Abstract
Objective
To use a systems paradigm to examine upper limb (UL) motor intervention elements driving biomarker and clinical measure improvement after stroke.
Methods
Databases were searched up to March 2022. Eligibility screening was completed by 2 authors. Studies using biomarkers and clinical measures pre- and post-upper limb intervention were included. Studies of adjunct interventions (eg, brain stimulation) were excluded. Cochrane Risk-of-Bias tools and Template for Intervention Description and Replication were used to rate studies. Studies were synthesized using a systems paradigm: intervention outcome was considered an emergent property of the systemic interactions of 4 intervention elements (demographics, type, quality, and dose) characterized by individual dimensions.
Results
Sixty-four studies (n = 1814 participants) containing 106 intervention groups (66 experimental; 40 control) were included. Combined biomarker and clinical outcomes defined 3 scenarios: restitution, mixed, and unchanged. The restitution scenario included more moderate-to-severely impaired participants in earlier recovery phases (<6 months). Interventions with graded difficulty were more frequently used in the restitution scenario compared with the unchanged scenario. No difference in quality or amount of therapy was identified when examining scenarios that demonstrated restitution compared to those that did not (mixed and unchanged).
Conclusions
A systems paradigm may be one of many approaches to understand UL motor restitution. This review found no single element consistently delivered improvements in biomarkers and clinical measures in the examined intervention groups. Complex patterns formed by multiple interacting intervention elements were observed in participants with and without restitution.
Introduction
Upper limb (UL) motor recovery remains a priority for stroke survivors 1 and has been identified as a key research area.1,2 Up to 80% of stroke survivors present with UL disability early after stroke. 3 While recent advances in acute stroke interventions, such as endovascular clot retrieval have dramatically changed the expectations for recovery, not all stroke survivors are eligible for these treatments. 4 For stroke survivors with persistent UL disability, 5 engagement in motor interventions is their pathway to recovery. This has yet to be fully optimized.
Current aspirational views of recovery focus on the ability of motor interventions to deliver restitution. 6 The international Stroke Recovery and Rehabilitation Roundtable defined restitution as a “return towards more normal patterns of motor control.” 6 As restitution reflects true recovery, it requires neurological recovery in parallel with motor recovery. 6 Empirical measurement of restitution after stroke has been challenging. The stroke recovery field has relied on biomarkers and clinical measures to provide a window to infer what may have occurred during the neurological and motor recovery processes. The Roundtable defined a biomarker as a “measure of the underlying molecular/cellular processes.” 7 When indexed longitudinally, improvement in brain biomarkers that measure structure or function may reflect neurological recovery. 7 Motor recovery may be examined using measures of impairment and activity 8 and both are important to uniquely examine change over time. Measures of UL impairment infer recovery of the individual’s body structure and function, while UL activity measures infer recovery of the individual’s ability to execute a task or action. 9 Therefore, to interrogate restitution (neurological + motor recovery), it is imperative for a given intervention to demonstrate parallel improvements in biomarkers, impairment measures, and activity measures.6,10,11 This novel strategy to interrogate restitution may yield a unique perspective that can guide the identification of successful UL motor interventions.
UL motor interventions should be considered complex by any definition.12 -14 Prior research15 -17 indicates that numerous elements, such as dose, 18 type, 19 and quality, 12 have been routinely applied in UL motor interventions to drive recovery. In clinical trials, these elements (eg, type and dose) and their multiple dimensions (eg, dose: duration and difficulty; type: treatment and delivery method) have largely been explored in a factorial paradigm, where effects of individual elements are studied either individually or as two-way interactions between elements. While this approach has often been unsuccessful in shifting recovery profiles,5,20 it reflects underlying feasibility constraints and the confirmatory nature of clinical trials. Importantly though, it has guided the evolution of our understanding of motor interventions from simple to complex.
A systems paradigm21,22 presents an alternative perspective on the inherent complexity of motor interventions.23,24 In such a paradigm, intervention outcomes would be conceptualized as emergent properties of the interaction between individual system elements and dimensions. It would postulate that restitution is the result of all the multidimensional elements of the intervention working in an integrated manner. No individual intervention element or dimension could be modified without affecting the resulting restitution outcome. Therefore, a systems paradigm places emphasis on the consideration of numerous elements and dimensions integral to the design and operation of said intervention, and acknowledgment of the elements’ interrelatedness.
Use of a systems paradigm, and subsequent shift in analytic emphasis, could help explain why similar interventions produce different outcomes in different contexts. This could bring more clarity as to how and why outcomes were achieved. Understanding how and why intervention elements drive restitution may guide the field to better exploit these elements and capitalize on restitution of UL function post-stroke. This may have downstream effects on lost wellbeing and premature mortality, experienced by stroke survivors. 25
Therefore, we aimed to identify and examine multidimensional elements of UL motor interventions in a systems paradigm and investigate their relationship to biomarkers and clinical measures of recovery after stroke via systematic review.
Methods
The definitions of key terms guiding the conduct of this systematic review are outlined in Table 1.
Definitions of Terms Used in This Systematic Review.

This systematic review was prospectively registered on the Prospective Register of Systematic Reviews (CRD42020206967). The Preferred Reporting Items for Systematic Reviews and Meta-Analysis 2020 statement provided the framework for reporting. 26
Information Sources and Search Strategy
Searches were conducted in MEDLINE, EMBASE, and Cochrane Controlled Register of Trials databases on March 13, 2022. Medical Subject Heading terms and a combination of relevant keywords were used (see Supplemental I).
Eligibility Criteria
The search yield was imported into Covidence 27 and duplicates removed. Eligibility was examined by 2 reviewers (MW, and one of NF or KH). All titles/abstracts and full texts were independently appraised. All studies were screened against predetermined eligibility criteria. Hand searching of included study reference lists and relevant reviews were completed to identify additional studies. We attempted to link identified abstracts from conference proceedings with full-text peer-reviewed papers where possible. Disagreements regarding eligibility were resolved through discussion (MW/NF/KH).
Design: Peer-reviewed randomized, non-randomized, or single group studies published in English that aimed to explore recovery of UL impairment or activity post-stroke in any setting (ie, inpatient, outpatient, home) and via any mode of delivery (ie, in-person, individual/group, telehealth) were eligible. Each study required a minimum of 10 participants, which is consistent with a prior Cochrane review. 28 Grey literature and all other designs were excluded, including single case and systematic reviews.
Participants: Studies that enrolled adult (≥18 years) human stroke survivors, of any stroke type or time post-stroke onset 6 were eligible. Preclinical studies (eg, animal studies) and non-stroke participants, including mixed diagnosis cohorts, were excluded.
Interventions: Interventions designed to improve UL motor function (impairment or activity) were eligible. Any study without an active intervention group was excluded. Any pharmacological, complimentary (eg, acupuncture), non-invasive brain stimulation (eg, transcranial direct current stimulation), priming (eg, repetitive transcranial magnetic stimulation), or brain driven robotic (eg, brain computer interface) interventions in combination with or without a motor intervention were excluded.
Outcomes: Studies must have included a biomarker that indexed brain structure or function, in addition to a measure of UL motor impairment or activity both pre- and post-intervention. Studies that measured general motor function (eg, activities of daily living) or participation 8 (eg, Stroke Impact Scale) were excluded, as well as studies with only a pre-intervention (baseline) biomarker.
Risk of Bias and Intervention Reporting
One author (MW) rated Risk of Bias (RoB) using Version 2 of the Cochrane Risk-of-Bias Tool 29 for randomized controlled trials (RCT) or the RoB In Non-Randomized Studies of Interventions 30 for non-RCTs and single group intervention trials. Each study was subject to a separate RoB examination for both clinical measures and biomarkers (Supplemental II and III). The highest RoB rating across all sections of either tool was reported as overall RoB (Table 4). Intervention reporting was examined using the Template for Intervention Description and Replication Checklist (TIDieR). 15 Twenty percent of the RoB and TIDieR ratings were double rated (KH).
Synthesis Methods
Elements and Dimensions Taxonomy
Each control and experimental group, herein referred to as intervention group, were deconstructed into dimensions based on TIDieR checklist 15 and dose articulation framework. 17 The TIDieR checklist was chosen due to the rigorous methods used in its development and alignment with intervention reporting. 15 The dose articulation framework was chosen due to its appreciation of the multidimensional nature of dose as an intervention element. Similar dimensions were collated under a consistent element. The system we sought to investigate contains four elements (demographics, type, quality, and dose) made from 16 different dimensions. If the study reported information relevant to the TIDieR item (and subsequent dimension) the information was recorded according to the taxonomy developed for this review (Table 2 and Supplemental IV). If there was no information reported relevant to the TIDieR item (and subsequent dimension) it was recorded as “not reported.” Table 2 defines the elements and dimensions extracted for each intervention group, corresponding TIDieR items, and example application of this taxonomy.
Taxonomy of Elements and Dimensions.
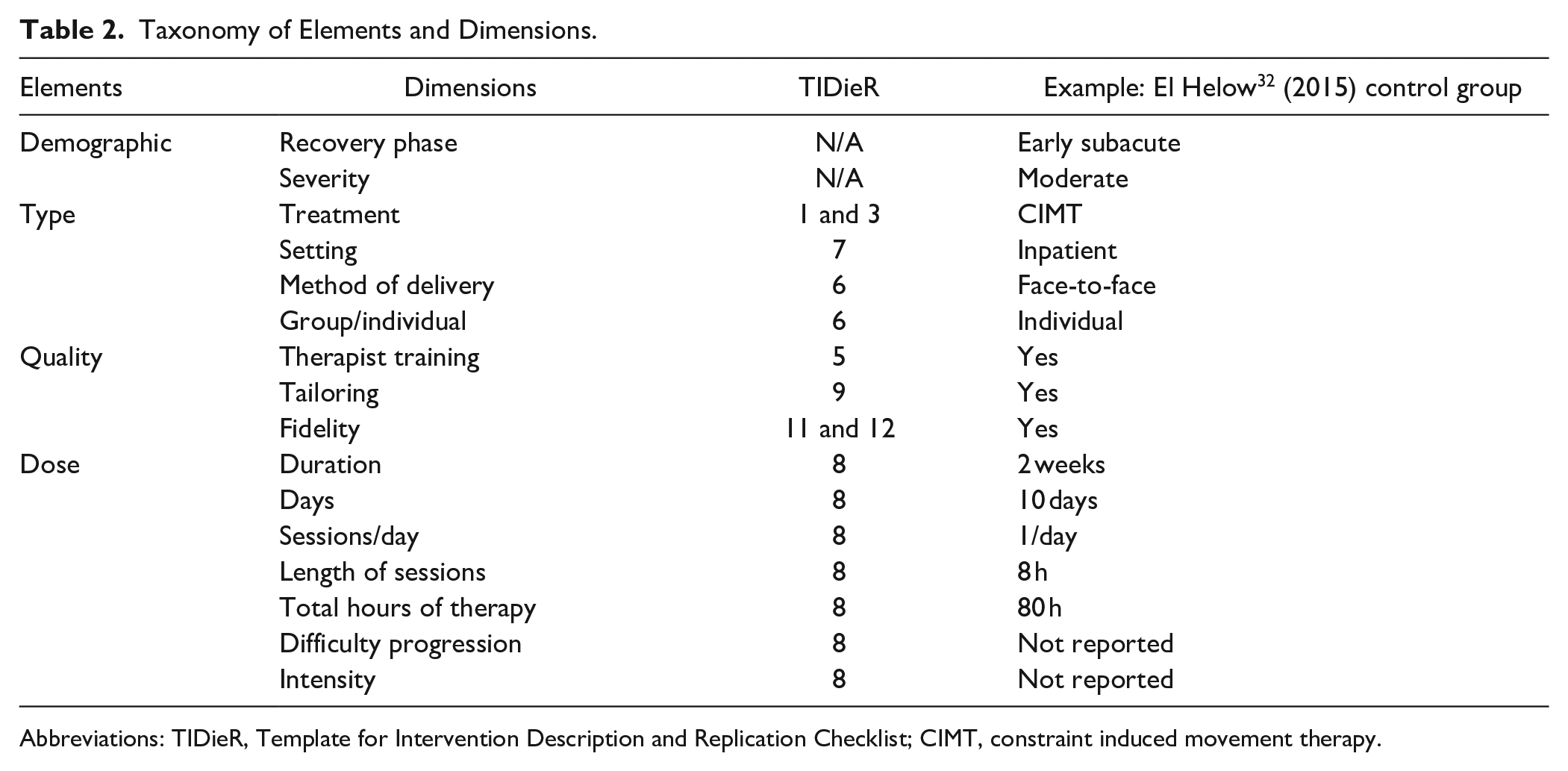
Abbreviations: TIDieR, Template for Intervention Description and Replication Checklist; CIMT, constraint induced movement therapy.
Classification of Recovery Phase
Studies were classified according to the Stroke Recovery and Rehabilitation Roundtable definitions: 6 acute, 1 to 7 days; early subacute, 7 days to 3 months; late subacute, 3 to 6 months; chronic, >6 months. Classification was made according to the intervention group’s mean time since stroke onset. If insufficient information was given the group was classified as “unable to classify.”
Classification of Severity
Studies were classified by participant severity using the mean of a clinical measure (UL eg, Fugl Meyer Upper Limb [FMUL], or general stroke scale eg, National Institutes of Health Stroke Scale) using established categories (Supplemental V). If studies did not use a clinical measure to define severity, study terminology was used: that is, mild, moderate, or severe. If there was no clinical measure that could be used to classify severity and no indication was given within the study, studies that stated an inclusion criterion of >10 degrees of voluntary finger extension were classified as mild, consistent with previous literature. 31 One other study was classified as moderate using baseline Action Research Arm Test summary scores using available literature. 32
Classification of Treatment Type
Treatment type were categorized as bilateral arm training, biofeedback, Bobath approach, constraint induced movement therapy (CIMT), electrical stimulation, “hands on” manual therapy techniques, mental practice or mirror therapy, music therapy or repetitive task training or task specific training, electromechanical interventions including robotics, strength training, virtual reality training, or standard therapy. 19 For data synthesis, similar treatment types were combined, such as repetitive task practice and task specific practice as both targeted high-volume task repetition.
Classification of Outcome Measures
Biomarkers were classified as measuring brain function or structure. 7 Changes in biomarkers, that were reported as statistically significant by each individual study and were in keeping with the changes outlined in Table 3, constituted improvement. Severity was an important consideration when defining improvements in biomarkers as mildly impaired stroke survivors may demonstrate different changes in biomarkers compared with moderate-severe survivors.33-35 Table 3 provides a summary of each biomarker, priority ranking, and justification for rating improvement.
Summary of Biomarkers.
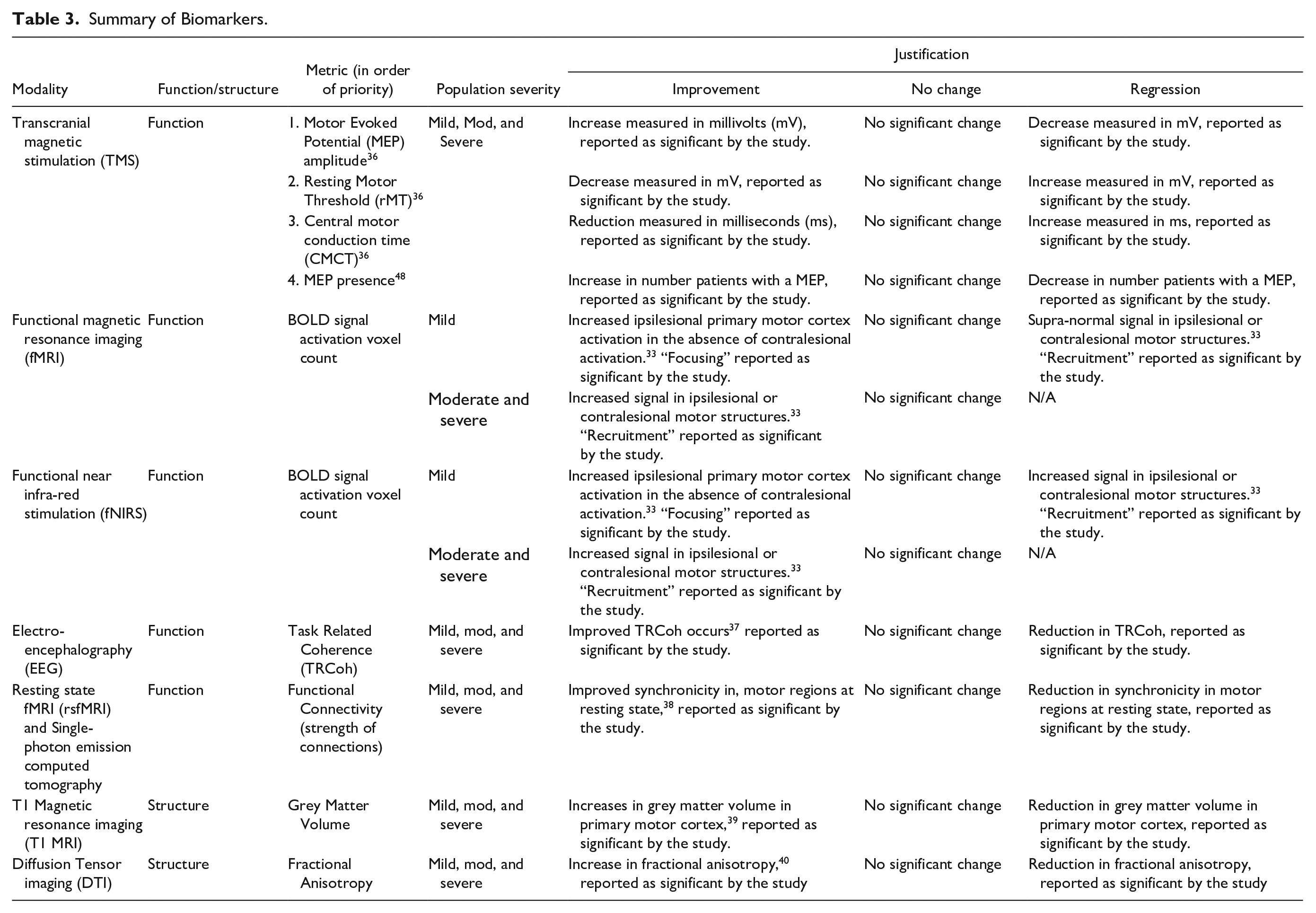
Clinical measures were classified as either impairment or activity 41 (Supplemental VI). To demonstrate improvement in clinical measures, each intervention group must have achieved the defined minimal clinically important difference (MCID) for the specific measure demonstrated by change from pre- to post-intervention. If no MCID was available in the literature, a 20% improvement was required. This 20% cut-off falls within evidence-based MCIDs reported for other UL measures post-stroke. 42 The applied MCIDs are outlined in Supplemental VI.
Data Extraction
Data from each study were extracted by 1 author (MW) and 20% were cross checked by a second author (KH). Two authors (MW/KH) discussed any unclear data to reach consensus. Intervention data were extracted in keeping with past reviews 17 and included participant demographics, intervention elements, biomarkers, impairment, and activity measures (Supplemental IV). If raw data were not provided, PlotDigitizerV2.6.9 interpretive software was used to extract the data from figures. If more data was required, the corresponding author was contacted (maximum 3 emails).
Data Analysis
Demographic and other data were collated in a custom-built Excel spreadsheet and summarized for overall number of participants and outcome measures used. Biomarkers and UL clinical measures were classified as improved, no change, or regressed (see Table 3 and Supplemental VI). Each dimension (Table 2) was analyzed dichotomously (reported or not). Dose dimensions were simplified to hours per day of therapy for consistency across studies. Dose was then dichotomized to whether the intervention group received a potentially important threshold for motor recovery (2 h/day). 43 Integrating outcomes on biomarkers and clinical measures classified each intervention group (ie, experimental and control) into 3 possible outcome scenarios:
(a) Restitution, groups that demonstrated parallel improvements in biomarkers, impairment, and activity measures.
(b) Mixed, groups that demonstrated improvements in either a biomarker, impairment, or activity measure in any combination.
(c) Unchanged, groups that demonstrated no change across biomarkers, impairment, and activity measures.
The restitution scenario elements were compared to a combined (mixed and unchanged) scenario. The mixed and unchanged scenarios were subsequently separated for a more nuanced comparison within each of these scenarios. Radar plots were created using Microsoft Excel version 2101 (Build 14827.20198). The results were descriptively reported.
Results
Search Results
The search strategy yielded 5317 studies after duplicates were removed (Cochrane Controlled Register of Trials n = 227, EMBASE n = 3784, MEDLINE n = 1306). A total of 201 full texts were reviewed. The PRISMA flow chart for the identification and inclusion of studies is provided in Supplemental VII. The final yield was 64 studies (RCT n = 34; non-RCT n = 6; single group n = 24). The reasons for exclusion at full text were related to the intervention (n = 51), study design (n = 46), sample size <10 (n = 24), duplication (n = 12), and absence of outcome measure (n = 4). A list of excluded references from full-text screening is provided in Supplemental VIII. From the 64 included studies, there were 106 intervention groups (66 experimental and 40 control). Summary of all included studies is provided in Supplemental IX and corresponding references are provided in Supplemental X.
Population Demographics
A total of 1814 participants were included (women n = 581, men n = 1069, not reported n = 164). Most intervention groups included participants with moderate motor impairment (n = 45, 42%) followed by mild (n = 38, 36%) and severe (n = 23, 22%). Participant groups were commonly in the chronic phase of recovery (n = 59, 56%) followed by early sub-acute (n = 31, 29%) and late subacute (n = 8, 8%). Of the 106 intervention groups: 12 used structural biomarkers, 99 used functional biomarkers, 70 used impairment measures, and 80 used activity measures.
Risk of Bias
Most studies were rated moderate RoB (n = 32, 50%), followed by high or critical (n = 26, 41%) and low (n = 6, 9%) (Table 4 and Supplemental II).
Results Summary.
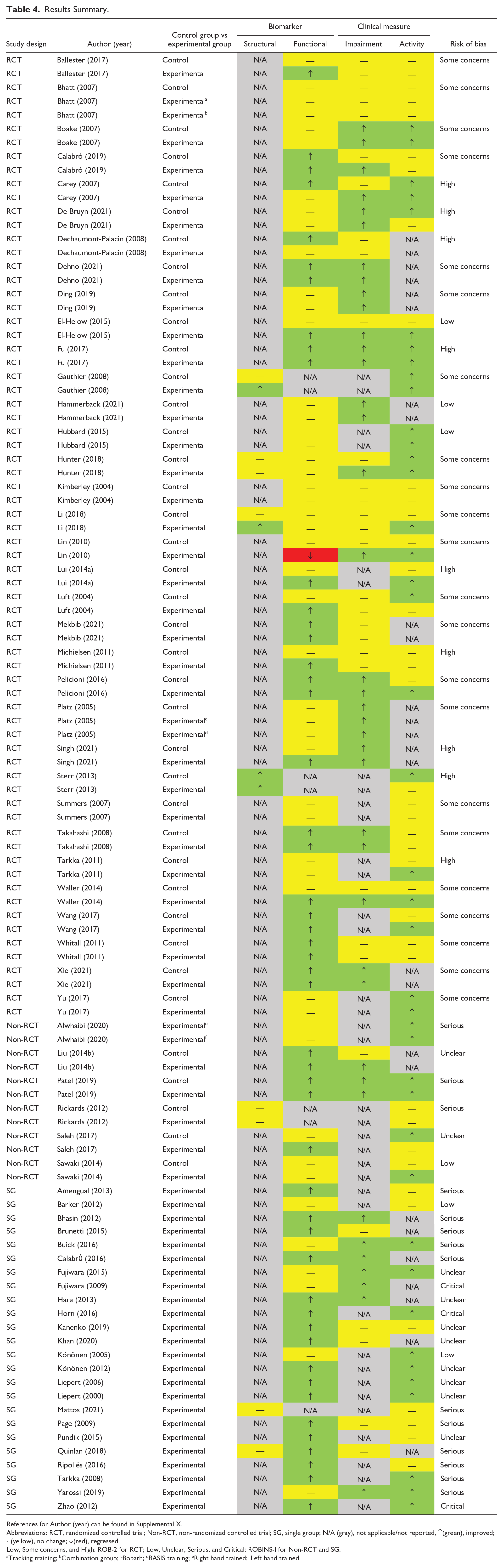
References for Author (year) can be found in Supplemental X.
Abbreviations: RCT, randomized controlled trial; Non-RCT, non-randomized controlled trial; SG, single group; N/A (gray), not applicable/not reported, ↑(green), improved; - (yellow), no change; ↓(red), regressed.
Low, Some concerns, and High: ROB-2 for RCT; Low, Unclear, Serious, and Critical: ROBINS-I for Non-RCT and SG.
Tracking training; bCombination group; cBobath; dBASIS training; eRight hand trained; fLeft hand trained.
Intervention Reporting
The median TIDieR score was 6. All studies reported intervention “name” that is, treatment type. The poorest items reported were: “who provided?,” that is, therapist training (11%), and “how well (planned)?” (10%) (see Figure 1A). The most frequently reported element was demographics (98%). The least frequently reported element was quality (33%). Reporting of elements and dimensions can be seen in Figure 1B.
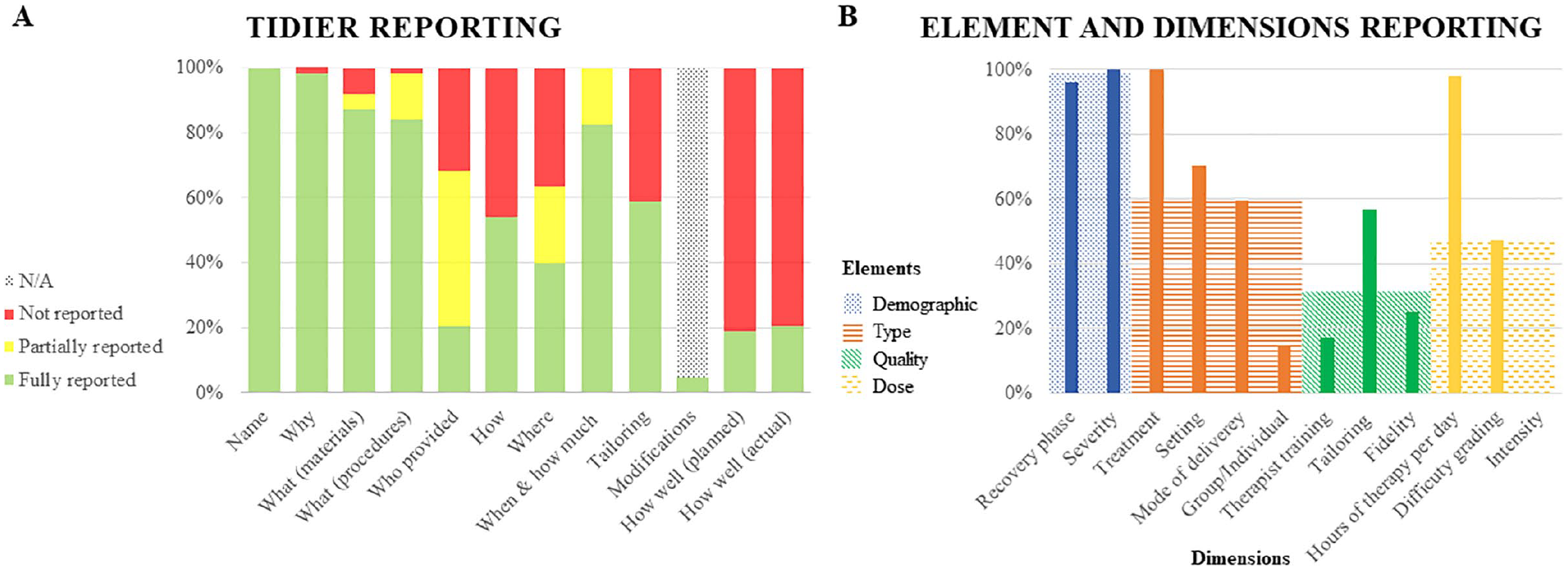
(A) TIDieR reporting. (B) Reporting of elements and dimensions.
Outcome Measures
Biomarkers
Structural biomarkers were included in 12 intervention groups using either diffusion-weighted diffusion tensor imaging (n = 6) or T1-weighted/MPRAGE structural magnetic resonance imaging (n = 6). Functional biomarkers were included in 99 intervention groups, with transcranial magnetic stimulation (n = 44) and functional magnetic resonance imaging (n = 40) used the most. Five intervention groups (5%) used both structural and functional biomarkers simultaneously.
An improvement in a structural biomarker was reported in only 4 intervention groups; remaining groups indicated no change. No groups regressed on a structural biomarker. Improvement on a functional biomarker was observed in 49 intervention groups and no changes were evident in 49 intervention groups. One intervention group 44 demonstrated a regression on a functional biomarker as defined by this review. This study demonstrated recruitment of supra-normal contralesional motor areas in a population with mild UL impairment. Of the 5 intervention groups that used both a structural and functional biomarker, 1 intervention group improved on its structural biomarker but demonstrated no change on the functional biomarker used. 45 The remaining 4 intervention groups demonstrated no change across both the structural and the functional biomarker used.
Clinical Measures
Only 44 intervention groups included both an impairment and activity measure. Impairment measures were included in 70 intervention groups, and FMUL (n = 58) was the most common. Activity measures were included in 80 intervention groups, and Wolf Motor Function Test (n = 35) was the most common. An MCID was achieved in 39 intervention groups that measured impairment and 41 intervention groups that measured activity. No intervention group regressed on either impairment or activity measures. Sixteen intervention groups achieved an MCID on both impairment and activity measures, while 19 intervention groups did not demonstrate an MCID across both measures. Five intervention groups demonstrated an MCID on an impairment measure while not achieving an MCID on the activity measure used in parallel. Four intervention groups demonstrated an MCID on an activity measure while not achieving an MCID in the impairment measure used in parallel.
Biomarkers and Clinical Measures
Only 4 intervention groups included all 4 measures: that is, a structural and functional biomarker, and an impairment and activity measure. None of these intervention groups demonstrated improvement across all 4 measures. There were 44 intervention groups that included a functional biomarker and both impairment and activity measures. Seven of these intervention groups improved across all 3 measures. Eleven intervention groups demonstrated no change in biomarker and did not achieve an MCID in either impairment or activity measures. Eight intervention groups improved on both impairment and activity measures despite no change on a functional biomarker. There were no groups that included a structural biomarker and both impairment and activity measures. A summary of results on biomarkers and clinical measures are provided in Table 4.
Intervention Element of Demographics and Its Dimensions
Both demographics dimensions (recovery phase and severity) were reported in 102 intervention groups (96%). Four intervention groups did not report their participant’s time since stroke.
Intervention Element of Type and Its Dimensions
All 4 dimensions of type were reported in 12 intervention groups (11%), 45 intervention groups (42%) reported 3 dimensions, and 32 intervention groups (31%) reported 2 dimensions. Seventeen intervention groups (16%) reported only type of intervention. Repetitive task training (n = 24) and CIMT (n = 15) were the most common intervention types. The most common setting for therapy delivery was center-based outpatient (n = 35) and most frequently reported mode of delivery was face-to-face (n = 55).
Intervention Element of Quality and Its Dimensions
All 3 dimensions of quality were reported by 7 intervention groups (7%), 19 intervention groups (18%) reported 2 dimensions, and 43 intervention groups (41%) reported 1 dimension. Thirty-seven intervention groups (35%) did not report any dimension of quality.
Intervention Element of Dose and Its Dimensions
No intervention group reported all 7 dimensions of dose due to no intervention group reporting intensity. Six dimensions of dose were reported in 40 intervention groups (38%), and a further 19 intervention groups (18%) reported 5 dimensions.
Examination of Motor Interventions as a System
Three scenarios were examined: 7 interventions groups were included in the restitution scenario; 11 intervention groups in the unchanged scenario; and remaining 86 intervention groups were included in the mixed scenario. Pooling each scenario’s reported dimensions was used to create a signature for each scenario, represented in Figure 2.
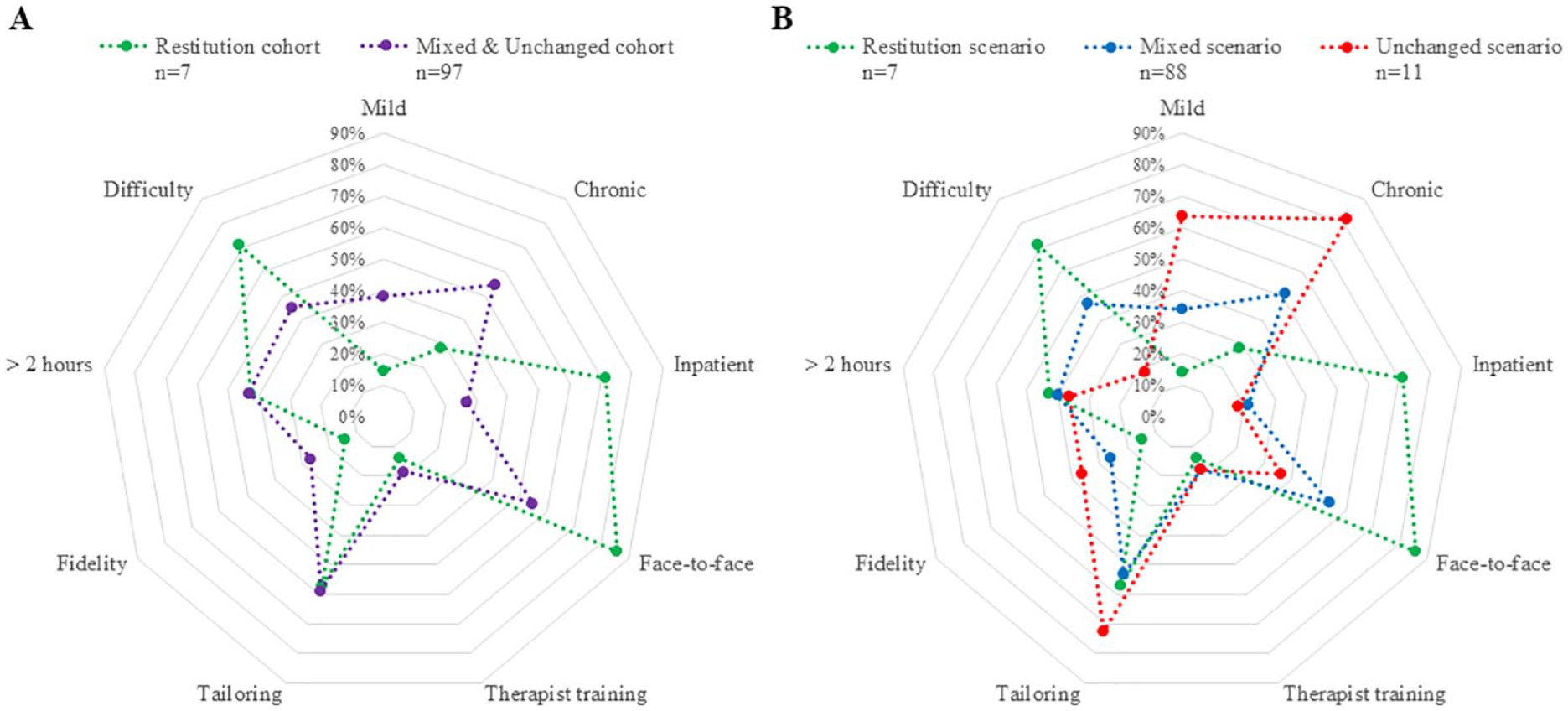
Signature of scenario signatures. (A) Restitution scenario versus combined scenario. (B) Restitution scenario versus unchanged scenario versus mixed scenario.
In the restitution scenario, the interventions (systems) were most frequently conducted face-to-face (86%) in an inpatient setting (71%) with moderate or severely impaired, non-chronic (<6 months) stroke survivors (Figure 2A). There were no other distinct differences in the restitution scenario signature compared to the signature of the combined (mixed and unchanged scenarios). Breaking down the mixed and unchanged into 2 separate scenarios demonstrated that graded difficulty was infrequently used as a dimension (18%) in the unchanged scenario compared to the signature of the restitution scenario (71%) (see Figure 2B). There was no one element or dimension that consistently delivered an improvement in biomarkers or clinical measures for stroke survivors across all scenarios.
Discussion
We aimed to examine multidimensional elements of UL motor interventions using a systems paradigm and identify their influence on biomarkers and clinical measures after stroke. At a macroscopic level, the restitution scenario included interventions that more frequently reported graded difficulty, were delivered face-to-face, and were delivered in an inpatient setting. The restitution scenario also included more moderate to severely impaired stroke survivors in the first 6 months of recovery. Further, an important finding at the level of intervention groups was that no intervention group demonstrated a regression on either an impairment or activity measure. Interestingly, 26 of the intervention groups with participants in the chronic phase of recovery (47%) demonstrated improvement in either an impairment or activity measure. These results support the notion that UL motor intervention can facilitate improvement, even in the chronic phase of stroke recovery.
The restitution scenario included mostly moderate to severely impaired intervention groups which may have allowed for more scope to improve on a given clinical measure. In contrast, mildly impaired stroke survivors, who were more commonly included in the unchanged scenario, may have had less capacity to improve on a given clinical measure due to possible ceiling effects. This may have impacted our ability to index an MCID for the purpose of this review. The restitution scenario also included more stroke survivors who were in earlier phases of recovery (<6 months), which suggests their outcome may have been supported by spontaneous biological recovery. 6 A time sensitive window of heightened potential for recovery is considered to last 3 to 6 months post stroke. 5 In this early period, the brain may be better able to capitalize on therapeutic opportunities. 46 Together, this highlights that UL intervention trials need to carefully consider who is recruited; that is, identifying important demographic data, both in severity and phase of recovery, as an element that may impact restitution.
Our analysis highlighted that there was more to a successful UL intervention than a single element, such as type, dose, or quality. The nuances of these results need to be carefully examined. Reporting of intervention dimensions was generally poor and with this volume of missing data, omission of measurement must not be mistaken for absence of effect. Until studies consistently report interventions using tools such as the TIDieR checklist, 15 the field may continue to struggle to translate findings into clinical practice. Interestingly, there was no treatment type that was represented more than another in the restitution scenario. Additionally, a consistent amount of therapist training (quality), tailoring of interventions (quality), and therapy per day (dose) was reported across scenarios. Multiple other dimensions (progressive difficulty, inpatient setting, and face-to-face delivery) were more prevalent in the restitution scenario compared to the unchanged scenario. The novelty of the current analysis, which is embedded within a systems paradigm, is the appreciation that restitution may be an emergent property of the combination of intervention elements and dimensions. In this systems paradigm, omission of an element or dimension may not simply reduce the magnitude of effect, it may change the outcome altogether and prevent the emergence of restitution. As exemplified above: even with adequate attention to the elements of dose, type, and quality of the intervention, inattention to the demographic element may see restitution not be realized. Considering multiple intervention elements essential to the design and operation of an intervention is in keeping with a recent trend in UL intervention studies that deliver improved clinical measures when conceptualized this way. 47 If interventions designed in a systems paradigm deliver clinical improvements to patients, therapists and researchers may equally benefit from this conceptual shift.
A second novel feature of this systematic review was the inclusion of clinical measures across multiple domains of the WHO-ICF in parallel with biomarkers. Our findings demonstrate the utility of this line of inquiry and support the need to prioritize collection of biomarkers and clinical measures across multiple UL motor domains of the ICF in future research. 11 Inclusion of biomarkers is key to help clarify how and why an intervention works by inferring what biological processes underpin the observed motor changes. Impairment and activity measures together provide more robust evidence for recovery than either one measure used in isolation. When improvement in a clinical measure was demonstrated, this review found that there was often a disconnect with improvements in biomarkers. This highlights the importance of selecting the right biomarker and clinical measures for the right population. For example: transcranial magnetic stimulation relies on intact corticospinal tract architecture, which is often compromised in more severely impaired stroke survivors; 48 and timed clinical measures (eg, box and block test) may more appropriately interrogate change in more mild populations as ordinal clinical measures (eg, Motor Assessment Scale) are more prone to ceiling effects. 49 Inclusion of kinematic measures would further advance the discussion of restitution and extend the findings of this review. While clinical measures are useful and easily accessible to monitor change, they cannot fully appreciate if recovery was achieved through compensation or restitution. 50 Kinematic measures have potential to discern between these 2 outcomes. Researchers have acknowledged the need to develop efficient and reliable methods for kinematic data collection and embed these measures within UL trials, and this remains a high priority. 50
This review has some limitations. Only studies with >10 participants were included. This may have led to exclusion of studies with small numbers reporting new or emerging interventions. However, small under-powered studies would have likely been rated at high RoB. Limiting to English excluded studies in other languages. The use of a 20% MCID for clinical measures with an undocumented MCID led to a higher standard than historically applied. 51 However, this threshold is a cautious interpretation that is within the range of 16% to 30% defined for other outcomes included in this review. 42 Interestingly, no reported results would have changed if a 10% threshold had been applied. Dichotomizing intervention dimensions reduces the nuance of the findings. Omission of measurement is not absence of effect and caution must be taken when interpreting these results (ie, Figure 2A and B). The routine use of established tools in UL trials may improve reporting of our interventions and advance our understanding of the impact that motor interventions have on recovery. 52 Finally, it is important to note that none of the studies included in this review intended their results to be examined in a systems paradigm, or for their outcomes to be presented this way. This limits the strength of the findings in this review as studies may have chosen to omit or emphasize certain aspects of their findings in the publication process.
Conclusion
A systems paradigm may be one of many approaches to understand UL motor restitution. This is in keeping with the finding that no single element of a motor intervention consistently led to improvements in biomarkers, impairment, or activity measures in the examined intervention groups. Given the difficulties with dramatically improving recovery profiles from UL motor interventions, different approaches should be explored to drive the field of stroke recovery forward in its thinking. While this review demonstrated that motor interventions can be conceptualized in a systems paradigm, it does not suggest that the proposed dimensions and elements are complete. Some elements were not able to be considered in our analysis, such as motivation, and these should be considered in future work. Careful selection of participants, intervention elements, clinical measures, and biomarkers remain not only key considerations but also key tools that may progress the field closer to understanding UL restitution.
Supplemental Material
sj-docx-1-nnr-10.1177_15459683221129273 – Supplemental material for Upper-Limb Motor Intervention Elements That Drive Improvement in Biomarkers and Clinical Measures Post-Stroke: A Systematic Review in a Systems Paradigm
Supplemental material, sj-docx-1-nnr-10.1177_15459683221129273 for Upper-Limb Motor Intervention Elements That Drive Improvement in Biomarkers and Clinical Measures Post-Stroke: A Systematic Review in a Systems Paradigm by Matthew Wingfield, Natalie A. Fini, Amy Brodtmann, Gavin Williams, Leonid Churilov and Kathryn S. Hayward in Neurorehabilitation and Neural Repair
Footnotes
Disclosures
Independent contractor work outside submitted work: AB, Biogen.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Person: National Health and Medical Research Council of Australia [KSH:1088449]; Heart Foundation of Australia [AB:GNT100784, GNT104748]. Institutional: Florey Institute of Neuroscience and Mental Health acknowledges the Victorian Government’s Operational Infrastructure Support Grant [KSH/AB].
Data Availability
Upon reasonable request to
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
