Abstract
Introduction
Amblyopia is a neurodevelopmental disorder of vision that occurs when the visual cortex receives decorrelated inputs from each eye during infancy. It typically affects one eye and is associated with a range of visual deficits, including poor visual acuity, 1 reduced contrast sensitivity, 2 and impaired binocular function. 3 Traditionally, amblyopia has been considered to be untreatable in adulthood, but current understanding of neural plasticity in the adult visual cortex challenges this idea. 4
There is increasing evidence that the monocular and binocular deficits in amblyopia are associated with an active suppression of inputs from the amblyopic eye within the visual cortex.5-8 For example, a number of neurophysiological 9 and neuroimaging studies10-12 have demonstrated that amblyopic eye viewing generates significantly less cortical activation in the visual cortex than fellow eye viewing. This is consistent with active suppression or inhibition of neural function, 13 which has been linked to GABAergic neural circuitry.14,15 Therefore, GABA (γ-aminobutyric acid)–mediated inhibition may represent a viable target for interventions aiming to improve vision in patients with amblyopia.
In this study, we aimed to assess whether transcranial direct current stimulation (tDCS), could modulate visual function in adults with amblyopia. tDCS involves the delivery of a weak direct electrical current to a targeted cortical region using sponge-coated electrodes placed on the head. The effects of tDCS are polarity dependent, whereby anodal tDCS (a-tDCS) tends to increase and cathodal tDCS (c-tDCS) tends to decrease neural excitability.16-21 In addition, a-tDCS reduces GABA and increases glutamate concentration in the human motor cortex 22 and greater reductions in GABA correlate with enhanced learning of motor tasks. 23 In contrast, c-tDCS reduces both GABA and glutamate 22 and these changes are not linked to enhanced learning. 24 In the visual cortex, a-tDCS, but not c-tDCS, can reduce psychophysically measured surround suppression, which is thought to be mediated by GABA. 25 On balance, a-tDCS appears to reduce the functional effects of GABA-mediated inhibition in the human brain whereas c-tDCS does not. We therefore hypothesized that a-tDCS would improve visual function in adults with amblyopia by reducing suppression of cortical inputs from the amblyopic eye.
To test this hypothesis, we measured monocular contrast sensitivity in each eye before, during, and after either a-tDCS or c-tDCS in a group of adult patients with amblyopia. We also used functional magnetic resonance imaging (fMRI) to measure the effects of a-tDCS on the activation of the visual cortex in a subset of patients. We reasoned that if a-tDCS reduced cortical inhibition associated with the amblyopic eye, then the stimulation would reduce the cortical response asymmetry in favor of the fellow eye that is characteristic of amblyopia.10,11
Methods
Participants
Thirteen adult participants with amblyopia (interocular acuity difference of at least 0.2 log MAR [minimum angle of resolution] with no organic cause) and no contraindications for fMRI or tDCS took part in the behavioral study (Table 1). One participant withdrew because of discomfort from the electrodes. Five available participants also completed a series of fMRI scans. Best refractive correction was worn throughout the study. The study was approved by the regional ethics committee and all study protocols were in accordance with the Declaration of Helsinki.
Participants’ Characteristics.
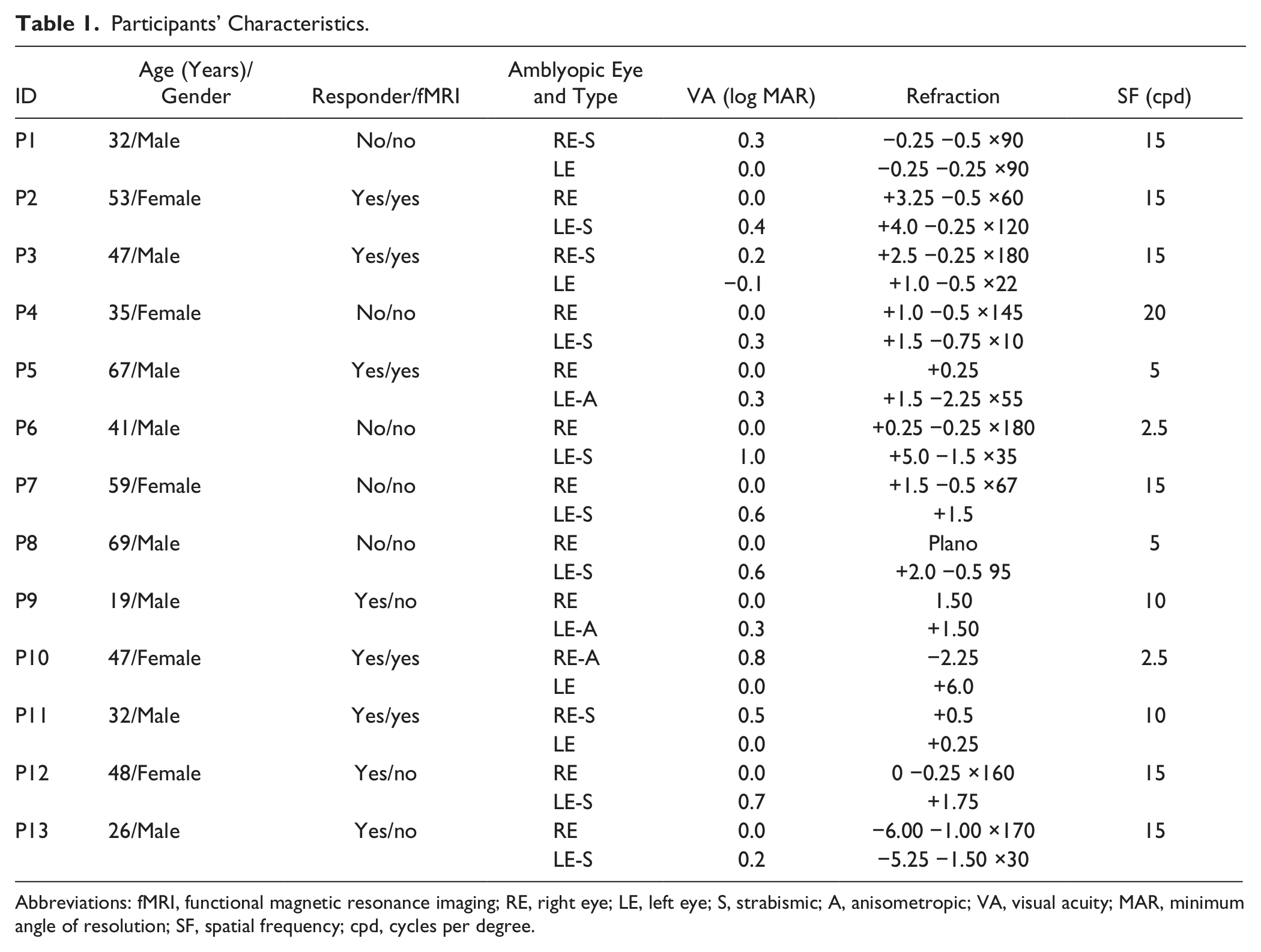
Abbreviations: fMRI, functional magnetic resonance imaging; RE, right eye; LE, left eye; S, strabismic; A, anisometropic; VA, visual acuity; MAR, minimum angle of resolution; SF, spatial frequency; cpd, cycles per degree.
Procedure
Contrast Sensitivity Measurements
Contrast sensitivity was assessed using a Gabor patch (radius = 1.3°, σ = 1.0°), presented on a uniform gray background (47 cd/m2) for 500 ms within a Gaussian temporal envelope (100 ms ramp up and 100 ms ramp down). Stimuli were generated using Psykinematix software 26 and presented on a Trinitron G520 CRT monitor. The viewing distance was 310 cm and a tight-fitting opaque patch was worn over one eye.
The experimental design is outlined in Figure 1. A 2-alternative forced choice paradigm (orientation discrimination: vertical vs horizontal) and a standard 1-up-2-down adaptive staircase procedure (step size = 25% before the first reversal, 7.5% increments, and 15% decrements after the first reversal) 27 was employed. 28 Contrast thresholds were calculated as the mean of the last 4 reversals out of a total of 6 reversals. In the first session, contrast was fixed at 40% and each participant’s amblyopic eye cutoff spatial frequency (SF) was measured. SF was then fixed and contrast sensitivity measured. The SF used for all subsequent sessions was within 1.5 octaves of the cutoff and allowed for stable contrast thresholds. Participants completed at least 2 hours of task familiarization prior to the tDCS measurement sessions. During tDCS sessions, which were separated by at least 3 days, contrast sensitivity for each eye was measured directly before, during, after, and 30 minutes after stimulation. A-tDCS and c-tDCS testing sessions were counterbalanced across participants.
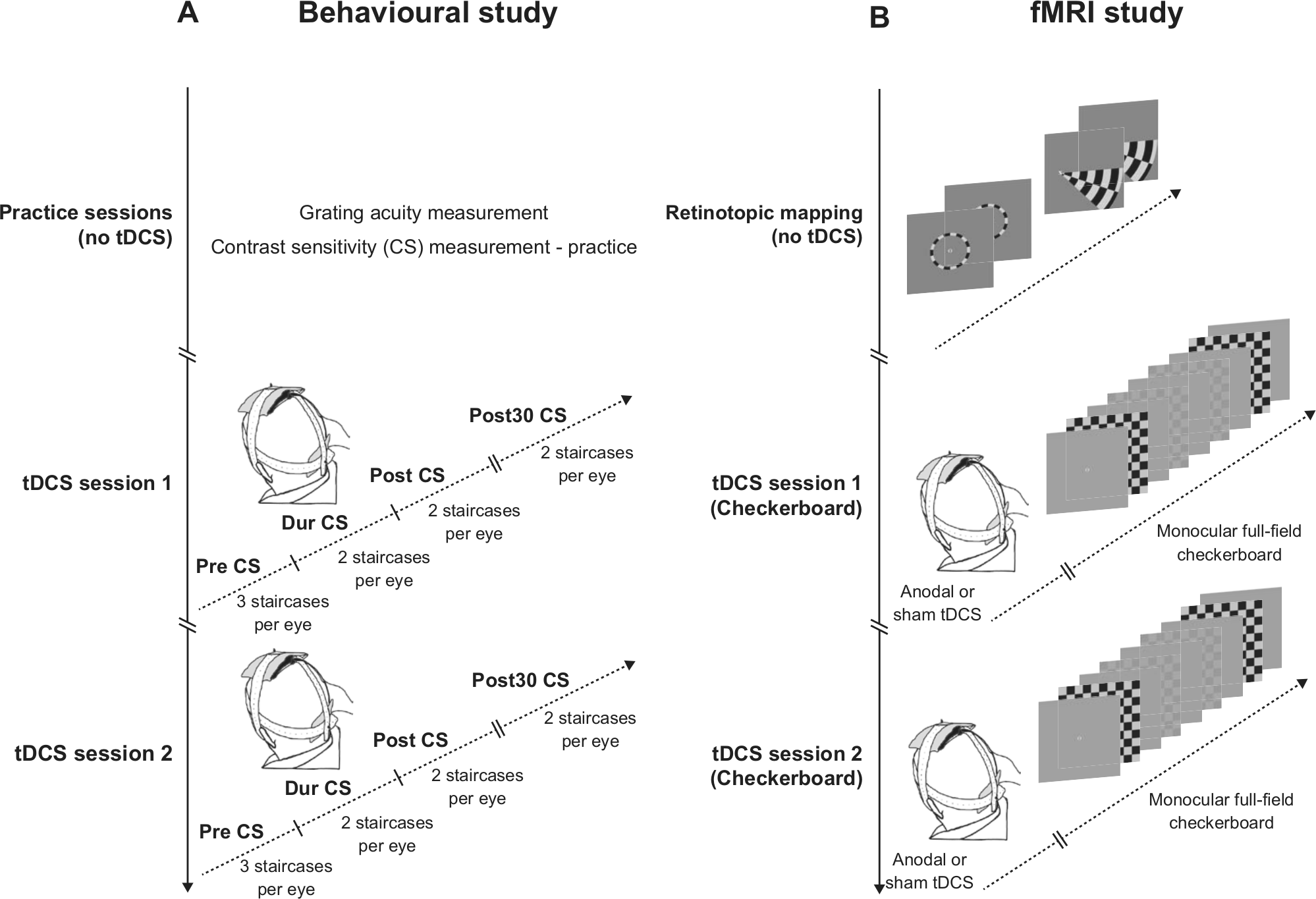
Experimental design. Panel A represents the behavioral experiment. Before commencing the transcranial direct current stimulation (tDCS) sessions, each participant completed 2 practice sessions. During these sessions participants’ grating acuity was also measured. In the tDCS sessions, contrast sensitivity (CS) of the amblyopic and fellow eye was measured before (Pre), during (Dur), directly after (Post), and 30 minutes after (Post30) anodal-tDCS (a-tDCS) and cathodal-tDCS (c-tDCS). The order of eyes (amblyopic first vs fellow first) and tDCS conditions (a-tDCS first vs c-tDCS first) was counterbalanced across participants. Panel B represents the functional magnetic resonance imaging (fMRI) experiment. In the first fMRI session, each participant was retinotopically mapped. In the second and third fMRI session, a-tDCS or sham tDCS (s-tDCS) was administered directly before fMRI. Counterphasing checkerboard stimuli were presented monocularly during these scanning sessions at 2 contrast levels.
Functional Magnetic Resonance Imaging
Five participants completed 3 fMRI scanning sessions. The first session consisted of retinotopic mapping using standard wedge and ring protocols. 29 Stimuli were presented monocularly and each eye was mapped separately. Borders of retinotopic areas and corresponding regions of interest were defined using an averaged map (left and right eye) in each individual participant. There was good agreement between the maps generated by each eye as has previously been reported. 29
The second and third scanning sessions assessed the effect of a-tDCS versus sham tDCS (s-tDCS) on the response of the visual cortex to inputs from the amblyopic versus fellow fixing eye. S-tDCS was used as a control as our contrast sensitivity data indicated that c-tDCS may have also altered the ratio of activation between the two eyes by reducing contrast sensitivity in the fellow eye. During fMRI, participants performed a reaction time task where they detected a change in the fixation point (“X” or “O”). This task was unrelated to the stimuli used to evoke blood oxygen level–dependent (BOLD) responses in the visual cortex. Task performance was constant across scans for all participants. One eye was occluded with a tight fitting eye patch during scanning and the viewing eye was alternated from scan to scan. tDCS was administered in a room next to the scanner and fMRI data acquisition began within 4 minutes of tDCS.
During the post tDCS scans, participants viewed a temporally modulated (8 Hz) broadband checkerboard stimulus (spatial frequency 0.25 cpd [cycles per degree], 18° × 16°) at 5% or 50% contrast. A block design was employed whereby each checkerboard was displayed for 10 TRs (20 seconds) separated by 10 TRs of mean luminance blank fixation. Each contrast was shown twice within each scan and there were 4 scans per session per eye.
Stimuli were presented on a MRI-compatible TFT-LCD screen viewed through a coil-mounted mirror. Scanning was performed on a 3.0 T Philips Achieva scanner equipped with an 8-channel head coil. Functional data were acquired using a T2*-weighted gradient echo EPI sequence (
Transcranial Direct Current Stimulation
Transcranial direct current stimulation was delivered using a portable stimulator (Chattanooga Ionto, Vista, CA) via a pair of sponge-covered rubber electrodes (Speds Medica S.r.l., Italy, stimulating electrode 72 × 60 mm, reference electrode 115 × 95 mm). A large reference electrode was used to reduce the current density and render the electrode inert. 30 The stimulating electrode was placed over Oz and the reference over Cz. The size and position of the electrodes were adopted from previous visual cortex tDCS studies.20,25,31
Transcranial direct current stimulation was ramped up over 31 seconds to an intensity of 2 mA and then kept constant for 15 minutes. In the sham condition used for the fMRI measurements, the current was ramped up and then turned off. 32 Sham stimulation was always anodal. Participants were masked as to whether tDCS was anodal, cathodal, or sham. The experimenter was not masked. Patients were not able to discriminate a-tDCS from c-tDCS but some patients were able to discriminate real tDCS from sham.
Data Analysis
Contrast Sensitivity
Contrast detection thresholds were converted to log contrast sensitivity and measurements made during, after, and 30 minutes after tDCS were normalized to the within-session baseline (the pre-tDCS measurement) by subtraction. A repeated-measures analysis of variance (ANOVA) with a factor of stimulation (anodal vs cathodal), eye (amblyopic vs fellow fixing), and time (during vs post vs post 30 minutes) was conducted on the normalized data. Post hoc 1-sample and 2-sample paired
Therapeutic effects of tDCS can vary significantly between individuals.33
-35 We therefore conducted a secondary analysis to assess whether our data set contained 2 distinct groups of patients; “responders” whose amblyopic eye contrast sensitivity improved and “nonresponders” who experienced no change or a reduction in amblyopic eye contrast sensitivity. To this end, we conducted a
Functional Magnetic Resonance Imaging data
Magnetic resonance imaging data analysis was conducted using BrainVoyager (Brain Innovation BV, Maastricht, Netherlands). Functional data were highpass filtered, motion corrected, and aligned to the anatomical data in Talairach space
36
using subroutines within BrainVoyager. Visual area boundaries were identified from the retinotopic mapping data following established methodology.
37
Regions of interest were generated for each visual area (V1, V2, V3, V3a, and V4) for each hemisphere, for each participant and were restricted to voxels that were responsive to the checkerboard stimuli (general linear model analysis, false discovery rate–corrected
To evaluate the effects of a-tDCS, we calculated the interocular BOLD ratio expressed as
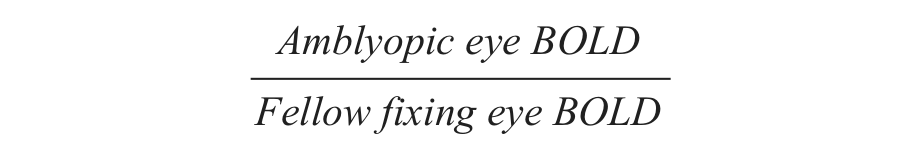
for each contrast for each ROI for each scanning condition. Values lower than 1 result from stronger functional activation for fellow fixing eye viewing. Conversely, values higher than 1 result from stronger functional activation for amblyopic eye viewing. The BOLD ratios were then compared between the a-tDCS and s-tDCS scanning sessions using a repeated measures ANOVA with factors of stimulation (anodal vs sham) and contrast (5% and 50%), conducted separately for each visual area (V1, V2, V3, V3a, and V4).
Results
There were no adverse effects during or following tDCS.
The Effects of tDCS on Contrast Sensitivity
There was a main effect of tDCS polarity on contrast sensitivity (
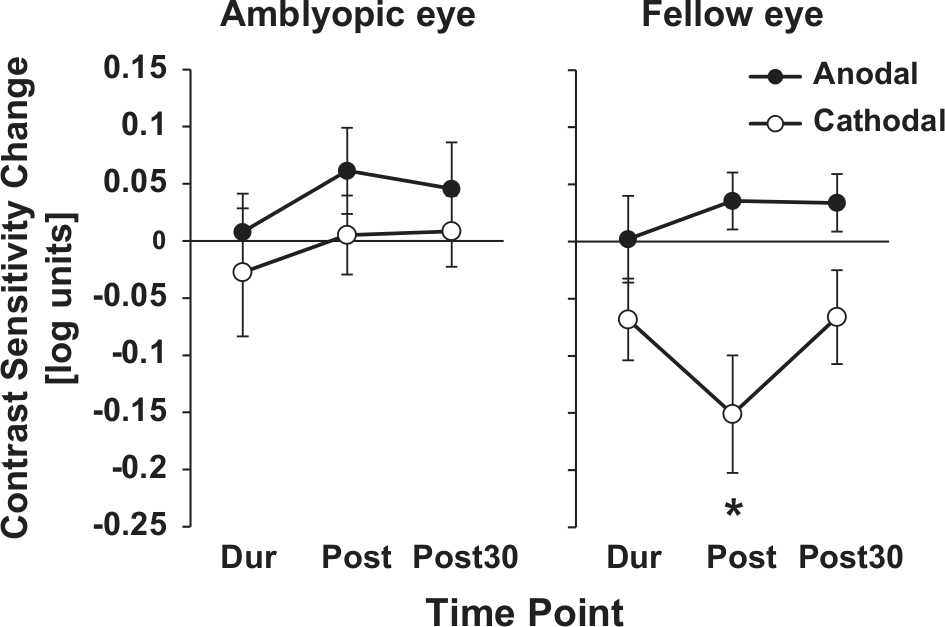
The effects of anodal (a-tDCS) and cathodal (c-tDCS) transcranial direct current stimulation on contrast sensitivity across the whole data set (N = 13). Contrast sensitivity data normalized to baseline for the amblyopic and fellow fixing eye. *
At a whole group level, a-tDCS did not induce reliable changes from baseline for either eye. On the other hand, c-tDCS decreased fellow fixing eye contrast sensitivity relative to baseline immediately after tDCS (
A
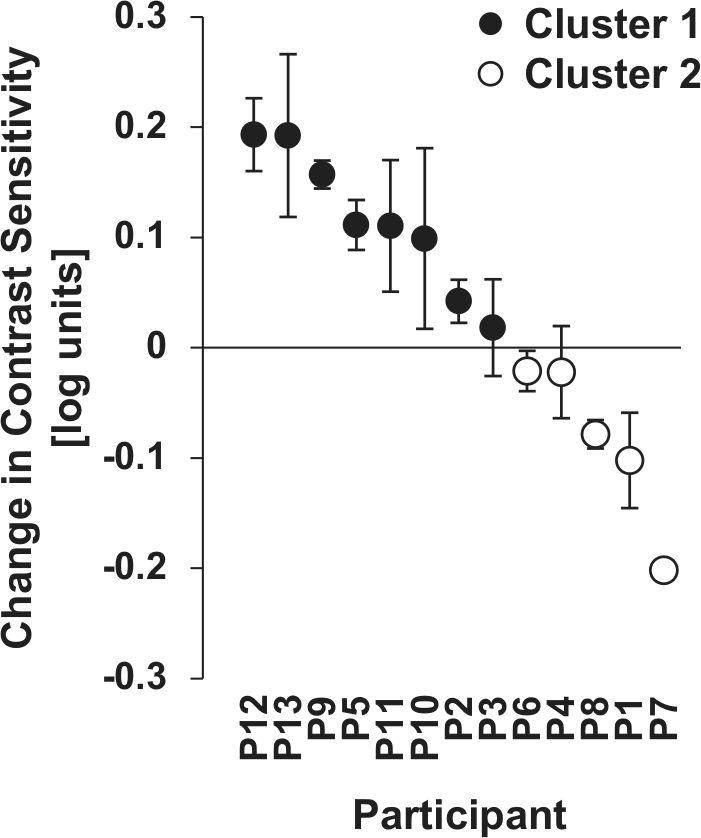
The results of a
An ANOVA conducted on the data for cluster 1, which represented “responders,” showed a significant main effect of stimulation (
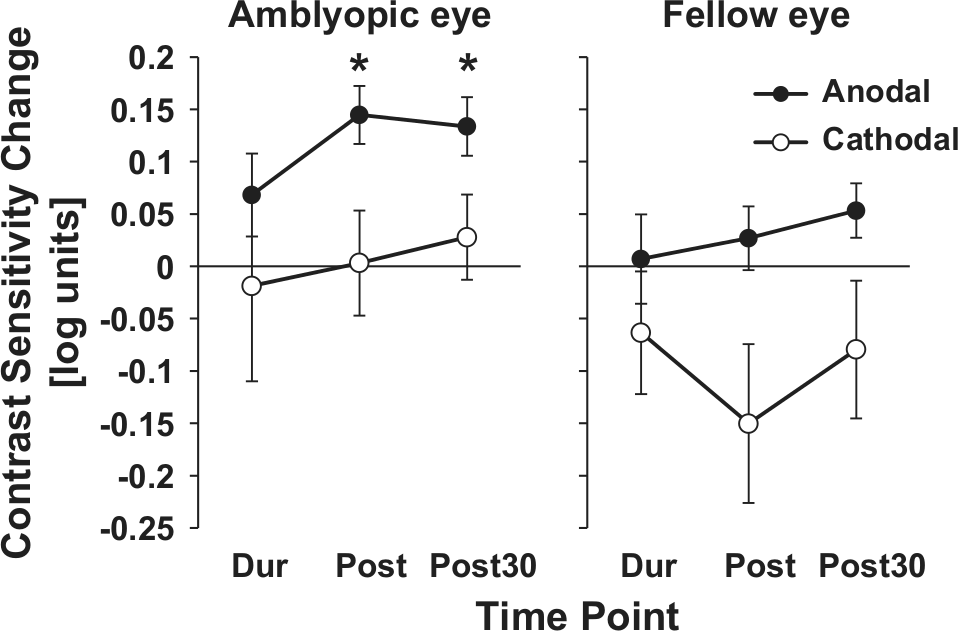
The effects of anodal (a-tDCS) and cathodal (c-tDCS) transcranial direct current stimulation on contrast sensitivity for “responders” (N = 8). Contrast sensitivity data normalized to baseline for the amblyopic and fellow fixing eye. *
An ANOVA conducted on the data from “nonresponders” in cluster 2 revealed a significant interaction between eye and stimulation (
The Effects of a-tDCS on Visual Cortex Activation
Five participants whose contrast sensitivity improved following a-tDCS completed fMRI measurements of visual cortex activation after both a-tDCS and s-tDCS (see Table 1 for participant details).
After s-tDCS, viewing through the fellow fixing eye resulted in significantly higher activation within both striate and extrastriate visual cortex than viewing through the amblyopic eye in agreement with previous studies10,11 (Figure 5). This bias toward stronger activation for fellow eye viewing was reduced by a-tDCS indicating that a-tDCS selectively influenced the cortical response to amblyopic eye inputs. Group data are shown in Figure 5 projected onto flattened representations of the occipital lobes to illustrate this effect. Regions that showed a significantly greater functional response to fellow eye viewing (collapsed across stimulus contrast) are shown in orange/yellow. There are large regions within striate and extrastriate visual cortex that show a significantly stronger response to fellow eye viewing after s-tDCS (Figure 5, top row). This bias in activation in favor of the fellow eye is clearly reduced after a-tDCS (Figure 5, middle row). The ratios of BOLD activation between the fellow and amblyopic eyes for individual participants using participant-specific ROIs are shown in Figure 5. A repeated-measures ANOVA conducted on the BOLD ratios revealed a significant effect of stimulation in visual areas V2 (
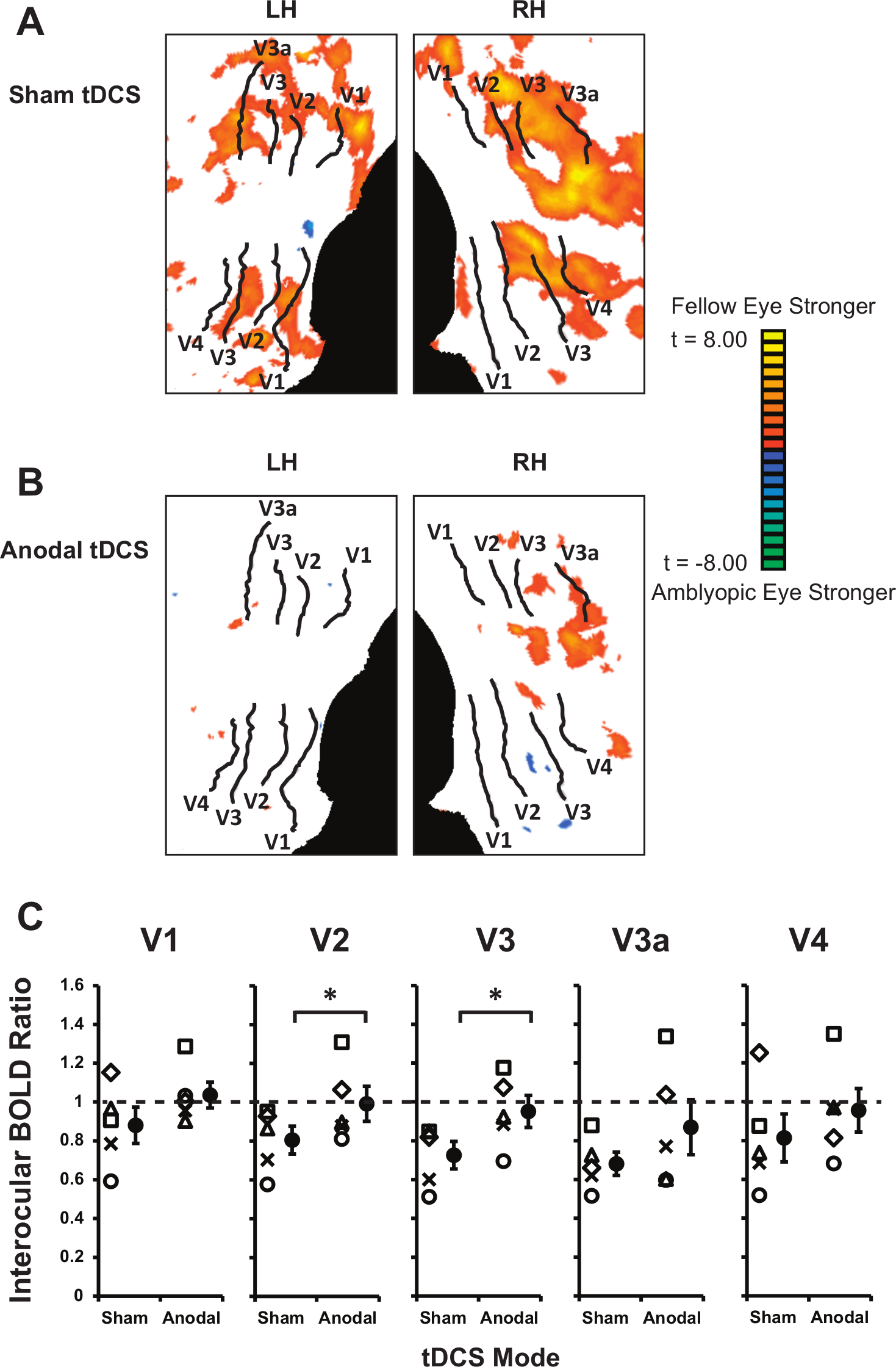
The effect of anodal transcranial direct current stimulation (a-tDCS) on cortical activation for fellow eye versus amblyopic eye viewing. Panels A and B represent a flattened visual cortex with boundaries of individual retinotopic areas for one representative participant (P2) marked on the left (LH) and right hemispheres (RH) for illustrative purposes only. The results of a group general linear model analysis comparing activation in response to fellow versus amblyopic eye viewing have been projected onto the cortical surface. “Hot” colors indicate a greater cortical response to the fellow eye than the amblyopic eye. The top panel represents functional magnetic resonance imaging (fMRI) data following sham tDCS (s-tDCS) and the lower panel following a-tDCS. The bias in cortical activation for fellow eye viewing is clearly reduced by a-tDCS. Panel C shows the interocular BOLD (blood oxygen level–dependent) ratios for individual subjects and the average BOLD ratio (solid symbols) for each visual area following sham and a-tDCS. A BOLD ratio <1 implies stronger activation for the fellow eye and a ratio >1 implies stronger activation for the amblyopic eye. *
Discussion
The aim of this study was to test the hypothesis that a-tDCS of the visual cortex would improve contrast sensitivity in adult patients with amblyopia by enhancing the cortical response to inputs from the amblyopic eye. We did not find a reliable effect of a-tDCS on contrast sensitivity for either the amblyopic or fellow fixing eye across all 13 participants. However, there was an effect of c-tDCS whereby contrast sensitivity in the fellow eye was reduced relative to baseline directly after tDCS. There were no measureable effects of c-tDCS on amblyopic eye contrast sensitivity. Subsequent analyses identified groups of responders and nonresponders to a-tDCS whereby responders showed a significant improvement in amblyopic eye contrast sensitivity following a-tDCS whereas nonresponders did not. fMRI measurements made on a group of 5 responders indicated that, in these participants, a-tDCS acted to equalize the response of the visual cortex to inputs from each eye.
Responders and Nonresponders to a-tDCS
Our results are in line with previous reports indicating that the therapeutic outcomes of noninvasive brain stimulation techniques differ across individual patients.28,34,35 In some studies, these differences are related to particular clinical characteristics such as the depth of amblyopia 28 or the severity of functional impairment after stroke. 35 However, in the present study we did not find any systematic differences in the clinical characteristics of responders and nonresponders.
There are, however, factors that were not assessed in the present study that may influence the effect of tDCS. It has been reported that BDNF polymorphisms may alter the response to tDCS whereby Met carriers may be less responsive to a-tDCS than Val/Val carriers. 39 In addition, individual skull and brain anatomy can affect the distribution of the electric current generated by tDCS,40,41 which may influence the effect of tDCS on cortical activation. 42 Other factors such as variations in neural connectivity 33 may also influence an individual’s response to tDCS.
Contrast Sensitivity Improvements in “Responders”
We found that a-tDCS could improve amblyopic eye contrast sensitivity in a subset of patients. It has been suggested that the visual deficits associated with amblyopia are the result of chronic suppression of inputs from the amblyopic eye to the visual cortex and that this suppression can be reduced by inhibiting GABA.14,15 As a-tDCS may act to reduce GABA-mediated suppression,22,23,25 the improved contrast sensitivity we observed in some patients could be a behavioral signature of reduced suppression within the amblyopic visual cortex.
Functional MRI data collected from 5 responders provide partial support for this interpretation of our results as a-tDCS acted to reduce the well-established bias of cortical activation in favor of inputs from the fellow eye.10,11 This is consistent with reduced inhibition of amblyopic inputs to the cortex.
Interestingly, the “balancing” of cortical activation between the eyes induced by a-tDCS was most pronounced for extrastriate areas V2 and V3. There is evidence that the effects of suppressive cortical interactions within the amblyopic visual cortex become amplified in extrastriate areas. 43 For example, Bi et al 44 found that interocular suppression was more pronounced in V2 than V1 of amblyopic monkeys. Furthermore, suppression within V2 was the strongest neural correlate of the visual deficits experienced by these animals. Similarly, extrastriate areas have been found to show greater deficits in cortical activation for amblyopic eye viewing in humans. 45 Therefore, the more pronounced effects of a-tDCS we found in V2 and V3 may reflect the presence of more extensive suppressive interactions in these areas.
The finding that c-tDCS did not induce improvements in amblyopic eye function within the group of “responders” is in agreement with previous work demonstrating that c-tDCS does not induce behavioural effects that are consistent with reduced GABA-mediated inhibition.24,25 The available data suggest that while c-tDCS does reduce GABA concentration, glutamate concentration is also reduced. 22 This may induce a reduction in excitation that moderates any effects of reduced inhibition.
Cathodal tDCS and Fellow Eye Contrast Sensitivity
Cathodal tDCS resulted in a reliable decrease in contrast sensitivity for fellow eye viewing. This finding is in agreement with previous studies showing reduced visuocortical excitability following c-tDCS.18 -20 However, not all studies report reduced excitability or behavioral performance following c-tDCS of the visual cortex.25,31,46 -48
In the present study, c-tDCS did not affect contrast sensitivity of the amblyopic eye. A possible explanation is that neural inputs from the amblyopic eye are already suppressed14,15 and therefore there is no scope for a further decrease in activity. This is in agreement with the current consensus that the effects of brain stimulation are strongly affected by the state of the stimulated neuronal population whereby neurons with recent history of suppression preferentially respond to excitatory stimulation.49 -51
Brain Stimulation and Amblyopia
It has previously been shown that both 1 Hz and 10 Hz repetitive transcranial magnetic stimulation (rTMS) of the primary visual cortex can improve contrast sensitivity in adult amblyopic eyes.7,28 rTMS may also act to balance the response of the visual cortex to inputs from the amblyopic and fellow eye7,28; however, it is difficult to directly compare the effects of rTMS and tDCS as there are a number of fundamental differences between the techniques. These include distinct mechanisms of action 52 and the focality of the stimulation effects. What is common to both the tDCS results reported here and the previous rTMS results is that improvements in amblyopic function are observed after short periods of stimulation. This suggests that both techniques act to unmask a latent capacity for improved neural processing of information from the amblyopic eye, which, presumably, is suppressed under normal viewing conditions.
General Conclusion
Anodal tDCS transiently improved contrast sensitivity in a subset of adults with amblyopia and equated the cortical response to inputs from the amblyopic and fellow eyes. It is our contention that this effect is mediated by a decrease in GABAergic inhibition of inputs from the amblyopic eye within the visual cortex.25,53 These results suggest that a-tDCS may be of use in the treatment in amblyopia in a subset of adult patients either alone or in combination with other interventions, as has recently been demonstrated for patients with hemianopia following stroke. 54 However, the fact that some participants showed an improvement in amblyopic eye contrast sensitivity following a-tDCS whereas others did not demonstrates that noninvasive brain stimulation may not be a “one-size-fits-all” solution. 35 To optimize the potential clinical use of tDCS in the treatment of amblyopia, we propose that the effect of tDCS on contrast sensitivity could be used to identify “responders” prior to commencing treatment.33,34
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was supported by grants from the Health Research Council of New Zealand, the Auckland Medical Research Foundation, and the University of Auckland to BT.
