Abstract
Background. Studies have demonstrated the efficacy of functional electrical stimulation in the management of foot drop after stroke. Objective. To compare changes in walking performance with the WalkAide (WA) foot-drop stimulator and a conventional ankle–foot orthosis (AFO). Methods. Individuals with stroke within the previous 12 months and residual foot drop were enrolled in a multicenter, randomized controlled, crossover trial. Subjects were assigned to 1 of 3 parallel arms for 12 weeks (6 weeks/device): arm 1 (WA–AFO), n = 38; arm 2 (AFO–WA), n = 31; arm 3 (AFO–AFO), n = 24. Primary outcomes were walking speed and Physiological Cost Index for the Figure-of-8 walking test. Secondary measures included 10-m walking speed and perceived safety during this test, general mobility, and device preference for arms 1 and 2 for continued use. Walking tests were performed with (On) and without a device (Off) at 0, 3, 6, 9, and 12 weeks. Results. Both WA and AFO had significant orthotic (On–Off difference), therapeutic (change over time when Off), and combined (change over time On vs baseline Off) effects on walking speed. An AFO also had a significant orthotic effect on Physiological Cost Index. The WA had a higher, but not significantly different therapeutic effect on speed than an AFO, whereas an AFO had a greater orthotic effect than the WA (significant at 12 weeks). Combined effects on speed after 6 weeks did not differ between devices. Users felt as safe with the WA as with an AFO, but significantly more users preferred the WA. Conclusions. Both devices produce equivalent functional gains.
Keywords
Introduction
Foot drop is a common symptom of central nervous system disorders, including stroke, spinal cord injury, and multiple sclerosis. Foot drop results in an inefficient gait pattern and often increases risk of falls as the foot does not effectively clear the ground because of weak or absent voluntary ankle dorsiflexion. 1 The conventional treatment of foot drop includes an ankle–foot orthosis (AFO), 2 a brace that passively limits ankle plantar flexion to neutral (0°). AFOs have several drawbacks (eg, they are bulky, not cosmetically pleasing, and restrict movement), which may lead to rejection of the devices.3-5
An alternative foot-drop treatment is functional electrical stimulation (FES) of the common peroneal (fibular) nerve to elicit ankle dorsiflexion during the swing phase of the step cycle. Several foot-drop stimulators are commercially available,3,5-8 and several reviews have been published on the effects of foot-drop stimulators in adults with hemiparesis.9-11 Foot-drop stimulators have an immediate orthotic effect, as the FES-induced ankle dorsiflexion improves gait biomechanics. In addition, prolonged FES use may result in physiological changes such as increased muscle strength, improved volitional control, increased joint range of motion,9,10,12 and increased walking speed, even when the device is turned off.5,6,8,13,14 Changes over time measured when the device is off have been referred to as a therapeutic effect.
Despite these documented benefits, foot-drop stimulators are not widely prescribed, likely because relatively few comparative studies of AFOs and foot-drop stimulators have been conducted. Existing studies are small scale and short term and have generally found no significant differences in walking speed between subjects using a foot-drop stimulator and an AFO after 1 session15,16 or 8 weeks.3,17 Kottink et al 7 performed a randomized controlled trial and found that the FES group had a 20% greater increase in walking speed (relative to baseline) than the AFO group after 6.5 months. The FES device in that study was a surgically implanted foot-drop stimulator.
To our knowledge, large-scale, comparative studies of orthotic and therapeutic effects achieved through use of surface stimulators and AFOs have not been conducted. The purpose of this study was therefore to compare outcomes (ie, walking speed, energy expenditure, perceived safety, and functional mobility) achieved with the WalkAide (Innovative Neurotronics, Inc, Austin, TX) foot-drop stimulator to those achieved with an AFO in a randomized controlled trial. Secondary outcomes also included information related to device preference. We hypothesized that (1) walking performance would be better (ie, higher speed and lower energy expenditure) with the device than without the device, for both devices, (2) improvements in walking performance over time would be greater with the WalkAide than an AFO, (3) orthotic effects would not differ between devices, and (4) more users would prefer a WalkAide than an AFO.
Methods
Subjects
We entered adults with hemiparesis and foot drop who were <1 year poststroke. This period is when patients would typically be fitted with a device for foot drop. Subjects were only included if they (1) had no prior experience with a foot-drop stimulator or an AFO, (2) could ambulate at least 10 m with or without an assistive device such as a cane or walker, (3) were medically stable, (4) had no expected change in medications for at least 6 months, (5) had adequate stability at the ankle during stance (with stimulation), and (6) had a Functional Independence Measure score ≥4 for ambulation.18,19 Subjects were excluded if they had (1) lower motor neuron injury resulting in inadequate response to FES; (2) history of falling more than once a week prior to the stroke; (3) fixed ankle contractures of 10° of plantar flexion with knee extended; (4) gait velocity greater than 1.2 m/s; (5) inability to operate the device safely and no caregiver assistance available; (6) need of an AFO for stance control; (7) morbid obesity reducing the response to FES because of adipose tissue (body mass index >40 kg/m2); and (8) preexisting conditions such as myocardial infarction, congestive heart failure, demand pacemaker, seizures, excessive dysesthetic pain, and severe lower extremity pathology. Enrollment occurred at 9 rehabilitation centers in the United States (see Supplementary Data A at http://nnr.sagepub.com/content/by/supplemental-data), from May 2005 to December 2008. All subjects gave written, informed consent according to the enrolling institutions’ review boards.
Study Design
This was a multicenter, randomized, controlled, parallel multi-arm, crossover trial, registered in clinicaltrials.gov (NCT00216320). The trial consisted of 2 consecutive phases of 6 weeks intervention with a crossover of devices at 6 weeks (Figure 1). Subjects were randomly assigned into 1 of 3 parallel arms. Envelopes with centrally randomized arm numbers were used to allocate subjects after they consented to participate. Subjects in arm 1 used the WalkAide first, then the AFO. Subjects in arm 2 used the AFO first, then the WalkAide. Subjects in arm 3 used an AFO in both phases. Subjects were tested at an initial visit (week 0) and at 3, 6, 9, and 12 weeks. Subjects in arms 1 and 2 were tested at 6 weeks with their phase-1 device (week 6), and the following day with their phase-2 device (labeled week 6.2). Phase-1 data included observations at weeks 0, 3, and 6; phase-2 data included observations at weeks 6.2, 9, and 12.
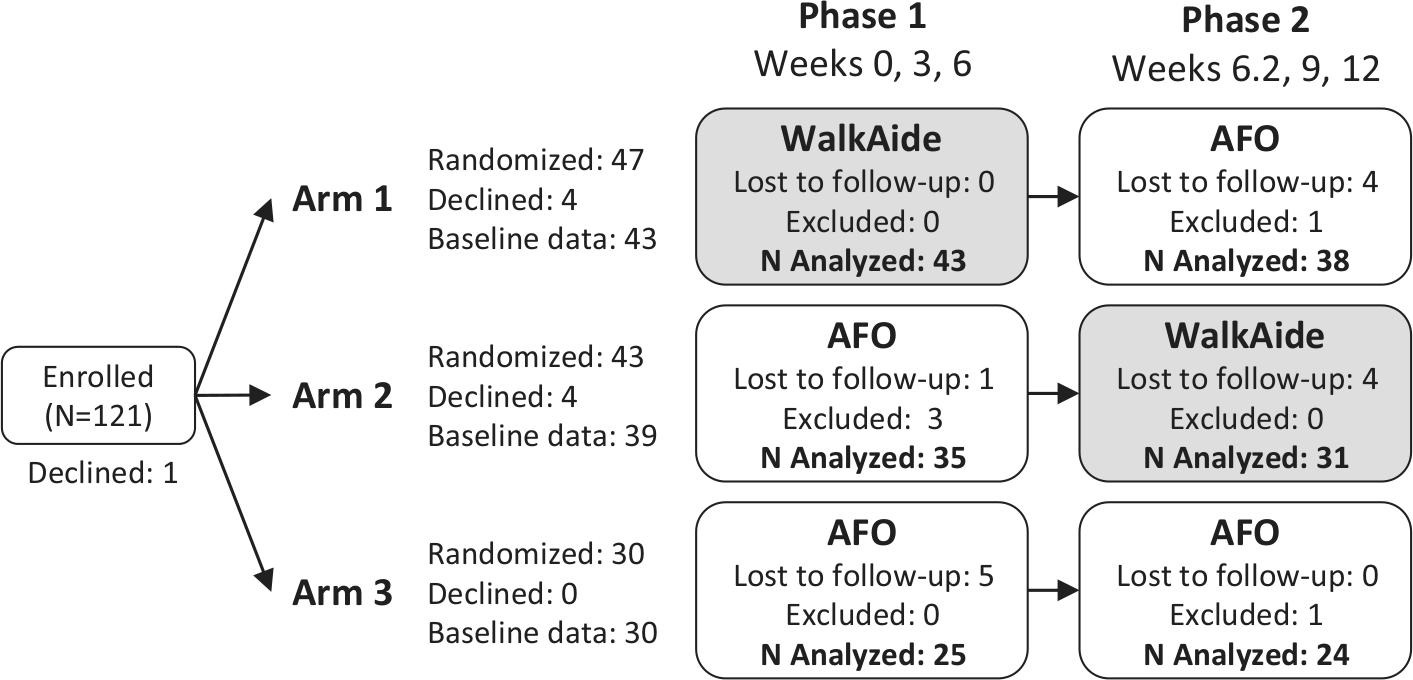
Subject flow diagram. No information was available on the number of subjects assessed for eligibility, the reasons why people declined participation after enrollment, and the reasons for dropout. A number of subjects were excluded from the analysis because of erroneously recorded data. All analysis, including the phase 1 analysis, was based on the subjects with data up to 12 weeks (AFO = ankle–foot orthosis).
At the end of phase 2 (week 12), subjects were given the opportunity to continue the trial for an additional 12 weeks. The subjects in arms 1 and 2 (ie, those who had experience with both devices) were provided with their device of choice. The arm-3 subjects continued to use an AFO. During each visit, walking performance was tested under 2 conditions—with (On) and without a device (Off)—assigned in random order.
Sample size was estimated assuming a standard deviation of 50% for figure-of-8 walking speed and an effect size of 25%. A sample size of 30 subjects per arm was desired, given a 2-sided 5% significance level, a power of 80%, and estimated attrition of 20%.
Intervention: WalkAide Foot-Drop Stimulator and Ankle–Foot Orthosis
Subjects were fitted with an AFO by a certified orthotist, or a WalkAide by a trained product specialist. After fitting, the subjects used the device on a daily basis at home and for walking in the community. The WalkAide is a self-contained FES device with built-in tilt sensor and attaches with a cuff to the leg below the knee. When the leg is tilted back at the end of stance, stimulation of the common peroneal nerve is initiated, producing dorsiflexion of the ankle to facilitate leg clearance during swing. When the leg is tilted forward at the end of the swing phase, stimulation is terminated. 6
Primary Outcome Measures
Figure-of-8 Walking Speed (Fig-8 Speed)
Subjects walked a marked, 10-m figure-8 for 4 minutes at their fastest, safe speed. The figure-8 test evaluates straight walking, bidirectional turning, and endurance. Average walking speed was calculated based on the total distance walked. 6
Physiological Cost Index
The Physiological Cost Index (PCI) was used as a measure of energy expenditure during walking. 20 Heart rate was registered before, during, and after the figure-8 test. The PCI was calculated as the difference between resting heart rate and active heart rate during walking, divided by average walking speed.
Secondary Outcome Measures
Ten-Meter Walking Speed (10-m Speed)
The test was performed twice per condition (ie, On and Off) and the average speed was calculated for each. 6
Modified Rivermead Mobility Index (Mobility Index)
Subjects were assessed on the ability to perform 8 movements, including transfers, standing, walking and climbing stairs. Scores ranged from 0 (unable to perform) to 5 (independent), with the overall score ranging from 0 to 40.21,22
Perceived Safety Level (Safety)
After the 10-m walking test, the subjects were asked to rate their perceived safety level with and without the device on a visual analog scale from 0 (very safe) to 10 (not safe at all).
Device Preference
At the end of phase 2, subjects in arms 1 and 2 were given the option to continue using either the AFO or the WalkAide for another 12 weeks. Their preference was recorded. The subjects were also asked to indicate the reason(s) for their preference on a form with 7 options (safety, convenience, confidence, comfort, function, ease of donning/doffing, other).
Data Analysis
To analyze changes between arms and phases, changes in outcome variables were calculated by subtracting the baseline value per phase measured in the Off condition. Baseline values (week 0 for phase 1, week 6.2 for phase 2) are designated as Time(0). Time(i) refers to the subsequent assessment points within each phase (weeks 3 and 6 for phase 1, weeks 9 and 12 for phase 2).
The orthotic effect for each outcome was calculated by subtracting values measured in the Off condition from those in the On condition at a given time (Time(i)On – Time(i)Off). The therapeutic effect was calculated by subtracting baseline values measured in the Off condition from those in the Off condition at a given time (Time(i)Off – Time(0)Off). Last, the combined effect was calculated by subtracting baseline values measured in the Off condition from those in the On condition at a given time (Time(i)On – Time(0)Off). Although changes over time may be due to a combination of intervention effects from the devices, spontaneous recovery, and training (see Discussion section), the terms therapeutic and combined effects were used for clarity.
Statistical analysis of subject outcomes included analyses of variance (ANOVAs) with up to 3 repeated measures factors (Phase, Time, OnOff) and 1 between-group factor (Arm). The factor OnOff refers to the difference between the On and Off conditions with regard to wearing the WalkAide or AFO. Consequently, 1-way ANOVAs were performed to analyze the simple main arm effect (ie, difference between AFO and WalkAide) at week 6 and week 12. Normality was tested for each Arm using the Kolmogorov–Smirnov statistic with Lilliefors’ significance level. When the condition of sphericity was not met (Mauchly’s test), Greenhouse–Geisser or Huynh–Feldt corrected ANOVA statistics were used if epsilon was respectively <0.75 or >0.75. Bonferroni-corrected post hoc tests were used for multiple comparisons. Frequency data were analyzed with χ2 tests. The significance level for statistical testing was .05, unless corrections were made for multiple testing, as indicated in the results. Statistical analysis was performed using SPSS (PASW Statistics 18, IBM, Armonk, NY).
Results
Subjects
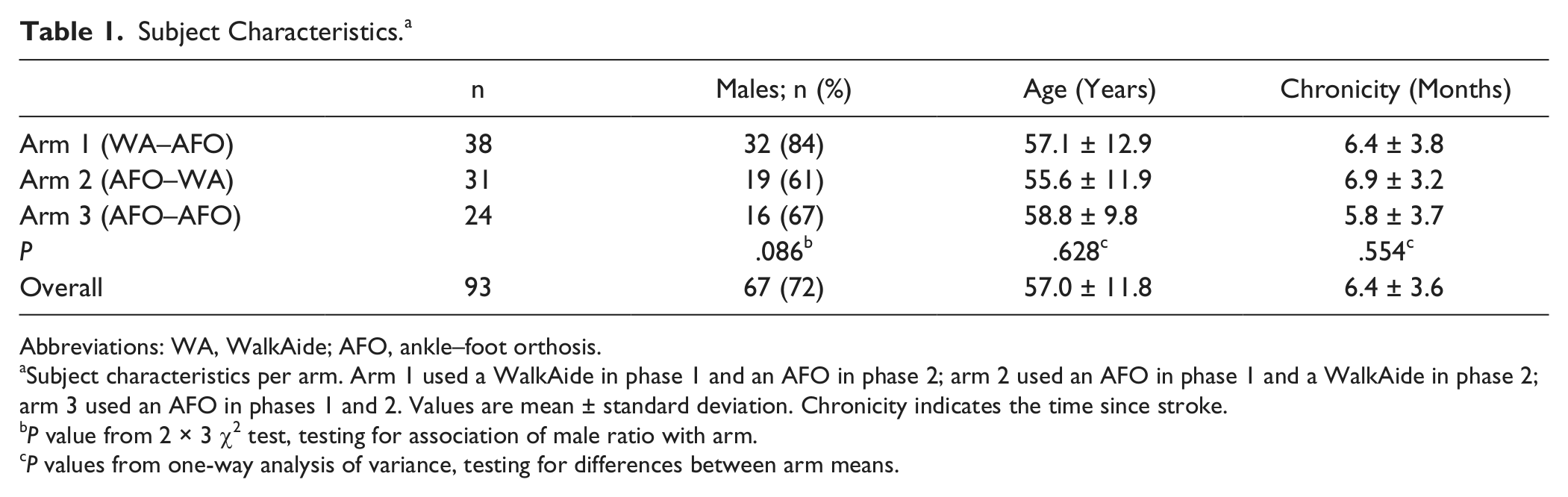
A total of 121 eligible subjects were enrolled. However, 9 subjects declined participation, 14 subjects were lost to follow-up, and 5 were excluded from the analysis (Figure 1). Final analysis was limited to 93 subjects who had data up to 12 weeks (n in arm 1 = 38; arm 2 = 31; arm 3 = 24). The number of subjects was lower for PCI and Mobility Index because of missing values, as indicated in the tables. Mean (±SD) age, time since stroke, and male ratio for all subjects were, respectively, 57 ± 12.9 years, 6.4 ± 3.6 months, and 67% males (Table 1). Subject characteristics (Table 1), Mobility Index, and the perceived safety level during walking (Table 2) did not differ significantly between arms at baseline.
Subject Characteristics. a
Abbreviations: WA, WalkAide; AFO, ankle–foot orthosis.
Subject characteristics per arm. Arm 1 used a WalkAide in phase 1 and an AFO in phase 2; arm 2 used an AFO in phase 1 and a WalkAide in phase 2; arm 3 used an AFO in phases 1 and 2. Values are mean ± standard deviation. Chronicity indicates the time since stroke.
P value from 2 × 3 χ2 test, testing for association of male ratio with arm.
P values from one-way analysis of variance, testing for differences between arm means.
Modified Rivermead Mobility Index and Perceived Safety Level. a
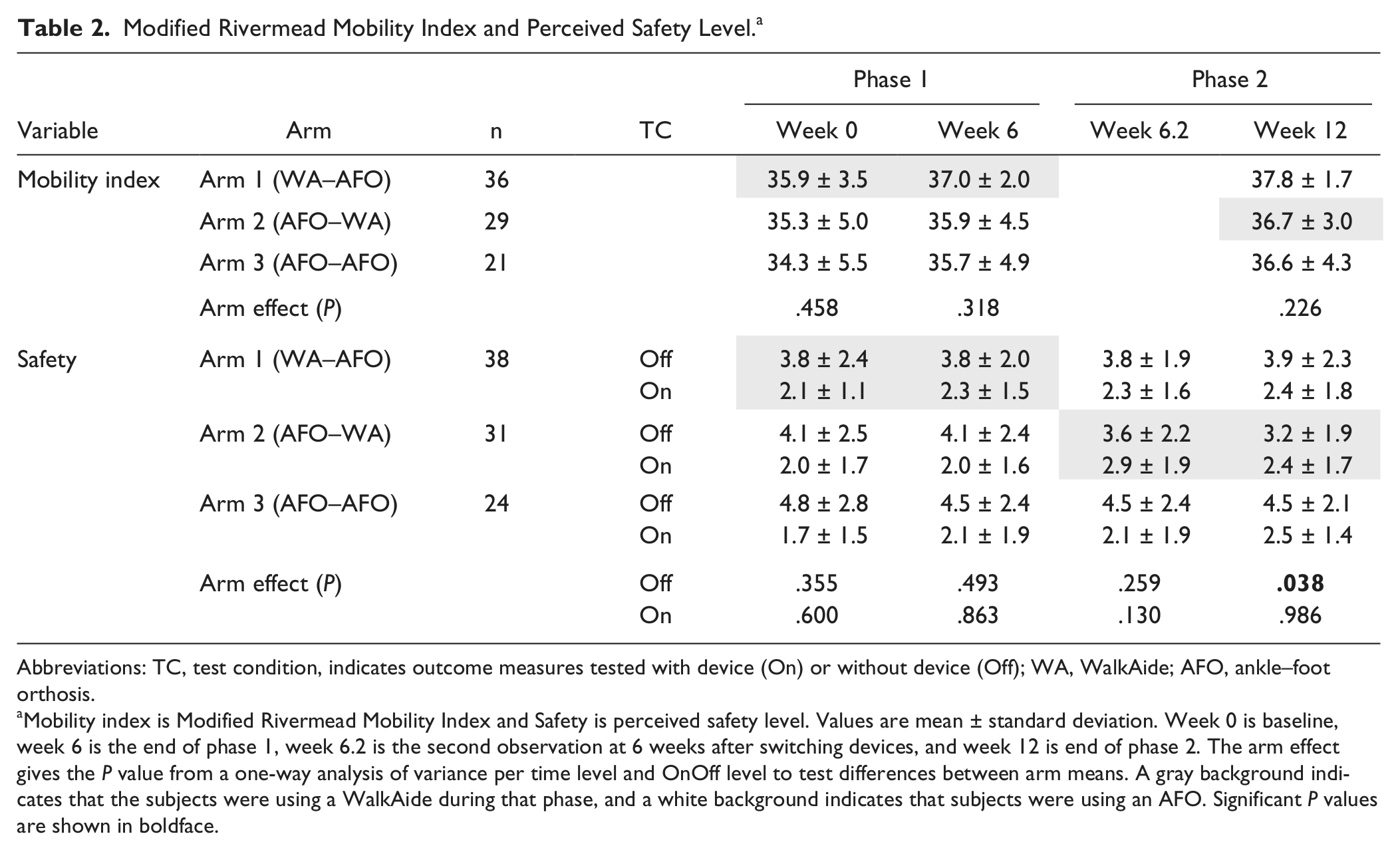
Abbreviations: TC, test condition, indicates outcome measures tested with device (On) or without device (Off); WA, WalkAide; AFO, ankle–foot orthosis.
Mobility index is Modified Rivermead Mobility Index and Safety is perceived safety level. Values are mean ± standard deviation. Week 0 is baseline, week 6 is the end of phase 1, week 6.2 is the second observation at 6 weeks after switching devices, and week 12 is end of phase 2. The arm effect gives the P value from a one-way analysis of variance per time level and OnOff level to test differences between arm means. A gray background indicates that the subjects were using a WalkAide during that phase, and a white background indicates that subjects were using an AFO. Significant P values are shown in boldface.
Walking Performance
Fig-8 and 10-m speeds without device at baseline were not significantly different between arms (arm effect, P = .21 and .32). However, despite randomization, PCI without device at baseline was significantly higher in arm 3 (AFO) than in arm 1(WalkAide; P = .024, Table 3 and Figure 2).
Figure-8 Speed, PCI, and 10-Meter Speed. a
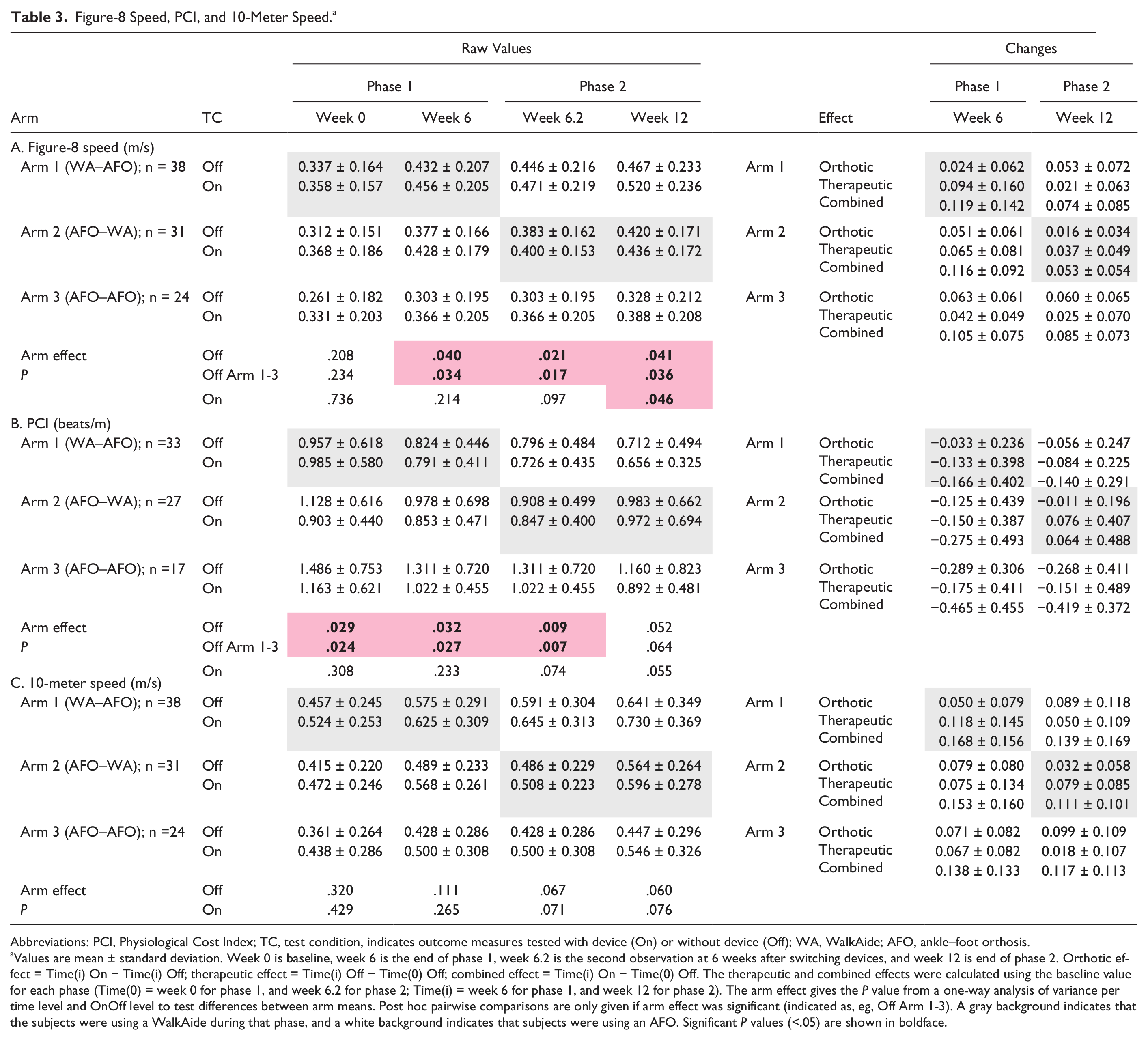
Abbreviations: PCI, Physiological Cost Index; TC, test condition, indicates outcome measures tested with device (On) or without device (Off); WA, WalkAide; AFO, ankle–foot orthosis.
Values are mean ± standard deviation. Week 0 is baseline, week 6 is the end of phase 1, week 6.2 is the second observation at 6 weeks after switching devices, and week 12 is end of phase 2. Orthotic effect = Time(i) On − Time(i) Off; therapeutic effect = Time(i) Off − Time(0) Off; combined effect = Time(i) On − Time(0) Off. The therapeutic and combined effects were calculated using the baseline value for each phase (Time(0) = week 0 for phase 1, and week 6.2 for phase 2; Time(i) = week 6 for phase 1, and week 12 for phase 2). The arm effect gives the P value from a one-way analysis of variance per time level and OnOff level to test differences between arm means. Post hoc pairwise comparisons are only given if arm effect was significant (indicated as, eg, Off Arm 1-3). A gray background indicates that the subjects were using a WalkAide during that phase, and a white background indicates that subjects were using an AFO. Significant P values (<.05) are shown in boldface.
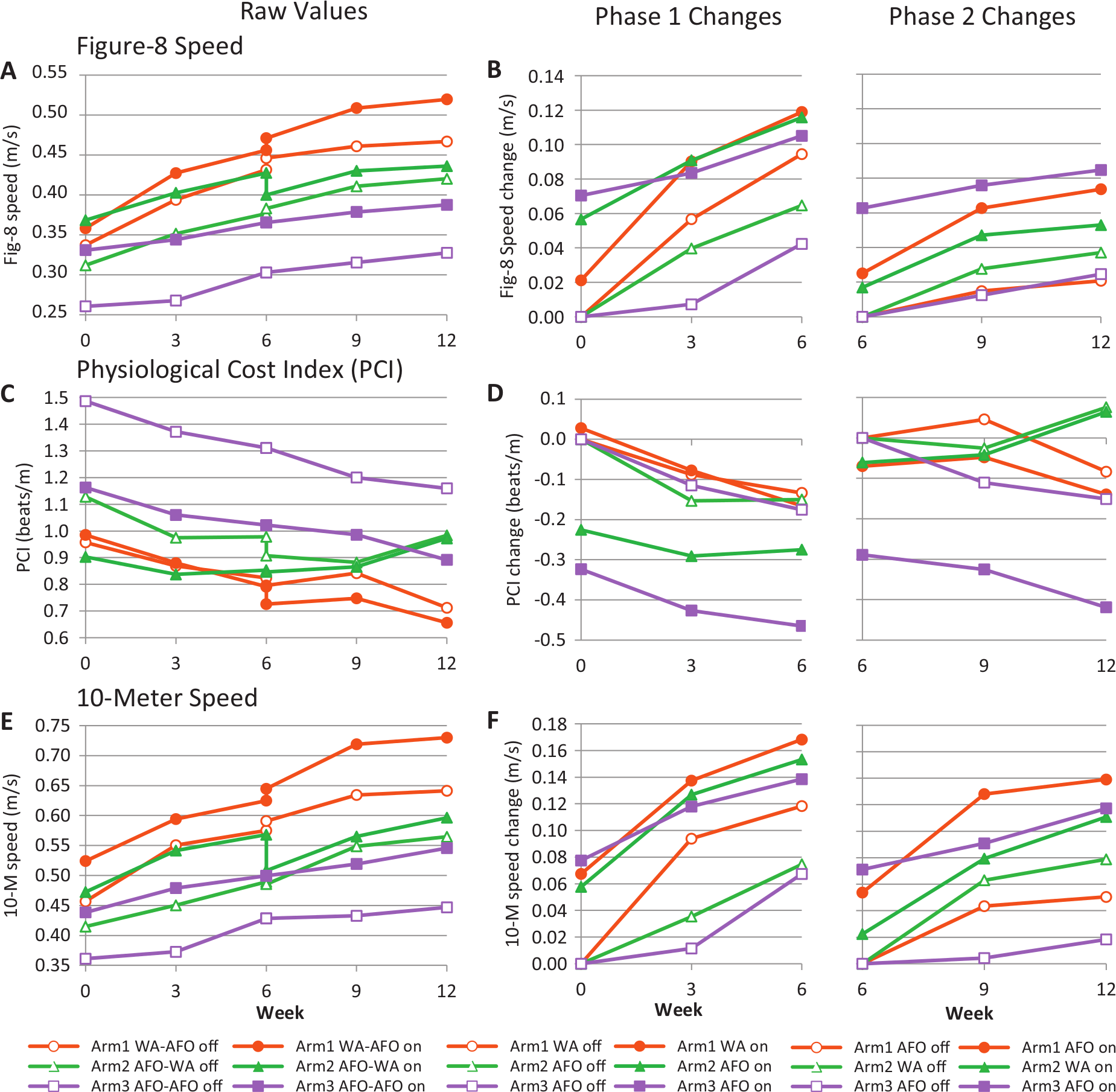
Figure-8 speed, Physiological Cost Index (PCI), and 10-meter speed. Panels A, C, and E show the outcome measures per arm from week 0 through week 12. Panels B, D, and F show the changes in phase 1 and phase 2 (note that the Y-axis scales start from 0). The changes were calculated by subtracting the value at baseline in Off condition (week 0 for phase 1, week 6.2 for phase 2). Subjects were tested at 0, 3, 6, 9, and 12 weeks. At 6 weeks, the subjects who switched device were tested with their phase-1 device, and again with their phase-2 device on the next day (week 6.2). At each time point, the subjects were tested with device (On, filled symbols) and without device (Off, open symbols). In between testing, the subjects used their ankle–foot orthosis (AFO) or WalkAide (WA) on a daily basis for walking at home and in the community. The legends indicate which device was used for each arm and phase, for example, arm 1 WA–AFO means that arm 1 used a WA in phase 1, and an AFO in phase 2. The statistical analysis corresponding with this figure is presented in Tables 4 and 5.
Fig-8 and 10-m speeds increased significantly over time in On and Off conditions in all arms in both phases (Time effect, Table 4, see Supplementary Data B at http://nnr.sagepub.com/content/by/supplemental-data). Consequently, both WalkAide and AFO had a significant therapeutic and combined effect on speed at 6 and 12 weeks. Both devices also had a significant therapeutic and combined effect on PCI, but only in phase 1. Furthermore, fig-8 and 10-m speeds were significantly higher in On than in Off condition for all 3 arms in both phases (OnOff effect, hypothesis 1, Table 4, see Supplementary Data B at http://nnr.sagepub.com/content/by/supplemental-data). Consequently, both WalkAide and AFO also had a significant orthotic effect on speed at 6 and 12 weeks. Only AFO had a significant orthotic effect on PCI.
Phase, Time, OnOff, and Arm Effect for Figure-8 Speed, PCI, and 10-Meter Speed. a
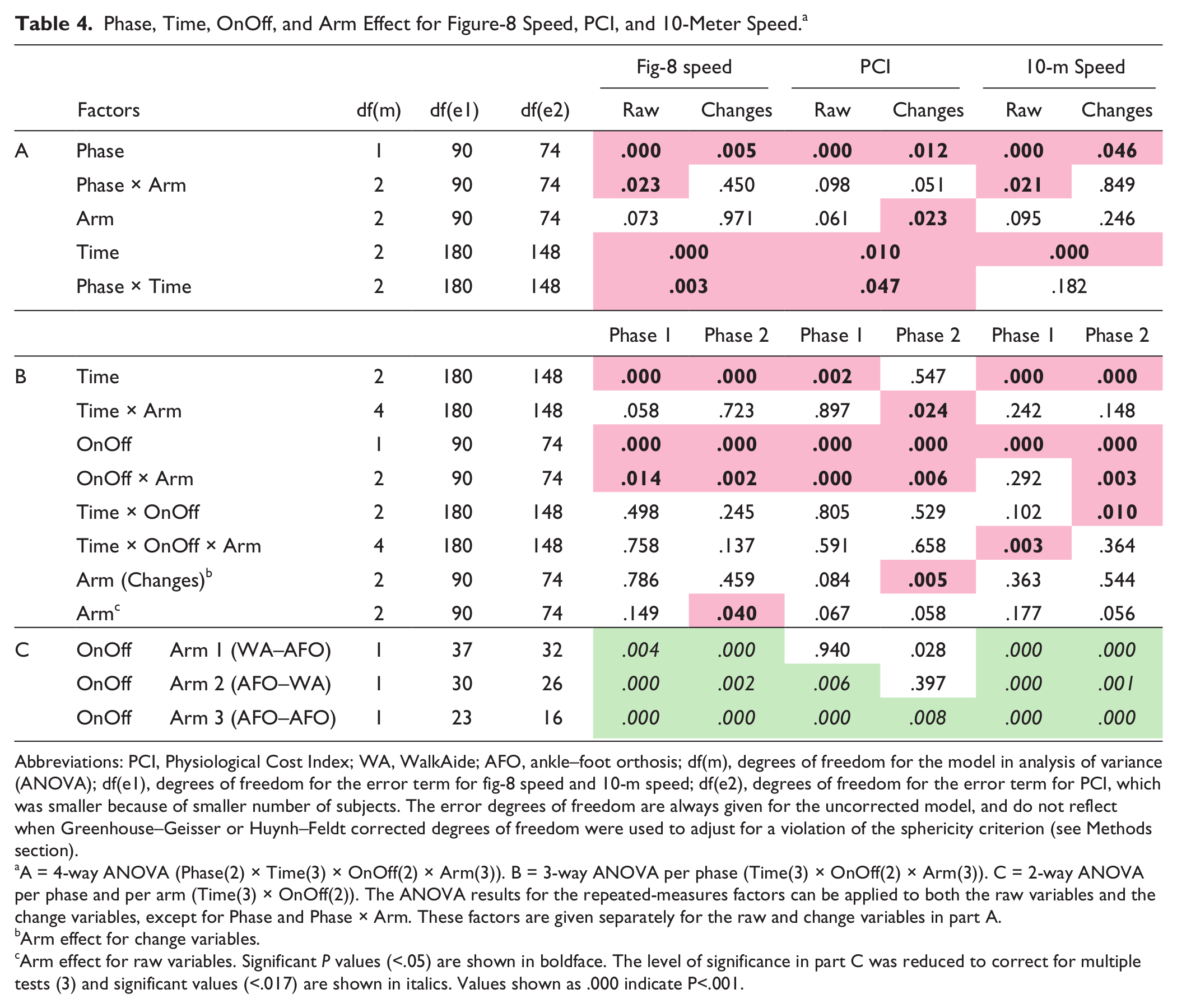
Abbreviations: PCI, Physiological Cost Index; WA, WalkAide; AFO, ankle–foot orthosis; df(m), degrees of freedom for the model in analysis of variance (ANOVA); df(e1), degrees of freedom for the error term for fig-8 speed and 10-m speed; df(e2), degrees of freedom for the error term for PCI, which was smaller because of smaller number of subjects. The error degrees of freedom are always given for the uncorrected model, and do not reflect when Greenhouse–Geisser or Huynh–Feldt corrected degrees of freedom were used to adjust for a violation of the sphericity criterion (see Methods section).
A = 4-way ANOVA (Phase(2) × Time(3) × OnOff(2) × Arm(3)). B = 3-way ANOVA per phase (Time(3) × OnOff(2) × Arm(3)). C = 2-way ANOVA per phase and per arm (Time(3) × OnOff(2)). The ANOVA results for the repeated-measures factors can be applied to both the raw variables and the change variables, except for Phase and Phase × Arm. These factors are given separately for the raw and change variables in part A.
Arm effect for change variables.
Arm effect for raw variables. Significant P values (<.05) are shown in boldface. The level of significance in part C was reduced to correct for multiple tests (3) and significant values (<.017) are shown in italics. Values shown as .000 indicate P<.001.
The observation that arms 2 and 3 (using an AFO in phase 1) also improved in Off condition suggested that those subjects experienced recovery from causes that we could not measure. The Mobility Index increased significantly over the 12 weeks for all 3 arms (Time, P < .001; Time × Arm, P = .83), which also suggested that recovery was occurring (Figure 3A). Figure 2A, C, and E show that the changes at the end of phase 1 carried over into phase 2, as there was no drop in performance while walking without a device at the crossover point (week 6.2).
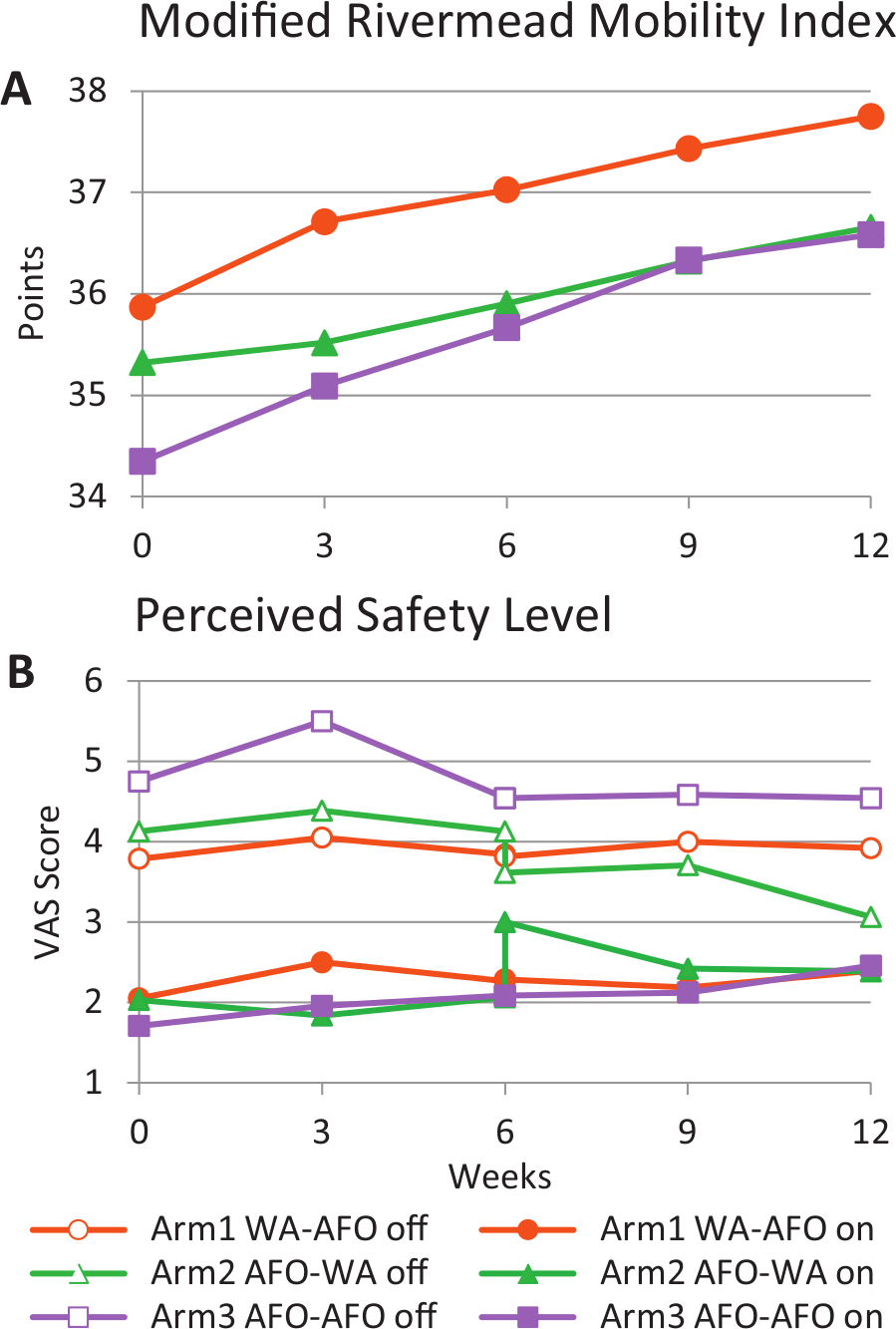
Modified Rivermead Mobility Index and Perceived Safety Level. Outcome measures per arm from week 0 through week 12. Perceived safety level was measured at each time point with device (On, filled symbols) and without device (Off, open symbols). The legends indicate which device was used for each arm and phase; for example, arm 1 WA–AFO means that arm 1 used a WA in phase 1 and an AFO in phase 2.
To eliminate the offset between the start of the 2 phases, and the offset between arms at baseline, change variables were calculated by subtracting the baseline value per phase (week 0 and week 6.2), as explained in the Methods section. The analysis described below is based on the change variables. Within-arm analysis of the change variables is presented in the Supplementary Data C (found at http://nnr.sagepub.com/content/by/supplemental-data).
Walking Performance: Comparisons Between WalkAide and Ankle–Foot Orthosis
Combined Effect
The increases in fig-8 and 10-m speeds over time while walking with the device On (hypothesis 2) were not significantly different between WalkAide and AFO users at the end of phase 1 (fig-8 speed, P = .89; 10-m speed, P = .75) or phase 2 (fig-8 speed, P = .25; 10-m speed, P = .66, see Table 5). The mean fig-8 speed increase at 6 weeks was 0.119 m/s for the WalkAide users (arm 1), and 0.116 m/s and 0.105 m/s for the AFO users in arms 2 and 3, respectively (Table 3 and Figure 2). The mean increases in 10-m speed at 6 weeks were higher than for fig-8 speed. As mentioned above, the combined effects were generally lower in phase 2. The decrease in PCI with the device On was not significantly different between WalkAide users (−0.166 beats/m) and AFO users in arms 2 and 3 (−0.275 beats/m and −0.465 beats/m, respectively) at 6 weeks (P = .089). However, the PCI decrease in phase 2 was significantly greater for the arm 3 AFO users (−0.419 beats/m) than for the arm 2 WalkAide users (0.064 beats/m, P < .001, Tables 3 and 5, Figure 2).
Comparison Between AFO and WA: Orthotic, Therapeutic, and Combined Effect on Figure-8 Speed, PCI, and 10-Meter Speed. a
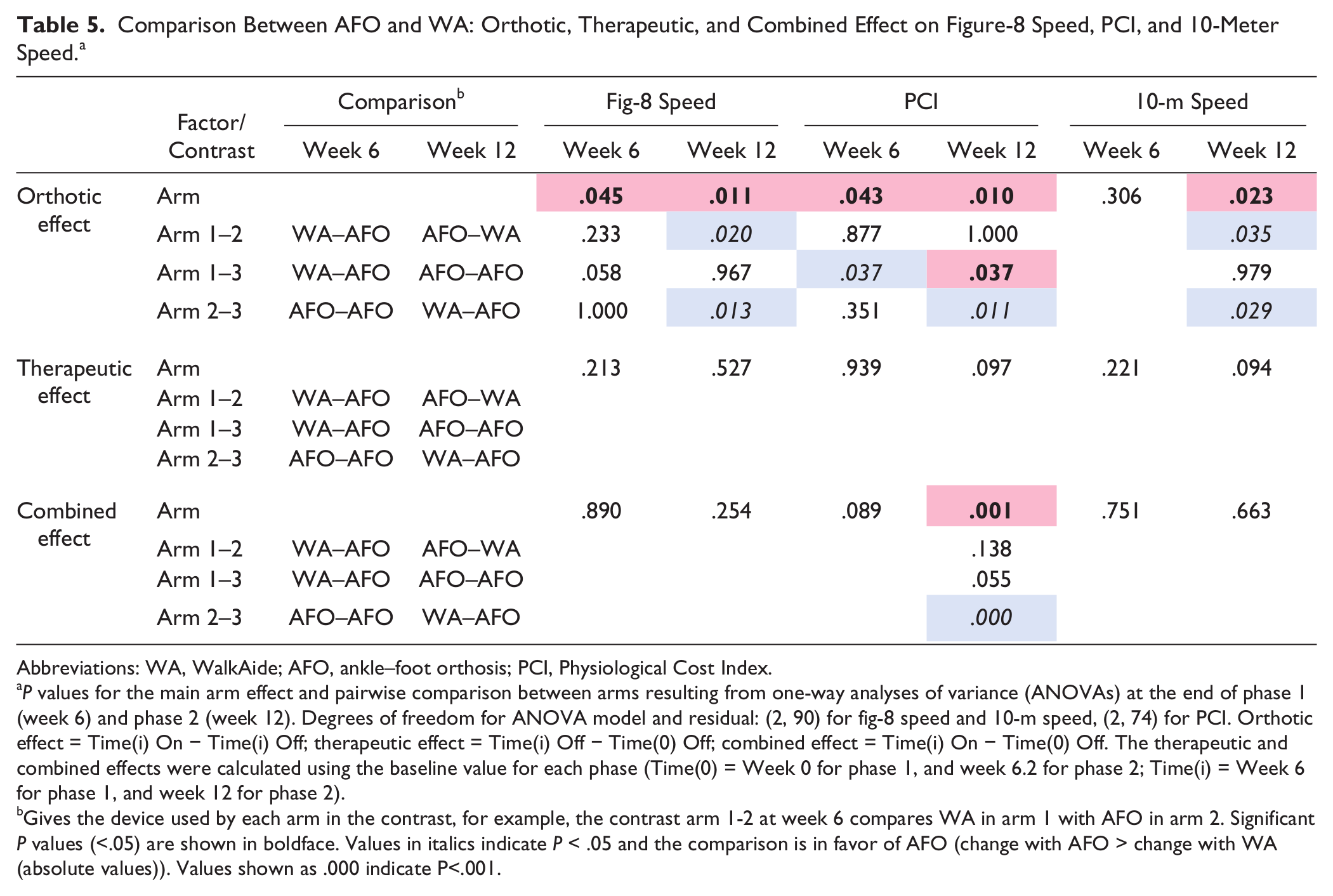
Abbreviations: WA, WalkAide; AFO, ankle–foot orthosis; PCI, Physiological Cost Index.
P values for the main arm effect and pairwise comparison between arms resulting from one-way analyses of variance (ANOVAs) at the end of phase 1 (week 6) and phase 2 (week 12). Degrees of freedom for ANOVA model and residual: (2, 90) for fig-8 speed and 10-m speed, (2, 74) for PCI. Orthotic effect = Time(i) On − Time(i) Off; therapeutic effect = Time(i) Off − Time(0) Off; combined effect = Time(i) On − Time(0) Off. The therapeutic and combined effects were calculated using the baseline value for each phase (Time(0) = Week 0 for phase 1, and week 6.2 for phase 2; Time(i) = Week 6 for phase 1, and week 12 for phase 2).
Gives the device used by each arm in the contrast, for example, the contrast arm 1-2 at week 6 compares WA in arm 1 with AFO in arm 2. Significant P values (<.05) are shown in boldface. Values in italics indicate P < .05 and the comparison is in favor of AFO (change with AFO > change with WA (absolute values)). Values shown as .000 indicate P<.001.
Therapeutic Effect
WalkAide users showed greater increases in walking speed over time when tested without a device than AFO users in both phases (hypothesis 2, increases not significant). The mean fig-8 speed increase in Off condition achieved with the WalkAide at 6 weeks (0.094 m/s) was up to twice as high as with the AFO in arms 2 and 3 (0.065 m/s and 0.042 m/s, Table 3 and Figure 2). However, differences between arms were not significant (arm effect, P = .53, Table 5). The trends were similar for 10-m speed (arm effect, P = .094). The therapeutic effect in phase 2 was generally lower than in phase 1. Changes in PCI for walking with a device Off were not significantly different between WalkAide and AFO users in either phase (Table 5).
Orthotic Effect
Ankle–foot orthosis users showed greater orthotic effects than WalkAide users at the end of phases 1 and 2 (hypothesis 3). The mean orthotic effect for fig-8 speed at 6 weeks was 2 times higher for the AFO users in arms 2 and 3 (0.051 m/s and 0.063 m/s) than for the WalkAide users (0.024 m/s, Table 3); however, the differences between arms were not significant (arm effect, P = .045, but no significant post hoc comparisons, Table 5). In phase 2, the orthotic effect was up to 3 times and significantly higher for the AFO users in arm 1 (0.053 m/s, P = .02) and arm 3 (0.060 m/s, P = .013) than for the WalkAide users (0.016 m/s). The orthotic effect on 10-m speed showed the same contrasts as for fig-8 speed in phases 1 and 2 (Tables 3 and 5). The orthotic effect on PCI was significantly higher in phases 1 and 2 for the AFO users in arm 3 (−0.289 beats/m and −0.268 beats/m) than for the WalkAide users (−0.033 beats/m and −0.011 beats/m, P = .037 and .011).
Perceived Safety Level
A 3-way ANOVA (Time × OnOff × Arm) per phase showed that there was no significant change in perceived safety level over time for any of the arms (for phases 1 and 2, respectively: Time, P = .22 and .82; Time × Arm, P = .84 and .27). Subjects in arm 2 felt significantly less safe with the WalkAide than with the AFO right after the switch at 6 weeks (visual analog scale 2.9 vs 2.0, P = .006, Figure 3, Table 2). However, there was no difference in perceived safety level between the WalkAide and AFO users while walking with the device On at the end of each phase (1-way ANOVA, arm effect, week 6, P = .86; week 12, P = .99). At the end of phase 2, WalkAide users in arm 2 felt significantly more safe than AFO users in arm 3 while walking without the device (visual analog scale 3.2 vs 4.5, P = .037).
Device Preference
At completion of phase 2, the number of subjects preferring to continue with a WalkAide, 45 out of 64 (70%), was significantly higher (P < .001, 1-way χ2, hypothesis 4) than the number preferring to continue with an AFO 19 (30%). Of the 35 arm-1 subjects who finished phase 2 with an AFO, 22 (63%) decided to switch back to the WalkAide for the next 12 weeks. Significantly fewer (21%, P = .001, 2-way χ2) of the 29 arm-2 subjects who finished phase 2 with the WalkAide elected to return to an AFO; most (79%) kept the WalkAide for another 12 weeks. The reasons given most often for choosing a device included function, confidence, comfort, convenience, easy donning and doffing, and safety by more than half for each preference.
Adverse Events
A total of 27 adverse events were reported by 21 of the 112 subjects during their participation in the trial. Seven of the 69 subjects (10.1%) using a WalkAide (arms 1 and 2) developed skin irritation localized under the electrodes, but none required medical treatment. Two of the 93 subjects (2.2%) using an AFO (arms 1, 2, and 3) developed skin irritation due to friction. Five falls were reported but only 2 were related to a device (1 was attributed to use of an AFO and 1 to use of a WalkAide). Three severe adverse events occurred that were not related to the interventions (fatal myocardial infarction, wrist fracture, and an unspecified cardiac condition).
Discussion
Comparisons Between WalkAide and Ankle–Foot Orthosis
The main finding of this study was that there was no significant difference between the WalkAide and AFO in terms of gains in walking speed tested with the device On after 6 weeks of use (arm effect was not significant for combined effect). This means that the overall functional gain experienced by the subjects at 6 weeks was similar for both devices. However, the mechanisms behind the measured functional gains seemed to differ. After 6 weeks in phase 1, the fig-8 speed increase with device On in the AFO users had a 2 times smaller contribution (although not significant) from therapeutic effect, but a 2 times greater contribution (also not significant) from orthotic effect than in the WalkAide users. The net result was that the combined effect for both devices was similar. The scenario was similar after 6 weeks use in phase 2 but with a larger and significant difference in orthotic effect between devices (AFO > WalkAide). The 10-m speed showed the same scenario, but differences in orthotic effect between devices were less pronounced (<60%) than for fig-8 speed. PCI changes did not show the difference in contributions from orthotic and therapeutic effects for WalkAide and AFO.
A previous study suggested that the therapeutic effect after long-term use of a foot-drop stimulator arises from strengthening of residual corticospinal connections that remain after a stroke. 12 In short, the authors suggested that electrical stimulation of sensory and motor pathways on a daily basis during walking can strengthen residual pathways, which results in increased strength and voluntary control. Consequently, people can walk faster and safer even when the stimulation is not being used. A passive brace such as an AFO does not facilitate these pathways and so has no therapeutic effect and no functional benefit when the brace is removed. On the other hand, because an AFO mechanically limits or completely blocks ankle movement, it provides robust, instantaneous and permanent support, regardless of user condition or attention level. The effects of the WalkAide and AFO were consistent with smaller studies in diverse participants with stroke.6,16,23
Perceived Safety and Device Preference
WalkAide users generally felt safer than AFO users when they were not using the device at the end of Phase 2. This is consistent with the idea that the WalkAide has a therapeutic effect resulting in increased muscle strength and voluntary ankle control. 12 The increased levels of control may reduce the risk of falling in situations where people typically do not use an orthosis (eg, to walk to the bathroom at night).
The majority of subjects who had used both devices for 6 weeks chose the WalkAide as the device they preferred to use for another 12 weeks after finishing phase 2. The majority of subjects who ended phase 2 using an AFO preferred to switch back to the WalkAide for the remainder of the study. Two other recent studies also reported that participants were more satisfied with their foot-drop stimulator than with their AFO regarding effort, stability, and safety of walking, quality of gait, walking distance, comfort of wearing, appearance, and ability to move ankle freely.17,24
Limitations
Crossover Design
This is the first randomized controlled trial that compared the walking performance of persons after stroke wearing the conventional intervention for foot drop (AFO) to those wearing an external foot-drop stimulator. The trial was designed as a crossover study so that both devices were used for 6 weeks by the same subjects, which mitigates intersubject variability. However, we found that changes in walking performance observed in phase 1 carried over into phase 2. This impeded intra-arm comparisons, as the effects in phase 2 were because of a combination of the phase-1 device (eg, in arm 1: WalkAide) and phase-2 device (AFO). Therefore, comparisons between interventions (WalkAide vs AFO) were limited to interarm analyses per phase. By calculating change scores relative to the baseline of each phase, we could analyze data from both phases in 2 independent blocks.
Subject Randomization
Despite randomization, subjects in arm 3 (AFO–AFO) differed from arm 1 (WalkAide–AFO) and arm 2 (AFO–WalkAide) in some respects at baseline (lower walking speed and mobility index, higher PCI). Although the use of change scores eliminated the offset between arms at baseline, comparisons of arm 3 with the other arms must be interpreted with caution. However, arms 1 and 2 were comparable at baseline and therefore allowed for a legitimate comparison between WalkAide and AFO, as both arms used both devices.
Early Intervention
This is also the first study comparing a foot-drop stimulator with an AFO in the earlier stages after stroke. The subjects were enrolled within a year after their stroke, which is earlier than most other studies (see overview in Stein et al 6 ). We believed that application of an earlier intervention might produce a greater therapeutic effect. Walking performance improved over time in all 3 Arms, including the subjects using an AFO. Therefore, the improvements might have been due to spontaneous, natural neurological recovery; training effects related to walking practice; or increased motivation. Therapeutic effects reported may represent specific intervention effects from the device, combined with natural recovery and training effects.
Subject Variability
Although some of the differences in walking parameters between arms were large, many comparisons were not significant because of the large between- subject variability as shown in Table 3. The coefficients of variation for fig-8 speed, 10-m speed, and PCI ranged from 38% to 73% across the 3 arms and time points. Such variability in walking performance within a year after stroke may be expected in subjects who had strokes of different severities.
Conclusion
Both the WalkAide and AFO had significant orthotic, therapeutic and combined effects. The WalkAide had a larger therapeutic effect over time, whereas the AFO had a larger immediate orthotic effect. Both devices produced similar functional gains after 6 weeks use (combined effect). People felt as safe with the WalkAide as with an AFO, but more people preferred the WalkAide.
Footnotes
Acknowledgements
We thank the staff at all participating medical centers for their help in collecting the data.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Stein and Kufta are consultants to Innovative Neurotronics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial was sponsored by Innovative Neurotronics Inc, Austin, Texas, which manufactures and sells the WalkAide foot-drop stimulator used in this trial. Innovative Neurotronics is a subsidiary of Hanger Orthopedic Group. Most ankle–foot orthoses used in this trial were fabricated by orthotists associated with the Hanger Orthopedic Group.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
