Abstract
Keywords
Introduction
From 55% to 75% of stroke survivors have a paretic arm 1 that may improve primarily within 6 months. 2 Further intensive training can lead to improved motor function and associated cortical reorganization. 3 Yet these training programs often make use of expensive apparatus 4 or require an intensive one-on-one interaction with a therapist, 5 which hinders implementation on a large scale. Mirror therapy may be a suitable alternative. Designed by Ramachandran et al, 6 mirror therapy was originally developed to diminish phantom limb pain in amputees. The reflection of the unimpaired arm in a mirror gave patients the sensation of having 2 moving arms, which led to a reduction in pain. In 1999, Altschuler et al 7 introduced mirror therapy for recovery of hemiparesis following stroke. In a crossover design, they showed that motor performance of chronic stroke patients improved. Although several additional studies were small and often not well controlled, 8-10 2 recent, high-quality, randomized controlled trials have also reported mirror therapy to improve motor function in patients with subacute 11 and acute 12 stroke.
Despite the encouraging clinical results, little is known about the underlying mechanisms of mirror therapy. Ramachandran et al 6 referred to a “learned paralysis” in the brain, which could possibly be “unlearned” as a result of the mirror illusion. 13 Other studies have attributed the positive effects of mirror therapy in stroke to motor imagery 8 or the mirror neuron system. 11 The prevailing idea is that observing mirrored movements causes additional neural activity in motor areas located in the affected hemisphere, which should eventually result in cortical reorganization and improved function. Research in healthy subjects has provided some evidence of such mechanisms, with the use of either transcranial magnetic stimulation 14,15 or functional magnetic resonance imaging (fMRI). 16 However, results of these studies are not conclusive, do not give insight into long-term neuronal changes, and do not necessarily apply to stroke patients.
The aim of this phase II study was 2-fold. First, we evaluated the effect of mirror therapy on upper extremity function in a group of chronic stroke patients. As the intervention had to be both effective and efficient, an unsupervised training program to be performed at home was developed and carried out with supervised weekly sessions. Our primary focus was on improvements in motor function, but to get a detailed insight in any effects of mirror therapy, outcomes were also measured at the other International Classification of Functioning, Disability and Health (ICF) 17 domains. Second, we used fMRI to examine whether mirror therapy could induce cortical reorganization.
Materials and Methods
Participants
After contacting 182 outpatients (hospitalized between January 1998 and September 2007) from the Rijndam Rehabilitation Centre in Rotterdam, the Netherlands, we enrolled 40 patients. Inclusion criteria were knowledge of the Dutch language, a Brunnstrom score for the upper extremity between III and V 18 (the 6 stages of the Brunnstrom score range from [I] flaccidity toward [VI] full-range voluntary extension and individual finger movements present but less accurate than on the opposite side), home dwelling status, and at least 1 year poststroke. Patients with neglect, comorbidities that influenced upper extremity usage, or a history of multiple strokes were excluded. For patients to participate in the fMRI experiment the following additional inclusion criteria applied: ability to perform a hand squeezing movement, no metal implants, no claustrophobia, and no severe obesity. The study was approved by the Medical Ethics Committee of the Erasmus Medical Centre, Rotterdam, and all patients gave written, informed consent before participating.
Sample Size
To calculate the necessary number of patients, we performed a power analysis on data from the literature. Assuming a clinical relevance of 10% on the Fugl-Meyer motor assessment 19 (FMA) score, a standard deviation of 3.2, 20 and a loss of patients at follow-up of 10%, we calculated that 20 patients in each group would be sufficient to have an 80% chance of detecting a statistically significant difference in improvements between the 2 groups.
Study Design
All participants were randomly assigned to either the experimental group receiving mirror therapy (mirror group) or the control group. One of the authors otherwise not involved in the intervention used a computer random number generator to create the randomization sequence and constructed sealed envelopes containing the assignments. Patients received their group allocation assignments after baseline measurements were performed, just before the first training session.
Measurements of upper extremity function were performed before intervention (baseline), right after intervention (posttreatment), and 6 months after intervention (follow-up). All assessments were made by the same investigator, who was blinded to group allocation. Blinding of the patients or the physiotherapist was not possible because of the nature of the therapy. Examinations with fMRI were done in scanning sessions at baseline and posttreatment. Posttreatment clinical measurements and fMRI examinations were performed within a week after the last treatment session.
Intervention
All patients participated in a 6-week training program. Both mirror and control groups performed bimanual exercises, with the difficulty of the exercises depending on the patients’ individual levels of functioning. Exercises were not only based on the Brunnstrom phases of motor recovery but also consisted of functional exercises such as moving objects. The control group had a direct view of both hands, whereas the mirror group practiced with the affected hand positioned behind the mirror while they looked at the reflection of the unaffected hand in the mirror. To ensure that patients focused at the mirror reflection of their unaffected hand instead of their moving unaffected hand itself, a cover was placed over their unaffected hand (Figure 1). Patients practiced at the rehabilitation center once a week under the supervision of a physiotherapist and were instructed to practice 5 times a week, 1 hour per day, at home. Home practice materials consisted of an instruction booklet with photographs and a digital video disk with film fragments of the exercises to be performed. Regular telephone calls were made by the physiotherapist to assure that patients complied with their exercise regimens. Furthermore, patients were instructed to keep detailed accounts of their practice schedules and experiences. These diaries were inspected by the physiotherapist during each training session in the rehabilitation center.

Setup for mirror therapy
Outcome Measures
As mirror therapy consists of exercises mainly on the level of body function, we chose as primary endpoint the difference in improvement between both treatment groups in motor function as measured with the upper extremity part of the FMA (including arm, wrist, and hand function measurements). Additionally, in the
fMRI Experiment
In all, 12 patients from the mirror group and 9 patients from the control group were eligible to participate in this part of the study. During the scanning sessions, patients lay on their backs in the scanner with their upper arms comfortably resting on the scanner table alongside their torsos, with their elbows flexed so that their hands were 20 cm apart above their waists. By means of 2 mirrors attached to the head coil above each patient’s head, patients were able to look in the direction of their feet and view both hands.
The experimental task consisted of 10 alternating 30-second periods of 5 rest and 5 active conditions (block design). In the active periods, patients had to open and close the affected hand; in the rest periods patients had to hold the hand still. Patients were instructed to pace the opening of their hand to a metronome with a rhythm of 0.5 Hz. The onsets of the rest and active conditions were indicated verbally by using simple words (start, rest) generated by a computer program (Matlab version 7.1; Mathworks, Sherborn, MA). Auditory stimuli were presented to the patients through MRI-compatible headphones. The hand movement was practiced before the scan session started.
Imaging was performed on a 3T MR system (HD platform, GE Healthcare, Milwaukee, WI). For anatomical reference, a high-resolution, 3-dimensional, inversion recovery, fast spoiled gradient echo, T1-weighted image was acquired (TR/TE/TI 10.7/2.2/300 ms, 18° flip angle, matrix 416 × 256, and field of view 250 × 175 mm2). For functional imaging, a single-shot, T2*-weighted, gradient echo echo-planar imaging (EPI) sequence was used (TR/TE 3000/30 ms, 75° flip angle, matrix 64 × 96, field of view 220 × 220 mm2). The imaging volume covered the entire brain, including the cerebellum.
Statistical Analyses
Clinical outcome
To test the study hypothesis, we used a generalized estimating equations approach. Under the assumption that missing data are random and not due to group allocation or treatment effect, this model estimates missing data values, thereby allowing the use of data from all participants, irrespective of whether they were measured at all time points. Each outcome measure was used as a separate response variable, and group (mirror vs control) and time (baseline vs posttreatment vs follow-up) were inserted in the model as predictors. The interaction of group × time was used to determine the efficacy of the intervention. Significance was set at .05.
fMRI data
The imaging data were analyzed using statistical parametric mapping software (SPM5; Wellcome Department of Cognitive Neurology, University College London, UK), implemented in Matlab version 7.1 (MathWorks, Natick, MA).
All functional images for each participant were realigned to the first scan of each session and then coregistered to the T1-weighted anatomical scan. Subsequently, images were transformed to standard Montreal Neurologic Institute space. To prevent warping around the lesions, we used a segmentation-based normalization approach. 26 Finally, normalized images were spatially smoothed by using a Gaussian filter of 8-mm full width at half maximum.
Preliminary analyses showed that the realignment parameters estimated during spatial preprocessing were sometimes correlated with task design. Therefore, we decided not to model the realignment parameters in the design matrix as regressors of no interest, as this would have resulted in canceling out task-related activation. Instead, we used the ArtRepair Toolbox, 27,28 which evaluates all volumes and detects the ones most affected by movement. Those volumes are repaired by interpolation to avoid side effects in the high-pass filter and then deweighted in the general linear model estimation to maintain unbiased estimates. The experimental block design was convolved with the canonical hemodynamic response function, and the resulting model was estimated using a high-pass filter at 128 seconds to remove low-frequency artifacts.
In the first-level analysis, contrast maps were calculated for the active periods versus rest for each patient and each session separately. In addition, contrasts were calculated for the pretreatment versus posttreatment sessions for each patient. Contrast images from patients with left-sided lesions were flipped about the mid-sagittal plane, so that the affected hemisphere corresponded to the right side of the brain for all patients.
Second-level analyses
We merged both groups and performed a random effect analysis on the baseline contrasts of task versus rest to show the typical activation patterns. To assess the differences between groups following therapy, we concentrated on effects within certain regions of interest (ROIs). Using the Anatomy Toolbox
29
and the Anatomical Automatic Labeling Atlas (AAL),
30
we defined ROIs for each hemisphere separately for the following areas: primary motor cortex (M1), dorsal premotor cortex (PMd), primary sensory cortex (S1), supplementary motor area (SMA), and cerebellum. M1 was constructed of Brodmann areas 4a and 4p; PMd of Brodmann area 6 excluding SMA and the ventral premotor cortex (area below
These ROIs were used to calculate weighted laterality indexes (wLI), which measure the relative amount of activation between the 2 hemispheres.
31,32
We chose this measure because previous research showed that it correlates with residual clinical deficit and is sensitive to detecting subtle therapy-associated changes in cortical activation.
33
Using this method, we first normalized for intersubject variations in global fMRI signal to reduce the chances of floor or ceiling values. For each condition, a threshold was defined as half the average value of the 5% highest
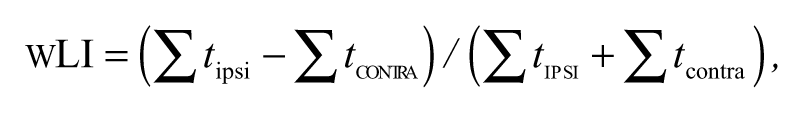
where the subscripts contra and ipsi, respectively, refer to contralateral and ipsilateral with respect to the side of the lesion. wLI values can range between −1 and 1, with higher values indicating a larger contribution of the affected hemisphere.
Changes in wLI within each ROI for the 2 groups separately were assessed using the Wilcoxon rank sum test. Differences in the amount of change in the wLI between groups before and after treatment were assessed with the Mann–Whitney
Finally, a correlation analysis (Spearman’s correlation coefficient) was performed to assess the relationship between changes in wLI and changes in FMA scores.
Results
Table 1 presents demographic and clinical characteristics of the 2 treatment groups, showing no differences. Figure 2 shows the flow chart of the study. None of the reported dropouts were because of group allocation or treatment effect. Figures 3 and 4 show the structural brain images of the participants of the fMRI subgroup.
Patient Characteristics
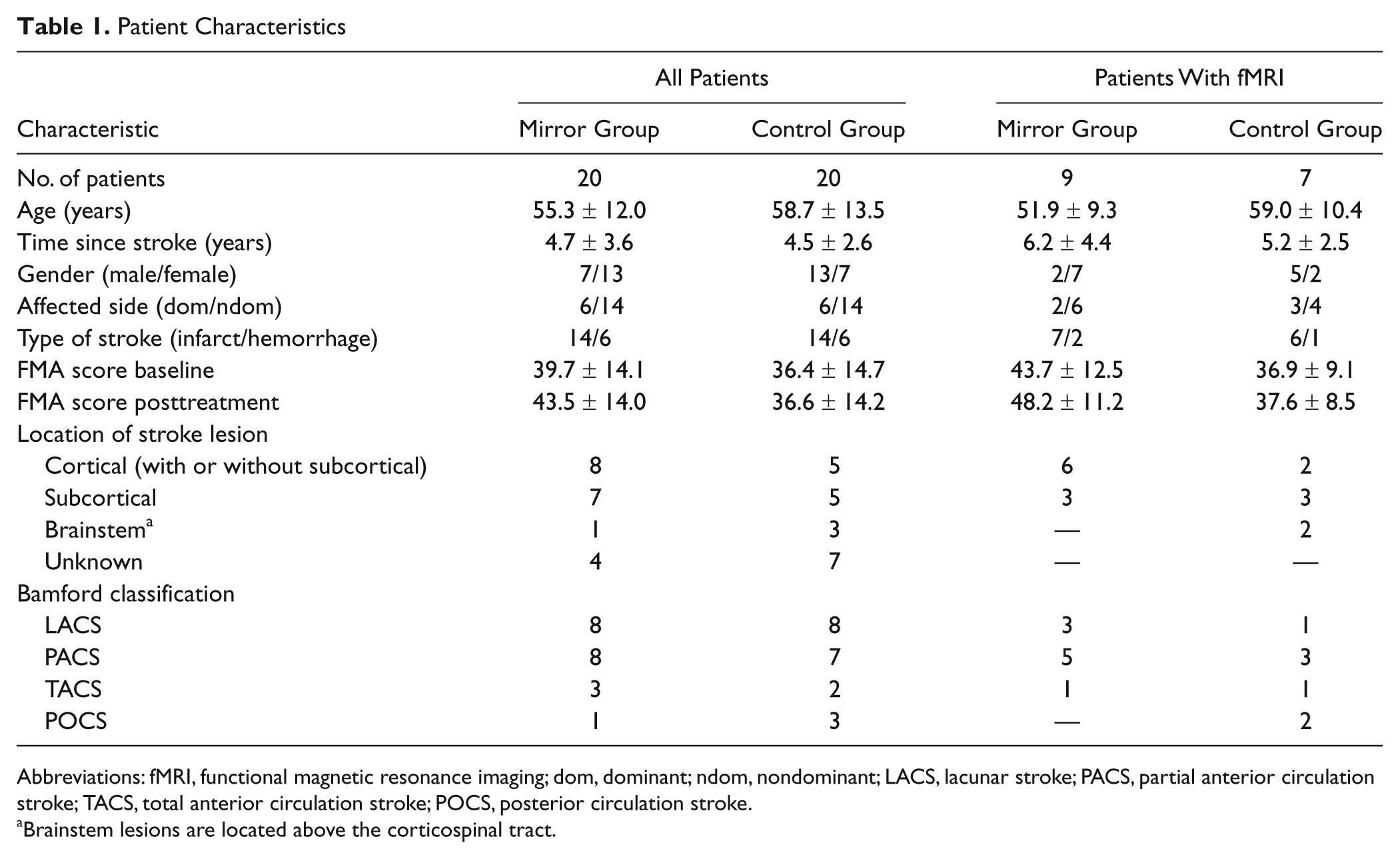
Abbreviations: fMRI, functional magnetic resonance imaging; dom, dominant; ndom, nondominant; LACS, lacunar stroke; PACS, partial anterior circulation stroke; TACS, total anterior circulation stroke; POCS, posterior circulation stroke.
Brainstem lesions are located above the corticospinal tract.
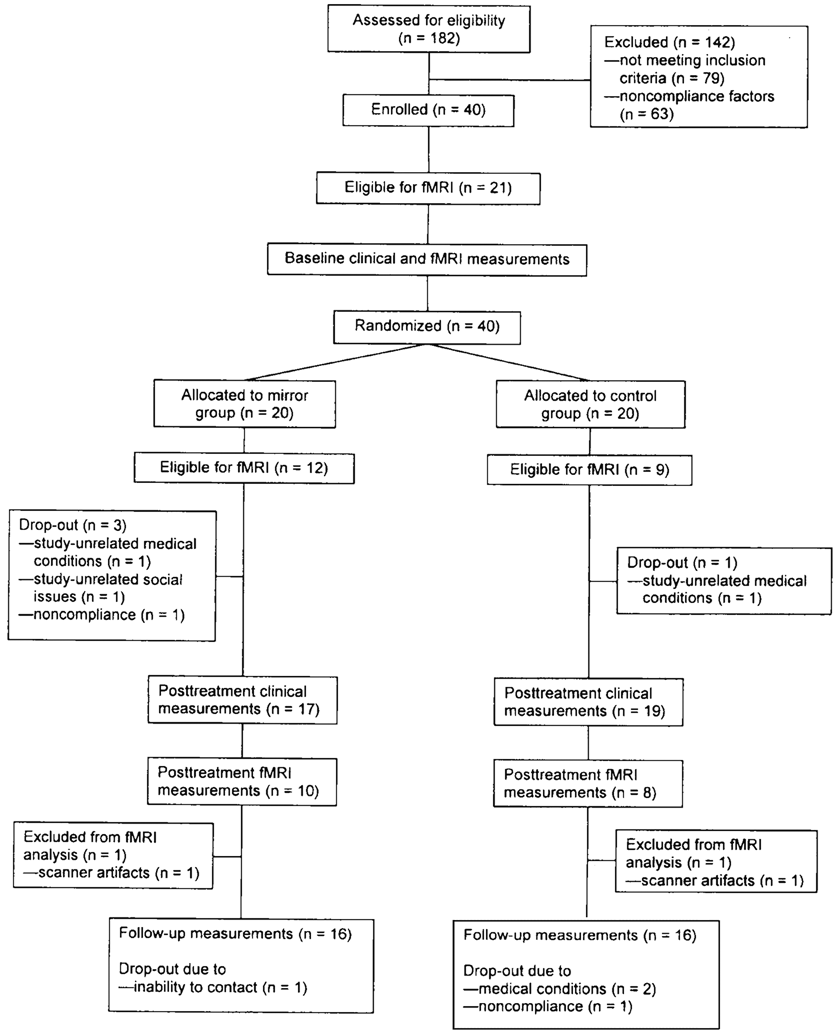
Flow chart
T1-weighted magnetic resonance imaging scans at the level of maximum infarct volume for each patient of the mirror group
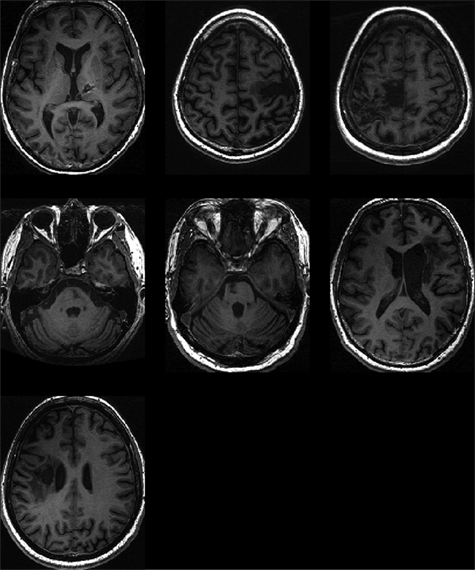
T1-weighted magnetic resonance imaging scans at the level of maximum infarct volume for each patient of the control group
Clinical Measures
All patients attended all 6 training sessions in the rehabilitation center, and the home-kept diaries showed no differences in total home-based practice time between the groups. All patients kept to their regimens, which resulted in an average home training time of 30 hours.
Analyses followed the intention-to-treat principle. Table 2 shows the estimated marginal means and standard deviations for all measurement tools at baseline, posttreatment, and follow-up as well as the between-group comparisons of the change scores from baseline to posttreatment and from baseline to follow-up. Posttreatment, the mirror group had improved significantly more on the FMA than the control group (
Mean Scores on the Clinical Outcome Measures and Their Changes Over Time
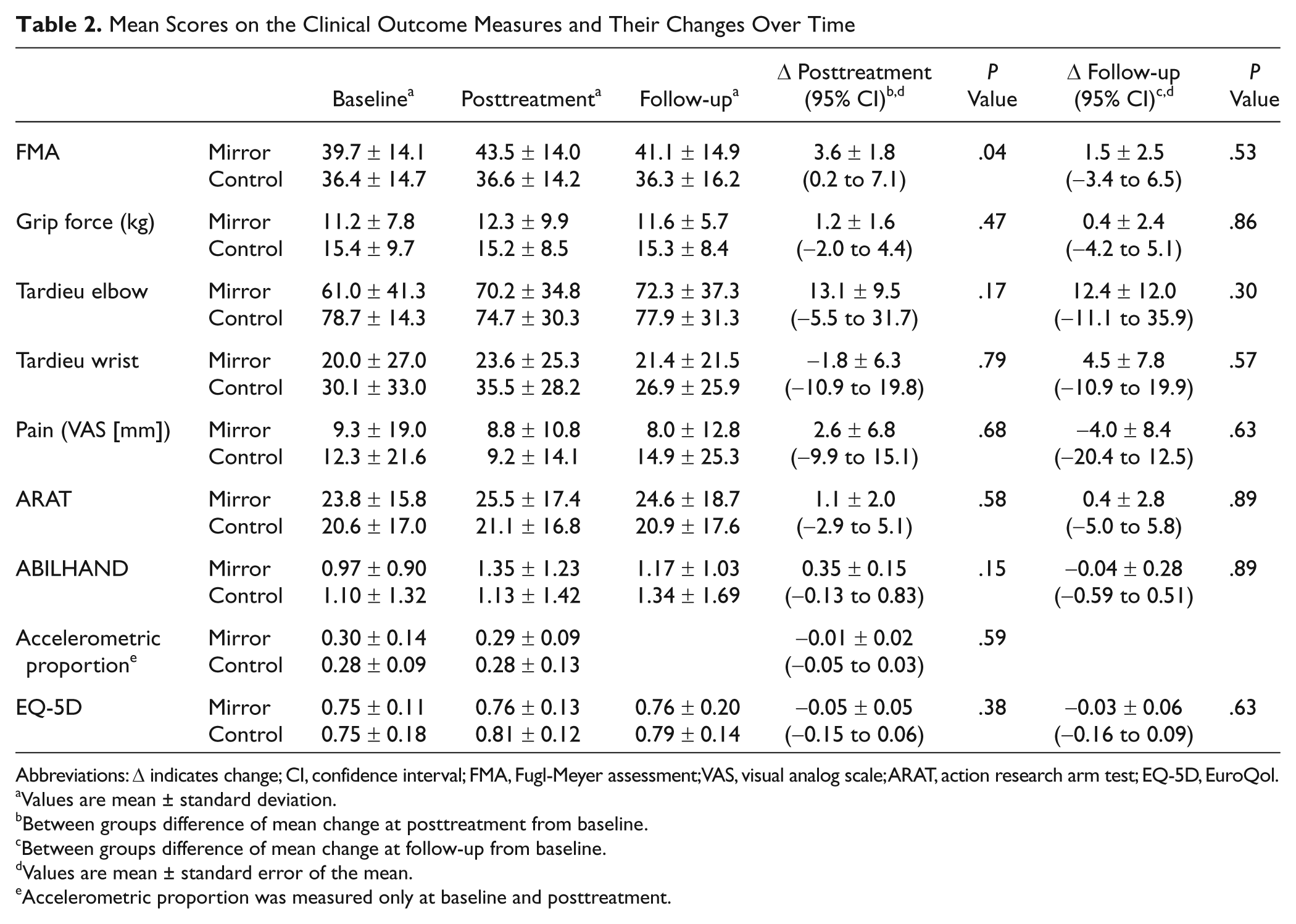
Abbreviations: Δ indicates change; CI, confidence interval; FMA, Fugl-Meyer assessment; VAS, visual analog scale; ARAT, action research arm test; EQ-5D, EuroQol.
Values are mean ± standard deviation.
Between groups difference of mean change at posttreatment from baseline.
Between groups difference of mean change at follow-up from baseline.
Values are mean ± standard error of the mean.
Accelerometric proportion was measured only at baseline and posttreatment.
Cortical Reorganization
In all, 3 patients who were scanned at baseline did not complete the training program and were not scanned posttreatment. Two other patients were discarded from further analysis because their data sets contained scanner artifacts. The remaining analyses were therefore conducted on data from 9 patients in the mirror group and 7 patients in the control group.
Figure 5 shows the activation maps as calculated in the random effects analysis of the task condition versus the rest condition. In general, in all sessions, activation patterns were in accordance with the expected activation for a hand motor task. Activity was observed bilaterally in the precentral and postcentral gyri (M1 and S1), the medial superior frontal gyrus (SMA), at the junction of the superior frontal sulcus and the precentral sulcus (PMC), and in the cerebellum.
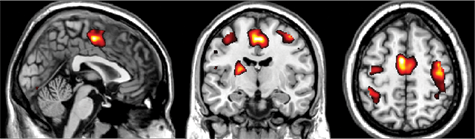
Activation map of the 2 experimental groups combined at baseline of the task condition versus rest (
Table 3 presents the wLI for each of the ROIs. Within M1, a difference in wLI change between the mirror group and control group was found. The activation in the mirror group shifted toward the affected hemisphere, whereas a small shift in activation toward the unaffected hemisphere was observed in the control group. In the other regions no differences in the wLI were observed between the 2 groups. No significant correlations were observed between pretreatment and posttreatment changes in wLI and FMA scores.
Voxel Count in Affected and Unaffected Hemisphere and Weighted Laterality Indices (wLI) for the Baseline and the Posttreatment fMRI Examinations and the Corresponding Changes for each of the 5 Regions of Interest a,b
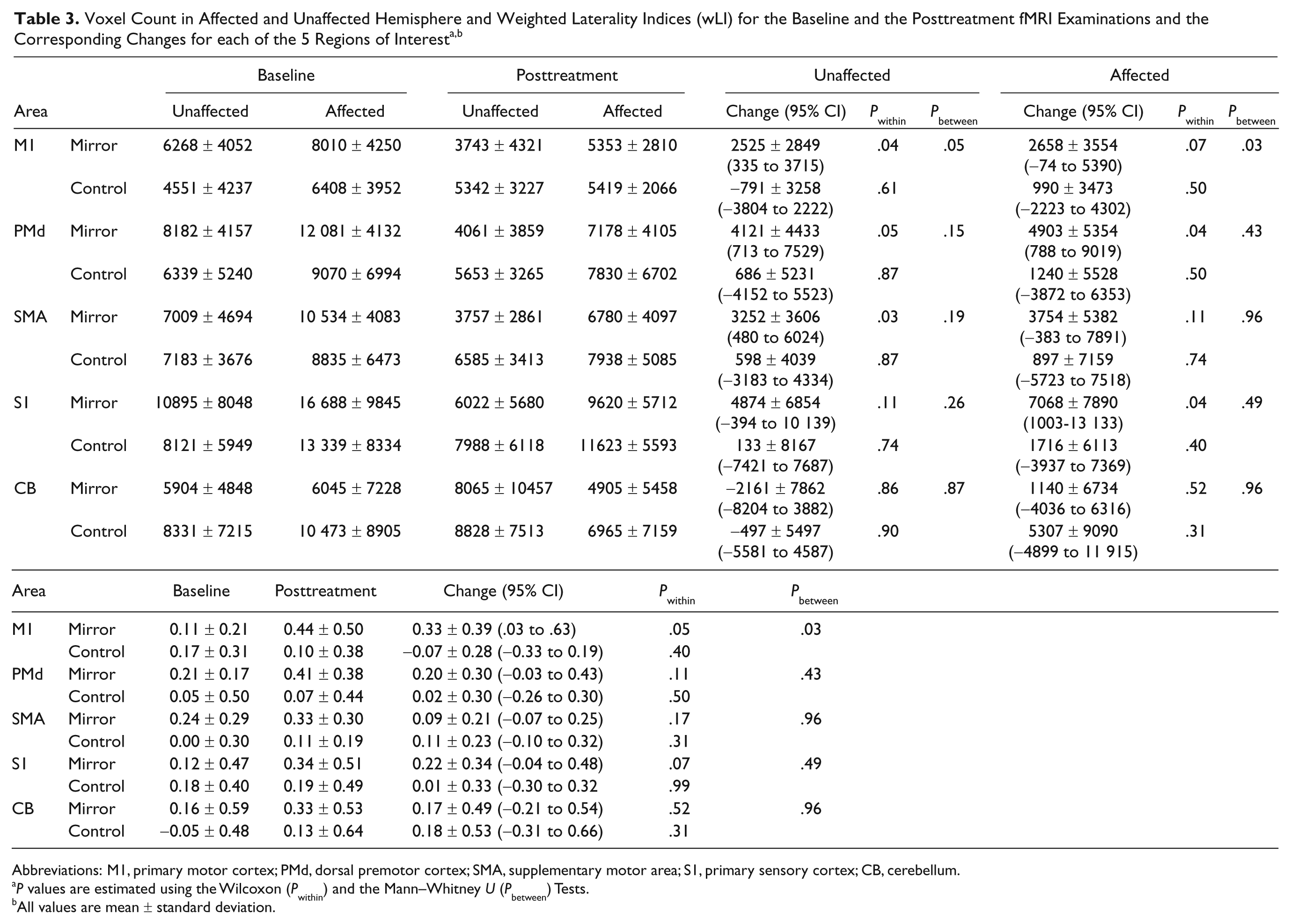
Abbreviations: M1, primary motor cortex; PMd, dorsal premotor cortex; SMA, supplementary motor area; S1, primary sensory cortex; CB, cerebellum.
All values are mean ± standard deviation.
Discussion
Our study has 2 important findings. We showed that in patients with chronic stroke, practicing with a mirror resulted in modest, but statistically significant, improvements in upper extremity motor function. This effect disappeared in the follow-up measurements and did not transfer to the ICF domains
The results on our clinical outcome measures are in agreement with previous studies on patients with subacute 11 and acute 12 stroke. The size of the FMA change in the present study is not very different from that in a study of patients with more acute stroke. Mirror therapy may thus be beneficial at all stages after stroke. The fact that the current study was based on unsupervised, home-based treatment is in this respect promising.
The effects we observed on motor function did not persist at the 6-month follow-up and did not transfer to any other ICF domains. This is in disagreement with the study of Yavuzer et al, 11 who reported improved motor function as well as improved activities of daily living also at 6-months’ follow-up. This discrepancy might be because of the more sensitive activities of daily living (ADL) measurement tool used by Yavuzer et al, as well as the difference in time post-onset. Whereas the subacute stroke patients in the study by Yavuzer et al were still in the rehabilitation center, the chronic stroke patients in our study were home based with daily living routines that were already well adjusted to their disabilities. Improvements in motor function may therefore cause less change in these routines. This lack of transfer to the activity and participation domain may also explain the lack of persistence of improvements in motor function: as patients do not involve the affected arm more in a home setting, therapeutic improvements will deteriorate more rapidly, following the “use it or lose it” principle. 34
The improvements we found in motor function did not reach the generally accepted clinically relevant level of 10%. 35 However, other training studies in chronic stroke patients have reported similarly small improvements, often using more complicated therapeutic strategies. 4,36 Although one could argue that the improvements we found are small and would not have survived a correction for the multiple comparisons (eg, a Bonferroni correction), from a clinical point of view, any improvements in this group are promising, especially considering that this was a short, easily implemented and nonintensive home training program.
A novel aspect of this study was that we evaluated the effect of mirror therapy on cortical organization. Our results suggest that after a period of mirror therapy the hemispheric activation balance shifts toward the affected hemisphere. This is in agreement with previous research showing a similar shift during the recovery following stroke 37 and after arm-tracking training. 38 At least several adaptation mechanisms occur following stroke, amongst them increased recruitment of the undamaged hemisphere. 39 Whether this increased recruitment is beneficial or detrimental remains controversial. 40 However, several studies have shown that increased activation in contralesional motor areas is associated with worse motor function, 31,41 whereas a more normal distribution of hemispheric activation is usually found in better recovered patients. 42 In our study, the mirror illusion seems to bring the disturbed activation balance within the hemispheres more toward normality. Our data show that this shift in hemispheric balance was mainly caused by a decrease in contralesional M1 activation, which is also in line with previous research. 31
We found no correlation between functional gains and changes in hemispheric balance. This may be because of our small sample size and the small changes we found on both measures. Although several studies have reported correlations between recovery of motor function and changes in brain activation patterns, 31,37,42 such relationships are not always found, 43,44 illustrating that the relationship between measures of brain reorganization and behavioral improvement remains complex. 45,46
The question that remains is which mechanism is responsible for the functional improvements and neural changes following mirror therapy. As mirror therapy resulted in larger improvements than the control intervention, the mirror does seem to have an additional effect beyond repetitive task-orientated training. 47 It is possible that by providing an image of a normal moving hand, the mirror illusion enriches the training environment and increases somatosensory input, thereby inducing excitability of the motor cortex. 40 Research in healthy subjects using transcranial magnetic stimulation has provided some proof of such a mechanism, although these results are not conclusive. 14,48 Alternatively, the mirror illusion might increase attention to the motor task, which is known to increase cortical activity 49 and is of importance during motor recovery following stroke. 42 In literature, mirror therapy is often linked with the mirror neuron system or motor imagery. One imaging study in healthy participants provided some proof for the activation of the mirror neuron system by the mirror illusion, but again these results were not conclusive. 16 Definite evidence for the contribution of specific neural areas to the effects of mirror therapy mirror therapy has to come from patient studies investigating neural activation patterns directly associated with mirror viewing.
The results of the present study should be interpreted with some caution. First, our treatment groups were of only moderate size, and the findings on neural activation changes are based on only a small number of participants. Because of the latter, we were unable to perform a random effects fMRI analysis to make population based interferences. Furthermore, our sample included patients with large lesions, including partial M1 damage. Previous fMRI studies have shown, however, that partial infarction of M1 still allows for functional recovery, accompanied by adaptive functional reorganization within spared M1. 44 In addition, some of the differences in activation patterns following treatment could be because of adaptation to the scanner or task. Although the overall decrease in activation observed in both hemispheres (Table 3) indicates that some adaptation occurred, it is not likely that changes in wLI are caused by such adaptation.
The generalizability of our results has limitations. First, our sample consisted mainly of nondominant hemisphere stroke. Second, although lesion size and motor deficits of our participants were considerable compared with patients in most fMRI research, we did exclude patients with complete hemiplegia. Whereas small lesions and modest motor deficits are preferable for making strong fMRI inferences, therapies such as mirror therapy may be especially relevant for patients with large motor deficits for whom virtually no treatment is available. 50
In conclusion, our phase II trial shows that mirror therapy improves motor function in chronic stroke patients more than a control intervention and leads to changes in cortical organization. However, the clinical improvements we found were small, did not persist 6 months after therapy, and were not reflected in ICF domains of activity and participation. A key question in future research is how to augment the effect of mirror therapy. The focus herein should be on identifying the optimal treatment regimens and subpopulations for the effects of mirror therapy to persist and translate to improvements in performance of daily activities and participation.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Fonds NutsOhra [SNO-T-0602-23]; Innovatiefonds Zorgverzekeraars [06-262]; Wetenschappelijk College Fysiotherapie [WU/2007/07] and Hersenstichting Nederland [15F07.54].
