Abstract
Background. Proof-of-principle studies have demonstrated transient beneficial effects of transcranial direct current stimulation (tDCS) on motor function in stroke patients, mostly after single treatment sessions. Objective. To assess the efficacy of multiple treatment sessions on motor outcome. Methods. The authors examined the effects of two 5-day intervention periods of bihemispheric tDCS and simultaneous occupational/physical therapy on motor function in a group of 10 chronic stroke patients. Results. The first 5-day period yielded an increase in Upper-Extremity Fugl-Meyer (UE-FM) scores by 5.9 ± 2.4 points (16.6% ± 10.6%). The second 5-day period resulted in further meaningful, although significantly lower, gains with an additional improvement of 2.3 ± 1.4 points in UE-FM compared with the end of the first 5-day period (5.5% ± 4.2%). The overall mean change after the 2 periods was 8.2 ± 2.2 points (22.9% ± 11.4%). Conclusion. The results confirm the efficacy of bihemispheric tDCS in combination with peripheral sensorimotor stimulation. Furthermore, they demonstrate that the effects of multiple treatment sessions in chronic stroke patients may not necessarily lead to a linear response function, which is of relevance for the design of experimental neurorehabilitation trials.
Keywords
Introduction
Different novel neurorehabilitation methods have been applied to facilitate motor recovery after stroke. 1 One of the most promising types of experimental therapies in the chronic phase is noninvasive brain stimulation, which includes techniques such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS).2-4 Both methods have been used to upregulate intact portions of the ipsilesional 2 or downregulate the contralesional motor cortex. 3 The latter approach is based on neurophysiological studies, which indicate an imbalance of interhemispheric interactions resulting in disinhibition of the contralesional hemisphere and increased inhibition of the ipsilesional motor cortex. 5 To simultaneously target both components of this imbalance, a bihemispheric tDCS approach has been proposed recently. 4 Its differential modulation of bilateral motor cortices might further enhance the effects of unihemispheric stimulation.6-8
Single-session tDCS experiments in chronic stroke patients demonstrated that the unihemispheric modulation of motor cortex excitability yields functional improvement of the affected upper extremity that outlasts the stimulation period.2,9,10 A recent multiple-session tDCS trial of stroke patients 6 as well as studies of healthy participants4,11 suggest that stronger and longer lasting effects can be obtained with repeated consecutive sessions. However, the influence of the number of experimental sessions on motor outcome after stroke has not yet been investigated. To address this question, we examined the improvements achieved during two 5-day intervention periods of bihemispheric tDCS and simultaneous physical/occupational therapy (PT/OT) in chronic stroke patients. We hypothesized that a second 5-day intervention period of tDCS and PT/OT would yield significant additive gains as compared with the first 5-day intervention.
Materials and Methods
Participants
A total of 10 chronic stroke patients underwent bihemispheric tDCS in combination with PT/OT for two 5-day interventions.4,6 Inclusion criteria were as follows: occurrence of first ischemic stroke at least 5 months prior to enrollment, no previous or subsequent cerebral ischemia, Medical Research Council (MRC) strength grade of ≤3/5 in extensor muscles of the affected upper extremity in the acute phase, no additional neurological or psychiatric disorders, and no concurrent use of CNS-affecting drugs. Group details of age, time poststroke, lesion volume, and motor impairment are given in the Table 1; individual lesion maps are provided in Figure 1. The study was approved by the local institutional review board, and all patients gave written informed consent.
Demographic, Imaging, and Behavioral Motor Data
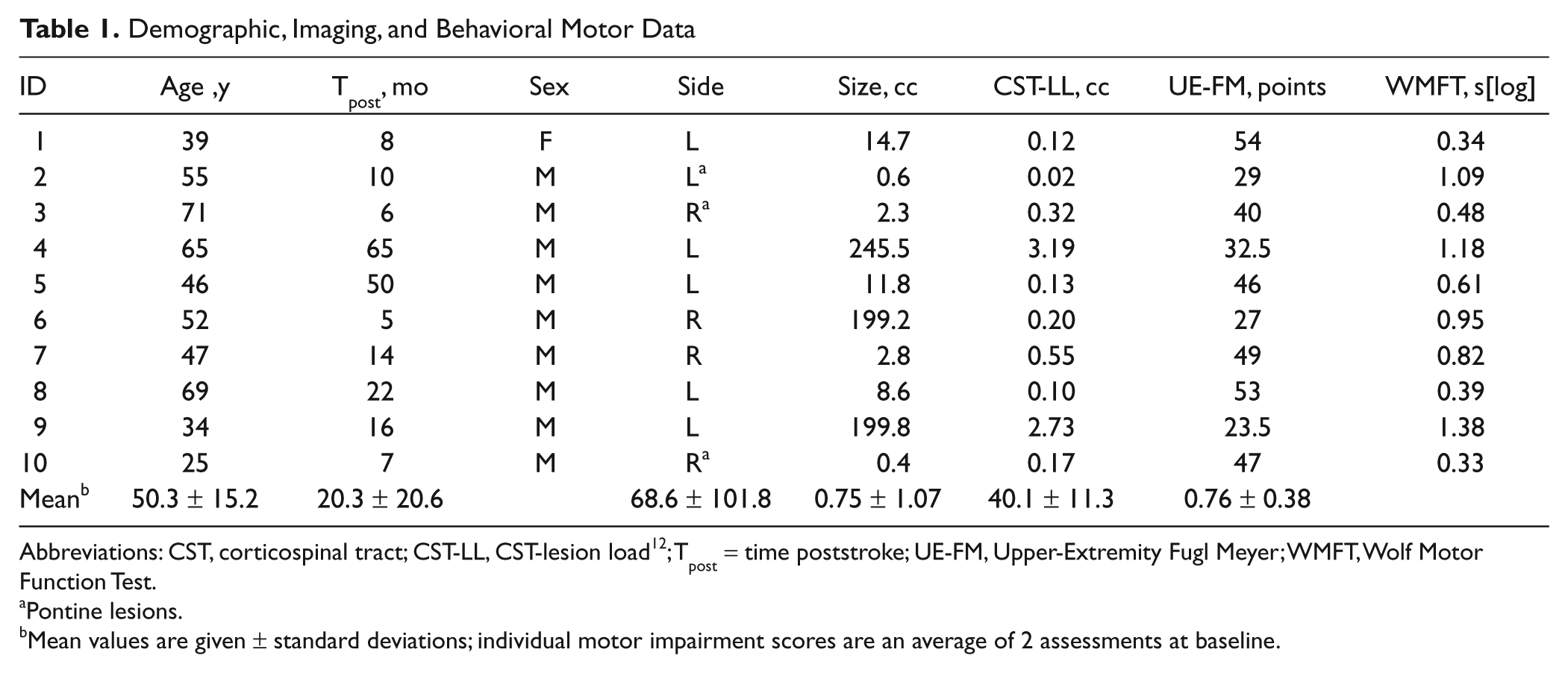
Abbreviations: CST, corticospinal tract; CST-LL, CST-lesion load 12 ; Tpost = time poststroke; UE-FM, Upper-Extremity Fugl Meyer; WMFT, Wolf Motor Function Test.
Pontine lesions.
Mean values are given ± standard deviations; individual motor impairment scores are an average of 2 assessments at baseline.
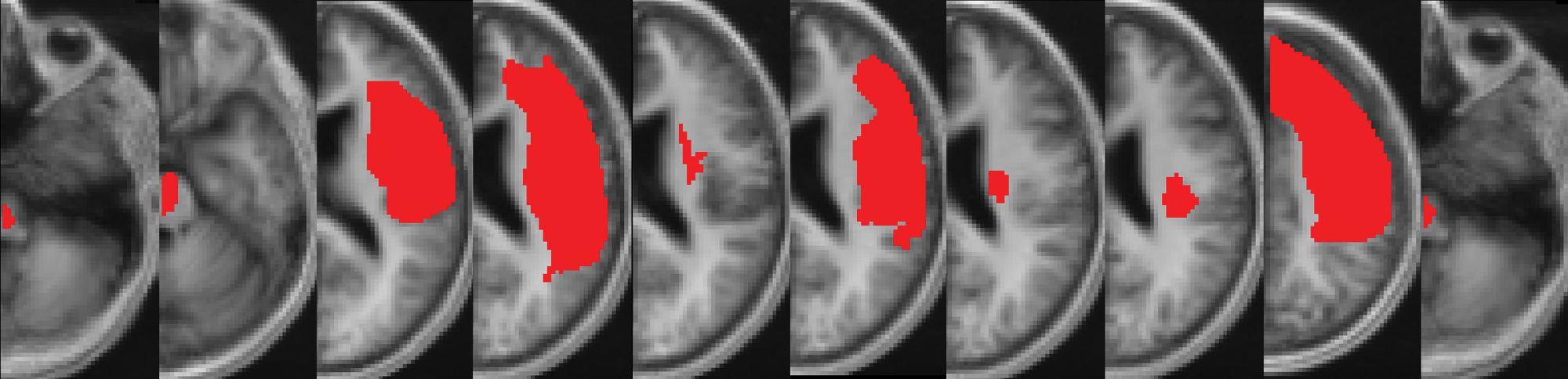
Individual lesion maps: after spatial normalization to the MNI space, individual lesion maps of patients were superimposed onto a canonical T1-weighted image. Right-hemispheric lesions were mirrored across the midline (detailed methods have been described previously 12 ).
Study Design
Participants underwent noninvasive brain stimulation (30 minutes) and simultaneous PT/OT (60 minutes) for 5 consecutive days. The therapist used a combination of PT and OT techniques, including functional motor tasks of the affected arm and hand to promote sensorimotor integration, coordination of movement, and goal-directed activities of practical relevance for the patient. This first treatment period was followed by a second 5-day intervention that was separated from the first by 2 to 29 days (mean 9.9 ± 9.4 days). Motor impairment and activity assessments were conducted prior to and after each of the 5-day intervention periods.
Of the 10 patients included in this study, 4 were randomly chosen from a separate double-blind, sham-controlled 5-day trial of tDCS and simultaneous PT/OT. 6 After finishing the trial, they underwent a second 5-day intervention (patients were still blinded as to whether they received real or sham stimulation). In addition, we recruited 6 de novo patients who also underwent two 5-day interventions of bihemispheric tDCS and PT/OT. All patients, the therapist, and the investigators who conducted the motor assessments were blinded as to whether the patients received real or sham stimulation.
Transcranial Direct Current Stimulation
Direct current was delivered through 2 saline-soaked surface gel–sponge electrodes using a Phoresor II Auto stimulator (IOMED, Salt Lake City, Utah). The stimulation consisted of 30 minutes of a 1.5-mA direct current with the anode placed over the ipsilesional motor cortex and the cathode over the contralesional motor cortex. 4 Stimulation sites were identified using the international 10-20 EEG electrode system.
Outcome Measures
Each patient underwent the Upper-Extremity Fugl-Meyer assessment (UE-FM) and the Wolf Motor Function Test (WMFT) on 2 different days prior to the intervention to ensure measurement stability at baseline. 13 The tests were repeated after each of the 2 intervention periods. In addition, those patients with more than 2 days in between interventions were reassessed before the second intervention started. The UE-FM is a standardized impairment scale with a maximum score of 66. 14 The WMFT consists of 15 time-based tasks and 2 tests of strength. 15 Similar to previous studies, completion times were logarithmized to account for skewed data distribution. 16 The resulting score has a maximum value of 2.08 s[log] with lower values reflecting better function of the affected arm.
To compare the outcome after the first and the second 5-day intervention periods, we used a repeated-measures general linear model with post hoc pairwise comparisons (controlling for age, time poststroke, and lesion size as covariates). We used the motor impairment scores of the 2 baseline assessments (PRE1 and PRE2), the scores after the first 5-day intervention (POST1), and the scores after the second 5-day intervention (POST2) as dependent variables. The rationale for using a single value at the time point POST1 for the respective motor assessments was that the scores after the first 5-day intervention period did not differ from the next assessment before the second 5-day intervention period in those patients who underwent their second 5-day intervention after an interval of 1 (n = 4), 2 (n = 1), or 3 (n = 1) weeks, as shown by 2-tailed paired t tests (UE-FM: t(5) = −.250, P = .813; WMFT: t(5) = .556, P = .602).
Results
All patients experienced the typical transient tingling sensation at the site of the electrodes. No patient reported any adverse effects during or after the stimulation.
The average improvement during the first 5-day intervention was −0.15 ± 0.06 s[log] in WMFT and 5.9 ± 2.4 in UE-FM scores. These values correspond to proportional changes of −23.6% ± 11.0% (WMFT) and 16.6% ± 10.6% (UE-FM), calculated as (Post − Pre) / (Pre × 100). The improvement during the second 5-day intervention was −0.08 ± 0.05 s[log] in WMFT (−16.9% ± 12.7%) and 2.3 ± 1.4 in UE-FM scores (5.5% ± 4.2%). The resulting overall change was −0.23 ± 0.09 s[log] in WMFT (−35.8% ± 16.3%) and 8.2 ± 2.2 in UE-FM scores (22.9% ± 11.4%) over the course of the 2 intervention periods (see Table 1 and Figure 2).
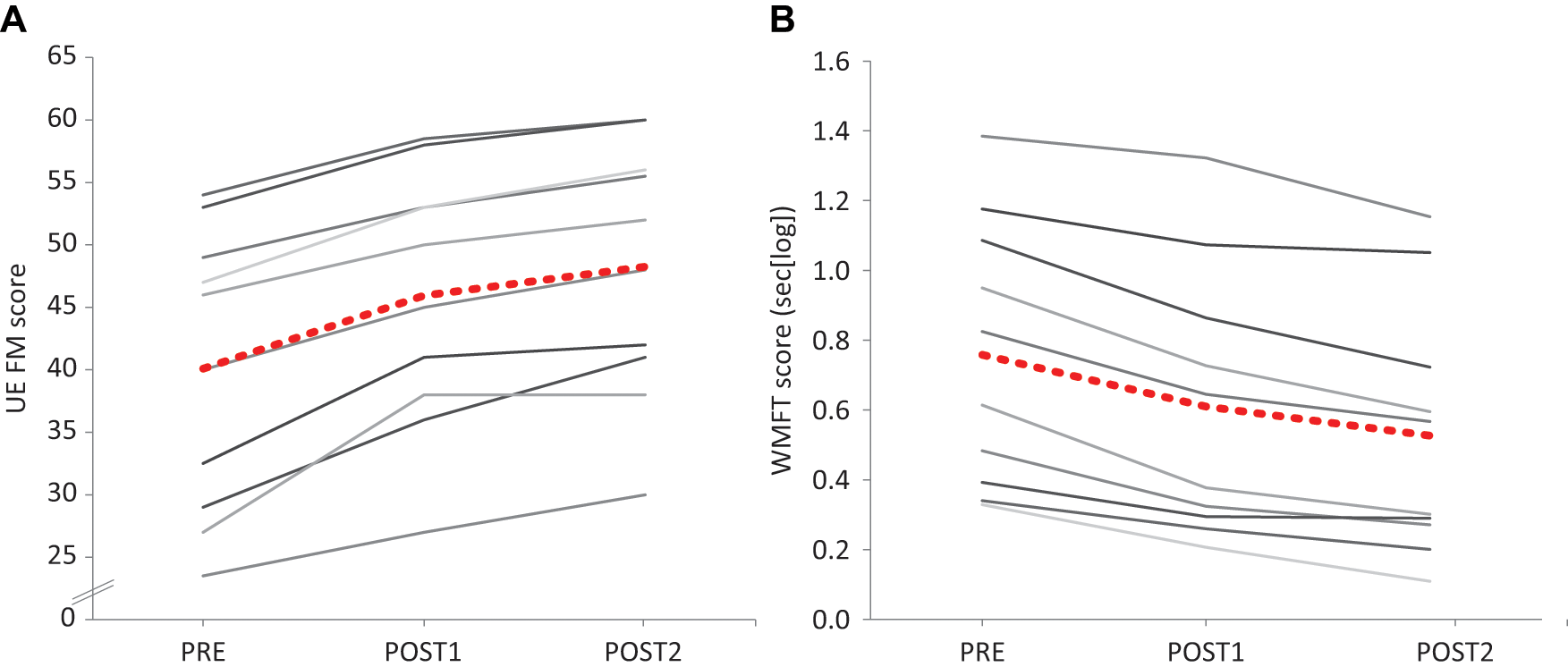
Improvement in motor function: Upper-Extremity Fugl-Meyer (UE FM; A) and Wolf Motor Function Test scores (WMFT; B) at baseline (PRE), after the first 5-day intervention (POST1), and after the subsequent 5-day intervention (POST2). Scores of individual patients are depicted in gray tones; the group mean is shown by red dots. Note that higher UE FM but lower WMFT scores indicate improvement in motor function.
The repeated-measures general linear model (controlling for age, time poststroke, and lesion size as covariates) yielded effects of the factor “TIME POINT” for WMFT (F = 4.552; P = .031, Huynh-Feldt corrected) and UE-FM (F = 2.953; P = .060). Post hoc pairwise comparisons revealed that the changes were greater during the first intervention than the second for both outcome measures: POST1 and POST2 were significantly different from either PRE1 or PRE2 (all P < .005, Bonferroni corrected), and POST2 was different from POST1 (all P < .006, Bonferroni corrected), whereas PRE1 and PRE2 did not differ from one another (WMFT: P = .161; UEFM: P = 1.000). Multiple regression analyses demonstrated that the length of the interval between the two 5-day interventions had no effect on any of the outcome measures (ie, raw and proportional changes; all P > .52).
Discussion
Bihemispheric tDCS in combination with peripheral sensorimotor activity led to substantial functional improvements in each of our 10 chronic stroke patients after 5 treatment sessions with additional gains in a subsequent 5-day intervention period. The most prominent functional gains were found after the first 5-day intervention. The mean improvement in this group of patients receiving bihemispheric tDCS and PT/OT was substantially greater compared with the sham control group of our recent randomized trial (mean UE-FM change of 1.2 ± 1.0 points and WMFT change of −0.05 ± 0.06 s[log] in a group of 10 patients receiving sham tDCS and PT/OT). 6 The second 5-day period yielded further improvements, resulting in significantly higher overall gains after 10 days as compared with the first 5-day period. However, the additional improvements during the second 5-day period were significantly lower than those observed during the first period. This finding indicates that the changes in impairment and activity scores were not linear over time, although this interpretation is somewhat restricted by the small number of participants included in our study. Taken together, the analyses of our data sets provide support for the use of multisession interventions but also suggest that the more sessions chronic stroke patients undergo, the less the change that is to be expected over time.
These results have important implications for future neurorehabilitation trials because they demonstrate the safety and efficacy of 5 as well as 10 sessions of noninvasive brain stimulation in combination with peripheral sensorimotor activities. Standard treatment durations of 2 weeks have been applied in previous neurorehabilitation trials, for example, using constraint-induced movement therapy (CIMT).1,16,17 The substantial functional improvement of all patients in the current study indicates that noninvasive brain stimulation in combination with peripheral sensorimotor activities may lead to long-term potentiation-like phenomena and greater synaptic plasticity than one of the interventions by itself.4,11 This notion is corroborated by the results of a previous study in which CIMT was administered consecutively (but not concurrently) with rTMS, which revealed no significant differences between real and sham stimulation on motor function in chronic stroke patients. 18 Furthermore, the tDCS montage used in the present study targets both sides of the above-mentioned model of an imbalance in interhemispheric interplay. Bihemispheric brain stimulation may thus potentiate the effects of anodal stimulation to the lesional hemisphere2,10 through additional modulation of interhemispheric interactions4,5 via cathodal stimulation to the contralesional motor cortex. 9 This notion is supported by the results of an rTMS study, which compared unihemispheric with bihemispheric motor cortex stimulation. 8 However, neither the rTMS trial nor our current study used electrophysiological measures to assess the presumed change in interhemispheric interactions, which limits the interpretation of the behavioral results.
In conclusion, the integration of consecutive multiple-session tDCSs into a rehabilitation program of standard peripheral sensorimotor stimulation yielded the most prominent improvements during the first 5-day period. Changes during the second 5-day intervention were smaller but still significant. These findings may help in designing future experimental neurorehabilitation trials.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from NIH/NINDS (NS045049).
