Abstract
Introduction
Tinnitus is defined as the perception of sound that does not occur in the environment. 1 Multiple overlapping and parallel brain networks such as the auditory cortex, somatosensory cortex, memory area (hippocampus, parahippocampus, and amygdala), perception (subgenual and dorsal anterior cingulate cortex, posterior cingulate cortex, precuneus, parietal cortex, and frontal cortex), salience (dorsal anterior cingulate cortex and anterior insula), and distress networks (subgenual and dorsal anterior cingulate cortex, anterior insula, and amygdala) are believed to be involved in tinnitus perception and reaction. 2 Tinnitus can lead to anger, frustration, poor communication, tension, lack of sleep,3,4 and negatively influences the overall quality of life of its sufferers. 5
Traditionally, hearing aids have been used for tinnitus management 6 and more recently, various neuromodulation techniques such as repetitive transcranial magnetic stimulation (rTMS), 7 neurofeedback, 8 transcranial electrical nerve stimulation (TENS) 9 , and transcranial direct current stimulation (tDCS)10-15 have been used with varying degrees of success. In this study, we hypothesized that priming of the central nervous system might achieve a stronger and faster acting benefit from hearing aids. Priming is an effect in which exposure to a stimulus influences a response to a later stimulus. Priming has been used in stroke patients for better clinical results 16 ; facilitating ipsilesional motor cortex excitability prior to motor practice with the paretic upper limb leads to greater functional improvements than motor practice alone. 17 According to Norena’s 18 central gain model of tinnitus, a reduction of central gain and peripheral drive might facilitate tinnitus management. We hypothesized that tDCS might assist in reducing central gain of tinnitus signal by facilitating the peripheral stimulation effects of hearing aids.
Transcranial direct current stimulation is a painless, safe and noninvasive neuromodulation technique. 19 Either the anode or cathode can be positioned over the target area, to facilitate or suppress cortical activity. 20 Anodal tDCS of left temporoparietal area (LTA)10-12 and dorsolateral prefrontal cortex (DLPFC)14,15,21 have been effective in transient tinnitus suppression. The LTA montage stimulates various cortical and subcortical areas, which, either by competition or inhibition results in the reduction of abnormal hyperactivity caused by tinnitus. 10 Underneath the LTA lies the primary auditory cortex (Brodmann area [BA] 41, 42), auditory association areas (BA 21, 22), and parts of the limbic system (amygdala and hippocampus), which are thought to be parts of the proposed neural network involved in tinnitus.22,23
Studies undertaken in the area of tinnitus and tDCS have usually investigated the effect of a single session11,19 or 2 sessions 10 of tDCS on tinnitus. Frank et al 24 and Faber et al 15 used multisession tDCS but did not find any long-term impact on tinnitus. Both studies15,24 stimulated the DLPFC with 1.5-mA current intensity and there is a possibility that not enough current reached other cortical and subcortical areas (auditory cortex and limbic system) because of the focal nature of the electric field and current density distribution offered by the DLPFC montage. 25 Recently, Shekhawat et al 12 conducted a dose–response study to optimize LTA tDCS parameters for tinnitus and proposed 2-mA current intensity and 20-minute duration as the most favorable intensity and duration settings for transient tinnitus suppression.
Tinnitus is usually associated with hearing loss 26 and hearing aids have been commonly used for tinnitus management for the past 6 decades. 6 Shekhawat et al 27 conducted a scoping review of the role of hearing aids for tinnitus management, and found that a large number of studies support the use of hearing aids although many of them provide a low level of evidence for the benefits of hearing aid use for tinnitus. There are several proposed mechanisms of effect for which hearing aids might assist in tinnitus management. These mechanisms include masking tinnitus, reversing tinnitus-related cortical reorganization, 28 providing compensation for the degree of hearing loss, downregulating central gain, 29 and reducing the communication stress associated with hearing loss. 30 Hearing aid use can lead to long-lasting tinnitus reduction, but its full effectiveness is often only achieved after 6 to 12 months of use.31,32
As it is well established that long-term hearing aid use can improve tinnitus,28,31,32 and it has also been shown that tDCS can lead to transient tinnitus suppression, a combination of the 2 treatments might offer benefits greater than each treatment alone. Modifying the stimulation parameters of tDCS by using a higher current intensity, and changing the site of stimulation from DLPFC to LTA, might also improve the effectiveness of tDCS for tinnitus suppression. We proposed that combining these 2 techniques (tDCS and hearing aids) may facilitate priming of the brain for sound therapy resulting in greater hearing aid benefit in a shorter period of time, much in the same way that priming in stroke results in greater functional improvement.16,17
Methods
This study was approved by the University of Auckland Human Participants Ethics Committee and was registered as a clinical trial on the Australian New Zealand Clinical Trial (registry number ACTRN12612000277842, http://www.anzctr.org.au/).
Participants
Forty participants (mean age = 59.18 years, range = 45-76 years) were recruited through the University of Auckland Hearing and Tinnitus Clinic and researchstudies.co.nz. “Research studies” is an online participant recruitment portal that connects research volunteers with research opportunities. Inclusion criteria for the participants were chronic tinnitus (more than 2 years), aidable hearing loss with no previous experience of hearing aid use, and a minimum score of 25 on the Tinnitus Functional Index (TFI). 33 Volunteers were excluded if they had any contraindications for undergoing tDCS (personal or family history of seizures, metal and electronic implants, pregnancy, heart conditions, brain surgery, and others) as screened by a neurologist. Twenty volunteers not meeting the inclusion criteria were excluded. Written informed consent was provided by all participants as per the Declaration of Helsinki.
Research Protocol
This was a double-blind, sham-controlled, randomized clinical trial. The first author carried out all experimental procedures and data analysis was performed with statistical support before being unblinded. A minimization method was used by the third author to randomize participants to a treatment group (20 participants) or control group (20 participants). Randomization was based on age, gender, TFI score, tinnitus duration, and severity of tinnitus. Both groups underwent 5 sessions of brain stimulation (real or sham tDCS) followed by hearing aid fitting and use for 6 months. Multiple evaluations were carried out (Table 1) at the following time points: 1 month pretreatment (first baseline), 1 week pretreatment (second baseline), before and after each tDCS session (5 tDCS/sham sessions), before hearing aid fitting, and 3 months and 6 months following hearing aid fitting. The progress of the clinical trial through various phases (Enrollment, Allocation, Follow-up, and Analysis) is shown in Figure 1.
Outcome Assessments Undertaken Across the 7-Month Long Clinical Trial.

Abbreviations: TCHQ, Tinnitus Case History Questionnaire, HADS, Hospital Anxiety and Depression Scale; TFI, Tinnitus Functional Index; TSNS, Tinnitus Severity Numeric Scale; THQ, Tinnitus Handicap Questionnaire; HHI, Hearing Handicap Inventory; MML, minimum masking level; VAS, visual analog scale; CGI, Clinical Global Impressions.
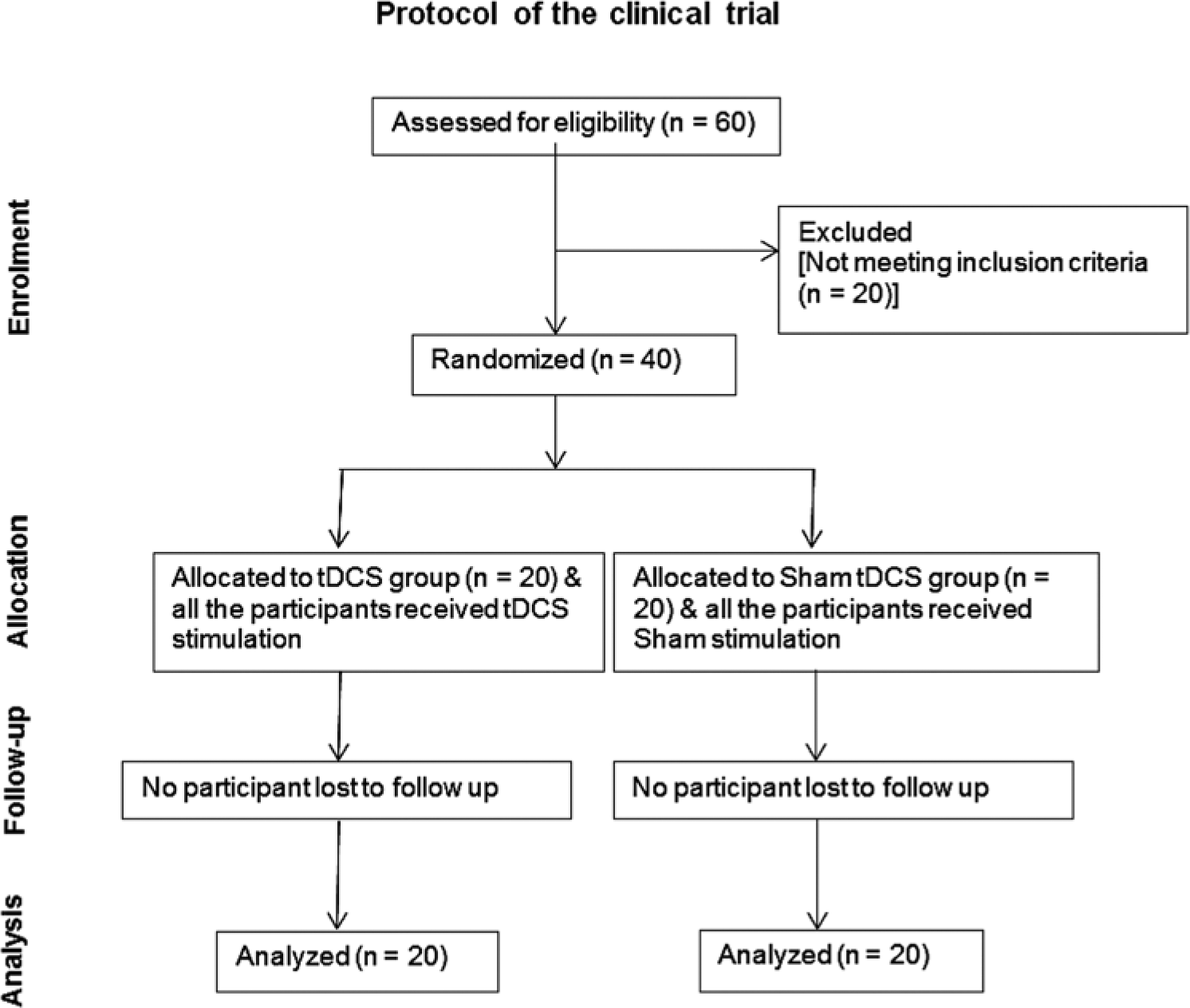
Flow diagram of the progress through the phases (Enrollment, Allocation, Follow-up, and Analysis) of the randomized clinical trial of transcranial direct current stimulation (tDCS) and sham tDCS groups.
Hearing Assessment
Hearing assessment was conducted in a sound treated room (ISO 8253–1:2010). Pure tone audiometry (0.25-16 kHz) was undertaken using a 2-channel audiometer (either GSI-61, Grason Stadler, Eden Prairie, MN; or AC40, Interacoustics, Assens, Denmark). Measurements (0.25-8 kHz) were made using standard earphones (TDH-50P; Telephonics) or insert headphones (E.A.RTONE 3A) and high-frequency (8-16 kHz) headphones (Sennheiser HDA 200). Audiometry was obtained using the modified Hughson–Westlake procedure. 34 Tympanometry was undertaken using a GSI (Grason Stadler) Immittance audiometer, and distortion product otoacoustic emissions (DPOAEs) were measured using an ILOV 6 (Otodynamics Ltd, Hertfordshire, UK) OAE analyzer.
Questionnaires Used for Clinical Evaluation
The following questionnaires—Tinnitus Case History Questionnaire (TCHQ), 35 TFI, Tinnitus Severity Numeric Scale (TSNS), 36 Hospital Anxiety and Depression Scale (HADS), 37 Tinnitus Handicap Questionnaire (THQ), 38 Hearing Handicap Inventory (HHI), 39 Clinical Global Impressions (CGI), 40 and the visual analog scale (VAS) 41 were used. Patient-reported ratings and any incidental observations were recorded at every assessment.
Psychoacoustic Tinnitus Assessment
Tinnitus pitch, loudness, and minimum masking level (MML) were assessed using testing software (The University of Auckland). Tinnitus pitch was assessed throughout the test frequency range of 0.25 to 16 kHz using a 2AFC method, in which pairs of tones were presented based on the configuration of audiogram and perceptual feedback. High-frequency circumaural headphones (Sennheiser HDA 200) were used for the entire pitch-matching procedure. Participants were asked to identify the tone that best matched their tinnitus pitch. Each tone was presented at a sensation level of 15 dBSL. Once the settings for a given pair of tones were established, the 2 tones were presented in an alternating manner until the participant indicated which one was closest to the pitch of their tinnitus. Pitch match was then compared to tones 1 octave above and below to rule out octave confusion.
The sensation level (loudness, dBSL) of tinnitus was measured by presenting sound at the tinnitus pitch and gradually increasing its intensity. Participants were instructed to indicate the level at which the loudness of the presented tone was equal to that of their tinnitus.
The MML measurement was similar to that of loudness measurement except that participants were instructed to indicate the level at which the presented sound masked (covered) their tinnitus perception.
Transcranial Direct Current Stimulation
Transcranial direct current stimulation was applied in accordance with the protocol described by Loo et al 42 and DaSilva et al. 43 A neuroConn DC stimulator (neuroConn GmbH, Ilmenau, Germany) was used for tDCS. The rubber electrodes had a surface area of 35 cm2 (anode) and 50 cm2 (cathode) as a smaller stimulating electrode can lead to a more focused stimulation and a larger reference electrode has minimal physiological effects on proximal structures and is less likely to affect the overall impact of target stimulation. 44 NaCl solution (0.85%) was used to soak the electrode sponges. The anode was placed above the LTA (halfway between C3 and T5 10 ) and the cathode was placed at the contralateral frontal scalp (halfway between F8 and T4 11 ) identified using the international 10-20 system. Impedance and voltage were monitored and were less than 5 kohm and less than 5 V, respectively, across all stimulation settings used. The neuroConn DC stimulator had a “study mode” where input codes were used (the researcher was blinded to these codes) and the settings generated were either a sham or actual stimulation. Parameters used for tDCS stimulation were 2-mA current intensity, 20-minute duration, on 5 consecutive days (Monday to Friday) with a gap of 24 hours between each stimulation session. The settings used for sham stimulation were a fade-in time of 8 seconds, followed by 30 seconds of direct current, followed by a fade-out time of 5 seconds. This was as per previous recommendations for effective sham setting for tDCS. 46
Hearing Aid Fitting
All the participants were fitted bilaterally with GN ReSound Live 571 open-fit hearing aids on the day following the final brain stimulation session (irrespective of which group they were in). A modified Desired Sensation Level (input/output) [DSL(I/O)] v5.0 was used as the amplification prescription target 47 and the fitting was adjusted according to participant’s comfort and preference. Care, maintenance, and use of the hearing aids was explained to participants during the hearing aid fitting session and they were recommended to use the hearing aids for a minimum of 8 hours per day in a variety of everyday listening situations. The “data logging” feature of the hearing aid was used to find the average number of hours of hearing aid use. Participants were told to contact the researcher if they required any further assistance before the next follow up (after 3 months). No tinnitus counseling was provided to either group. Hearing aids were well received.
Data Analysis
The data were analyzed using the statistical analysis system (SAS) version 9.3. Outcome variables measured at follow-up (post tDCS, 3 months, and 6 months) were analyzed using a mixed-effects model for repeated measures MMRM (SAS 9.3 MIXED procedure). When assessing hearing aid use, follow-up was defined as at 3 months and 6 months, post tDCS was used as baseline covariate. Group, visit (as categorical variable), group and visit interaction, tinnitus duration, age, tinnitus severity, and baseline TFI scores were assessed as fixed effects, baseline measure as a covariate, and participants were assessed as a random effect in the mixed-effects model. The within-subject errors were modeled using an unstructured (co)variance structure. The Kenward–Roger method was used to estimate the denominator degree of freedom for fixed effects. A generalized linear mixed effects model technique (SAS 9.3 GLIMMIXED procedure) was used to analyze variables (TSNS, CGI, and VAS-Tinnitus loudness) measured at follow-up (post tDCS, 3 months and 6 months for TSNS, after each tDCS session [5 sessions] for CGI and measured pre and post each tDCS for VAS). Multinomial distribution with cumulative logit link function was used. Variables with
Results
The results of the randomization and minimization procedure on participant characteristics for each group are shown in Table 2.
Profile of Research Participants in Sham Transcranial Direct Current Stimulation (tDCS) and tDCS Group. a

Both the groups were well matched and there was no significant difference between the 2 groups on all the mentioned parameters.
The overall hearing status of participants in the 2 groups is shown in Figure 2. Participants in both the groups had a sloping mild to severe sensorineural hearing loss and the 2 groups did not differ from each other significantly except for the right ear at 2 kHz, where the sham tDCS group had significantly better hearing than the tDCS group,
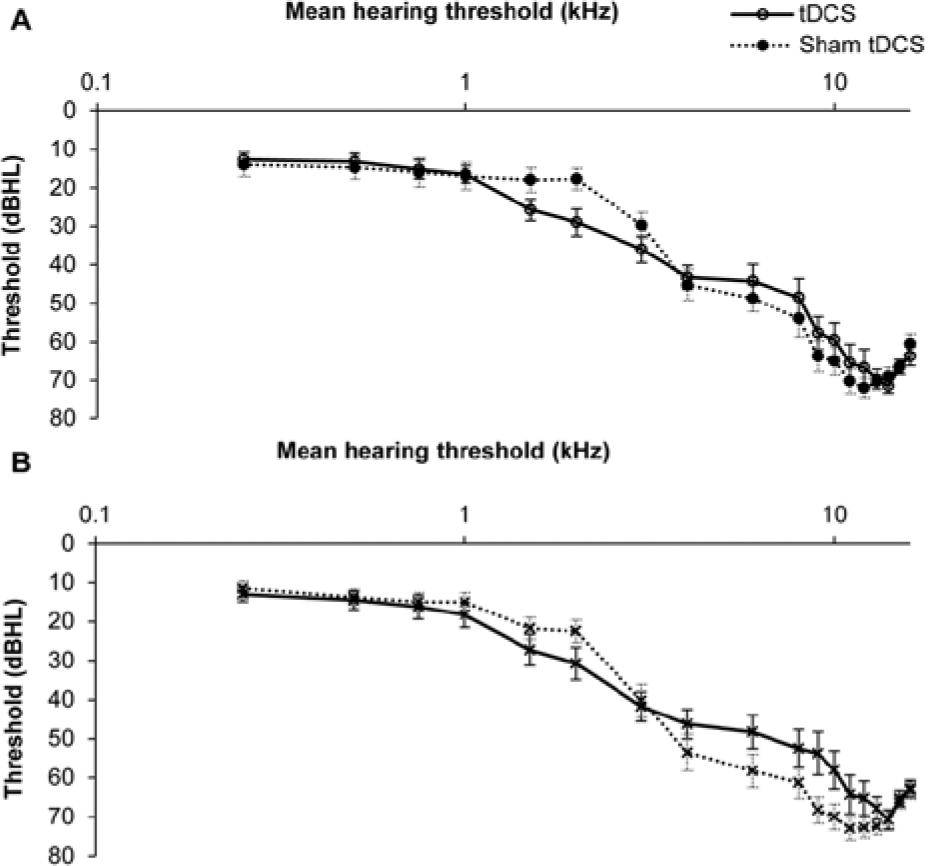
Mean hearing thresholds of right (A) and left ear (B) for participants in sham transcranial direct current stimulation (tDCS) and tDCS groups. The error bars represent ±1 standard error of the mean.
During the 5 stimulation sessions, participants in both groups rated their tinnitus loudness using the VAS on a 10-point rating scale. Loudness rating was undertaken 3 times in each session. The first rating was done immediately after arrival and then after 10 minutes the second rating was done, the rationale for doing the rating twice before stimulation was to document the effect of change in environment (day-to-day environment to sound treated room) on the tinnitus. The second rating, after arriving in the sound treated room, was used as the baseline measure to compare with poststimulation ratings (third rating) which was done immediately after the stimulation. The change in average loudness rating for both the groups is shown in Figure 3A.
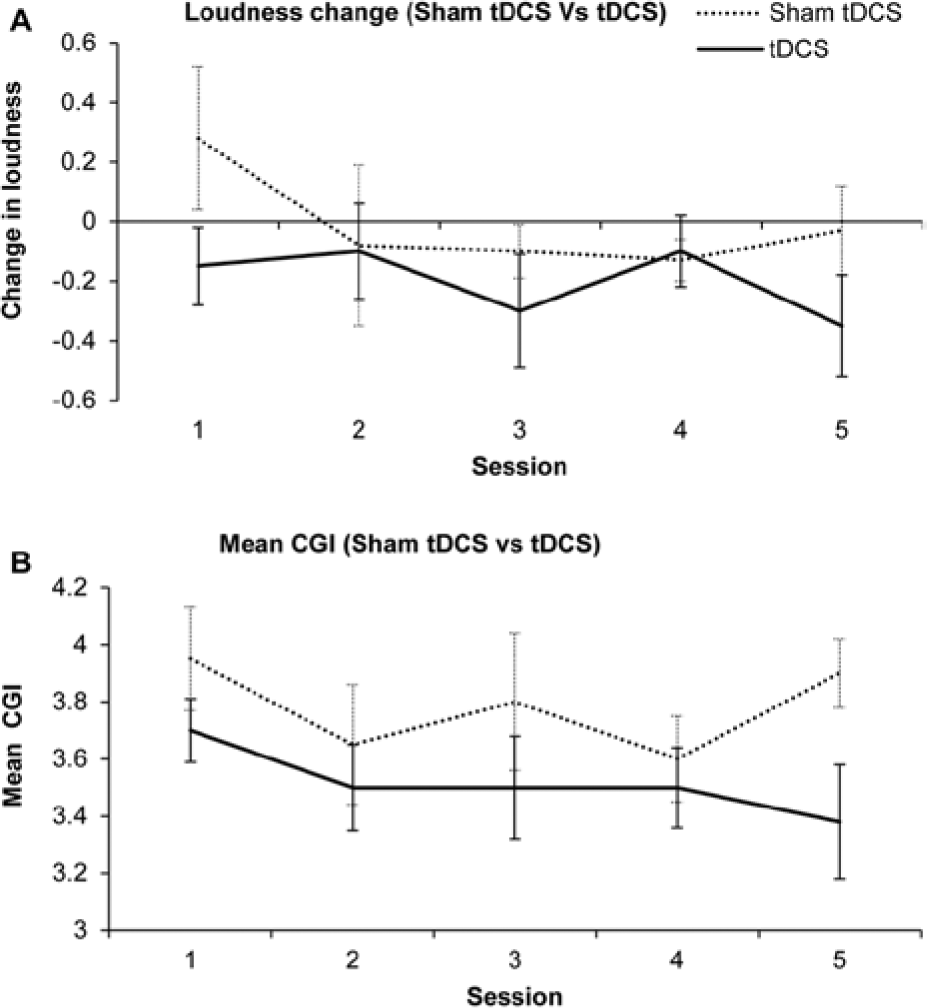
Change in loudness in transcranial direct current stimulation (tDCS) and sham tDCS groups (A) followed by 5 sessions of stimulation. Change in loudness was calculated as follow: baseline change was calculated by subtracting loudness rating immediately after arrival (baseline 1) from loudness rating after 10 minutes of arrival (baseline 2); poststimulation loudness change was calculated by subtracting loudness rating immediately before stimulation from loudness rating immediately after stimulation. Positive values represent increase in tinnitus loudness (worsening), negative values represents reduction in tinnitus loudness (improvement). (B) Mean Clinical Global Impressions (CGI) for sham tDCS and tDCS groups during 5 sessions of stimulation. On the CGI rating scale, point 4 represents “no change” and point 3 represents “minimally better.” The error bars represent ±1 standard error of the mean.
There was no significant difference between the 2 groups for loudness change during the 5 stimulation sessions using the statistical model. However, based on the raw data, there was a consistent trend of reduction in tinnitus loudness with maximum loudness change occurring after the fifth stimulation session for the tDCS group. The sham tDCS group failed to show a consistent reduction in tinnitus loudness; during the first session the loudness increased (tinnitus worsened) after stimulation, during the second to fourth sessions there was some reduction in tinnitus loudness and finally after the fifth session the poststimulation rating was worse than the prestimulation baseline.
The CGI was measured after completion of each stimulation session to document participant’s perception of the effectiveness of stimulation on their tinnitus (Figure 3B). There was no significant difference in the 2 groups in their mean CGI ratings, although the mean CGI ratings for the tDCS group decreased with the lowest rating at the end of the fifth stimulation session. The average ratings for the sham tDCS group were close to “4” reflecting no change.
The primary outcome measure used in this trial was the TFI and there was a significant reduction in the overall TFI score with time,
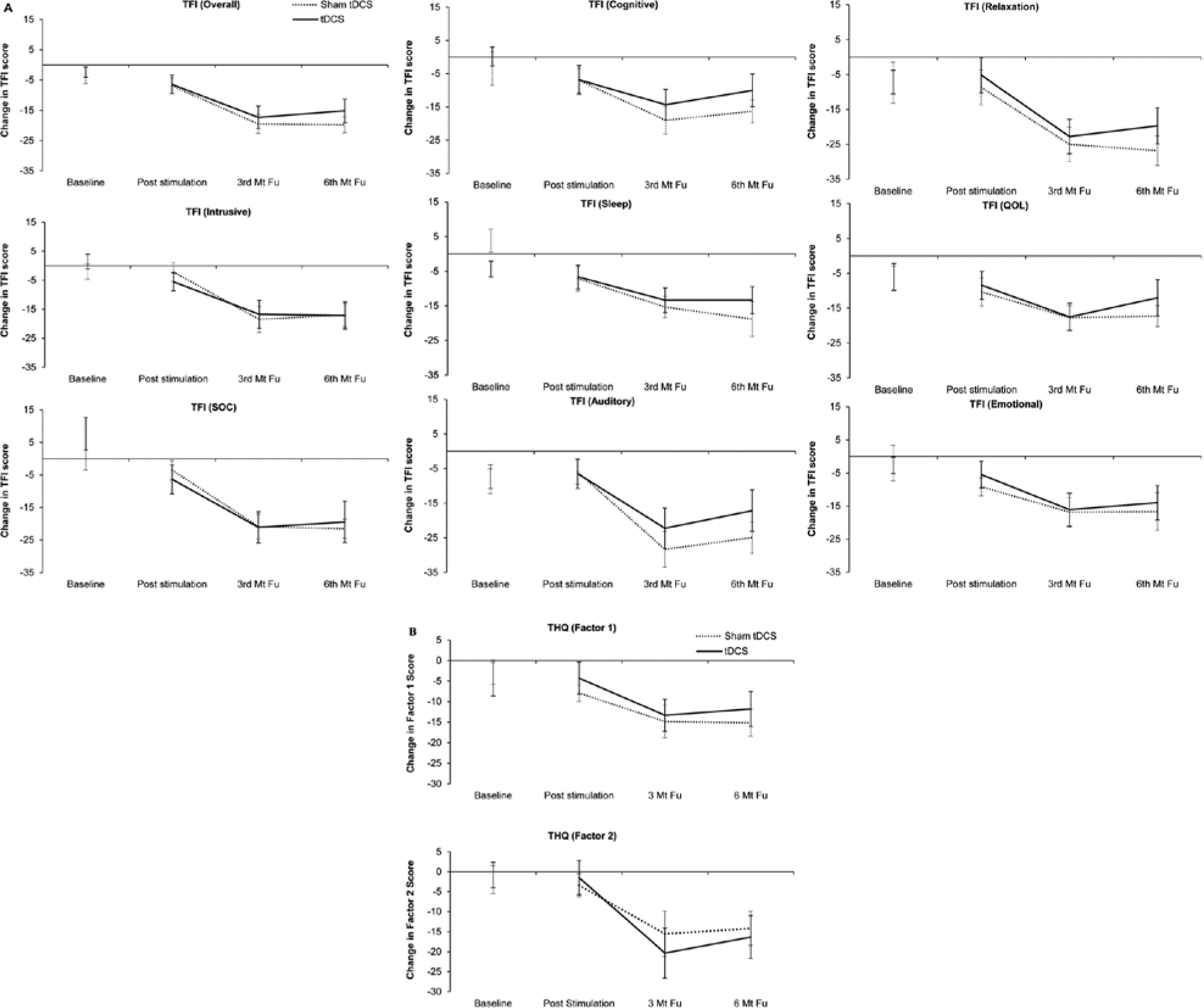
Change in (A) Tinnitus Functional Index (TFI) and (B) Tinnitus Handicap Questionnaire (THQ), baseline, poststimulation, 3- month follow-up, and 6- month follow-up in sham transcranial direct current stimulation (tDCS) and tDCS groups. Change in baseline was calculated by subtracting first baseline obtained one month prior to stimulation session from second baseline obtained one week before starting stimulation. Change in poststimulation, 3- month follow-up, and 6- month follow-up was calculated by subtracting the second baseline obtained 1 week before starting stimulation from post tDCS, 3-month follow-up, and 6-month follow-up. Positive values represent worsening in tinnitus symptoms, and negative values represent improvement in tinnitus symptoms. The error bars represent ±1 standard error of the mean.
Along with the TFI, several other questionnaires (THQ, HHI, TSNS, HADS) were used as secondary measures. The change in THQ was correlated with change in TFI (factor 1,
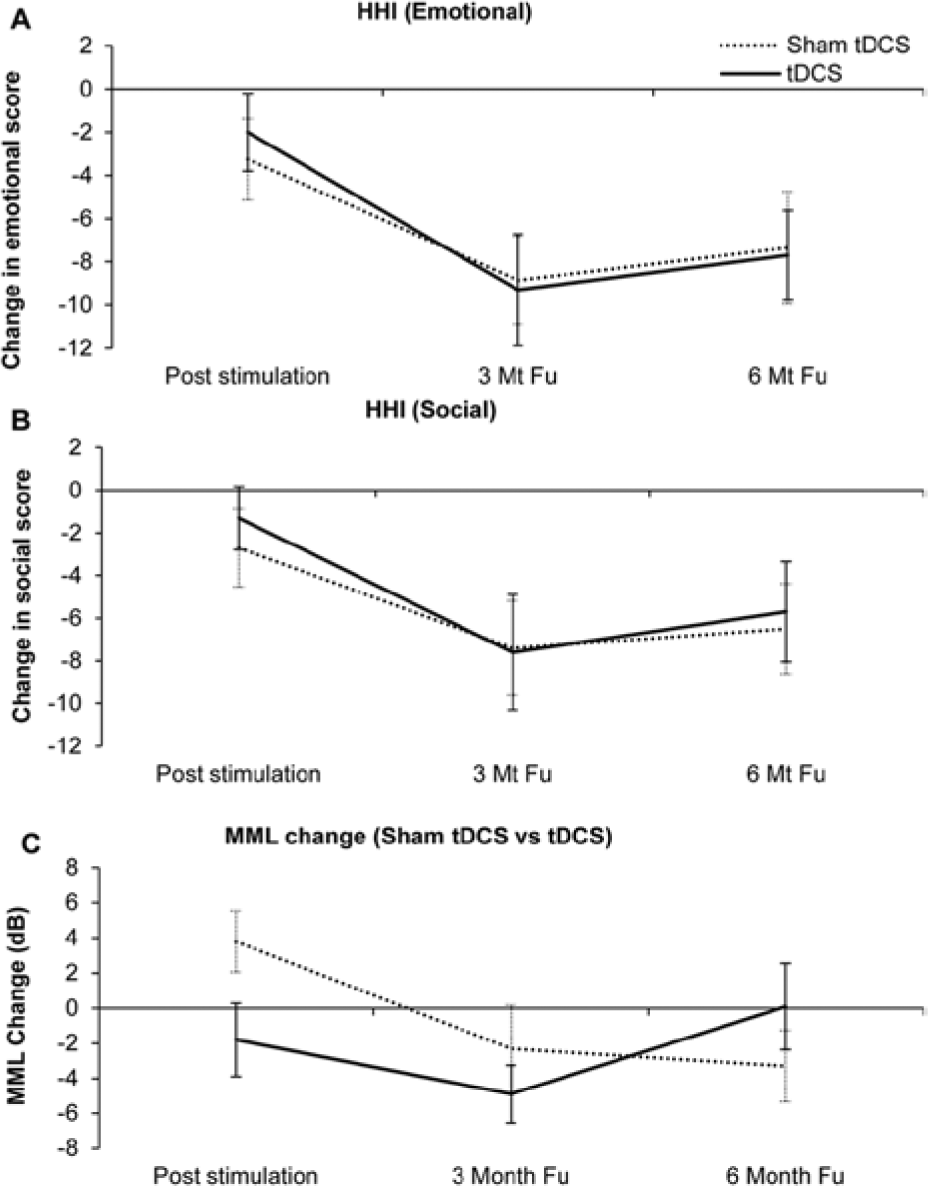
(A and B) Change in Hearing Handicap Inventory (HHI) poststimulation, 3-month follow-up, and 6-month follow-up in sham transcranial direct current stimulation (tDCS) and tDCS groups. (C) Change in minimum masking level (MML) poststimulation, 3-month follow-up, and 6-month follow-up in sham tDCS and tDCS groups. Change in scores was calculated by subtracting baseline values measured before starting stimulation sessions from poststimulation, 3-month follow-up, and 6-month follow-up values. The error bars represent ±1 standard error of the mean.
For the TSNS, there was a significant reduction in the overall tinnitus score with time,
Discussion
The main finding of this study was that hearing aids can significantly reduce the negative impact of tinnitus on quality of life. There was some preliminary evidence suggesting that tDCS may have resulted in lower MMLs (reducing the amount of sound need to cover tinnitus); however, such results need to be replicated. It was hypothesized that multiple sessions of tDCS might diminish peripheral drive, by modulating neural correlates of tinnitus at various cortical and subcortical areas possibly through a top-down approach.48,49 Hearing aids were used for 6 months in an attempt to reduce central gain, likely through a bottom-up approach, by increasing afferent activity diminished by hearing loss.
The use of hearing aids led to a significant reduction in tinnitus handicap as measured with the TFI. The maximum amount of benefit was achieved after 3 months of hearing aid use, unlike many other studies which observed maximum benefit after 6 to 12 months of hearing aid use.31,32,50 The 3 subscales of TFI showing the largest reductions after hearing aid use were auditory, sense of control, and relaxation. This suggests that along with providing benefit in hearing, the hearing aids were also beneficial in inducing relaxation and a sense of control for coping with tinnitus. The THQ also reduced in a similar manner to the TFI. The sham tDCS group experienced a 9.53-point reduction in THQ score, which is comparable to masking alone (Henry et al 51 ) and counseling alone (Searchfield et al 32 ). The tDCS group experienced a 13.94-point decrease. This study addressed a number of shortcomings of previous tinnitus-focused hearing aid studies. 27 In the present study, hearing aids were programmed specially for tinnitus instead of optimizing them for communication only52,53; this trial was a controlled trial, purely assessing the impact of hearing aids and tDCS, not a combination of hearing aids with counseling54,55 and multiple measurement methods, including psychoacoustic tinnitus assessment were used providing convergent validity.53,56 All participants were fitted with binaural hearing aids of the same design. The hours of hearing aid use were recorded using data logging, enabling control for participant compliance.
We hypothesized that tDCS might improve the effectiveness of hearing aids; for the majority of measures, this hypothesis was not supported. The one measure showing a difference between tDCS and sham groups 3 months following hearing aid fitting was the MML. The MML was at its lowest for the tDCS group at the 3-month follow-up, while the maximum reduction for the sham tDCS group was measured at 6-month follow-up. A possible interpretation is that tDCS hastened the maximum suppression possible by sound achieved by the hearing aids, but this advantage disappeared after 6 months.
The change in measured tinnitus loudness mirrored the MML change (ie, was in the opposite direction). An increase in measured tinnitus loudness for both the groups could be because of the attention drawn to their tinnitus as suggested by adaptation level theory 57 rather than a true increase in tinnitus magnitude. While the hearing aid effects on tinnitus dominated any improvement due to tDCS, there were trends toward reduction in subjective tinnitus loudness, mean CGI, and MML with tDCS.
All the brain stimulation sessions were well tolerated and none of the participants reported any discomfort or side effects. So far only 2 clinical studies15,24 have investigated the impact of multisession tDCS on tinnitus perception. Both these studies have used DLPFC as the site of stimulation and neither of them found any lasting impact of tDCS on tinnitus perception.
In our trial, all the 6 items of the TSNS (overall, strong, uncomfortable, annoying, ignore, and unpleasant) reduced with a maximum effect after 3 months of hearing aid use; however, the 2 groups (tDCS and sham tDCS) did not differ from each other significantly. The post–brain stimulation differences in the ratings were small. The hearing aids had a strong effect; any tDCS effects may have been “washed out” by larger effects of the acoustic stimulation. There was large variability between participants in the results, which has also been seen in other trials10,11,13,24 and it could potentially be due to various forms of tinnitus and the likelihood of differences in the neural networks and connections in participants with tinnitus. 58
All the participants in our trial had hearing loss; the tDCS group had significantly worse hearing at 2 kHz in the right ear (in the conventional frequency range) compared with the sham tDCS group. Fregni et al 10 found an inverse relationship between severity of hearing loss and responsiveness toward tDCS. The slightly greater hearing loss in our tDCS group and 3 years higher average tinnitus duration (though not statistically significant) may have reduced the magnitude of benefit from the brain stimulation sessions. Further trials could be undertaken on tinnitus sufferers with normal or near normal hearing to explore the potential of this effect.
This trial is the first reported attempt to prime the auditory central nervous system for hearing aid–based tinnitus relief. It showed little beneficial effect of priming; however, it is too early to conclude that priming the auditory system for change does not have potential, given that only one tDCS stimulation protocol was used in this study. Different options for tDCS need to be explored for use with hearing aids. One possibility could be to try variations in the spacing of tDCS sessions, such as every 2 to 4 weeks along with hearing aid use. This protocol might increase the possibility of regular modulation of neural excitability that could be exploited by hearing aid use. The impact of tDCS and acoustic stimulation on residual inhibition (RI; brief tinnitus suppression following cessation of the masker 59 ) could also be investigated to explore whether the use of tDCS could prolong RI. RI usually lasts for less than a minute, but the brief respite patients can have from their tinnitus can be strong (complete absence of tinnitus). 60 Research investigating the impact of tDCS on RI might provide further insight into the better utilization of tDCS with sound therapy.
Conclusion
Hearing aids (without tinnitus counseling) resulted in significant improvement in tinnitus-related quality of life after 3 months of use. The hearing aid benefits were independent of tDCS. This trial did not reveal any statistically significant benefit of tDCS over hearing aid use alone. Five sessions of tDCS resulted in a significant reduction in MMLs compared with sham, a consistent trend for lower loudness and CGI scores was observed but the effects were not statistically significant. Further investigations of tDCS, or other neuromodulation techniques such as rTMS, transcranial vagal nerve stimulation, and neurofeedback, may find that priming the auditory system for hearing aid use can be clinically beneficial. Different tDCS protocols, including combined LTA and DLPFC as site of stimulation and providing multiple tDCS sessions spaced throughout the hearing aid use (every 2 to 3 months’ time) might provide benefits not seen with tDCS only provided prior to hearing aid fitting.
Footnotes
Acknowledgements
We would like to thank the Neurological Foundation of New Zealand for a small project grant, Professor Alan Barber for neurological screening of participants, and Dr Arier Chi Lun Lee for statistical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A small project grant was received from the Neurological Foundation of New Zealand. Hearing aids for this study were donated by GN ReSound.
