Abstract
Background. Evidence supports peroneal nerve functional electrical stimulation (FES) as an effective alternative to ankle foot orthoses (AFO) for treatment of foot drop poststroke, but few long-term, randomized controlled comparisons exist. Objective. Compare changes in gait quality and function between FES and AFOs in individuals with foot drop poststroke over a 12-month period. Methods. Follow-up analysis of an unblinded randomized controlled trial (ClinicalTrials.gov #NCT01087957) conducted at 30 rehabilitation centers comparing FES to AFOs over 6 months. Subjects continued to wear their randomized device for another 6 months to final 12-month assessments. Subjects used study devices for all home and community ambulation. Multiply imputed intention-to-treat analyses were utilized; primary endpoints were tested for noninferiority and secondary endpoints for superiority. Primary endpoints: 10 Meter Walk Test (10MWT) and device-related serious adverse event rate. Secondary endpoints: 6-Minute Walk Test (6MWT), GaitRite Functional Ambulation Profile, and Modified Emory Functional Ambulation Profile (mEFAP). Results. A total of 495 subjects were randomized, and 384 completed the 12-month follow-up. FES proved noninferior to AFOs for all primary endpoints. Both FES and AFO groups showed statistically and clinically significant improvement for 10MWT compared with initial measurement. No statistically significant between-group differences were found for primary or secondary endpoints. The FES group demonstrated statistically significant improvements for 6MWT and mEFAP Stair-time subscore. Conclusions. At 12 months, both FES and AFOs continue to demonstrate equivalent gains in gait speed. Results suggest that long-term FES use may lead to additional improvements in walking endurance and functional ambulation; further research is needed to confirm these findings.
Keywords
Introduction
Every year 795 000 individuals in the United States and 15 million worldwide experience a new stroke.1,2 With a 75% overall survival rate 1 and an estimated yearly cost of 3.6 billion dollars, 3 stroke has a significant and serious impact on public health and health care costs. Seventy percent of stroke survivors regain the ability to walk, but residual impairments such as spasticity, muscle weakness, and poor balance may persist and limit functional ambulation. 4 Foot drop, a significant gait impairment affecting approximately 20% of all stroke survivors, is a consequence of spastic hemiparesis from stroke. 5 Foot drop, typically due to a combination of ankle dorsiflexor muscle weakness and plantarflexor muscle spasticity, results in a slower, less efficient gait and increases fall risk. 6 Walking limitations have a continued and pervasive impact on functional mobility, community integration, and quality of life (QoL) for stroke survivors; 4 many accomplish independent ambulation only by using an assistive device. 4
Traditionally, the standard of care for foot drop is the ankle foot orthosis (AFO). 7 An AFO, usually made of polypropylene or carbon fiber, is a lower leg brace worn to hold the foot and ankle in the neutral position. Published evidence demonstrates the effectiveness of AFOs for improving speed, 8 gait stability,9,10 symmetry,9,11 and static balance 12 in individuals with neurologic gait impairment. However, AFOs have the disadvantages of limiting ankle mobility 13 and decreasing muscle activity,9,14 which may impair dynamic balance.12,13 Functional Electrical Stimulation (FES), the use of neuromuscular electrical stimulation to activate muscles during functional tasks, is an alternative to the AFO. 15 FES applied to the peroneal nerve during the swing phase of ambulation provides active dorsiflexion and can reduce foot drop by facilitating voluntary muscle activity, 16 and improving gait quality and symmetry.15,17 Disadvantages of FES are increased possibility of skin irritation,18,19 difficulty with electrode placement,18,20,21 and stimulus discomfort. 19
Published literature also supports the efficacy of peroneal nerve FES. Several studies have demonstrated that FES utilization can increase gait speed,15,20,22-24 decrease energy expenditure during walking,15,20,22-24 and improve gait symmetry.15,17,25 Previous evidence in support of both AFOs and FES originates from nonrandomized pre–post intervention studies, but 4 recently published randomized controlled trials (RCTs) comparing FES to AFOs have added higher quality evidence to the literature.7,26-28 Additionally, several studies have investigated the long-term effects of FES with follow-up periods ranging from 6 to 7 months7,27,29 to 1 year,16,20-22,25,28,30 with the longest describing a follow-up of 10 years. 31 Only 3 of these long-term studies were RCTs that compared FES to AFOs.7,27,28 Sheffler et al 7 compared use of FES to a control group (wearing AFO or no device) for only 12 weeks, but reassessed subjects at 12 and 24 weeks after device wear ended to determine the durability of changes in motor impairment, functional mobility, and QoL. Kluding et al 27 followed subjects for 7.5 months of full-time device wear and Bethoux et al 28 for 6 months; both studies assessed changes in gait speed and function, endurance, balance, and QoL. However, there is currently no published RCT comparing FES and AFOs in terms of safety and efficacy up to 1 year.
This article reports the results of a 12-month follow-up to the RCT. Primary endpoint (6 months) analysis results were previously published by Bethoux et al. 28 The main objective of this follow-up analysis was to assess if the noninferiority of the WalkAide (WA; Innovative Neurotronics, Austin, TX) compared to the AFO at 6 months, on gait speed and serious adverse event (SAE) rate, was still observed at 12 months. The secondary objective was to assess the magnitude of changes in efficacy measures at 12 months and to compare them to the changes observed at 6 months.
Methods
Study Design
This multicenter, unblinded, parallel-group RCT (ClinicalTrials.gov #NCT01087957) was approved by the Western Institutional Review Board and conducted in 30 rehabilitation research centers in the United States. 28 Individuals with foot drop secondary to stroke meeting eligibility criteria (see Table 1) were enrolled and randomized into 1 of 2 groups, FES or AFO. Screening, consent, randomization, and device fitting processes are described in Bethoux et al. 28 Subjects included in this study were required to complete all baseline and 12-month assessments; long-term changes in gait speed, quality, endurance, and functional mobility were compared. This study assessed changes in outcome measures between screening (without device) or baseline (with device) and the 12-month follow-up visit (with device).
INSTRIDE Inclusion and Exclusion Criteria.
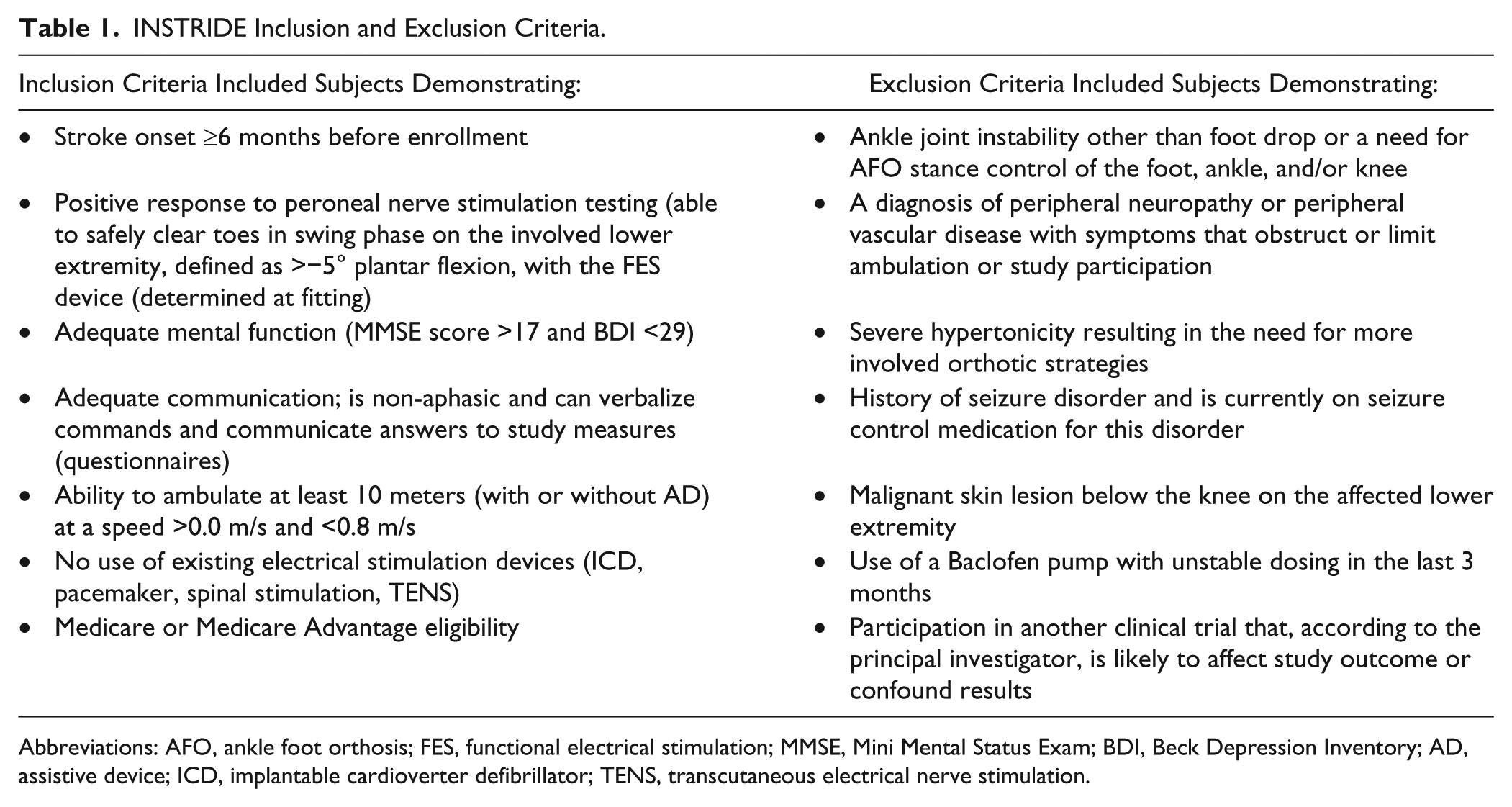
Abbreviations: AFO, ankle foot orthosis; FES, functional electrical stimulation; MMSE, Mini Mental Status Exam; BDI, Beck Depression Inventory; AD, assistive device; ICD, implantable cardioverter defibrillator; TENS, transcutaneous electrical nerve stimulation.
Clinical Evaluation and Measurement
Subjects were fitted with either FES or an AFO for use during community and household ambulation within 2 weeks of randomization. The FES device, the WalkAide (WA; Innovative Neurotronics, Austin, TX), is a commercially available battery-operated, single-channel electrical stimulator approved by the US Food and Drug Administration and CE marked in Europe as treatment for foot drop. The WA, worn on the proximal lower leg, utilizes a tilt sensor and accelerometer to trigger ankle dorsiflexion and control the duration and timing of peroneal nerve stimulation during the swing phase of gait. Individuals randomized to the AFO group continued wearing their own prescribed orthosis if determined by an on-site certified orthotist to adequately alleviate foot drop. AFOs provided for participants needing a new or replacement orthotic device were articulated or solid based on the orthotist’s clinical assessment and judgment. For all walking performance tests, subjects were instructed to use their assigned device and any necessary assistive device (eg, cane, walker).
The predefined primary outcomes of this 12-month follow-up analysis were gait speed, calculated from time to complete the 10-Meter Walk Test (10MWT), and device-related SAE rate. The 10MWT, validated for the stroke population, is accepted as a responsive, functional measure of a patient’s gait speed over short distances typical of a household setting. 32 The device-related SAE rate was utilized to measure the safety of each device over long-term use. SAEs were defined as serious deteriorations in a subject’s health that resulted in life-threatening illness, injury, or permanent impairment of body structure or function and required inpatient hospitalization, prolongation of existing hospitalization, or resulted in medical or surgical intervention to prevent permanent impairment. To be considered device-related, the SAE was determined to have occurred as a direct result of wearing the study device as assigned. SAE severity and their relatedness to the device were adjudicated by an independent Clinical Events Committee, based on information provided by each study site. We hypothesized that the WA would prove noninferior to the AFO at 12 months on the primary outcomes defined above.
The secondary outcomes, the 6-Minute Walk Test (6MWT), GaitRite Functional Ambulation Profile (FAP), and Modified Emory Functional Ambulation Profile (mEFAP), provided an assessment of walking endurance over longer distances (6MWT), 33 gait quality (FAP), 34 and the ability to perform functional ambulation tasks (mEFAP). 35 All secondary outcomes were shown to be reliable and valid for the stroke population.33-35
The 6MWT was performed on a standardized course. Subjects walked for 6 minutes along a 30-meter course with turns at each end; the course was marked every 3 meters to allow accurate measure of distance walked. The GaitRite FAP was calculated using the GaitRite instrumented mat; subjects walked across the mat 5 times and the trials averaged to produce the GaitRite FAP score. The mEFAP consisted of 5 subtasks: (a) a 5-m walk on a hard floor, (b) a 5-m walk on carpet, (c) a Timed Up and Go, (d) the navigation of a standardized obstacle course, and (e) the ascent and descent of 4 stairs. The total mEFAP was calculated using the sum of the 5 subtasks. To adjust for varying functional levels, the mEFAP uses multipliers to account for use of assistive devices or assistance. For this study, all subtask times were multiplied by 2 to account for the study devices (AFO or WA). If subjects used an assistive device, the multiplier for the use of the study device was added to that of the assistive device use. Therefore, use of a cane with the study device was assessed a multiplier of 5; use of a hemi walker or quad cane with the study device was assessed a multiplier of 6. Total and subtask mEFAP scores are expressed as seconds after adjustment with the multipliers.
Initial measurements were collected at the screening visit (without device) for the 10MWT and at the baseline visit (2 weeks later, with device) for the 6MWT, FAP, and mEFAP, and compared to 12-month measurements with device. All GaitRite walking trials utilized standardized instructions for walking on the mat and were processed and analyzed by a central lab.
Sample Size Calculation
A power analysis was performed for the original 6-month analysis based on estimates required to adequately power the primary endpoints. 28 The device-related SAE rate yielded the largest sample size estimate, 198 per group, based on a power of 80% to detect a difference between groups in device-related SAE rate of no more than 3% (1-sided Blackwelder’s noninferiority test of difference in proportions, α = .05). Correction for an estimated attrition rate of 20% resulted in a minimum total sample size of 495 (approximately 248 per group).
Statistical Analysis
Study data were analyzed using both per-protocol (only complete subject data analyzed) and intention-to-treat (ITT) analyses (all subject data analyzed with missing data points calculated using multiple imputation). The 2 analyses showed minimal differences in results; to preserve randomization and mitigate impact of bias resulting from subject dropout, only the ITT results are presented, similar to the 6-month analysis. 28 To account for potential clustering at the site level, all ITT analyses were conducted on multiply imputed data via mixed-effect regression models with the treatment arm as the fixed effect and the study center as the random effect.
The study protocol mandated a noninferiority analysis of the primary endpoints and, if noninferiority was met, a superiority analysis of the chosen secondary endpoints. 28 The WA and AFO groups were compared at 12 months on the primary efficacy endpoint of gait velocity and the primary safety endpoint of device-related SAE rate. The noninferiority margin was considered met if the WA group demonstrated, with 95% confidence, both a change in gait velocity no more than 0.2 m/s below that of the AFO group (based on the classification of ambulation categories poststroke by Perry et al 36 ), and a device-related SAE rate no greater than 3% above that of the AFO group.
Several secondary endpoints were scheduled for a 12-month analysis providing noninferiority was met: 6MWT distance, total and subtask mEFAP scores, and FAP score standard t tests. Blackwelder’s t or Z tests were used to test changes from baseline or percent change from baseline. Due to multiple comparisons, a Bonferroni adjustment set the level of statistical significance for secondary endpoints at P = .002. Changes in Perry ambulation category classification were demonstrated using a McNemar’s test.
Results
Subjects
A total of 384 subjects, out of the 495 enrolled, completed the 12-month follow-up visit (180 in the WA group and 204 in the AFO group; see Table 2 for subject characteristics). Ninety-six subjects (55 WA and 41 AFO group subjects) dropped out in the first 6 months. Only 15 additional subjects exited between 6 and 12 months (7 WA and 8 AFO group subjects). Figure 1 details participant flow from baseline to 12 months and identifies sources of dropout. Initially, only gait speed (P = .05) and body mass index (BMI; P = .006) were significantly different between groups. Despite randomization procedures, initial gait speed in the WA group was 0.04 m/s slower than that of the AFO group. While statistically significant, this difference was not considered clinically significant since it was not large enough to differentiate between ambulatory classification category, 36 nor did it exceed the range of minimal clinically important differences (MCIDs) for gait speed established for the stroke population.37,38 The difference in BMI (lower for the WA group) was not sufficient to differentiate between BMI categories 39 and was not considered clinically significant. These variables were therefore considered balanced in the analyses.
Subject Demographics and Characteristics at Screening a .
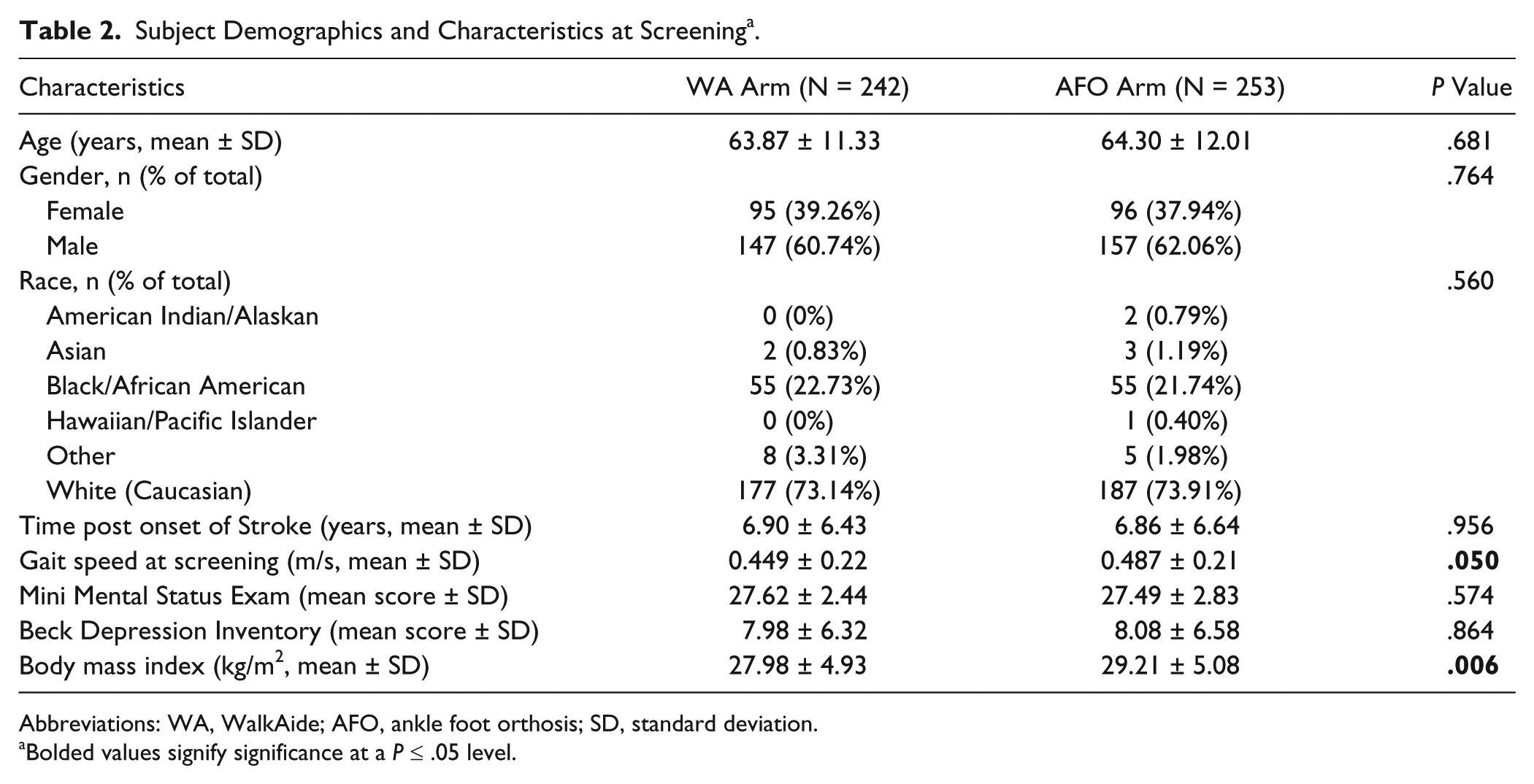
Abbreviations: WA, WalkAide; AFO, ankle foot orthosis; SD, standard deviation.
Bolded values signify significance at a P ≤ .05 level.
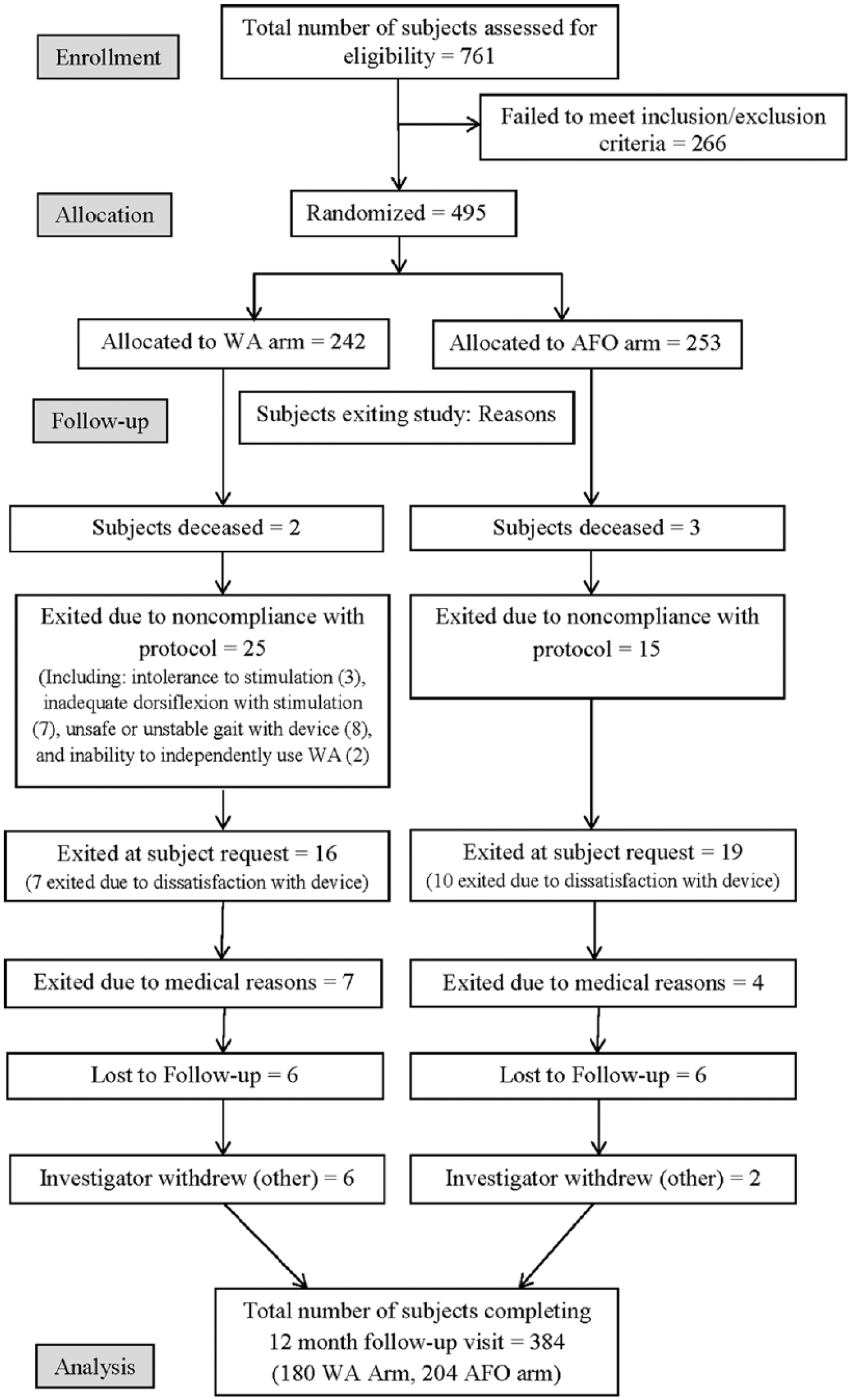
Participant flow diagram from baseline to 12 months.
Change in Primary Endpoints Between Initial and 12-Month Visits
The results presented examine the magnitude of changes between the initial assessment and the 12-month time point. The WA was noninferior to the AFO at 12 months on both gait velocity and device-related SAE rate. The mean 12-month difference in gait velocity between groups was 0.026 m/s (95% confidence interval = −0.02, 0.07). The lower confidence interval limit, −0.02, exceeded the set inferiority margin (−0.2 m/s) demonstrating that the WA was noninferior to the AFO. Both the WA and AFO groups showed statistically significant increases in gait velocity at 12 months as compared to screening measures (0.198 and 0.172 m/s, respectively, P < .001); no statistically significant between-group differences were observed (see Table 3).
Changes in Primary and Secondary Endpoints (Multiply Imputed Data N = 495).
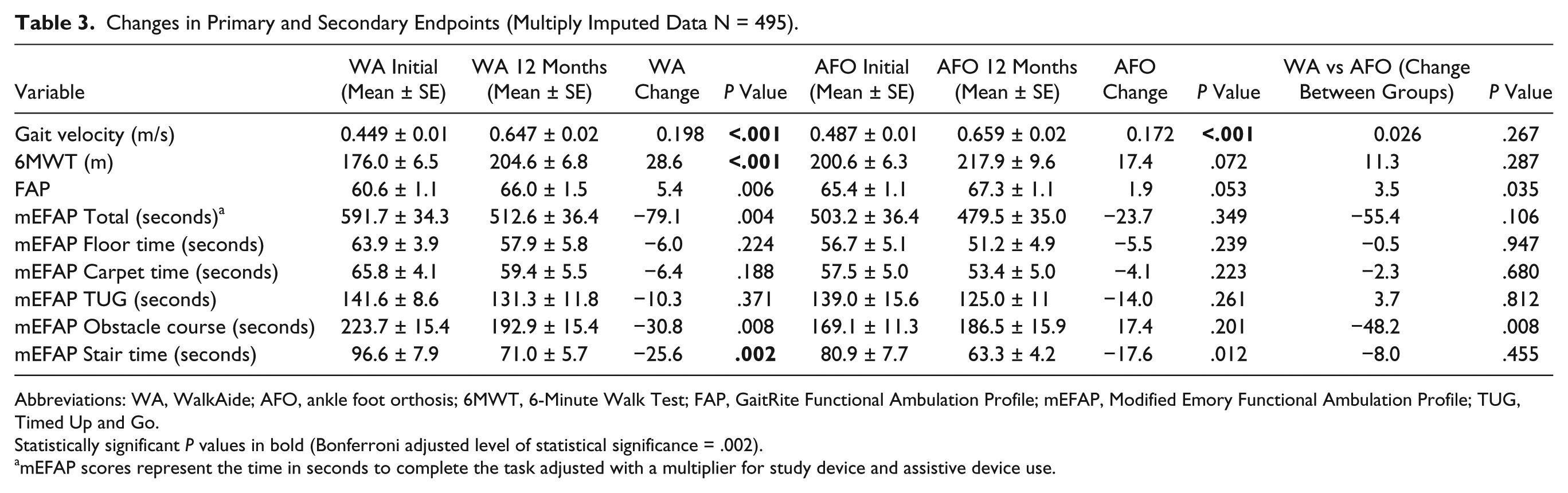
Abbreviations: WA, WalkAide; AFO, ankle foot orthosis; 6MWT, 6-Minute Walk Test; FAP, GaitRite Functional Ambulation Profile; mEFAP, Modified Emory Functional Ambulation Profile; TUG, Timed Up and Go.
Statistically significant P values in bold (Bonferroni adjusted level of statistical significance = .002).
mEFAP scores represent the time in seconds to complete the task adjusted with a multiplier for study device and assistive device use.
At 12 months there were 3 device-related SAEs in the AFO group and 1 in the WA group. The percentage of device-related SAEs (number of device-related SAEs divided by total number of SAEs per group) was 3.6% for the WA group and 13.6% for the AFO group, demonstrating that the WA was noninferior to the AFO. The number of device-related SAEs per group was too small to be statistically relevant; therefore, no between- or within-group device-related SAE rate differences were computed. All SAEs in this study were falls, and were adjudicated by a Clinical Events Committee (CEC) for relatedness to the study device. The CEC deemed all falls to be “possibly related” regardless of whether the study device was worn at the time of the fall or not. All 3 subjects in the AFO group experiencing serious falls were wearing their study device at the time; the one subject experiencing a serious fall in the WA group was not wearing their study device at the time of the fall. Table 4 summarizes all adverse events occurring between baseline and 12 months with a frequency ≥2%.
Summary of Adverse Events: Baseline to 12 Months.
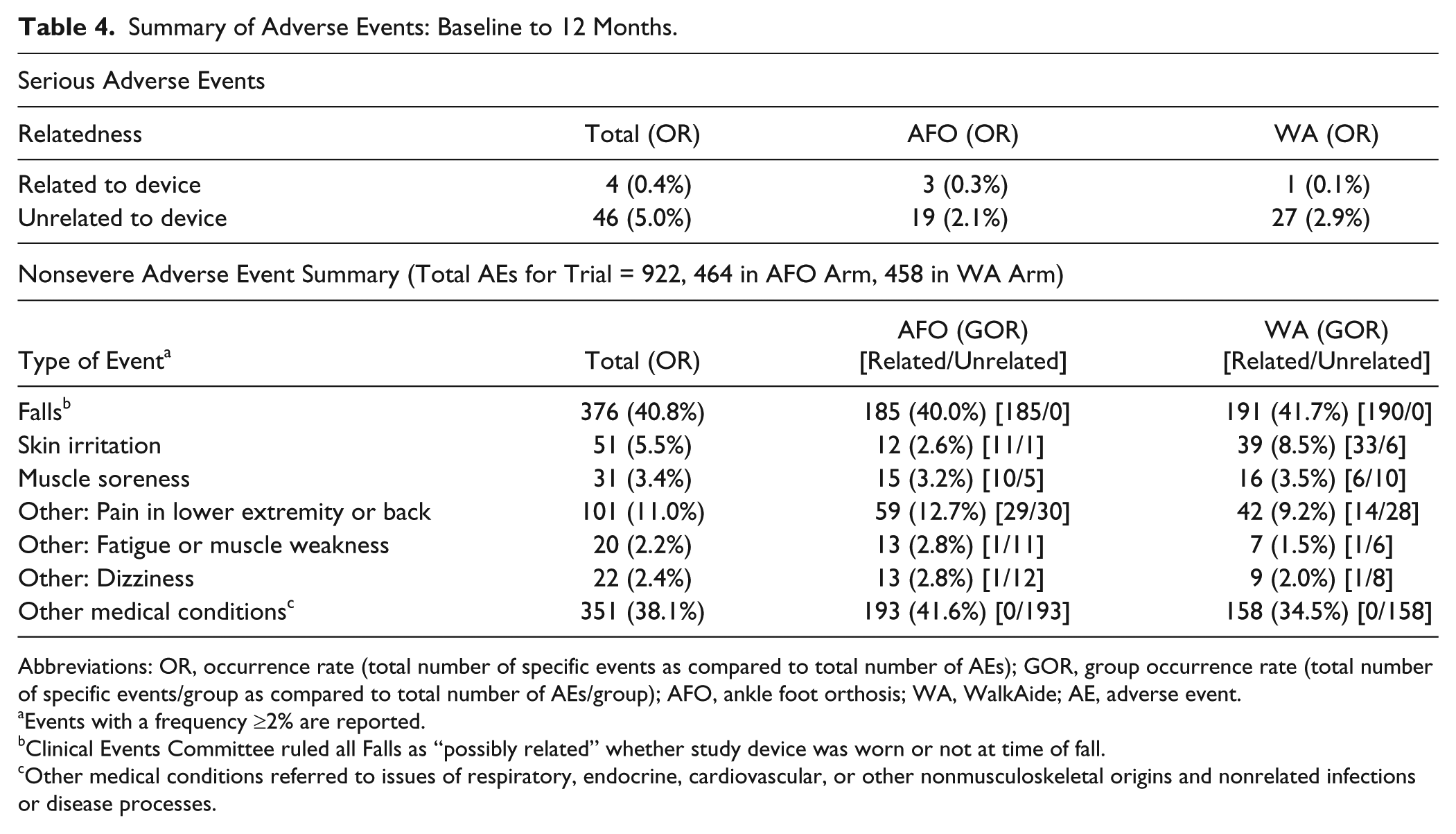
Abbreviations: OR, occurrence rate (total number of specific events as compared to total number of AEs); GOR, group occurrence rate (total number of specific events/group as compared to total number of AEs/group); AFO, ankle foot orthosis; WA, WalkAide; AE, adverse event.
Events with a frequency ≥2% are reported.
Clinical Events Committee ruled all Falls as “possibly related” whether study device was worn or not at time of fall.
Other medical conditions referred to issues of respiratory, endocrine, cardiovascular, or other nonmusculoskeletal origins and nonrelated infections or disease processes.
Change in Secondary Endpoints Between Baseline and 12-Month Visits
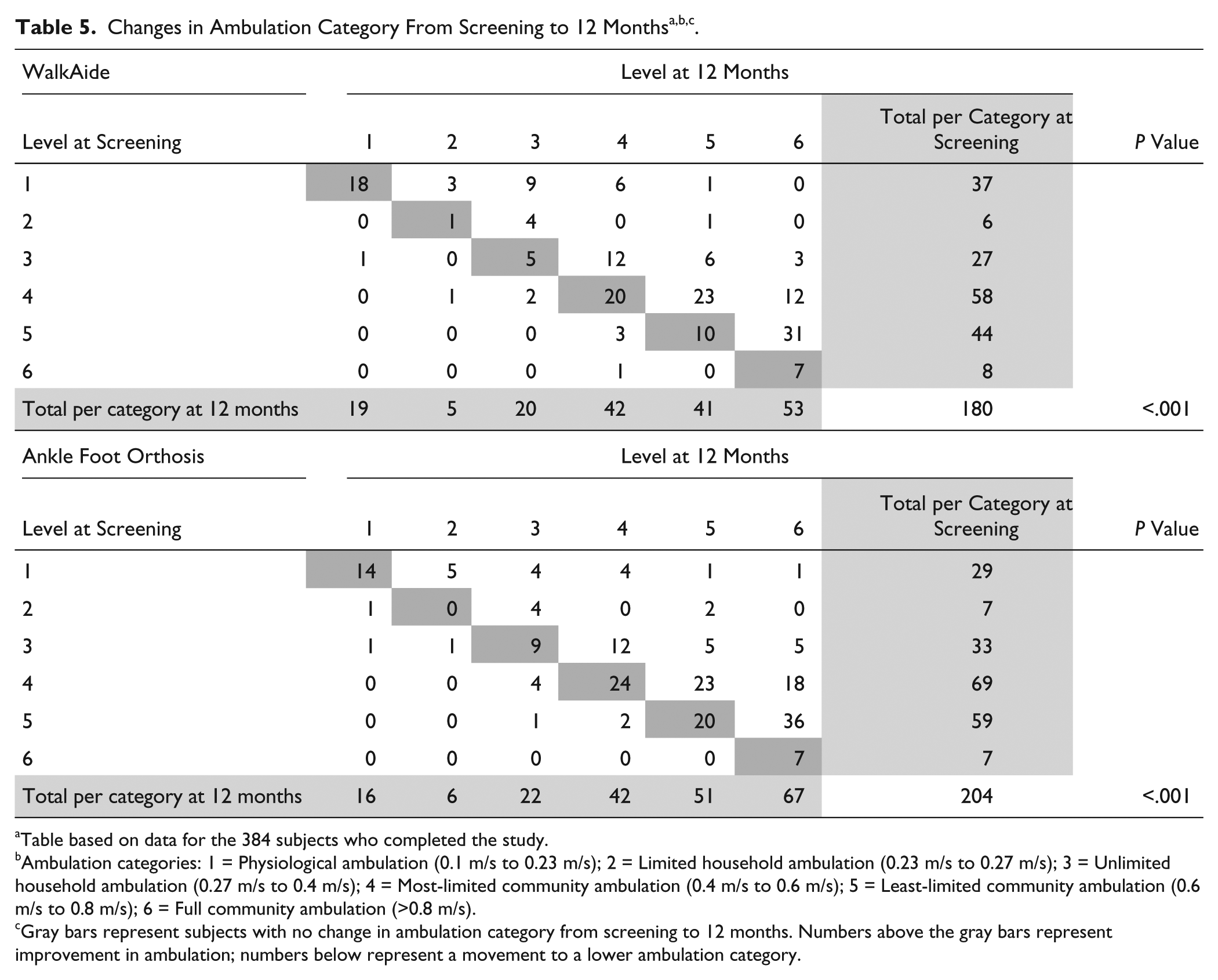
Table 3 details results for the 6MWT, FAP, and mEFAP between baseline and 12 months. The WA group alone demonstrated a statistically significant increase for the measures of 6MWT distance (P < .001) and mEFAP Stair time (P = .002). The GaitRite FAP score did not change significantly for either group. No statistically significant between-group differences were found for 6MWT, FAP, or mEFAP scores.To demonstrate the effects of changes in gait speed on functional ambulation, subjects were grouped post-analysis into functional ambulation categories 36 by gait velocity (Table 5 details subject categorization). A McNemar’s test showed statistically significant improvement in ambulation category between screening and 12 months for both the WA and AFO groups (P < .001).
Table based on data for the 384 subjects who completed the study.
Ambulation categories: 1 = Physiological ambulation (0.1 m/s to 0.23 m/s); 2 = Limited household ambulation (0.23 m/s to 0.27 m/s); 3 = Unlimited household ambulation (0.27 m/s to 0.4 m/s); 4 = Most-limited community ambulation (0.4 m/s to 0.6 m/s); 5 = Least-limited community ambulation (0.6 m/s to 0.8 m/s); 6 = Full community ambulation (>0.8 m/s).
Gray bars represent subjects with no change in ambulation category from screening to 12 months. Numbers above the gray bars represent improvement in ambulation; numbers below represent a movement to a lower ambulation category.
Changes Between 6 and 12 Months
Comparisons between the 6-month and 12-month values failed to show any statistically significant within- or between-group differences for all primary and secondary endpoints.
Discussion
The results of this study show the WA to be noninferior to the AFO for gait velocity and device-related SAE rate after 1 year of device utilization. The very small number of device-related SAEs demonstrates that both the WA and AFO are equally safe and that long-term use poses no increased risks to users. The 6- to 12-month comparisons showed no significant changes past 6 months of device wear, implying that gains in this study occurred before 6 months and that these gains proved stable across the 12-month period.
For individuals poststroke, walking ability has important implications for activity levels and health status, 4 and improving walking ability constitutes the most frequently stated goal. 38 Gait speed in particular is not only an important indicator of ongoing gait impairment, functional mobility, and level of disability, it is also considered the most efficient way to classify ambulation status poststroke. 40 Ambulation categories based on gait speed have been linked to the impact of stroke on QoL; individuals who transition from one ambulation category to another exhibit meaningful changes in Stroke Impact Scale domain scores. 40 However, to be considered clinically meaningful there must be a sufficient magnitude of change in gait speed. To that end, MCIDs for gait speed have been established for the stroke population and range from 0.1 37 to 0.16 m/s. 38 In this study, both the WA and AFO groups showed gait speed increases above the established MCID range (0.198 and 0.172 m/s, respectively, at 12 months). Statistically and clinically significant changes were noted in both groups at both the 6-month 28 and 12-month time points. These results indicate that both the WA and the AFO have a clinically significant impact on gait speed that continues with long-term device use. Other nonrandomized, pre–post intervention FES studies have demonstrated long-term, cumulative changes in gait speed in individuals poststroke. Laufer et al reported improvements in gait speed for 16 individuals with chronic hemiparesis ranging from 29% improvement after 2 months of FES wear to 52% at 12 months. 25 Stein et al studied 26 subjects with chronic hemiparesis reporting increases in gait speed of 15% at 3 months, 32% at 6 months, and 47% at 12 months. 20 There is, however, little prior evidence examining the long-term results of AFO wear for people with stroke. 41 To our knowledge this is the first RCT to compare changes in gait speed due to FES or AFO utilization over the course of a full year.
We utilized Perry ambulation categories to rank subjects by gait speed. These categories, separated by a maximum change of 0.2 m/s, classify ambulation status from physiologic to full community ambulation. 36 Mean gait speed at screening fell within the category of most-limited community ambulation for both WA and AFO groups (0.449 and 0.487 m/s, respectively). Both groups demonstrated mean gait speed increases sufficient for transition into the least-limited community ambulation category (0.647 and 0.659 m/s, respectively), representing significant improvement in community mobility. Also, a statistically significant number of subjects transitioned into higher ambulation categories in both the WA and AFO groups at 12 months (P < .001). These changes were shown to be significant at both 6 months 28 and 12 months, indicating that early improvement is sustained and that the WA and AFO are equally beneficial as long-term treatments for foot drop. These findings agree with the results of Taylor et al 31 and O’Dell et al, 42 both of which show significant transitioning to higher ambulation categories with the use of FES in patients poststroke. These results also add to the literature for both FES and AFO utilization and show the impact of device-related changes in gait speed on household and community ambulation.
Walking endurance was measured by the 6MWT. At 12 months, mean changes for the 6MWT were statistically significant only within the WA group (26.83 m; P < .001). Though the magnitude of change did not meet the criterion established by Perera et al for substantial meaningful change (50 m), 37 the improvement noted in the WA group may represent clinically relevant change. The literature supports other estimates of true clinical change, as opposed to statistical or random change. Flansbjer et al suggested that a 13% change in 6MWT can indicate a smallest real difference (smallest difference indicating true clinical change), 32 and the Perera et al study also identified a small meaningful change in 6MWT distance to be within the range of 19 to 20 m for subjects with stroke. 37 For the WA group, the 12-month percent change from baseline (16.3%) exceeds the smallest real difference. Also, the increase in distance (28.6 m) fell above the proposed thresholds for smallest meaningful change. These results suggest that though the higher range of meaningful change was not reached, the differences noted in the 6MWT could be attributed to real improvements in subject performance, and warrant further research.
Home and community environments often present architectural barriers or obstacles that challenge gait for individuals poststroke. 35 The mEFAP measures ambulatory ability in functional, real-world environments commonly encountered in household and community ambulation. At 12 months, the WA group demonstrated a statistically significant decrease in time to complete the mEFAP Stair time subtask (−26.6 seconds, P = .002). While not reaching statistical significance with the Bonferroni adjustment, there were trends for within-group improvement on the mEFAP Total time and Obstacle Course time in the WA group, and on the mEFAP Total time in the AFO group, which may warrant further research. To our knowledge, the only other RCT that has utilized the mEFAP to compare the use of FES to an AFO was the study published in 2013 by Sheffler et al. 7 That study utilized the total mEFAP scores to compare FES and AFOs on the performance of functional ambulation tasks after 12 weeks of full-time wear and 6 months after device wear was completed. 7 Sheffler et al found a significant within-group improvement in the mEFAP score with both FES and AFO, but no significant between-group difference at 12 weeks or at the 6-month follow-up. 7 Sheffler et al looked at the status of their subjects without FES or an AFO before and after the intervention (training effect). Our study differs in that observed changes were measured with the study devices on at each time point, which will have affected the magnitude of the differences observed. Our study also differs from that of Sheffler et al in that our length of treatment was significantly longer (1 year as compared to 12 weeks) and our study looked at mEFAP subtask scores as well as the total mEFAP score. The changes in mFEAP subtask scores in our study could suggest that long-term device wear may have a greater cumulative, positive effect on functional mobility than shorter term usage. Also, using both the total and subtask scores of the mEFAP may allow for more specific representation of changes in functional ability. The improvement of performance noted in our study for the WA group on some mEFAP subtasks may imply that the increased range of motion and muscle activity afforded by FES could positively affect the ability of an individual poststroke to manage challenging gait environments, although no significant between-group differences for change were noted.
Study Limitations
Several limitations noted in the original publication 28 continue to be pertinent to this follow-up analysis. Data collection, management, and processing were rigorously standardized across 30 sites but confounding effects due to multiple sites are possible. All study personnel were trained to administer the measures, although actual interrater reliability was not assessed. This study was unblinded, which may have contributed to measurement bias. Subjects were not stratified by gait velocity, and there was a small but statistically significant difference in gait velocity between groups at baseline. Also, the actual dropout rate of 22% was slightly higher than predicted dropout rate (20%). However, we employed an ITT analysis with multiple imputations to account for missing data, preserve randomization, and mitigate selection bias risk. Device wear compliance was not systematically tracked, so overall patterns of wear are unknown. Even though we enrolled subjects with a wide range of gait speeds, we did not recruit subjects with a gait speed above 0.8 m/s; therefore, our results cannot be generalized to this group of poststroke patients. Finally, the trial was designed to collect the secondary endpoints beginning after device fitting, preventing us from evaluating the separate orthotic or training effects in the classical sense. While this design did enable us to measure the magnitude of post-device fitting improvement over 12 months as it pertains to ambulation ability and quality, it may limit comparisons with other studies.
Conclusions
The results of the 6-month RCT indicated that the WA and AFOs are equivalent in facilitating clinically significant improvements in gait speed. 28 These 12-month follow-up results contribute additional support to this finding, demonstrating that for both devices, these clinically relevant improvements in gait speed remain stable over the long term. The WA shows equivalence to the AFO in measures with clinical relevance to gait performance and in long-term safety, and should therefore be considered an appropriate alternative to bracing for individuals with foot drop secondary to chronic stroke.
Footnotes
Acknowledgements
We thank the following Primary Investigators for their participation: Ajay Arora, MD, Morton Plant Healthcare System, Clearwater, Florida; Everett Heinze, MD, St David’s Medical Center, Austin, Texas; David Patterson, MD, Casa Colina Centers for Rehabilitation, Pomona, California; Aamir Rasheed, MD, United Health Services, Johnson City, New York; Bruce Solomon, DO, FirstHealth of the Carolinas, Pinehurst, North Carolina; Robert Taylor, MD, Cardinal Hill Rehabilitation, Lexington, Kentucky.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Francois Bethoux was a consultant with Innovative Neurotronics, Inc. for the INSTRIDE trial (honoraria paid to his institution), and Helen L. Rogers is an employee of Innovative Neurotronics, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Innovative Neurotronics, Inc.