Abstract
Keywords
Memory deficits are common consequences of neurological injury and disease and can be ameliorated through cognitive rehabilitation. 1 Explicit-memory training (EMT), which involves the conscious use of strategies, significantly improves learning and memory in healthy elderly 2,3 and various patient populations. 1 Although some evidence supports the use of EMT in Alzheimer disease (AD), 4-6 consensus is lacking. 7
Mild cognitive impairment (MCI), characterized by cognitive deficits but intact daily functioning, is often a precursor to AD. 8 Therefore, MCI may allow a window of opportunity for interventions that prolong functional independence. However, patients with MCI 9,10 and older adults with below-average memory functioning 11 demonstrated limited improvement following EMT. These studies taught patients multiple mnemonic strategies that may have been difficult for patients to select and use during posttraining memory testing. 10 A single mnemonic strategy applicable to multiple types of information may be more beneficial for patients with cognitive deficits. 12 This is an especially important area of investigation given the rapidly growing elderly population and the continuum between normal aging, MCI, and AD.
Identifying the neural correlates of EMT may assist clinicians in selecting, developing, and using patient-specific mnemonic techniques. Neurologically healthy (typically young) adults show improved memory test performance and increased activation within the middle and inferior frontal gyri 13-16 as well as the hippocampus and medial parietal cortex 17 after EMT. Thus, EMT may provide a mechanism through which patients can facilitate performance by recruiting distributed memory-related brain regions. To our knowledge, there have been no neuroimaging studies of EMT in patients with MCI or AD.
Assessing memory using face–name associations provides an ecologically valid measure that is dependent on the explicit-memory system and is difficult for patients with MCI. 18-21 We previously reported the behavioral results of a pilot study in which patients with MCI demonstrated improved memory for face–name associations after EMT. 22 Furthermore, we found that training-associated memory test improvement was positively correlated with memory for the mnemonic cues but negatively correlated with the number of required training trials, suggesting that the behavioral improvement was attributable to EMT rather than merely repeated exposure to the stimuli. We therefore hypothesized that EMT would lead to training-specific increases in activation and connectivity within explicit memory–related regions, such as those identified in previous EMT studies. 13-17 Alternatively, decreased activation in sensory and memory areas would be evident if training-related improvements were a result of repeated stimulus exposure (repetition suppression) or perceptual priming. 23-25
Methods
Participants
A total of 6 right-handed, white patients were recruited from the Atlanta Veterans Affairs Medical Center and the Emory University Alzheimer’s Disease Research Center. They were among the 8 patients in our previous behavioral report. 22 Each patient had been diagnosed with amnestic, multidomain MCI according to Petersen criteria 8 at a consensus conference that included neurologists, neuropsychologists, and other key clinical staff. Individual demographic and neuropsychological data are shown in Table 1. Patients completed the Dementia Rating Scale-2 (DRS-2) after consenting to the study in order to grossly reassess cognitive functioning. Language was the most common nonmemory domain affected (4 of 6 patients); executive dysfunction and attentional deficits were less commonly found. Although the nature and severity of these nonmemory deficits varied across patients, it is important to note that every patient demonstrated training-related behavioral improvement (Table 1). Exclusion criteria included a history of other neurological diseases (eg, stroke, epilepsy, traumatic brain injury), psychiatric disorders (eg, severe depression, bipolar disorder, schizophrenia), and current or past alcohol or drug abuse. The study was approved by the institutional review board of Emory University and the R&D Committee of the Atlanta VAMC. All participants gave written informed consent.
Demographic, Neuropsychological, and Task-Specific Performance Improvements for the 6 Patients Who Took Part in the Study a
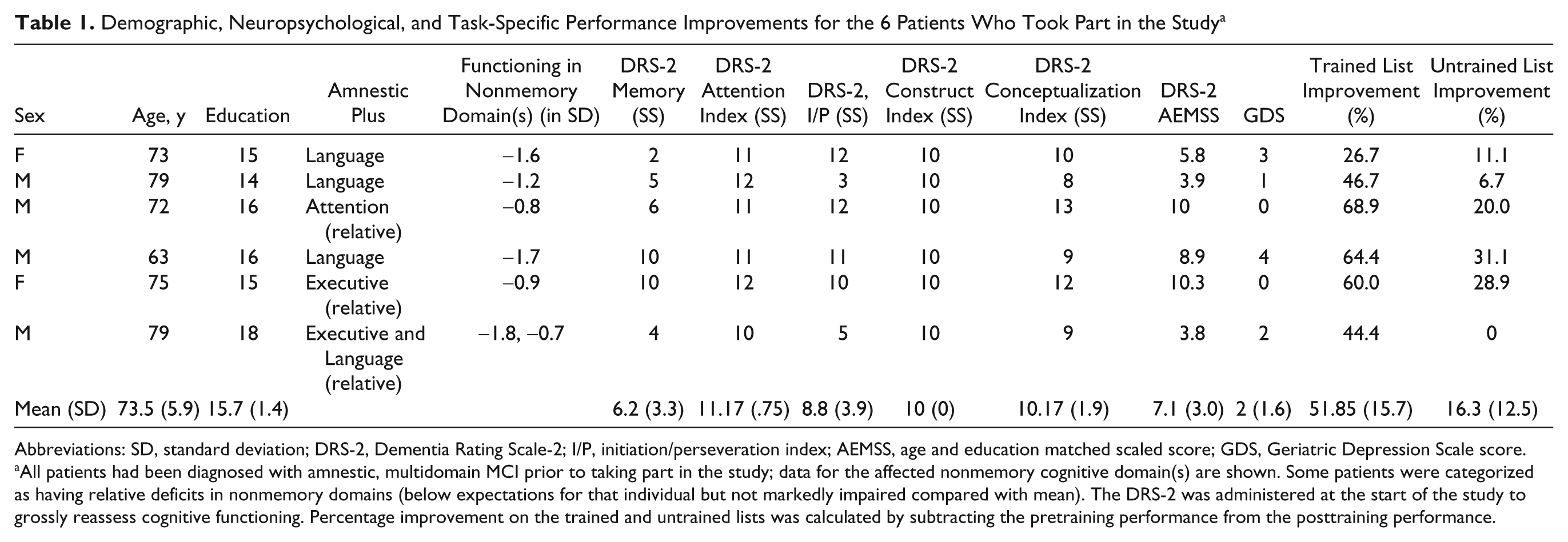
Abbreviations: SD, standard deviation; DRS-2, Dementia Rating Scale-2; I/P, initiation/perseveration index; AEMSS, age and education matched scaled score; GDS, Geriatric Depression Scale score.
All patients had been diagnosed with amnestic, multidomain MCI prior to taking part in the study; data for the affected nonmemory cognitive domain(s) are shown. Some patients were categorized as having relative deficits in nonmemory domains (below expectations for that individual but not markedly impaired compared with mean). The DRS-2 was administered at the start of the study to grossly reassess cognitive functioning. Percentage improvement on the trained and untrained lists was calculated by subtracting the pretraining performance from the posttraining performance.
Stimuli
As detailed earlier, 22 a total of 92 faces were selected from a previously used set. 19 These faces were transformed to grayscale and randomly paired with a gender- and age-appropriate name (based on popularity by approximate birth decade). Ninety of these face–name pairs were divided into 2 lists of 45 that were matched for gender, race, and approximate age (by decade). The remaining 2 faces (one of each gender) served as control stimuli during functional magnetic resonance imaging (fMRI) scanning.
General Procedures
The procedures have been described earlier 22 and are summarized here. Each patient completed a total of 5 sessions within a 2-week period. During the first (pretraining) and fifth (posttraining) sessions, patients underwent fMRI scanning as they encoded the face–name associations. Approximately 15 minutes after each scan was completed, patients took a 4-choice recognition memory test (outside the scanner) involving all 92 face–name pairs. Stimuli were presented within the scanner using a laptop PC running Presentation software (Neurobehavioral Systems Inc, Albany, California). The postscan memory test used a PC-based program specifically developed for this study. During sessions 2, 3, and 4, patients received EMT.
As detailed earlier, 22 patients were trained using 1 of the 2 lists of 45 pairs, in counterbalanced order across patients. This list is hereafter referred to as the trained list, the other being the untrained list. Patients learned 15 face–name pairs during each training session. For each pair, patients were required to spontaneously recall the name on 3 consecutive trials, with a maximum of 10 trials to reach this criterion. Training used a modified Biographical Information Module from the Ecologically Oriented Neurorehabilitation of Memory (EON-Mem) program. 12 Although this program teaches patients to self-generate cues, we provided cues to standardize procedures across patients. For each face–name pair, patients were directed to a salient facial feature (visual cue) and given a nickname that often rhymed with the actual name (verbal cue) linking the feature to the name. Patients were instructed to associate the visual and verbal cues by creating mental images that exaggerated and emphasized the relationship between these cues. On subsequent training trials, patients first recalled the visual cue, then the verbal cue, and finally the corresponding name.
fMRI Procedures
Imaging parameters
MR scans were performed on a Siemens Trio 3T MRI scanner (Siemens Medical Solutions, Malvern, Pennsylvania), using a 12-channel head coil. For blood oxygenation level–dependent (BOLD) contrast, T2*-weighted functional images were acquired using a single-shot, gradient-recalled, echo-planar imaging sequence with the following parameters: repetition time (TR) 2000 ms, echo time (TE) 30 ms, field of view (FOV) 220 mm, flip angle (FA) 90°, 27 axial slices of 4 mm thickness, in-plane resolution 3.4 × 3.4 mm2, and in-plane matrix 64 × 64. High-resolution anatomical images were acquired using a 3D MPRAGE sequence (TR 2300 ms, TE 3.9 ms, inversion time 1100 ms, FA 8°) consisting of 176 sagittal slices of 1 mm thickness (FOV 256 mm, inplane resolution 1 × 1 mm, in-plane matrix 256 × 256). Once the scanner achieved magnetic stabilization for each run, it triggered the computer running Presentation software to synchronize stimulation and scan acquisition.
fMRI paradigm
The same block design paradigm was used pretraining and posttraining, comprising alternating 20 s rest and 30 s active blocks, similar to previous studies of face–name encoding. 20,21 There were 3 types of active blocks, comprising face–name pairs from (1) the trained list, (2) the untrained list, or (3) the 2 control stimuli repeated in alternation. During active blocks, 5 face–name pairs were shown for 5 s each, with a 1 s interstimulus interval. Patients were instructed to remember the name paired with each face; no responses were required because this could have distracted patients from using the trained strategies. The MRI technologist (RFS) entered the room between functional runs to ensure that the patient was performing the task; no problems were reported by any patient. Memory performance was assessed on a postscan test, as in other imaging studies of memory. 20,21 Each block type occurred 3 times during each of 3 functional runs, so that the 45 pairs from the trained and untrained lists were each displayed once. Repeated stimuli were seen a total of 45 times.
Imaging Data Analysis
Image processing and analysis were performed using BrainVoyager QX v1.6.3 (Brain Innovation, Maastricht, The Netherlands). Functional runs were motion corrected in real time using Siemens 3D-PACE (prospective acquisition motion correction). For each participant, the functional images were realigned to the first image of the series. Images were preprocessed using trilinear interpolation for motion correction, sinc interpolation for slice scan time correction, and high-pass temporal filtering to 3 cycles/run. They were then coregistered with anatomical images and transformed into Talairach space. 26
For group analysis, transformed data were spatially smoothed with an isotropic Gaussian kernel (full width half maximum = 4 mm), and baseline periods were normalized based on the percentage signal change. A mask was created using all activated voxels within any condition relative to baseline (uncorrected
Activation maps were generated for 3 contrasts described below. For each activation site, regions of interest (ROIs) constrained to be no larger than 125 mm3 (5 × 5 × 5 mm3 cube) were created following the methods described in the preceding paragraph. The following contrasts were examined.
First, to compare our results with previous studies of face–name encoding, we combined the trained and untrained stimuli from the pretraining scan into a single group of “novel” stimuli (because training had yet to take place). Activation in response to these stimuli was compared to that for the repeated face–name pairs (novel > repeated) using a balanced contrast.
Second, we used the interaction between list type (trained, untrained) and session (pretraining, posttraining) to tease out training-specific effects while controlling for nonspecific factors, including practice effects from scanning twice. This interaction identified regions whose activation increased or decreased between sessions, more for the trained list than the untrained list (trained [post > pre] > untrained [post > pre]). We refer to this interaction as training-specific changes.
Third, we assessed nonspecific factors, such as practice effects and/or generalization of the EMT strategies, by analyzing the comparable task-by-session interaction for the untrained and repeated stimuli (untrained [post > pre] > repeated [post > pre]). We refer to this interaction as nonspecific changes.
Effective Connectivity Analysis
We used Granger causality analysis (GCA), which relies on the principle of temporal precedence/lag between time series,
29
to investigate effective connectivity (ie, directional interactions between brain regions during task performance
30
). A multivariate implementation of GCA was used, as first applied to electrophysiological data
31
and subsequently by our group to fMRI data.
32-38
Because effective connectivity obtained from GCA is potentially contaminated by leakage of instantaneous correlation into the causal domain, we used a refined method termed
The time series data from the selected ROIs were averaged across voxels within each ROI, normalized separately for each run and participant by dividing by the grand mean for that run, and concatenated across all runs and participants to form a single vector per ROI. Then, a modified vector autoregressive model was computed. 36,37 From this model, a CPGC connectivity matrix was derived comprising path weights for all potential interactions between the selected ROIs. As noted previously, 33-35,37 interactions between any 2 ROIs that are mediated through another ROI in the matrix are filtered out by this approach, as are zero-lag correlations that may include task-induced correlations because of simultaneous onsets of activation as opposed to true functional connectivity.
Performing data-driven GCA (as in the present study) can require a large number of ROIs, which complicates interpretation. Therefore, we used a network reduction method to eliminate redundancy in the network, as detailed earlier. 34 This was done by removing ROIs that did not significantly reduce the overall network connectivity when eliminated and repeating this process iteratively until no further ROIs could be removed without significantly affecting overall network connectivity. This is a principled method of network reduction that is data driven rather than arbitrary.
Within this reduced network, we then examined connectivity specific to blocks for trained stimuli, separately for pretraining and posttraining scans. We calculated the statistical significance (
Results
Behavioral Results
A repeated-measures analysis of variance (ANOVA) revealed significant main effects of time (
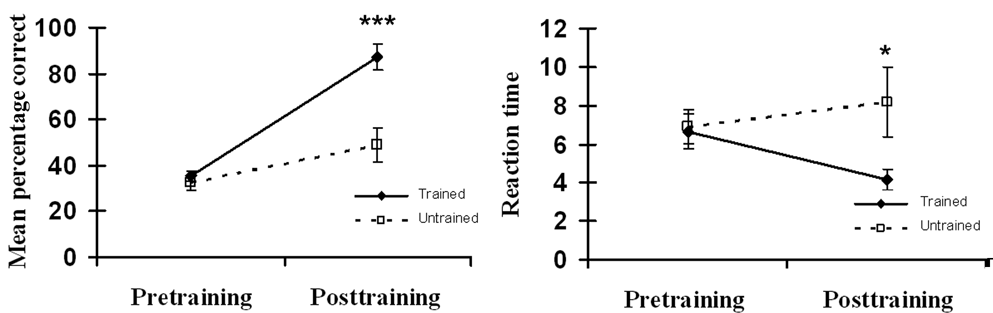
Accuracy (left) and reaction time (right) on the face–name association memory test. Error bars: standard error of the mean.
Analyses of reaction times (RTs) revealed no significant main effects (time:
fMRI Activation Changes
Novel versus repeated
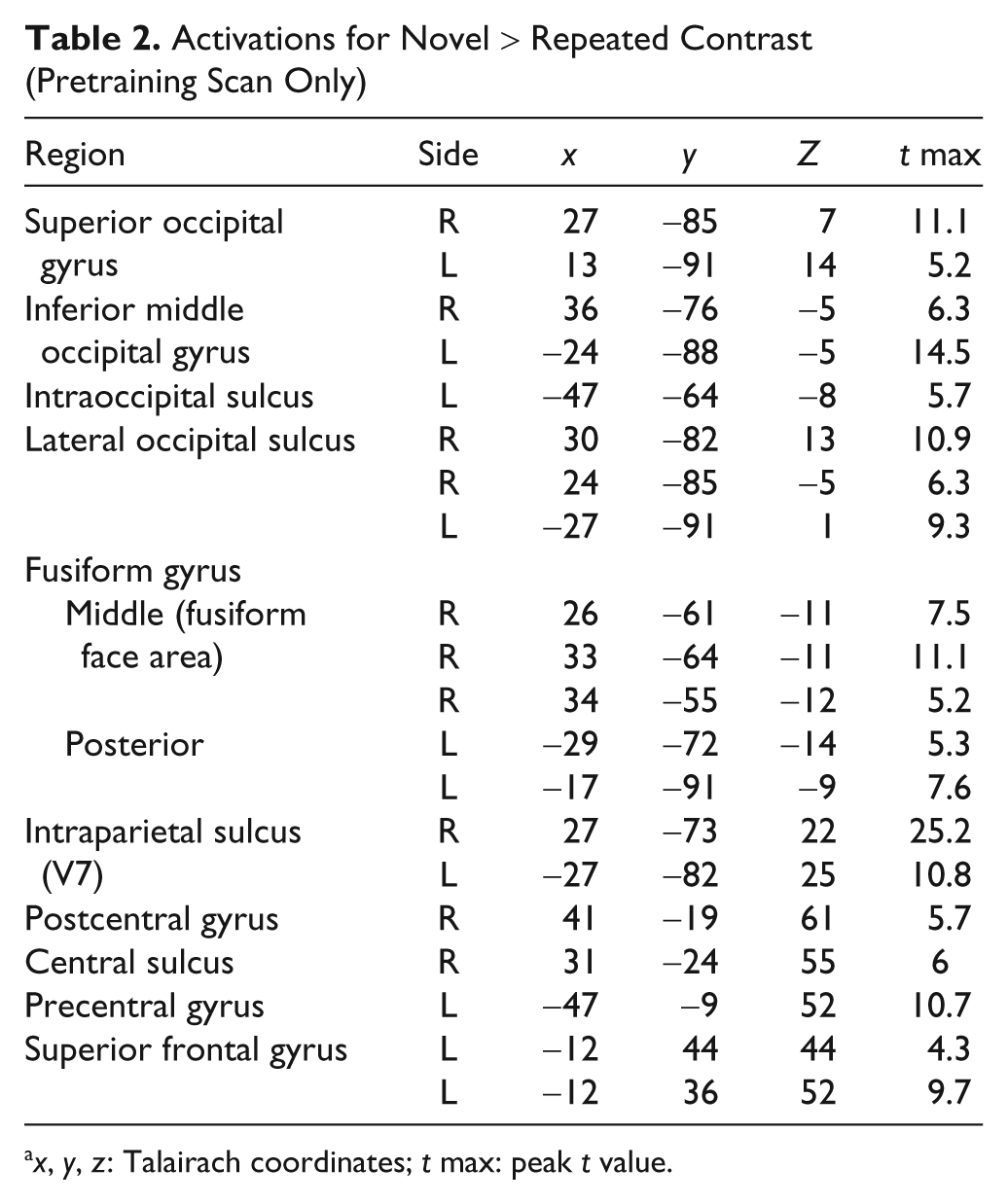
In the pretraining session, encoding novel relative to repeated face–name pairs evoked widespread bilateral activations within the occipital cortex and the fusiform gyrus. Right-fusiform activations were consistent with the location of the fusiform face area, as described previously.
39
Left-fusiform activations were in posterior portions of the gyrus. In addition, activity was found in the left hippocampus (Talairach
Activations for Novel > Repeated Contrast (Pretraining Scan Only)
Training-specific changes
Training-specific increases in activation were found within a widespread network (Table 3; Figure 2). Many of these changes were within the medial frontal (frontopolar gyrus, rostral anterior cingulate cortex [ACC]), medial parietal (posterior cingulate cortex [PCC] and precuneus [PCu]), and medial occipital cortex (infracalcarine and supracalcarine, lingual gyrus [LG]). Increases were also found in more lateral regions, including the left frontal operculum (FO, in the vicinity of Broca’s area), around the left temporoparietal junction (angular gyrus; posterior superior temporal sulcus), and more inferiorly in the left temporal cortex (middle and inferior temporal gyrus). Examination of the BOLD signal time courses confirmed that all these areas showed greater posttraining increases in activation for trained compared with untrained stimuli. Several of these medial frontal and parietal regions and the lateral temporoparietal regions comprise what has become known as the default network. 42 Two regions, both in the middle occipital gyrus (Table 3), showed increases after training that were greater for the untrained than the trained list. Within the regions reported above, the supracalcarine activity was least consistent across patients (3/6 patients), whereas all other cortical increases or decreases in activity following training were present in at least 4 of the 6 patients; the increases were evident in all 6 patients in the majority of regions.
Training-Specific Activations From Interaction (Trained [Post > Pre] > Untrained [Post > Pre]) a
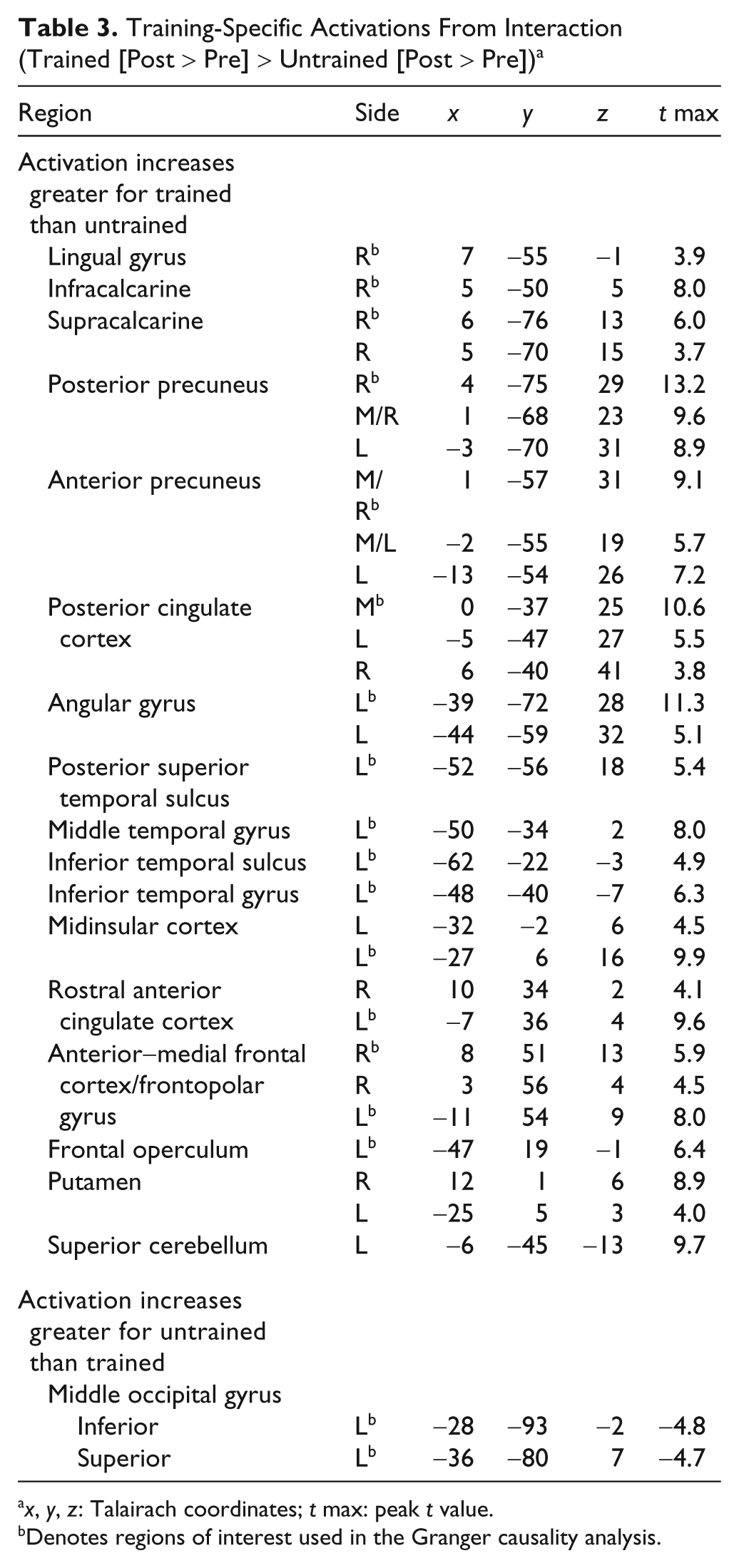
Denotes regions of interest used in the Granger causality analysis.
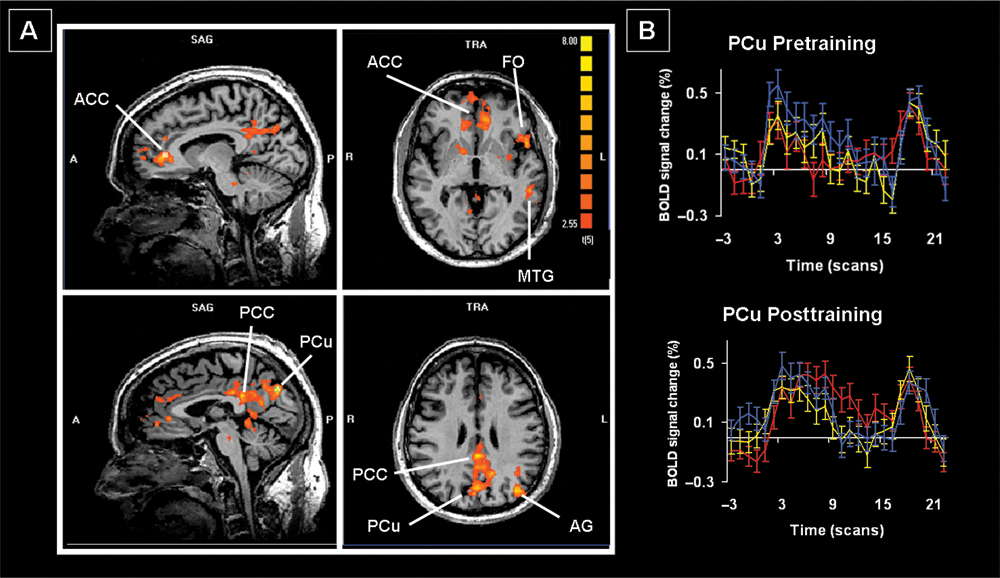
A. Training-specific changes as revealed by the interaction trained stimuli (Post > Pre) > untrained stimuli (Post > Pre). B. Sample time-course data from the PCu for the trained (red), untrained (yellow), and repeated stimuli (blue). Abbreviations: ACC, anterior cingulate cortex; AG, angular gyrus; FO, frontal operculum; MTG, middle temporal gyrus; PCC, posterior cingulate cortex; PCu, precuneus.
Nonspecific changes
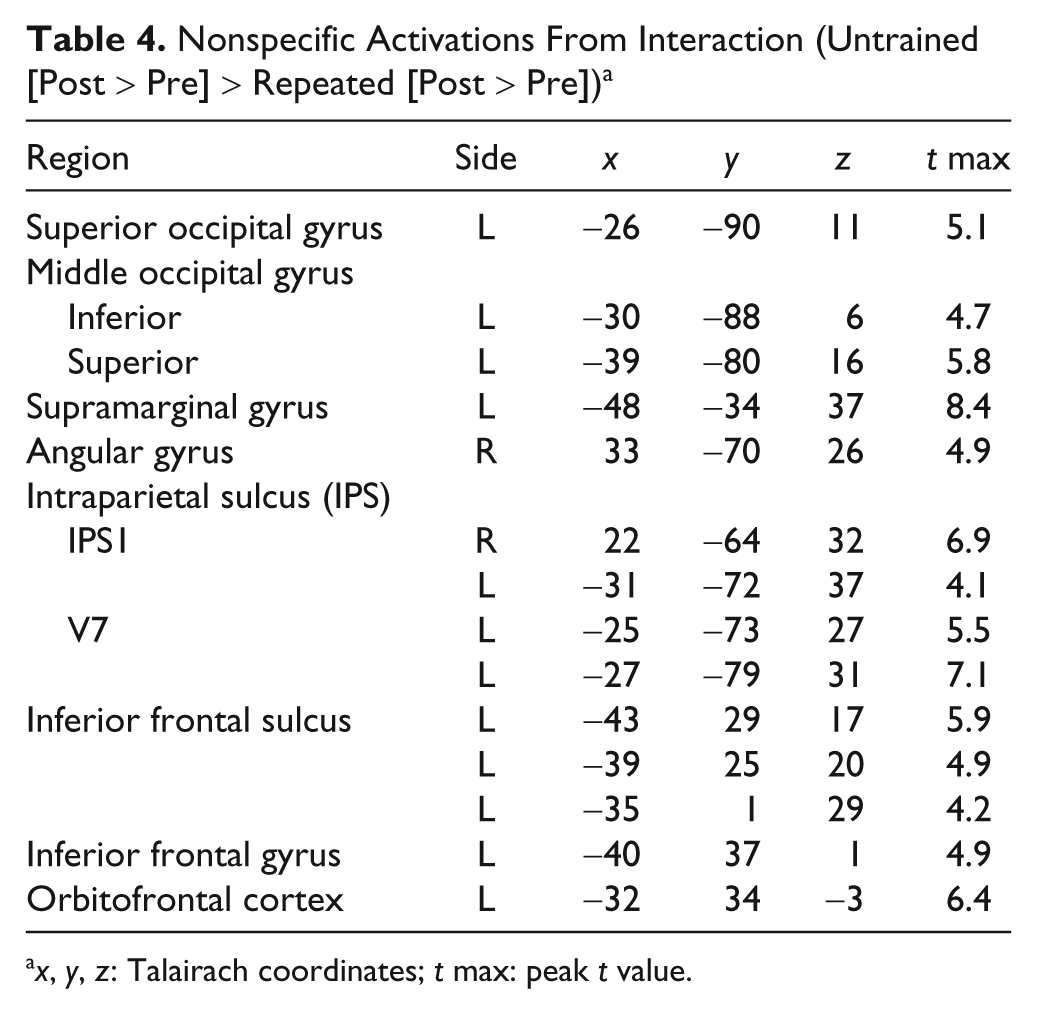
Nonspecific changes included increased activation within the left occipital and inferior frontal cortex and bilaterally in the inferior parietal cortex (regions IPS1 and V7 by Swisher et al 41 ; Table 4; Figure 3). The BOLD signal time courses were indistinguishable for the trained and untrained stimuli in most of these regions and were consistently greater than those for the repeated stimuli. There were no regions of significantly decreased activation. At least 5 of the 6 patients demonstrated activation in all the areas identified on this contrast.
Nonspecific Activations From Interaction (Untrained [Post > Pre] > Repeated [Post > Pre]) a
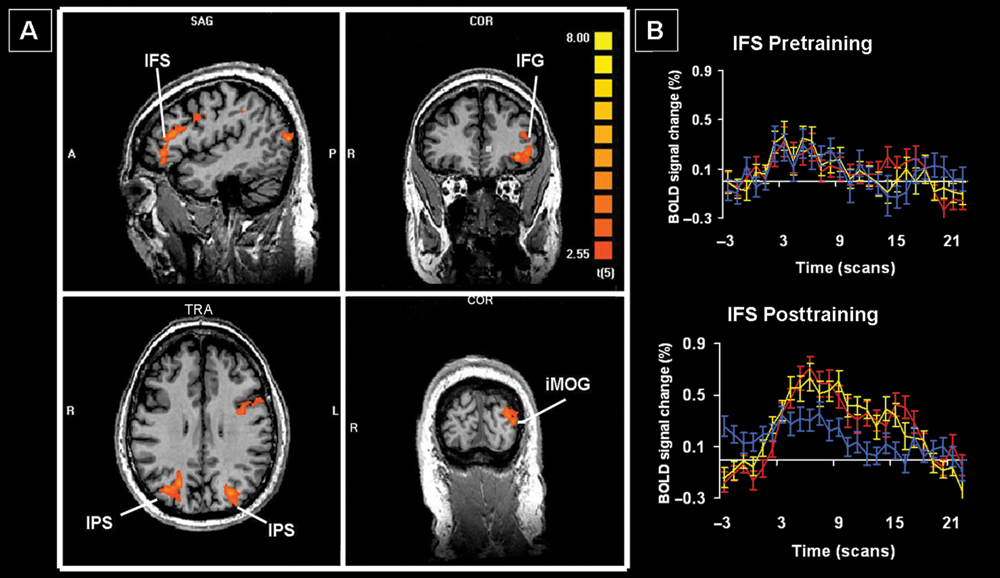
A. Nonspecific changes as revealed by the interaction untrained stimuli (Post > Pre) > repeated stimuli (Post > Pre). B. Sample time-course data from the IFS for the trained (red), untrained (yellow), and repeated stimuli (blue). Abbreviations: IFG, inferior frontal gyrus; IFS, inferior frontal sulcus; iMOG, inferior middle occipital gyrus; IPS, intraparietal sulcus.
Effective Connectivity Changes
In all, 13 of the 19 ROIs survived network reduction; significant path weights during presentation of trained stimuli are shown in Figure 4 and Table 5. The number of significant paths increased from 7 during pretraining to 12 posttraining. The middle temporal gyrus (MTG) was a primary “driver” of activation in other areas during both scans; however, some of the targets changed to include 2 occipital (LG and inferior middle occipital gyrus) and 2 medial parietal regions (anterior and posterior precuneus [aPCu and pPCu]) posttraining, compared with a more distributed pattern of targets pretraining (1 occipital, 1 medial parietal, 1 lateral temporal, and 1 medial frontal). The superior middle occipital gyrus and aPCu emerged as other significant drivers following EMT, whereas these regions did not show any significant connectivity with other areas before training.
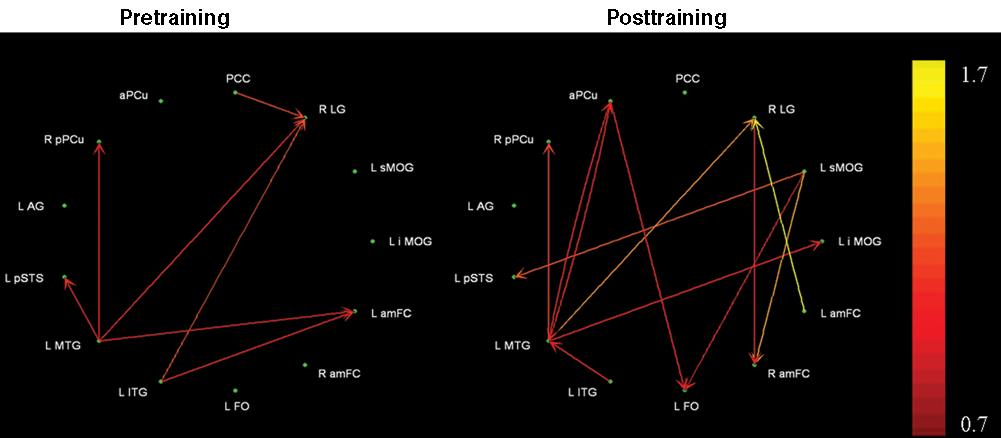
Effective connectivity during encoding of trained stimuli pretraining (left) and posttraining (right).
Significant Path Weights During Encoding of Trained Stimuli a
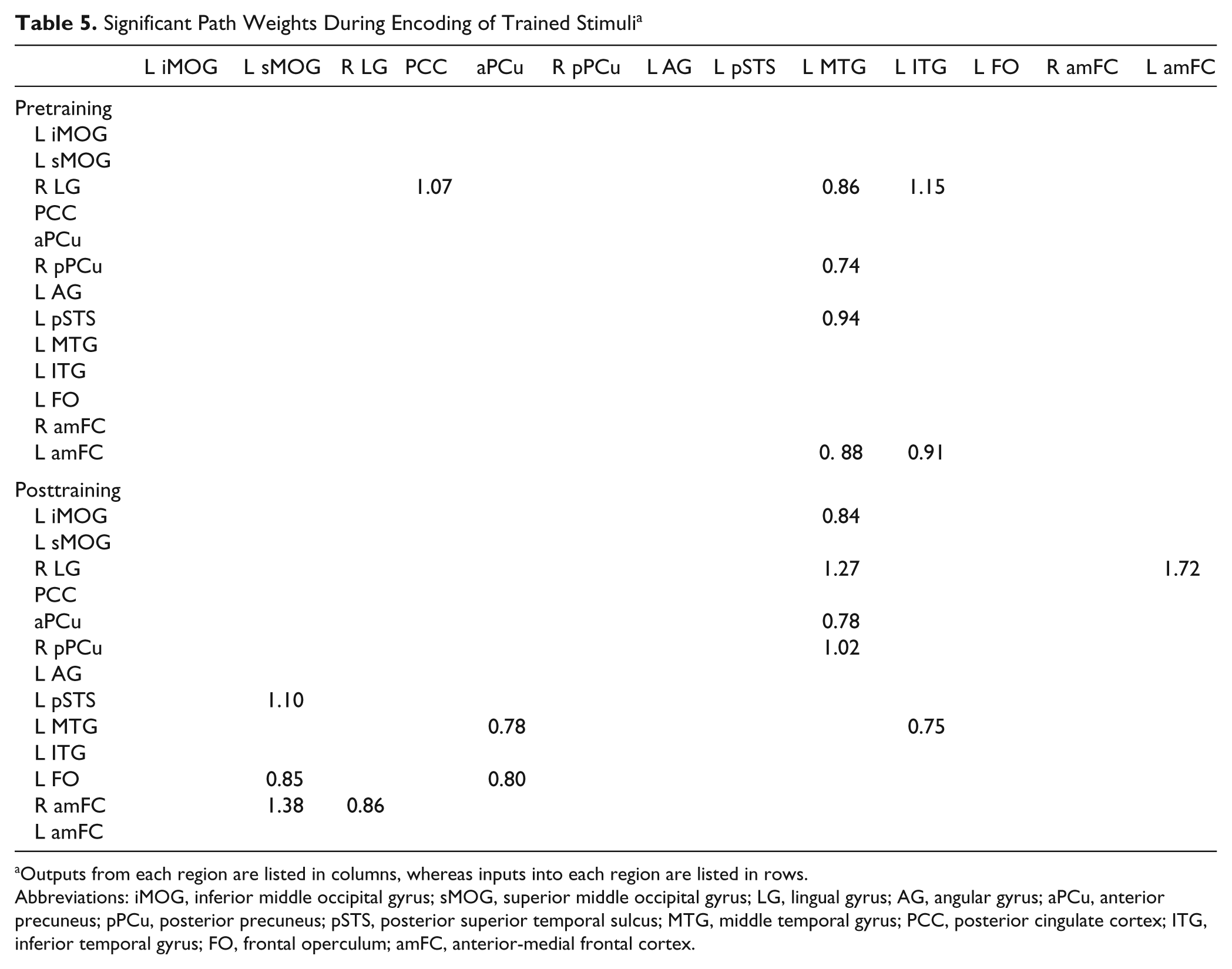
Outputs from each region are listed in columns, whereas inputs into each region are listed in rows.
Abbreviations: iMOG, inferior middle occipital gyrus; sMOG, superior middle occipital gyrus; LG, lingual gyrus; AG, angular gyrus; aPCu, anterior precuneus; pPCu, posterior precuneus; pSTS, posterior superior temporal sulcus; MTG, middle temporal gyrus; PCC, posterior cingulate cortex; ITG, inferior temporal gyrus; FO, frontal operculum; amFC, anterior-medial frontal cortex.
Discussion
This pilot study demonstrated that significant behavioral improvement after EMT in patients with MCI is associated with training-specific increases in activity within medial parts of the frontal, parietal, and occipital cortex as well as lateral areas around the temporoparietal junction and left frontal operculum. Additionally, training generally resulted in increased connectivity. A separate network involving lateral frontal and parietal regions was common to trained and untrained stimuli. It is important to note that the pretraining pattern of activation was consistent with previous studies of face-name encoding, 21,40 showing that our imaging paradigm is comparable to previous research.
Behavioral Changes
As reported earlier, 22 our results join only a few other studies indicating that EMT can be beneficial in patients with MCI 9 and early AD. 4,6 Our focused intervention presumably reduced the confusion caused by using multiple mnemonic strategies 10 and allowed patients to implement the trained strategies. We reported earlier 22 that there were significant correlations between recall of names and of mnemonic cues, supporting the patients’ anecdotal reports that they were attempting to use the strategies during the posttraining scan.
Training-Specific fMRI Changes
The most robust training-specific increases were within the medial frontal and parietal cortices and around the temporoparietal junction, which are part of the default network 42 and are abnormal in MCI and AD. 42,43 This network is posited to control self-directed, internally based processes, including episodic memory 42,44 —processes likely used by EMT strategies. Previous research has identified 3 primary regions through which the default network is posited to interact with other brain areas. 42
First, the PCC interacts with the medial temporal lobe memory system. 42 Dysfunction in this region may be at least partially responsible for the memory deficits in MCI and AD, 45 consistent with the lack of PCC connectivity at either time point. The PCC and its neighbor, the PCu have extensive reciprocal connections. 46 Increased activity in these regions corresponds with better explicit-memory functioning, 45-47 whereas dysfunction is related to worse memory functioning 45 and increased rate of conversion from MCI to AD. 48 EMT results in increased activation in these regions in neurologically healthy participants. 13,15,17 The aPCu demonstrated increased connectivity posttraining because it formed a reciprocal connection with the MTG and drove activation within the left FO. This may reflect the role of the PCu in mental imagery, 46 an important mnemonic strategy used in EMT, or heightened personal relevance of information. 46,49 Finally, the PCu is thought to mediate top-down control of attention during memory retrieval, 50 as would be required when patients search for specific cues. The increased connectivity with the left FO could reflect the use of verbally based cues. Thus, EMT appears to have provided a mechanism through which processes mediated by the PCC and PCu could be used.
The left temporoparietal junction is another region identified as containing key default network areas. 42 This region is posited to mediate the bottom-up capture of attentional resources by memory-relevant cues, 50 which could occur as patients identify the salient stimulus features. The angular gyrus is important for language processing, which may explain the greater left hemisphere activation, given our verbally laden strategies. However, the angular gyrus did not demonstrate significant connectivity, whereas other language areas (FO and MTG) increased connectivity posttraining. The MTG appeared especially important for the face–name association task because it demonstrated the most connectivity at both time points, and a similar MTG region was more active for familiar than novel face–name associations in patients with MCI. 40 A recent meta-analysis suggested that the MTG and ITG are part of the semantic memory network and may play a particular role in supramodal processing and concept retrieval. 51 These findings suggest that our EMT procedures may have used semantically related memory areas. Future studies will be necessary to determine whether EMT bypasses or augments the dysfunctional episodic memory system in favor of the relatively less dysfunctional semantic network in patients with MCI. 52,53
The medial frontal cortex (ACC, frontopolar cortex) is the third important default network region and may be responsible for coordinating multiple cognitive processes 42 ; a role that accords with the frequency with which these areas show increased activation in functional neuroimaging studies. 50 Here, these regions exhibited significant posttraining increases in activation and changes in connectivity, suggesting that EMT facilitated the coordination of multiple cognitive processes.
Nonspecific fMRI Changes
Significant posttraining increases in activation were observed in lateral frontal and parietal cortical areas, with similar activation magnitudes for both trained and untrained stimuli. These changes may reflect practice effects given reexposure to the same stimuli seen pretraining or attempts to generalize the trained strategies to the untrained stimuli. The most notable changes were within the inferior frontal gyrus and inferior frontal sulcus. These areas have been implicated in the resolution of interference among conflicting stimulus attributes 54 and the rehearsal of internally based representations. 55,56 Additionally, increased activation was found within the superior middle occipital gyrus and fusiform face area—regions previously found more active after healthy individuals were trained to attend to specific facial features. 57,58 Together, these findings tend to support our patients’ reported attempts to generalize trained strategies to the untrained stimuli, especially because practice effects typically result in decreased activation. 18-20,59 Further work will be needed to definitively resolve the basis of these nonspecific changes.
Conclusions and Implications
This pilot study provides the first evidence, to our knowledge, that the beneficial effect of focused EMT for patients with MCI results from the increased use of distributed neural networks mediating explicit-memory functions (as hypothesized). Mere repetition of stimuli during training is unlikely to be solely responsible for the behavioral improvement because (1) we previously reported an inverse correlation between the number of training trials and subsequent memory test improvement 22 and (2) we primarily found increased activation following training as opposed to the well-established repetition suppression effect of the BOLD signal 23,25,59,60 that persists in AD. 24
The level of effort required by a task could affect activation patterns. Although using EMT strategies is effortful, we do not think that this can account for our findings because patients demonstrated significant improvements in both accuracy and RT, which suggests that the memory test was easier posttraining. EMT is, however, likely to be more difficult for those with MCI than for healthy older people. Ongoing studies by our group suggest that MCI patients demonstrate greater post-EMT increases in activation than healthy elderly individuals. However, it is currently unclear whether these changes represent compensatory activation or a relative restoration of normal activation that is associated with task performance. 52
Despite our small sample size, the robust behavioral and neural changes are quite encouraging and should serve as the basis for future studies. We anticipate that the importance of EMT for patients with MCI and AD will escalate once pharmacological agents that modify or arrest disease progression are developed and that synergistic use of such agents and rehabilitative interventions 61 will become more prevalent.
Footnotes
Acknowledgements
We wish to thank Dr Felicia Goldstein for her assistance with patient recruitment.
The contents of this manuscript do not represent the views of the Department of Veterans Affairs or the United States Government. Portions of this work were presented at the 2008 annual meetings of the International Neuropsychological Society, the American Academy of Clinical Neuropsychology, and the Society for Neuroscience. Each author provided significant intellectual contribution to warrant authorship and declares that he/she has seen and approved the final version of this manuscript. Dr Benjamin M. Hampstead had full access to all the data in the study; Dr K. Sathian had final responsibility for the decision to submit for publication.
The author(s) declared a potential conflict of interest as follows: Dr Anthony Y. Stringer is author of the Ecologically Oriented Neurorehabilitation of Memory (EON-Mem) program and receives royalties from sales. No other author has any conflict of interest.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This material is based on work supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, and Rehabilitation Research and Development Service through grants B4602H and B6366W to BH, and B3323K and B4954N to AM. This work was also funded by the Atlanta VAMC RR&D Center of Excellence. Support to KS from the Atlanta VAMC and from National Institutes of Health (NIH) grant K24 EY017332 and to XH from Georgia Research Alliance and NIH grant R01EB002009 is also gratefully acknowledged.
