Abstract
Background. Proportional motor recovery in the upper limb has been investigated, indicating about 70% of the potential for recovery of motor impairment within the first months poststroke. Objective. To investigate whether the proportional recovery rule is applicable for upper-limb somatosensory impairment and to study underlying neural correlates of impairment and outcome at 6 months. Methods. A total of 32 patients were evaluated at 4 to 7 days and 6 months using the Erasmus MC modification of the revised Nottingham Sensory Assessment (NSA) for impairment of (1) somatosensory perception (exteroception) and (2) passive somatosensory processing (sharp/blunt discrimination and proprioception); (3) active somatosensory processing was evaluated using the stereognosis component of the NSA. Magnetic resonance imaging scans were obtained within 1 week poststroke, from which lesion load (LL) was calculated for key somatosensory tracts. Results. Somatosensory perception fully recovered within 6 months. Passive and active somatosensory processing showed proportional recovery of 86% (95% CI = 79%-93%) and 69% (95% CI = 49%-89%), respectively. Patients with somatosensory impairment at 4 to 7 days showed significantly greater thalamocortical and insulo-opercular tracts (TCT and IOT) LL (P < .05) in comparison to patients without impairment. Sensorimotor tract disruption at 4 to 7 days did not provide significant contribution above somatosensory processing score at 4 to 7 days when predicting somatosensory processing outcome at 6 months. Conclusions. Our sample of stroke patients assessed early showed full somatosensory perception but proportional passive and active somatosensory processing recovery. Disruption of both the TCT and IOT early after stroke appears to be a factor associated with somatosensory impairment but not outcome.
Introduction
Upper-limb motor function is frequently impaired after stroke and displays a wide range of recovery from a complete flaccid and nonfunctional upper limb to (nearly) normal function. 1 Early prediction of recovery is clinically relevant to steer goal setting and treatment selection 2 and can be useful to guide recruitment for clinical trials in the early phase. 3 Initial motor impairment, assessed by clinical scales such as the Fugl-Meyer Upper Extremity assessment (FM-UE), is considered a key predictor of motor recovery, as shown in several review studies concerning poststroke prognosis.4,5 More recently, the concept of proportional recovery has been introduced, demonstrating that after removing a subpopulation of patients with severe motor impairment and poor to no recovery (typically called nonfitters), in the remaining population (fitters) a strong proportional relation exists between initial impairment and recovery.6-12 In one of the first studies on this topic, Prabhakaran et al 6 collected FM-UE at 24 to 72 hours and again at 3 months poststroke. For the sample excluding patients with severe initial impairment, they found that patients at 3 months obtained about 70% (95% CI = 57%-80%) of their maximal potential recovery, defined as the difference between the initial FM-UE score and the maximum score (66) of the scale. 6
Recent studies have investigated neural correlates underlying the proportional recovery rule, with a focus on corticospinal tract (CST) integrity.3,8,12,13 Early neuroimaging and neurophysiology biomarkers have been found, such as fractional anisotropy asymmetry index at the level of the posterior limb of the internal capsule and motor-evoked potential (MEP) responses to transcranial magnetic stimulation (TMS). A recent large cohort study 3 confirmed the earlier study of Byblow et al 8 by showing that patients with MEP responses to TMS will recover proportionally regardless of baseline impairment, whereas patients without MEP responses are expected to have limited to no recovery.
Further studies have confirmed the proportional recovery rule3,7 and investigated the applicability in other domains besides upper-limb motor outcome, such as visuospatial neglect9,11 and aphasia,11,14 suggesting a common underlying recovery paradigm, which applies to motor and cognitive functions. 11 Finally, Smith et al 10 investigated proportional recovery from lower-limb motor impairment using lower-limb FM assessments at 3 days and 3 months. Their results showed proportional recovery of 74% (95% CI = 60%-88%) from lower-limb impairment. Interestingly, there was no identifiable nonfitters group—that is, patients who did not fit the proportional recovery rule. 10
A common deficit in the upper limb besides motor impairment is a somatosensory deficit. Meyer et al 15 investigated the prevalence of disorders in somatosensory modalities in the first 4 to 7 days poststroke and reported impairment in somatosensory modalities in 78% of patients. Recent studies focusing on this topic showed that somatosensory deficits have significant relations with unimanual and bimanual upper-limb motor impairment and recovery.16,17 So the question is whether the proportional recovery rule is applicable for somatosensory impairment. Therefore, we investigated patients at 4 to 7 days and 6 months after stroke and determined whether there is full or proportional somatosensory recovery. We analyzed the different components of somatosensory impairment: (1) somatosensory perception, defined as the ability to perceive a somatosensory stimulus (eg, touch, pressure); (2) passive somatosensory processing, defined as the ability to discriminate between somatosensory stimuli (eg, sharp/blunt discrimination, proprioception); and (3) active somatosensory processing (eg, stereognosis), defined as the ability to recognize an object by the integration of somatosensory functioning and active manipulation. We hypothesized, based on earlier longitudinal work from our group, 15 that there is full somatosensory perception recovery, but proportional passive and active somatosensory processing recovery. Because we obtained magnetic resonance imaging from our patients, we were further able to test the hypothesis that somatosensory impairment would be shown in patients with disruption of sensorimotor pathways and that the amount of disruption would relate to somatosensory outcome at 6 months.
Methods
For the present study, 38 consecutive patients were included from a previous study recruiting from the acute stroke unit of the University Hospitals Leuven (Belgium) and Cliniques Universitaires Saint-Luc, Brussels, Belgium, between October 2012 and September 2014. 15 Six patients did not complete the assessment at 6 months and were, therefore, excluded from the analysis. Inclusion criteria were as follows: first-ever stroke (ischemic or hemorrhagic); assessment within the first week after stroke onset; presence of somatosensory and/or motor impairment, as detected by the Erasmus MC modification of the revised Nottingham Sensory Assessment (Em-NSA) 18 and FM-UE 19 ; and sufficient cooperation to perform the assessment. Patients were excluded if presenting a prestroke Barthel Index 20 score <95 out of 100; other serious neurological conditions with permanent damage, such as subdural hematoma, tumor, encephalitis, or trauma that leads to symptoms similar to that of a stroke; and serious communication, cognitive, or language deficits, which could interfere with the assessment protocol. Ethical approval was obtained from the Ethics Committee of both university hospitals. Written informed consent was signed prior to participation.
Clinical Assessment
Patients were evaluated by one trained research therapist (SM). Assessments were performed within the first week (between day 4 and 7 poststroke) and at 6 months after stroke. At the first assessment, patient characteristics were collected, including data regarding age, gender, hand dominance, time poststroke, stroke severity (National Institutes of Health Stroke Scale [NIHSS] 21 ), lateralization, and type of stroke (ischemic or hemorrhagic).
The clinical evaluation comprised the FM-UE, Em-NSA, and the stereognosis component of the NSA (NSA-stereognosis) for the upper limb, assessed at both time points. Clinical scales were selected according to established validity and reliability together with a balance between clinical utility and psychometric properties. 22
The FM-UE 19 assesses motor impairment as a whole for the upper extremity, including shoulder, elbow, wrist, and hand assessment, from reflex activity to voluntary activation. The total score for the FM-UE ranges between 0 and 66, with a higher score representing less upper-limb motor impairment. Excellent reliability and validity have been reported for investigating motor impairment.19-21,23
Somatosensory impairment was assessed by the Em-NSA, 18 in which 5 distinct somatosensory modalities in the affected upper limb are evaluated: light touch, pressure, pinprick, sharp-blunt discrimination, and proprioception. Light touch is tested with cotton wool, pressure with the index finger, pinprick with a toothpick, and sharp-blunt discrimination with alternating the index finger and a toothpick, all at predefined points of contact. Proprioception is assessed during passive movements of the different joints of the upper limb. Each site is assessed 3 times and graded on an ordinal scale as a 0 (patient fails to detect sensation on all 3 occasions), 1 (identifies test sensation, but not on all 3 occasions), or 2 (correctly identifies the test sensation on all 3 occasions), and this is repeated over several sites in each modality. Scores for each modality range on a scale from 0 (complete somatosensory impairment) to 8 (no somatosensory impairment). The total score for the Em-NSA ranges from 0 to 40, with a higher score representing less upper-limb somatosensory impairment. A score of less than 40 indicates somatosensory impairment.18,24 The Em-NSA has good to excellent intrarater and interrater reliability 18 and validity 25 and is a recommended measure of sensation in neurological conditions. 22 Light touch, pressure, and pinprick were grouped as measures of somatosensory perception. Sharp-blunt discrimination and proprioception were clustered as measures of passive somatosensory processing impairment.
Active somatosensory processing was evaluated through stereognosis, which assesses the ability to identify an object by manipulation with eyes closed. Stereognosis assessment was based on the original NSA, in which participants are asked to identify 11 common objects by touch and manipulation in the affected hand. When needed, assistance to the manipulation of objects in the hand was given by the assessor. Total scores range from 0 to 22. A cutoff score of less than 19 was used for the presence of stereognosis impairment. 15 The stereognosis section of the NSA shows a moderate to good test-retest reliability in people with stroke. 26
Imaging Acquisition, Lesion Segmentation and Overlay, and Probabilistic Fiber Tracking
Together with being assessed clinically at the first assessment 4 to 7 days poststroke, our patients underwent an imaging protocol within the first week poststroke, and subsequent imaging analysis was performed, as reported elsewhere. 27 Magnetic resonance images of the brain were obtained with a Philips 3T Achieve scanner. Either 3D or 2D fluid-attenuated inversion recovering imaging (FLAIR) data and diffusion-weighted images (DWIs) were acquired. Parameter settings for FLAIR sequences were as follows: echo time = 350 ms, repetition time = 4800 ms, inversion time = 1650 ms, field of view = 250 × 250 mm2, slice thickness = 1.12 mm, and gap = 0.56 mm. Parameter setting for DWI sequences were as follows: number of slices = 58, number of gradient directions = 60, b-value = 1300 s/mm2, echo time = 72 ms, repetition time = 12 s, slice thickness = 2.5 mm, gap = 2.5 mm.
Thalamocortical tract (TCT), insulo-opercular tract (IOT), and CST probabilistic fiber tracking were obtained from DWIs of 24 healthy, age-matched volunteers, as described elsewhere. 27 A 3T Siemens SkyraMRI scanner (Siemens, Erlangen, Germany) and 32-channel head coil were used. A total of 75 axial slices were obtained covering the whole brain with gradients (b = 1500 s/mm2) applied along 64 noncollinear directions with the following sequence parameters: repetition time = 10 000 ms, echo time = 82 ms, field of view = 256 × 204, slice thickness = 2 mm, and in-plane resolution = 2 × 2 mm2. All data sets were corrected for eddy currents and head motion. Common somatosensory tracts were created using voxels that were found in at least 50% of the participants. Individual pyramidal tracts were also created analogously using the precentral cortex as seeding mask and waypoints in the posterior internal capsule (MNI coordinates z = 5 to z = 7), cerebral peduncle, and pontomedullary junction (z = −44 to z = −48). Only streamlines starting from the primary motor cortex and passing all 3 masks were kept. Further exclusion masks were used to avoid interhemispheric trajectories, aberrant pathways, and loops via subcortical or cerebellar structures. Default settings of the tracking algorithm (probtrackx) were used (curvature threshold: 0.2; step length: 0.5 mm; distance correction: off). The resulting volumes of interest were overlaid on lesion volumes to measure TCT, IOT, and CST lesion load (TCT-LL%, IOT-LL%, and CST-LL%, respectively), defined as the percentage of tract volume (voxel) that was within the volume of the stroke lesion. 3
Statistical Analysis
Patient characteristics were analyzed and presented through descriptive statistics. Clinical scores of somatosensory perception, and passive and active somatosensory processing impairment obtained at 4 to 7 days and 6 months poststroke were used to calculate (1) potential recovery, defined as the difference between the score obtained at 4 to 7 days and the maximum score of the scale, and (2) observed recovery, defined as the difference between the scores obtained at 6 months and scores at 4 to 7 days. Subsequently, scatterplots were derived displaying potential and observed recovery. We applied the same analysis for motor impairment (FM-UE), as validation of our sample, expecting to find proportional motor recovery in line with previous literature. On the scatterplot of motor recovery, we identified visually fitters and nonfitters groups for proportional recovery and investigated the existence of these groups through agglomerative hierarchical clustering analysis (Ward’s method, squared Euclidean distance). On the scatterplots of somatosensory perception and processing recovery, there was no nonfitters group identified through hierarchical clustering analysis. We noted 1 possible outlier for somatosensory perception and passive somatosensory processing: a patient with very severe somatosensory impairment and a poor somatosensory recovery. We applied a standard test for outliers (standardized residuals), which confirmed this as an outlier and removed this patient from subsequent regression analyses. For clarity, we will refer to fitters/nonfitters grouping when discussing motor recovery and to the whole group when discussing somatosensory recovery.
Next, a univariate linear regression analysis was conducted for somatosensory recovery (whole group) and motor recovery (fitters) separately, with potential recovery included as the explanatory variable and observed recovery as the outcome variable. The resulting slope of the regression equation represents, then, the somatosensory and motor recovery model. Bootstrapping (single-sampling method, 1000 samples, 95% CI) was performed to test the robustness of regression models.
Finally, to study neural correlates of somatosensory impairment, between-group differences for patients with normal and decreased somatosensory performance at 4 to 7 days were investigated for TCT-LL%, IOT-LL%, and CST-LL% obtained within 1 week poststroke using nonparametric tests (Mann Whitney U). For somatosensory domains that showed proportional recovery, univariate and multivariate regression analyses were conducted, evaluating whether TCT-LL%, IOT-LL%, and CST-LL%, in combination with the clinical score at 4 to 7 days, contributed to predicting somatosensory outcome at 6 months. Level of significance was set at P < .05. Analyses were performed using IBM SPSS Statistics for Windows, Version 24 (Armonk, NY, USA).
Results
In total, 32 patients were included in the present analysis. Median (interquartile range [IQR]) age was 68 (61-80) years; 53% of patients were male; and 84% had an ischemic stroke. Out of 32 patients, 23 (72%) had a right-sided lesion. All patients were evaluated at 4 to 7 days (median: 6, IQR: 5-7 days) and at 6 months (median: 183, IQR: 181-185 days) from stroke onset. Stroke severity, as evaluated by the NIHSS, showed a median of 8 (IQR: 5-13) out of 42 points. Upper-limb somatosensory impairment, as assessed by Em-NSA showed median scores of 33 (IQR: 13-40) out of 40 at 4 to 7 days and 40 (IQR: 39-40) out of 40 at 6 months. Median upper-limb motor impairment (FM-UE) was 20 (IQR: 2-55) out of 66 at 4 to 7 days and 59 (IQR: 10-64) out of 66 at 6 months. Our sample ranged from severely to mildly impaired patients, and further characteristics are provided in Table 1.
Patient Characteristics. a
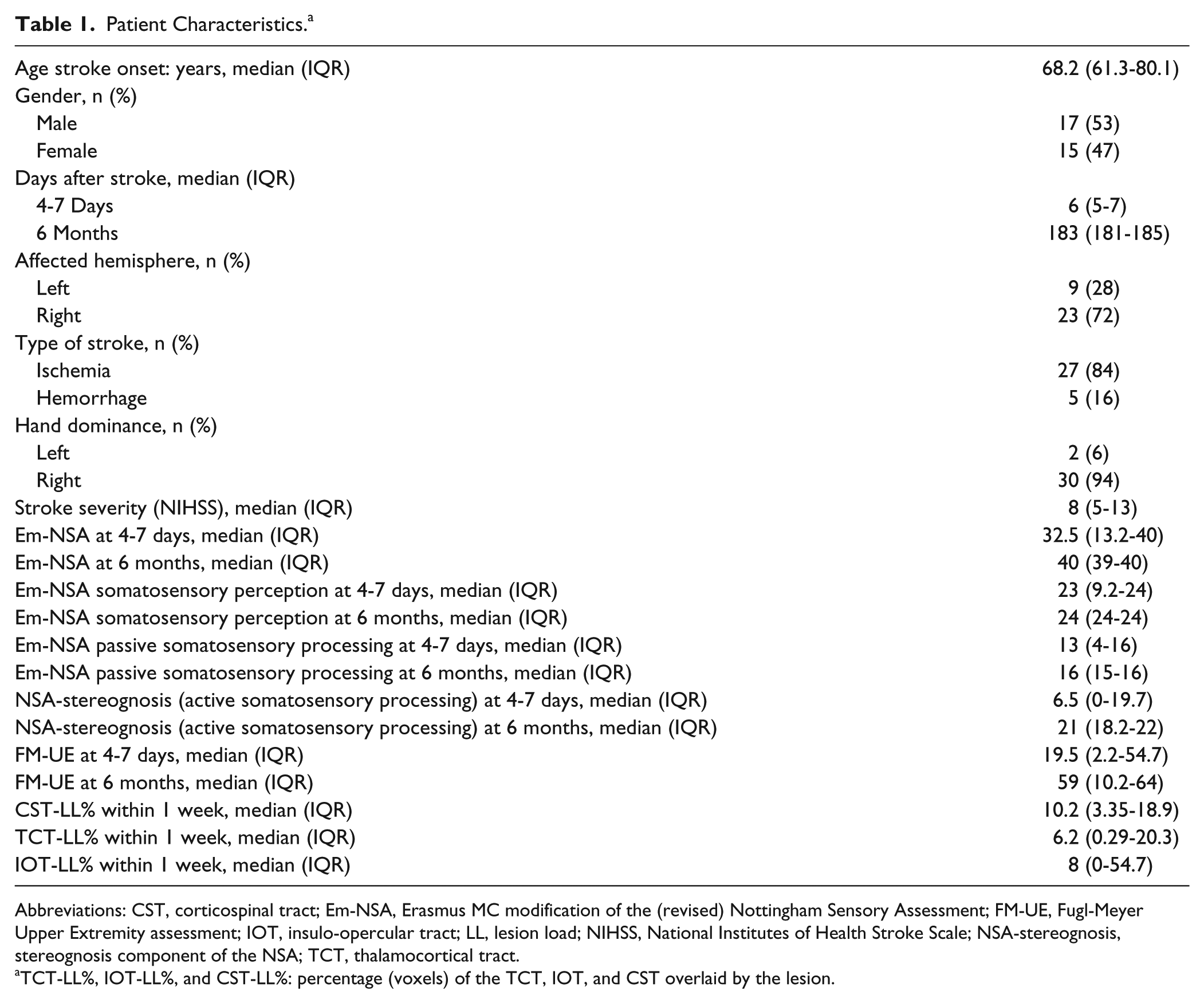
Abbreviations: CST, corticospinal tract; Em-NSA, Erasmus MC modification of the (revised) Nottingham Sensory Assessment; FM-UE, Fugl-Meyer Upper Extremity assessment; IOT, insulo-opercular tract; LL, lesion load; NIHSS, National Institutes of Health Stroke Scale; NSA-stereognosis, stereognosis component of the NSA; TCT, thalamocortical tract.
TCT-LL%, IOT-LL%, and CST-LL%: percentage (voxels) of the TCT, IOT, and CST overlaid by the lesion.
Scatterplots presenting potential and observed recovery are provided in Figure 1. For motor recovery (1A), nonfitters are indicated as gray triangles. Scatterplots for somatosensory perception, and active and passive somatosensory processing are presented in Figures 1B, 1C, and 1D, respectively. For clarity, we highlighted the nonfitters group from the motor recovery scatterplot in the somatosensory scatterplots by means of the same gray triangles.
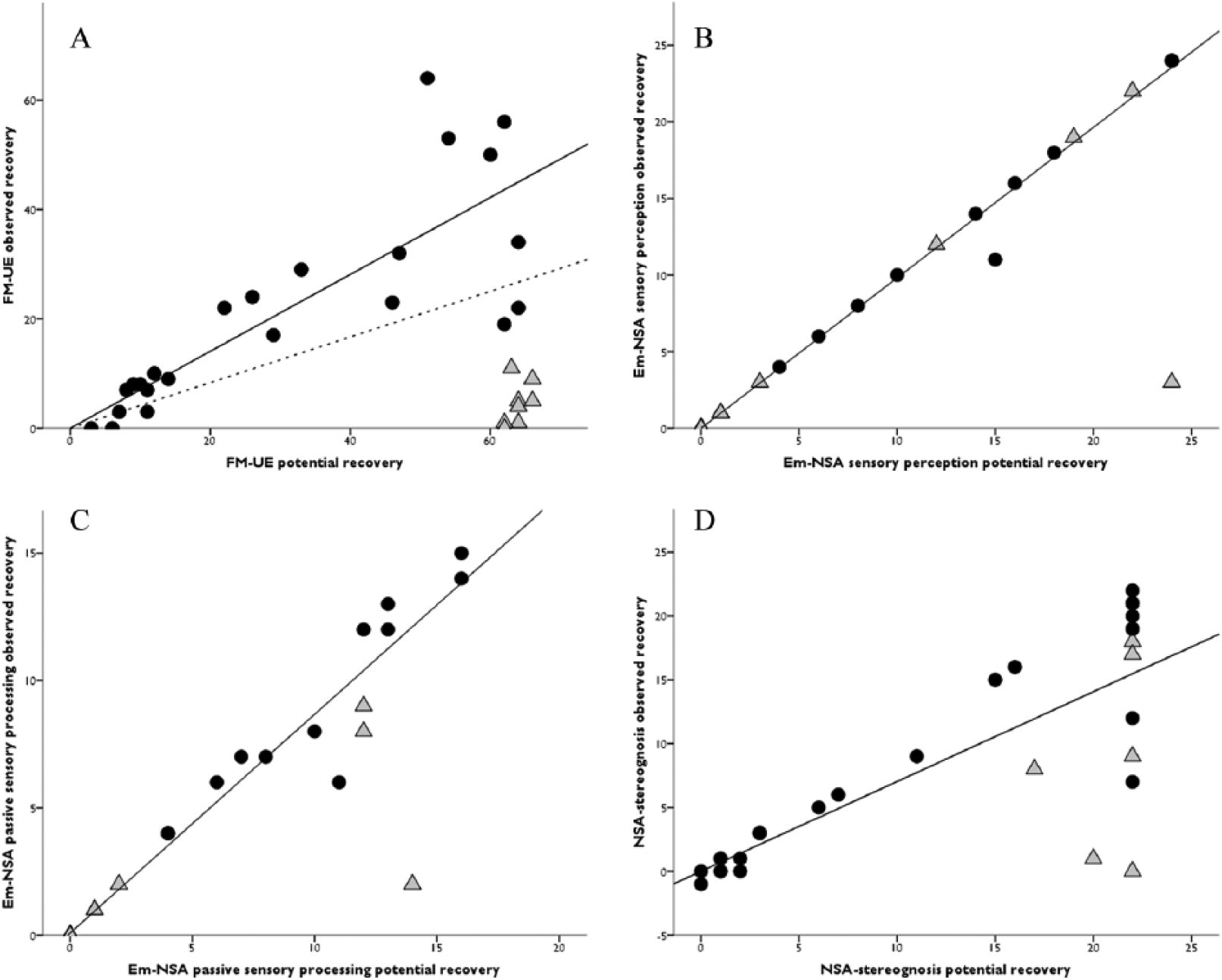
Scatterplots for motor (A), somatosensory perception (B), passive somatosensory processing (C), and active somatosensory processing recovery (D). Potential recovery: difference between maximum score and the score obtained at 4-7 days; observed recovery: difference between the score obtained at 6 months and at 4-7 days. Black dots: fitters for motor recovery; gray triangles: nonfitters for motor recovery. For motor impairment, nonfitters were excluded from regression analysis; for somatosensory perception and passive somatosensory processing, 1 outlier was detected and excluded, which was also a nonfitter for motor recovery. Both hierarchical clustering and standardized residuals failed to detect any outliers or nonfitters group for active somatosensory processing impairment. Solid line: linear regression analysis; dotted line: linear regression for the whole group for the motor model.
Table 2 presents the univariate linear regression analysis results for somatosensory and motor impairment. For fitters for motor recovery (n = 23, 72%), the proportional recovery model for observed recovery showed a β coefficient of 0.68 (95% CI: 0.48-0.87) for potential recovery with an explained variance (adjusted R2) of 70% (P < .001; Figure 1A). For the whole group for somatosensory perception, the model showed a β coefficient of 0.99 (95% CI: 0.96-1.01) with an explained variance of 99% (Figure 1B). For the whole group for passive somatosensory processing, the model showed a β coefficient of 0.86 (95% CI: 0.79-0.93) with an explained variance of 95% (P < .001; Figure 1C). And for the whole group for active somatosensory processing, the model showed a β coefficient of 0.69 (95% CI: 0.49-0.89) with an explained variance of 61% (P < .001; Figure 1D). Bootstrapping confirmed the robustness of regression models for passive somatosensory processing (standard error [SE] = 0.04; CI: 0.76-0.93; P < .01), for active somatosensory processing (SE = 0.09; CI: 0.51-0.86; P < .01), and for motor recovery (SE = 0.11; CI: 0.45-0.89; P < .01). All y-intercepts were almost 0 (−0.03 to 0.36).
Univariate Linear Regression Analysis Results for Somatosensory and Motor Impairment Recovery Models.
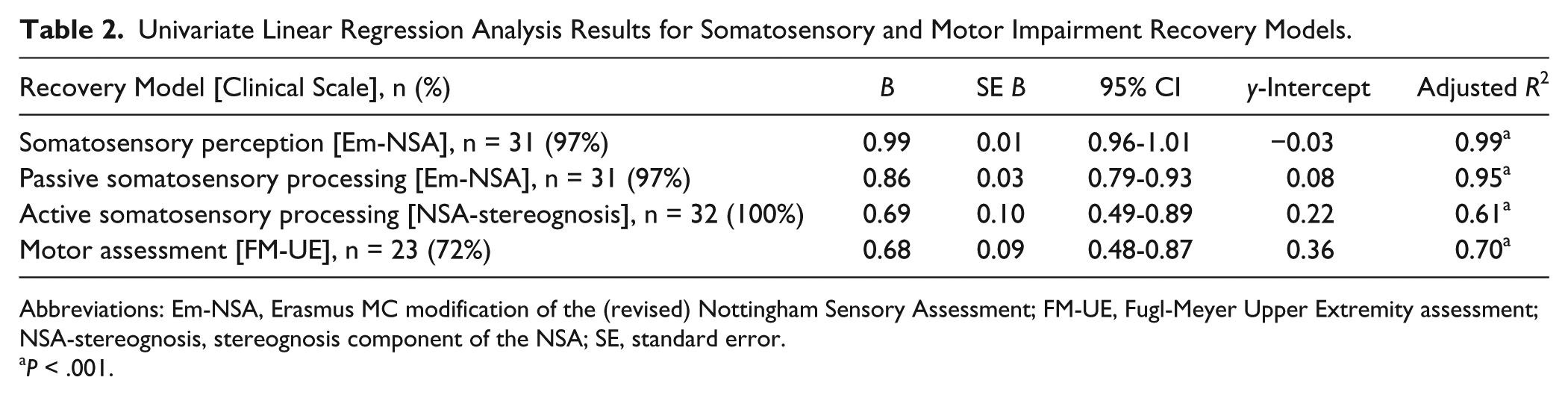
Abbreviations: Em-NSA, Erasmus MC modification of the (revised) Nottingham Sensory Assessment; FM-UE, Fugl-Meyer Upper Extremity assessment; NSA-stereognosis, stereognosis component of the NSA; SE, standard error.
P < .001.
Figure 2 presents the lesion overlay plots for our stroke sample (A); the TCT, IOT, and pyramidal tract display in healthy volunteers (B1); and a 3-dimensional visualization of these tracts (B2). LLs within sensorimotor tracts were compared between patients with normal and decreased somatosensory performance (Table 3). At 4 to 7 days, patients with impaired somatosensory perception showed significantly higher LL for all sensorimotor tracts (P < .00) in comparison with patients without impairment. For passive and active somatosensory processing, impaired patients showed a significantly higher LL in the TCT and IOT (P < .05) compared with patients without dysfunction.
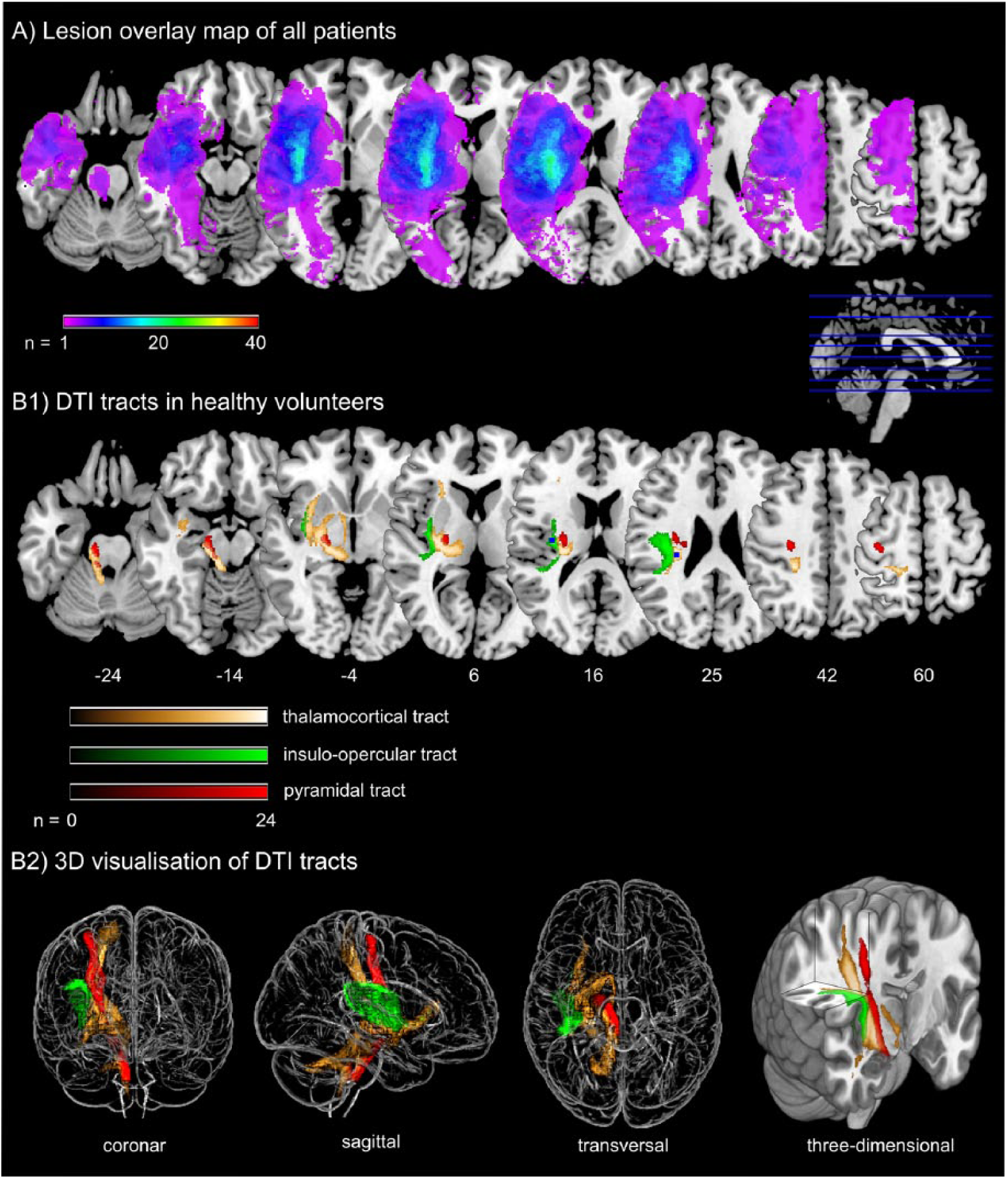
Lesion overlay plot from stroke patients and diffusion tensor imaging (DTI) reconstruction for healthy individuals: A. Individual lesion volumes were overlaid to show white matter regions most frequently involved. Maps are overlaid on a T1-template in MNI space 1 × 1 × 1 mm3. All lesions were flipped to the left hemisphere. MNI coordinates of each transverse section (z-axis) and a sagittal slice for visualization are given. Color scale for lesion overlay indicates the number of patients presenting with a lesion in this voxel. B1. Three different probabilistic fiber tracts of core sensorimotor brain pathways were taken from 24 healthy age-matched volunteers. Color scales for probabilistic fiber tracking indicate the number of volunteers presenting with the tract in this voxel. B2. “Glass brain” visualization and a half-split 3-dimensional model of the 3 tracts is shown.
Comparison for TCT, IOT, and CST LL Between Patients With Impaired and Normal Somatosensory Perception and Processing. a
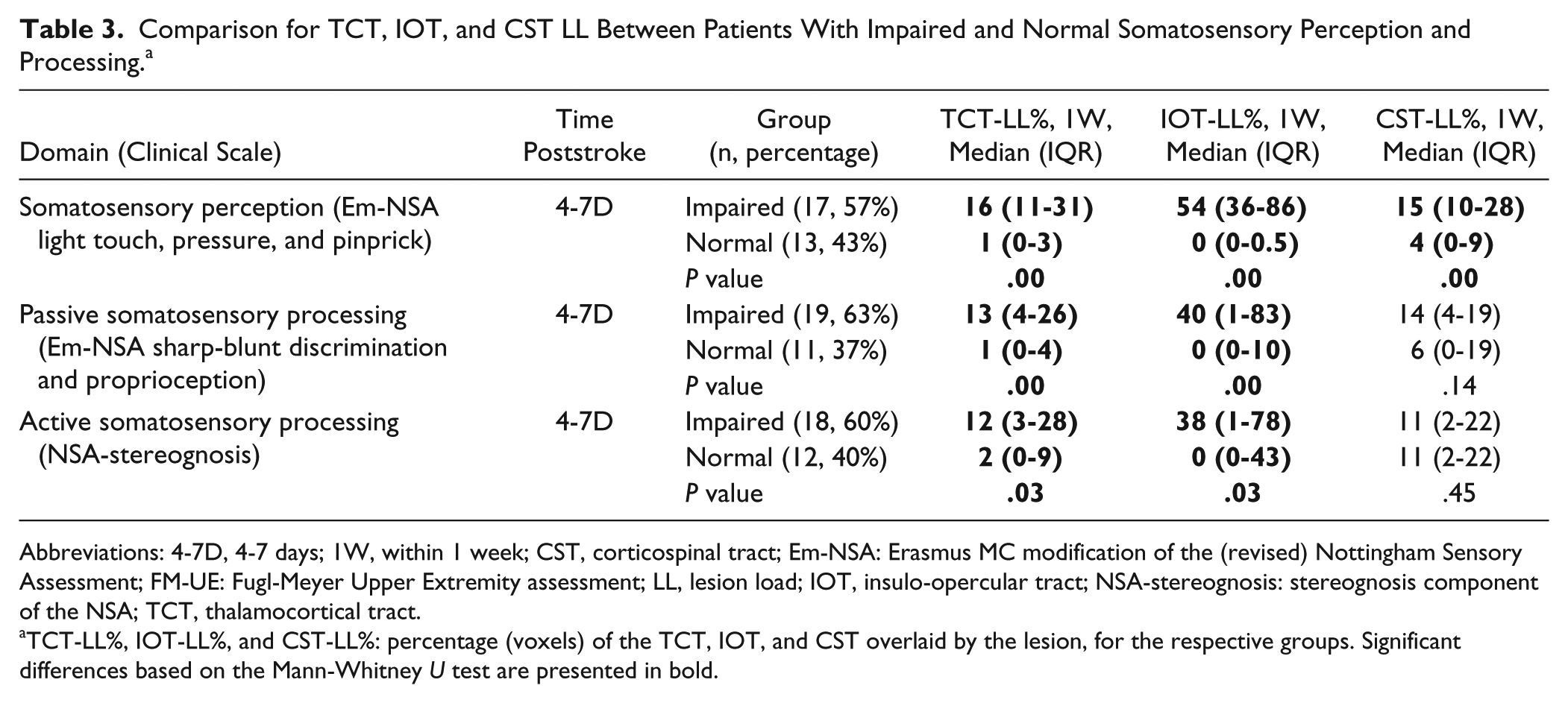
Abbreviations: 4-7D, 4-7 days; 1W, within 1 week; CST, corticospinal tract; Em-NSA: Erasmus MC modification of the (revised) Nottingham Sensory Assessment; FM-UE: Fugl-Meyer Upper Extremity assessment; LL, lesion load; IOT, insulo-opercular tract; NSA-stereognosis: stereognosis component of the NSA; TCT, thalamocortical tract.
TCT-LL%, IOT-LL%, and CST-LL%: percentage (voxels) of the TCT, IOT, and CST overlaid by the lesion, for the respective groups. Significant differences based on the Mann-Whitney U test are presented in bold.
Univariate analysis for passive somatosensory processing at 6 months demonstrated a significant relation with passive somatosensory processing score at 4 to 7 days (adjusted R2 = 0.27; P = .002), TCT-LL% (adjusted R2 = 0.21; P = .01), IOT-LL% (adjusted R2 = 0.11; P = .04), and CST-LL% (adjusted R2 = 0.18; P = .01). When combining all variables in a multivariate analysis, only passive somatosensory processing score at 4 to 7 days was retained as a significant variable. Univariate analysis for active somatosensory processing at 6 months demonstrated only a significant relation with active somatosensory processing score at 4 to 7 days (adjusted R2 = 0.23; P = .003). TCT-LL% (adjusted R2 = 0.05; P = .13), IOT-LL% (adjusted R2 = 0.00; P = .31), and CST-LL% (adjusted R2 = 0.03; P = .18) did not show a significant relation with active somatosensory outcome at 6 months.
Discussion
The aims of the present study were to assess whether the proportional recovery rule is also applicable for upper-limb somatosensory impairment after stroke and whether neural correlates of somatosensory impairment and outcome could be identified. Results from data collected at 4 to 7 days and 6 months showed that there is full recovery for somatosensory perception but proportional recovery for passive and active somatosensory processing. At 4 to 7 days, both the TCT and IOT showed a greater LL in impaired patients compared with patients without somatosensory dysfunction for somatosensory perception, and passive and active somatosensory processing. A significant relation was demonstrated for sensorimotor tract disruption at 4 to 7 days and passive somatosensory processing outcome at 6 months; however, there did not appear to be an additional explanatory value above the passive somatosensory processing score at 4 to 7 days when entered in a multivariate model.
We included analysis of proportional recovery for upper-limb motor impairment to demonstrate initial validity of our data. Indeed, our results confirm previous literature in this area because we found a proportional recovery of 68% (95% CI: 48%-87%). Again, as earlier identified, our study also showed that there is a group of patients presenting with substantially less recovery than predicted (9/32 = 28%) and, thus, not following the proportional motor recovery model. In comparison, Winters et al 7 recognized a group of 65 out of 211 patients (31%) having substantially less recovery than predicted. Thus, these results confirm the representative value of our sample to investigate upper-limb somatosensory recovery.
We classified somatosensory modalities into 3 domains; somatosensory perception, and passive and active somatosensory processing. For somatosensory perception—that is, the exteroceptive modalities—results showed essentially full recovery (99%, 95% CI including 1). Earlier results indeed showed that prevalence of the exteroceptive impairments in light touch, pressure, and pinprick only existed in 6% or fewer of patients at 6 months after stroke. 15
For passive somatosensory processing, we observed proportional recovery (86%, 95% CI: 79%-93%). Passive somatosensory processing comprised sharp/blunt discrimination and proprioception. In earlier work, 15 proprioceptive deficits only existed in 3% of patients at 6 months, whereas sharp/blunt discrimination impairment was seen in 22% of the sample. Thus, our proportional recovery model could largely be steered by a sharp/blunt discrimination deficit. Sharp/blunt discrimination and proprioception were grouped together because both require passive detection of somatosensory input and discrimination of this input (ie, sharp or blunt stimulus, or position or movement sense in one or the other direction in case of proprioception). This is different from the exteroceptive modalities where only awareness of sensory input is required (ie, whether the touch, pressure, or pinprick stimulus is felt). In comparison to proportional recovery models for other domains, there is no nonfitter group identified, although we recognized 1 outlier. Proportional recovery for passive somatosensory processing might indicate that a biological repair from a somatosensory impairment is reflected in a clinical change, although it should be noted that the level of proportional recovery (86%) is relatively higher in comparison to other domains. Confirmation of our findings is, therefore, required.
For active somatosensory processing, measured through stereognosis, we also observed proportional recovery (69%, 95% CI: 49%-89%). Stereognosis was classified as a different somatosensory domain because it requires detection of somatosensory input and discrimination as well as an active (motor) component when manipulating the objects in the hand. It should be noted that for patients in our sample who were unable to move the affected hand at all, the assessor passively moved the hand when testing stereognosis. The level of proportional recovery for active somatosensory processing (69%) is well in line with the reported rate of proportional recovery for visuospatial neglect, 11 aphasia,11,14 and lower-limb motor impairment. 10 The proportional recovery for active somatosensory processing might (partly) be determined by the motor component required and, thus, by motor impairment recovery. Future research should attempt to disentangle the sensorimotor coupling in upper-limb recovery after stroke.
Neural correlates of impairments in the different somatosensory domains at 4 to 7 days demonstrated that greater LL in the TCT and IOT were found in patients with impairment in comparison to patients without impairment. In earlier work using voxel-based symptom-mapping analysis, 27 voxels with a significant association to somatosensory impairments were grouped in 2 core brain regions: the sensory component of the superior thalamic radiation and the parietal operculum close to the insular cortex. Furthermore, in our study, for patients with somatosensory perception impairment, a greater CST LL was discovered than in patients without impairment. This is somehow surprising, because in line with the argument raised above, one might expect a stronger relation between disruption of the CST and active somatosensory processing because of the latter requiring motor activity. However, other motor tracts such as the reticulospinal pathway might be more involved in the motor component of active somatosensory processing. As for the link between motor tract disruption and somatosensory perception impairment, this might be steered by the lesion sites and sizes in our sample, and future studies should unravel this finding.
When relating somatosensory impairment and sensorimotor tract disruption early after stroke with sensorimotor outcome at 6 months, LL of the sensorimotor tracts was significantly related for passive sensorimotor processing only. No model was evaluated for somatosensory perception because there is essentially full recovery, with nearly normal scores at 6 months. In multivariate models for passive and active somatosensory processing at 6 months, only the clinical processing score early after stroke was retained, with an explained variance of 23% to 27%. This appears in line with motor prognostic models where imaging variables have limited predictive value. For proportional motor recovery models, imaging parameters have prognostic value, determining nonfitters. However, we did not find nonfitter groups for proportional somatosensory processing recovery. The limited explained variance should encourage future research in understanding the early determinants of somatosensory outcome.
Some limitations of our study need to be considered. The sample size was limited; nevertheless, we obtained robust models, evaluated with bootstrapping analysis, and our results confirmed previous literature concerning proportional motor recovery.3,6,7 Nonetheless, the present results are based on recruitment from 2 centers in 1 country. Therefore, future, large international cohort studies are needed to confirm the proportional recovery model in the somatosensory domains, focusing on patients with somatosensory impairment because our sample included also participants without somatosensory deficit. Furthermore, we analyzed both ischemic and hemorrhagic stroke patients and recognize that previous studies included only ischemic patients. However, Stinear et al 3 recently generalized the proportional recovery model in a large cohort of both ischemic and hemorrhagic patients.
Conclusions
The present study is the first to demonstrate full recovery for upper-limb somatosensory perception impairment but proportional recovery for passive and active somatosensory processing impairment in the upper limb after stroke. Patients with somatosensory impairment in the very early phase after stroke show greater LL in both the TCT and IOT. Disruption of sensorimotor tracts early after stroke was significantly related with passive somatosensory processing outcome at 6 months but does not appear to provide additional predictive value above passive somatosensory processing measured early.
Footnotes
Authors’ Note
Leonardo Boccuni and Sarah Meyer are joint first authors.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Verheyden reports grants from Promobilia Foundation, Sweden; grants from Foundation Van Goethem-Brichant, Belgium; and grants from Research Foundation Flanders (FWO), Belgium, during the conduct of the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants of the Promobilia Foundation, Sweden (Grant Number 15060); the Foundation Van Goethem-Brichant, Belgium; and Research Foundation Flanders (FWO), Belgium.
