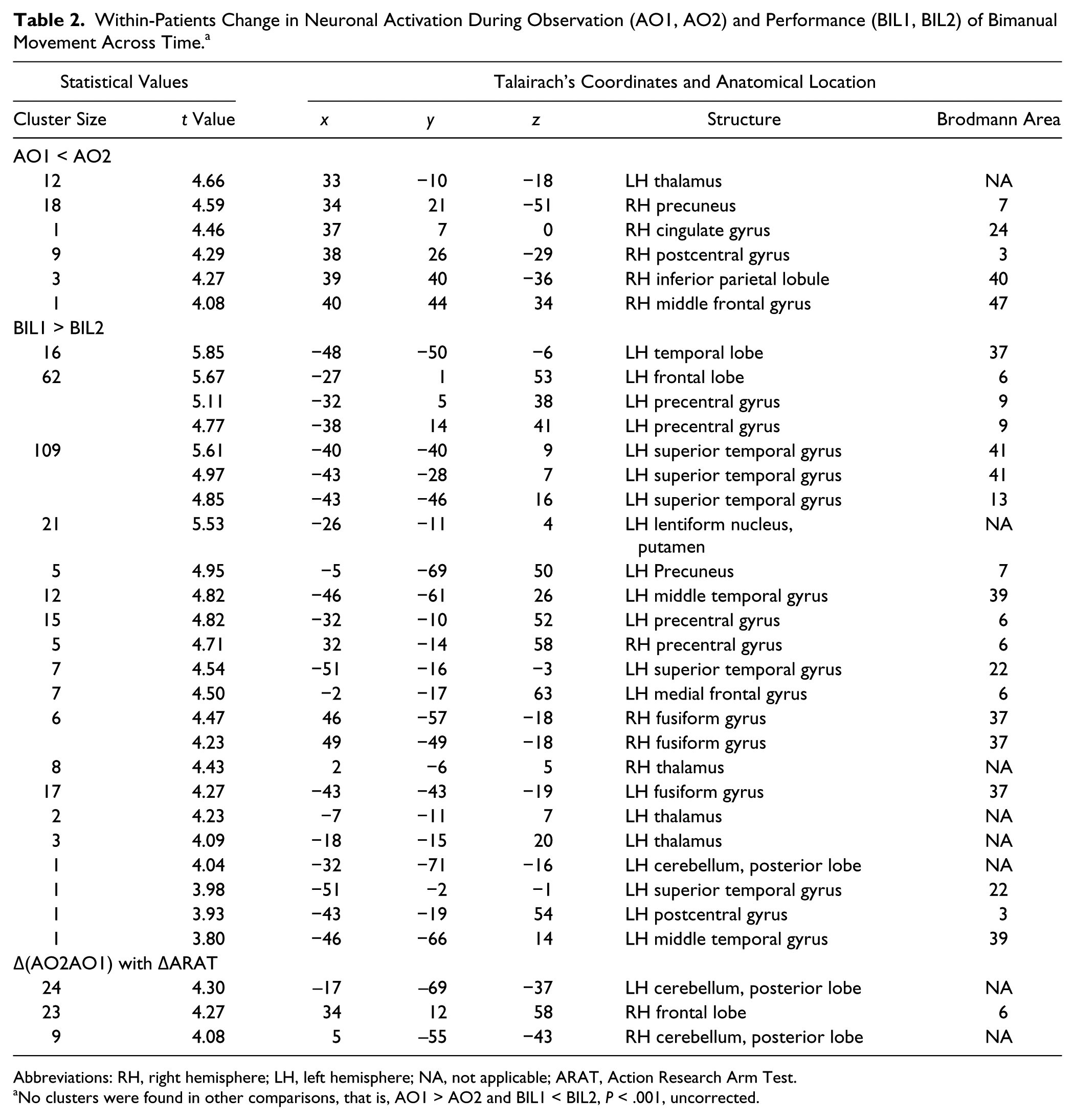
Abbreviations: RH, right hemisphere; LH, left hemisphere; NA, not applicable; ARAT, Action Research Arm Test.
No clusters were found in other comparisons, that is, AO1 > AO2 and BIL1 < BIL2, P < .001, uncorrected.
Patients, ~2 Weeks Poststroke (AO1)
Imaging data of the action observation condition at first examination revealed, as expected, much involvement of occipital and temporal visual areas bilaterally, with activation maxima in the middle and inferior temporal gyrus, and the occipital lobe, probably including the extrastriate body area (EBA), sensitive to the perception of body parts. The patients also showed activation in distinct parietal and frontal areas that are associated with the mirror neuron system. Notably, the inferior and superior parietal lobes (BA40, BA7) and the inferior frontal gyrus (BA44) were involved, as well as the primary motor cortex (BA4; Figure 1).
Patients, ~3 Months Poststroke (AO2)
At the second exam, the general extent of activated clusters had increased considerably. Most activated clusters were observed in the inferior temporal gyrus and the ventral anterior nucleus of the thalamus, which provides major input to the premotor cortex, and the medial dorsal nucleus associated with memory. Other movement-related areas, such as the premotor, supplementary, and motor cortex (BA4, BA6) showed large spatially extended responses to action observation.
A tendency of increase in activation over time was observed in distinct regions particularly in the “unaffected” (right) hemisphere and the left thalamus, while no significant clusters were found in the reverse comparison AO1 > AO2 (Table 2; P < .001, uncorrected). Further investigations in predefined regions of interest (small volume analysis) showed in particular a significant increase in activation in the thalamus (P < .05, false discovery rate corrected). Interestingly, changes in ARAT scores correlated with changes in the neuronal activation during the action observation task both in the premotor area and in the cerebellum (Table 2; P < .001 uncorrected). An ROI analysis for smaller volume investigations confirmed these correlations in the left cerebellum (P = .043 for cluster of 21 voxels using family-wise error correction), but significant results were not found in the other regions that were predefined for analysis, that is, the thalamus, the premotor cortex, the inferior parietal cortex, and the pars opercularis.
Control Participants (AO)
The control subjects showed activation including the occipital cortex and distinct areas in frontal and parietal lobes corresponding to areas associated with the mirror neuron system (BA40) and premotor cortex (BA6). The largest extent of activated clusters was found in the posterior lobe of the cerebellum, usually associated with fine motor control. Among the areas responding in healthy controls and patients at both exams were the inferior and superior parietal lobes, premotor areas and insula.
Neuronal Activation When Executing the Bimanual Task
When executing the bimanual task, the cerebellum, premotor cortex and supplementary motor cortex were strongly activated in both patients and controls. Tendencies of decreased activations were particularly seen in areas of the precentral gyrus (BA6; Table 2; P < .001, uncorrected). This finding was also seen in a small volume analysis in the precentral gyrus (P < .05, false discovery rate corrected; Figure 2).
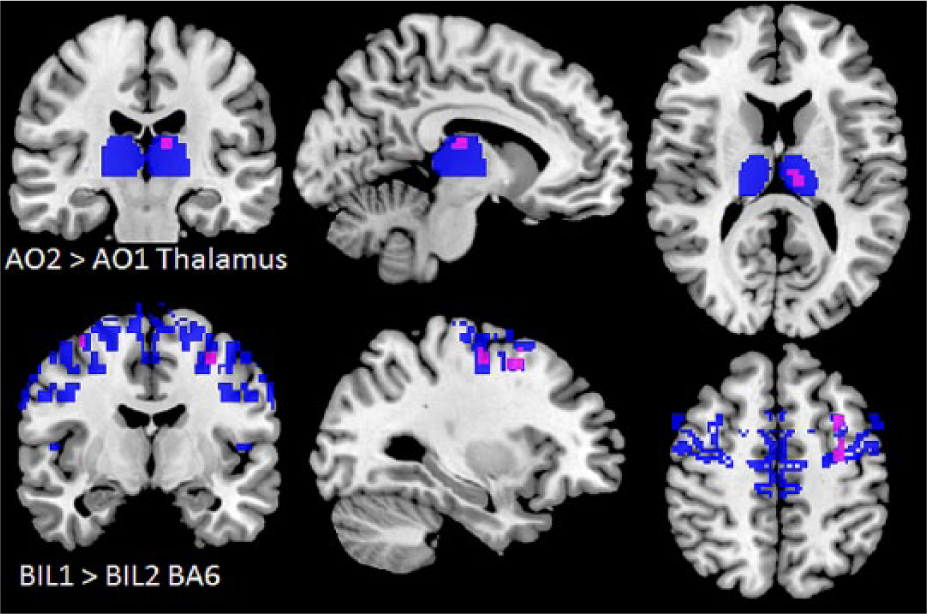
Within-patients change in neuronal activation during observation (AO1, AO2) and performance (BIL1, BIL2) of bimanual movement across time in small volume corrected analysis in predefined regions of interest (blue) in the cerebellum, the thalamus, the premotor cortex (BA6), the inferior parietal cortex, and pars opercularis (BA44), respectively. Only significant differences, P < .05, false discovery rate corrected, are shown (pink).
Contrary to changes in the action observation task, patterns of activation thus were decreased in time and became more symmetric across cerebral hemispheres in time, as indicated by the lateralization index, LI2weeks = 0.5, LI3months = 0.1, LIcontrols = −0.2 (Supplementary Material, Figure S3). It is noteworthy that the spatial extent of the neuronal activation when performing the task was almost twice that of purely observing the task both in patients and controls (Supplementary Material, Figure S3, eg, AO vs BIL).
Overlap Between Observation and Execution
To reveal areas activated both in the action observation condition and during performance, a conjunction analysis was performed (Table 3, Figure 3) wherein activated clusters during the action observation were masked with the corresponding bilateral performance in control participants and patients, respectively. A common factor for all participants was a pronounced activation of the inferior parietal lobe. Motor-related areas, such as primary motor cortex, premotor cortex, and insula responded to all conditions, as well.
Conjunction of Neuronal Activation in Areas Active Both During Performance and Observation of Bimanual Movement for Patients, 2 Weeks and 3 Months Poststroke Onset, and Controls. a
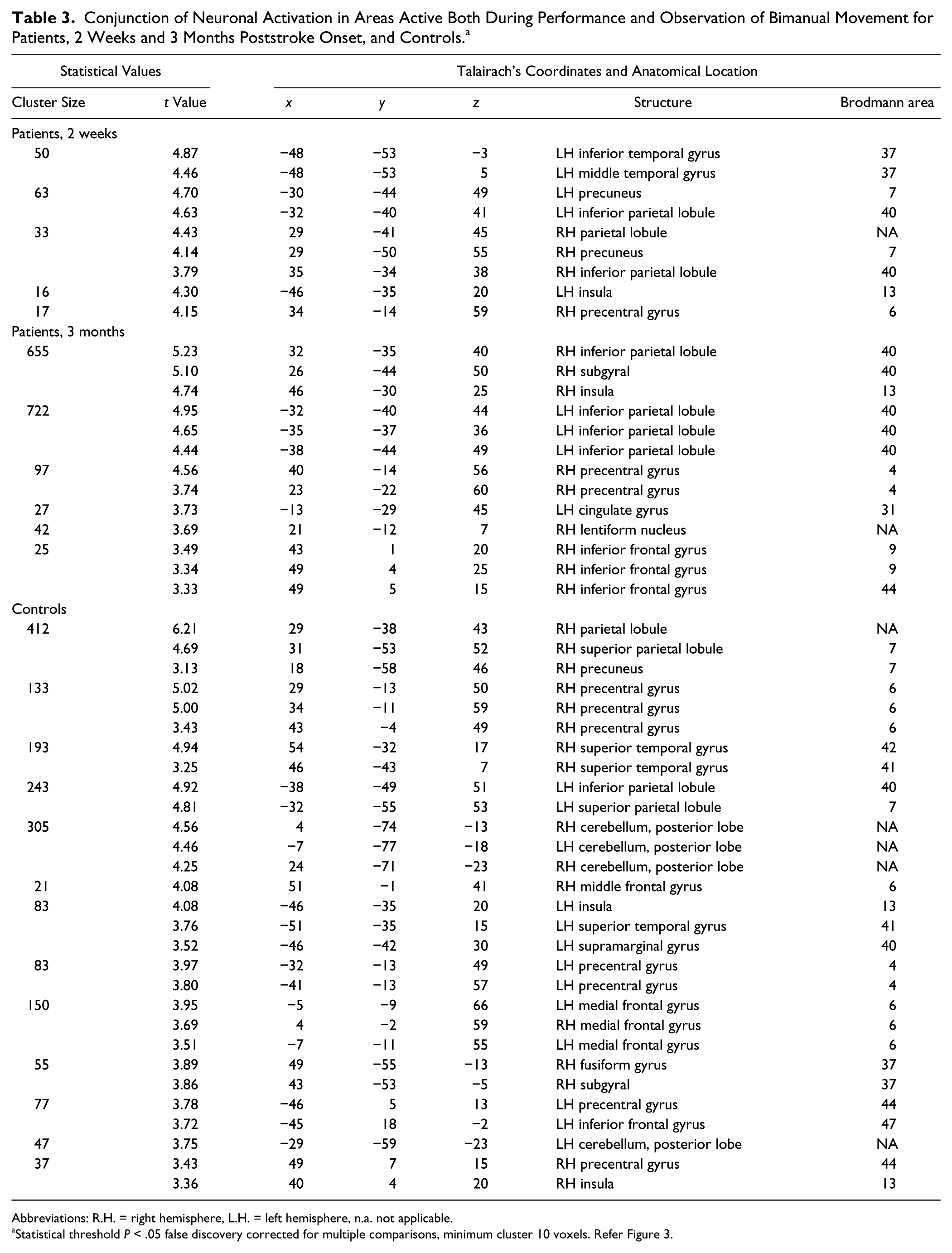
Abbreviations: R.H. = right hemisphere, L.H. = left hemisphere, n.a. not applicable.
Statistical threshold P < .05 false discovery corrected for multiple comparisons, minimum cluster 10 voxels. Refer Figure 3.
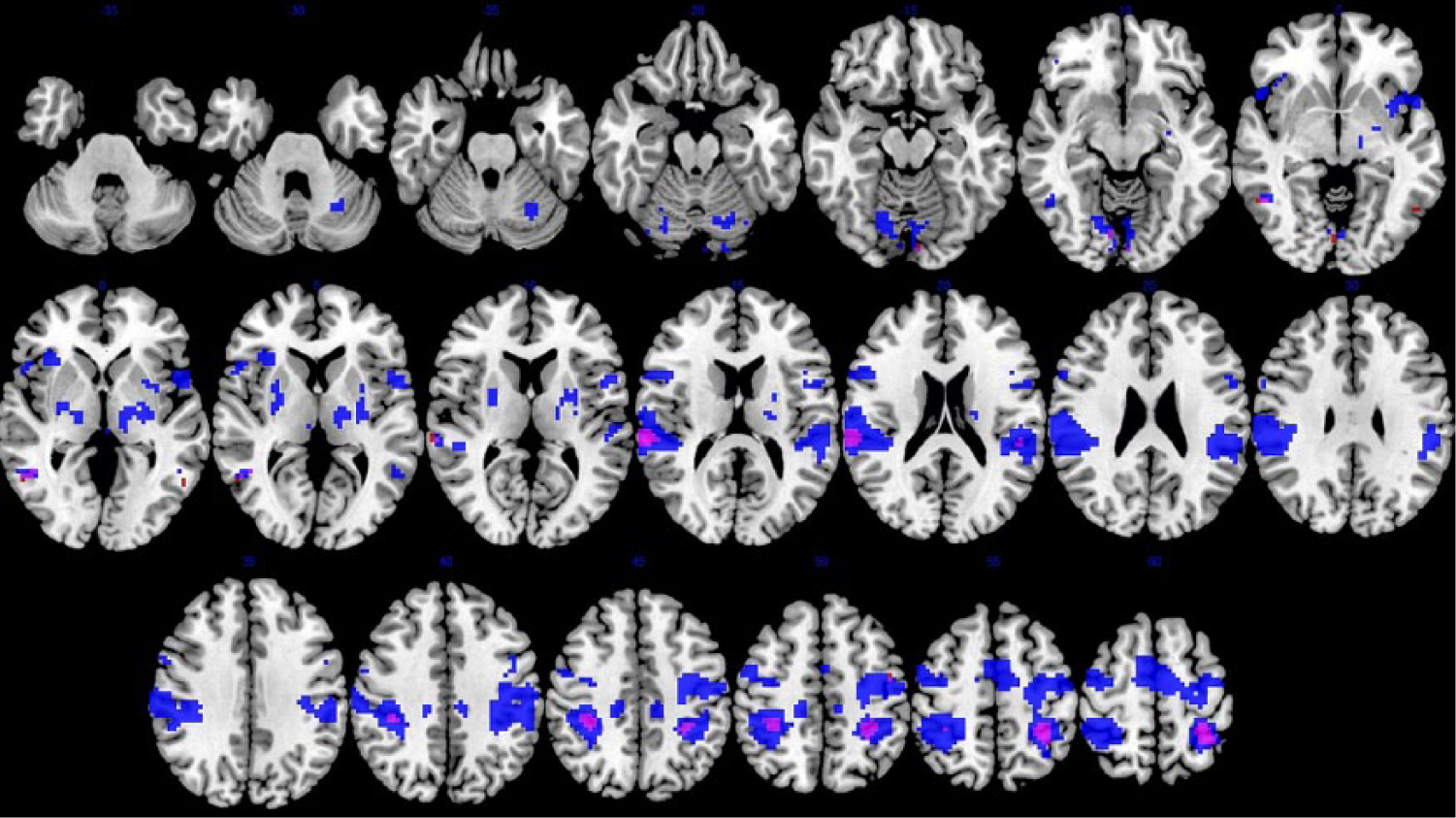
Conjunction in neuronal activation from observing and performing bimanual movement. View of axial slices in the patient group; 2 weeks poststroke (red) and about 12 weeks poststroke (blue). Statistical threshold P < .05, false discovery rate corrected. See Table 3 for details.
Discussion
To our knowledge, this is the first longitudinal fMRI study to include both an observation and an execution task in stroke patients. While the spatial distribution of the neuronal activation decreased after 3 months when executing a bimanual motor task, watching the same task being performed actually showed increased patterns of activation. Interestingly, the increased clusters overlap with areas typically associated with mirror neuron systems, that is, inferior frontal gyrus, the premotor cortex, and the inferior parietal lobule, and it was also seen that improved arm motor function scores were correlated with increased activation in the premotor areas and cerebellum. Thus, the neuronal response to action observation appears to change during the course of recovery and is seemingly associated with good recovery.
Similar findings of upregulation poststroke have also been described with respect to reorganization of language poststroke. 18 Language function improved in accordance with increased activation of a bilateral language network involving the right Broca-homologue in the subacute phase (2 weeks after stroke onset). A continuous upregulation of left hemispheric inferior frontal gyrus and middle temporal gyrus occurred throughout the recovery process. In the chronic phase, that is, more than 4 months poststroke, a reshift to the left hemisphere and a decrease of activation was observed. The longitudinal follow-up in the current study may still be considered part of the subacute stroke phase. Because of the lack of information on the chronic phase, it is unknown if the observed upregulation in the bimanual observation task is a transient compensatory mechanism similarly to what is observed in the language network.
Interestingly, lateralization was more pronounced during observation than execution of a bimanual motor task early after stroke, and developed to the expected bilateral activation pattern after 3 months. Increased lateralization has previously been reported in a study where patients observed movements of the affected right hand. 19 The patterns were then lateralized toward the ipsilateral left hemisphere and differed from findings in healthy controls.
Both during observation and execution of the bimanual motor task, changes in the involvement of thalamus were observed during recovery. Thalamus activation was not observed in the control participants. Although the thalamus has not typically been ascribed mirror properties, it is regarded as an important relay station. Extensive connections extend from the ventral anterior nuclei to prefrontal areas, and a major input to premotor areas from the ventral lateral nuclei is associated with voluntary movements.20,21(pp184-187) The strong thalamic response 3 months poststroke may be interpreted as a potential compensatory mechanism, emphasizing the importance of somatosensory feedback for functional recovery.
We observed a task related downregulation, as well as a shift toward more symmetric activation, in the action execution task over time. Changes in neuronal motor networks has been reported in several fMRI studies in patients with stroke.2,22 Ward et al 23 found that a decrease of task-related activation correlated with the rate of recovery. Askim et al 24 reported increased lateralization and a shift of activation back to ipsilesional sensorimotor cortex in patients 3 months poststroke. It has been shown that some patients may rely on extended cortical and subcortical structures to create motor output, while well-recovered patients recruit additional bihemispheric sensorimotor, cerebellar, and other specific areas when performing a motor task. 25 Buma et al 26 reviewed neuroimaging studies of recovery of arm function within 6 months poststroke. They identified a trend toward unilateral overactivation, a posterior shift in activation in the primary motor cortex and bihemisheric activation of motor and nonmotor areas early after stroke, which seems to decrease concomitant with recovery. Accordingly, our well-recovered patients demonstrated a tendency toward decreased activation at 3 months, particularly in premotor areas.
In the current study, improvement of arm motor function was correlated with an increase of activation in the premotor area (BA6) and the cerebellum when observing the bimanual motor task. Cerebellar and premotor functions are regarded as critical for bimanual coordinated movements, 27 and the role of the cerebellum for motor recovery has also been emphasized in a longitudinal fMRI study by Small et al. 28 Johansen-Berg et al. 29 found correlations of motor function improvement and increased activation in premotor and cerebellar areas when performing a motor task. Our findings may indicate that motor representations in relevant areas can be more easily evoked with improved motor function.
Did the Observed Changes Reflect Mirror Activity?
The concept of mirror neurons is still under debate. However, when adhering to the definition by Rizzolatti et al, 30 who described “visuomotor neurons” discharging both when an action is executed and when it is merely observed, it can be claimed that several of the activations observed in the present study reflected mirror activity. The actual execution of the bimanual task was associated with a decrease of activation after 3 months, in contrast to the observation task. This lends strength to the assumption that mirror activity and not a general upregulation was observed. Pronounced involvement of the inferior parietal lobe and premotor areas was found both in patients and control subjects. These results are in accordance with the results from a meta-analysis by Molenberghs et al 9 where mirror-related activity in the inferior parietal lobe and in premotor areas was found in most studies included. The inferior parietal lobe is concerned with movement planning and its special relevance for object-related actions has been demonstrated. 31 Caspers et al 32 found a stronger involvement of the inferior parietal lobe in studies where the observation of hand actions included objects as compared to hand movements only. The inferior parietal lobe is regarded as an essential part of the mirror neuron system; thus, the use of an object in our study may have contributed to even stronger activation. Premotor area 6 has consistently been ascribed mirror properties in action observation and imitation, as well.9,32 However, we cannot claim that the activated clusters observed actually comprise mirror neurons. Gazzola and Keysers 33 proposed the neutral term shared voxels to describe neurons that respond to both execution and observation. In a study from 2009, they analyzed unsmoothed single-subject data from healthy subjects when executing and observing actions. Interestingly, shared voxels were observed in similar areas as in the current study, such as the premotor cortex (BA6), the parietal cortex, the inferior and middle temporal gyrus (BA37), and the cerebellum.
Implications for Rehabilitation After Stroke
Mirror neurons have been presented as a possible avenue to restore motor function after stroke.34,35 The concomitant involvement of precentral, postcentral, and parietal areas and inferior frontal gyrus when observing a motor task early and at a later stage after stroke corroborates the possibility of potentially enhancing motor recovery by action observation. However, putative mirror activity is apparently more widespread in humans than in monkeys and can be altered by different factors. Earlier studies with healthy persons suggest that familiarity with the observed movement, the observed movement being part of one’s own motor repertoire and the intention to imitate play a role for the activation elicited.35-37 In patients, the localization of the stroke is likely to be a limiting factor when affecting cortical areas associated with mirror activity. On the other hand, widespread mirror activity may provide multiple accesses to different brain areas, and observation may facilitate motor relearning even in paralytic patients. 34 More clinical and imaging studies are needed to explore the potential of an observation / execution network for rehabilitation and to determine which patients may benefit.
Limitations of the Study
Only mild to moderately impaired patients took part, primarily with subcortical stroke or cortical strokes with mainly motor and little other cortical impairments, so the results cannot be generalized, for instance, to more severely impaired patients. Furthermore, the lack of an indicator of quality in motor performance constitutes a potential confounder. Differences in blood oxygenation level–dependent response between healthy controls and patients when executing the bimanual task could be attributed to differences in exertion of force and pace, although this should probably not influence the observation condition. The execution and observation were always presented in the same order, which comprises the risk of a systematical bias. Scanning healthy control participants on two occasions would have been valuable. Multiple fMRI assessments and a longer follow-up period would have provided further insight to the potential reorganization processes. The necessity to choose a task simple enough to be performed by patients with reduced dexterity and independent of handedness and lesion location was a limiting factor. To watch the same task on a video clip was monotonous; some participants reported difficulties in maintaining attention. More interesting video clips could have elicited different activation patterns.
The interpretation of the change in fMRI activation in our study remains difficult. The observed longitudinal changes are not specific for mirror motor neuron networks but rather reflect a common underlying mechanism of spontaneous neurological change. Also, the progress of time in itself is an important factor for recovery after stroke as described by Kwakkel et al, 38 and it is likely that improvement of function is accompanied by changes in neuronal activation. Processes promoting recovery, such as the reperfusion of penumbral tissue in the early days after stroke and compensation with the help of redundant networks and a remapping of cortical representations in neighboring areas at a later stage are reflected in different activation patterns.39,40 Additional assessments of the microvasculature, that is, perfusion, would have been valuable.
Further research should address longitudinal changes of potential mirror neurons at more points in time, including long-term follow-up 6 and 12 months after stroke. It would be important to study changes in patients with more severe paresis and poor recovery and compare them with well-recovered patients. A broader array of observed tasks, including object-related, unilateral, bilateral and ecologically valid tasks could help to determine what kind of observation elicits the strongest responses in patients with stroke at different points in time, thus helping provide targeted rehabilitation.
In summary, plasticity changes in potential mirror neurons were observed during the course of recovery, corresponding to an improvement of arm motor function. More research is needed to determine if these changes in activation may constitute a possible access to the motor system.
Footnotes
Acknowledgements
We thank all the patients and volunteers for their participation in the study. We also thank Professor Kenneth Hugdahl for valuable comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Grieg Foundation and the Rieber Foundation. The first author received a grant from the Norwegian Fund for Post-Graduate Training in Physiotherapy.
