Abstract
Background. Evidence supports peroneal nerve functional electrical stimulation (FES) as an effective alternative to ankle-foot orthoses (AFO) for treatment of foot drop poststroke, but few randomized controlled comparisons exist. Objective. To compare changes in gait and quality of life (QoL) between FES and an AFO in individuals with foot drop poststroke. Methods. In a multicenter randomized controlled trial (ClinicalTrials.gov #NCT01087957) with unblinded outcome assessments, 495 Medicare-eligible individuals at least 6 months poststroke wore FES or an AFO for 6 months. Primary endpoints: 10-Meter Walk Test (10MWT), a composite of the Mobility, Activities of Daily Living/Instrumental Activities of Daily Living, and Social Participation subscores on the Stroke Impact Scale (SIS), and device-related serious adverse event rate. Secondary endpoints: 6-Minute Walk Test, GaitRite Functional Ambulation Profile (FAP), Modified Emory Functional Ambulation Profile (mEFAP), Berg Balance Scale (BBS), Timed Up and Go, individual SIS domains, and Stroke-Specific Quality of Life measures. Multiply imputed intention-to-treat analyses were used with primary endpoints tested for noninferiority and secondary endpoints tested for superiority. Results. A total of 399 subjects completed the study. FES proved noninferior to the AFO for all primary endpoints. Both the FES and AFO groups improved significantly on the 10MWT. Within the FES group, significant improvements were found for SIS composite score, total mFEAP score, individual Floor and Obstacle course time scores of the mEFAP, FAP, and BBS, but again, no between-group differences were found. Conclusions. Use of FES is equivalent to the AFO. Further studies should examine whether FES enables better performance in tasks involving functional mobility, activities of daily living, and balance.
Keywords
Introduction
Stroke represents a major public health problem, with an annual incidence of 795 000 cases in the United States 1 and 15 million worldwide. 2 Seventy-five percent of strokes occur in people aged 65 years or older. 2 Stroke remains the fourth leading cause of death and the leading cause of serious, long-term disability in the United States. 2 With more than 7 million stroke survivors, 1 an estimated $34.3 billion was spend on direct and indirect cost of stroke-related care in 2008. 3
Seventy percent of stroke survivors regain the ability to walk, but functional ambulation is limited in many because of residual spastic hemiparesis, and often requires the use of an assistive device for independent ambulation. Walking limitations have a continued and significant impact on functional performance and quality of life (QoL) for stroke survivors. 4 An estimated 20% of all stroke survivors experience foot drop, a consequence of spastic hemiparesis from stroke. 5 Foot drop, typically because of a combination of weakness of the ankle dorsiflexor muscles (agonists) and spasticity of the plantarflexor muscles (antagonists), results in a slower, less efficient gait and increases the risk of falls. 6
The current standard of care for foot drop is the ankle-foot orthosis (AFO). An AFO, usually made of polypropylene or carbon fiber, is a brace worn on the lower leg to hold the foot and ankle in the correct position. AFOs restrict the natural range of motion and flexibility of the ankle and foot resulting in limited walking ability on uneven terrains, and may be uncomfortable to wear. 7 Functional electrical stimulation (FES), the use of neuromuscular electrical stimulation to activate muscles during functional tasks, is an alternative to the AFO. FES to the peroneal nerve provides active dorsiflexion during the swing phase of ambulation and can reduce foot drop by facilitating increased voluntary muscle activity, 8 and improving the quality and symmetry of gait.9,10
There is strong support for FES both in clinical guidelines and in recently published literature. The Department of Veterans Affairs/Department of Defense (VA-DoD) and the American Heart Association (AHA)/American Stroke Association (ASA) both published clinical practice guidelines for stroke rehabilitation in 2010.3,11 The VA-DoD guidelines recommend the use of FES as an adjunctive treatment for motor relearning, at the acute and chronic phase (level B evidence). 3 The AHA/ASA guidelines acknowledge published evidence showing improvement of gait with FES, when compared with or as a supplement to other rehabilitation modalities (neuroprosthetic effect). 11 In several studies, peroneal nerve FES was associated with increased gait velocity,7,9,12,13 decreased energy expenditure with gait,7,12-14 and improved gait symmetry.9,10,15 Evidence in the literature primarily comes from nonrandomized, pre–post intervention studies with small sample sizes and short follow-up periods. Few studies cover all dimensions of the International Classification of Functioning, Disability and Health (ICF). The recent publication of 3 larger randomized controlled trials (RCTs) comparing the use of FES to an AFO has added higher quality evidence to the literature.16-18
The purpose of this investigation was to measure the effects on gait performance and QoL of the WalkAide FES system (WA) compared with an AFO in individuals with foot drop secondary to hemiparetic stroke eligible for Medicare or Medicare Advantage benefits.
Methods
Study design
This study was an unblinded, parallel-group RCT of subjects with foot drop due to stroke conducted across 30 sites in the United States (Supplementary Table 1 [available online at http://nnr.sagepub.com/content/by/supplemental-data] provides detailed eligibility criteria). All subjects were consented and asked to sign an authorization to use and disclose information. Subjects meeting all inclusion and exclusion criteria were enrolled and randomized into 1 of 2 groups, WA or AFO, using a centralized computer-generated randomization scheme built into the electronic data capture system for this study. Subjects were followed for 6 months on measures of gait performance, functionality with activities of daily living, balance, and quality of life.
Clinical Evaluation and Measurement
At screening, sites performed a medical history and a peripheral nerve stimulation test, collected demographic information, and completed the Mini Mental State Exam, 19 Beck Depression Inventory, 20 and a neurological evaluation to ensure inclusion and exclusion criteria were met. Subjects’ walking ability and gait speed were also assessed using a 10-Meter Walk Test (10MWT), during which subjects were allowed to use their usual assistive devices, but no physical assistance or external support/bracing was allowed. The 10MWT was used as an inclusion/exclusion criterion (subjects ambulating faster than 0.8 m/s were excluded), and as the initial (pre–device fitting) value in the primary endpoint analysis of change in gait velocity from screening to 6 months.
Eligible subjects were randomized and scheduled for fitting of the study device within 2 weeks of randomization. Study devices were the WA (Innovative Neurotronics, Austin, TX) or an AFO. The WA is a battery-operated, single-channel electrical stimulator approved by the US Food and Drug Administration as treatment for foot drop. The device consists of a cuff worn around the proximal part of the lower leg, which holds the control module and surface electrodes. The WA uses a tilt sensor and accelerometer to trigger ankle dorsiflexion and control the timing and duration of peroneal nerve stimulation during the swing phase of gait. After initial fitting, programming, and patient education performed by a trained clinician, patients are able to use the WA to facilitate walking in daily activities. Fitting for the AFO was performed by a licensed orthotist; subjects coming in to the study with AFOs that met the standard of care were able to continue in their own orthosis. AFOs provided for subjects or AFOs fabricated as replacements were custom molded and either articulated or fixed at the ankle based on the professional opinion of the orthotist and clinical needs of the subject. Fitting for the WA was performed by a WA-certified orthotist or licensed physical therapist. After completing a 2-week progressive wearing schedule relevant to each device, subjects were instructed to wear their device on a full time basis (ie, for all walking activities throughout the day).
Study measurements were performed at screening (without device), baseline (postfitting, with device) 1, 3 and 6 months (with device); the primary endpoint for data analysis was at 6 months (Supplementary Table 2 [available online at http://nnr.sagepub.com/content/by/supplemental-data] details measurement periodicity). For all walking performance tests, the subjects were instructed to use their usual assistive device (eg, cane, walker), and to use the same device at each visit. The primary endpoints for the trial were the following: gait velocity (10MWT), 21 a composite score consisting of the sum of the Mobility, Activities of Daily Living/Instrumental Activities of Daily Living (ADL/IADL) and Social Participation domain scores of the Stroke Impact Scale (SIS), and the device-related serious adverse event (SAE) rate. Secondary endpoints were the following: 6-Minute Walk Test (6MWT), GaitRite Functional Ambulation Profile (FAP), Modified Emory Functional Ambulation Profile (mEFAP), Berg Balance Scale (BBS), Timed Up and Go (TUG), Stroke-Specific Quality of Life (SSQoL), and individual SIS domain scores (Strength, Mobility, Communication, Emotion, Memory and Thinking, Social Participation, ADL/IADL, and Hand Function).
Secondary measures were chosen in order to accurately assess general ambulation ability, performance in functional mobility tasks, dynamic balance, and parameters describing QoL and the impact of the disability from stroke. The chosen gait measures provide an assessment of walking endurance over longer distances (6MWT), 22 gait quality (FAP), 23 and ability to perform functional ambulation tasks (mEFAP). 24 The measures chosen to assess balance, the BBS and TUG, are designed to test static and dynamic balance during functional tasks, including walking.25,26 Values for the TUG were extracted from the mEFAP battery. The impact of disability from stroke was assessed via the SIS and the SSQoL, both of which provide a multidimensional assessment of the impact of stroke on physical function as well as stroke-related QoL.27,28
All the measures used have published reliability values, and standard instructions were used across all sites during data collection. All study personnel were trained to the administration of these tests, but actual interrater reliability was not assessed. All of the GaitRite walking trials were processed and analyzed by a central lab, and the instructions for walking on the mat were standardized.
Sample Size Calculation
The sample size needed to adequately power the study was estimated for each primary endpoint (gait speed, SIS composite score, and device-related SAE rate), and the largest estimate retained. Estimates were for a power of 80% to detect a difference between groups in gait speed of ≥0.1 m/s (2-sided t test, α = .025), a difference in SIS composite score of ≥15 points (2-sided t test, α = .025), and a difference in device-related SAE rate of no more than 3% (1-sided Blackwelder’s noninferiority test of difference in proportions, α = .05). The device-related SAE rate yielded the largest sample size estimate, 198 per group. This number was corrected for an estimated attrition rate of 20%, establishing the sample size estimate for this study at 495.
Statistical Analysis
Study data were analyzed using per-protocol (completers) and intention-to-treat (ITT) analyses with missing data points calculated using multiple imputation. The results of the 2 analyses differed minimally; to preserve randomization and mitigate impact of bias resulting from subject dropout, the ITT analyses results are presented. To account for potential clustering at the site level, the ITT analysis on all endpoints were conducted on multiply imputed data via mixed-effect regression models in which the treatment arm was the fixed effect and the study center was the random effect.
A noninferiority analysis was conducted at 6 months comparing the WA and AFO groups on the primary efficacy endpoints of gait velocity, the composite of the SIS Mobility, ADL/IADL and Social Participation domain scores, and the primary safety endpoint of the device-related SAE rates. The composite SIS score was chosen as a primary endpoint because selected domains accurately depict changes in functional ability, independence with ADLs, and community mobility. Moriello et al 29 demonstrated that the majority of SIS domain items were linked to ICF domains. For the purposes of this study, this SIS composite score was considered most relevant and most likely to demonstrate changes in functional mobility attributable to device use. SAEs were defined as serious device-related deteriorations in a subject’s health resulting in a life threatening illness, injury or a permanent impairment of body structure or function requiring inpatient hospitalization or prolongation of existing hospitalization, or resulting in medical or surgical intervention to prevent permanent impairment to a body structure or function. The noninferiority margin was considered met if the WA group demonstrated each of the following with 95% confidence: a change in gait velocity no more than 0.2 m/s below that of the AFO group (based on Perry’s classification of ambulation categories poststroke 30 ), a change in SIS composite score no lower than 15 points below that of the AFO group (based on estimations of clinically meaningful change in SIS score 27 ) and a device-related SAE rate no greater than 3% above that of the AFO group.
If noninferiority was met, multiple secondary endpoints were scheduled for analysis at 6 months: 6 MWT distance, total and specific mEFAP task scores, GaitRite FAP score, ambulation category classification, 30 BBS score, TUG time, specific SIS domain scores, and SSQoL score. Changes from baseline or percent change from baseline were tested using t tests or Blackwelder’s t or Z tests. A Bonferroni adjustment for multiple comparisons was performed setting the level of statistical significance at P = .001. The changes in ambulation category classification were demonstrated by a McNemar’s test.
Results
Subjects
A total of 761 potential subjects were screened, of whom 266 failed to meet inclusion and exclusion criteria and 495 were enrolled at 30 rehabilitation centers nationwide between April 27, 2010 and April 26, 2012. A total of 399 subjects completed the study; 187 in the WA group and 212 in the AFO group. Figure 1 details participant flow through the study and sources of dropout. Table 1 outlines demographic information and screening measures for all subjects enrolled in the study. The only significant differences between groups initially were body mass index (BMI; P = .006) and gait speed (P = .05). Despite randomization procedures, gait speed in the WA group was slower than that of the AFO group by 0.04 m/s. Although this difference was statistically significant, it was not considered clinically significant, since it was not large enough to differentiate between ambulatory classification 30 ; nor did it differ within the range of minimal clinically important differences (MCID) for gait speed established for the stroke population.31,32 The difference in BMI (which was lower in the WA group) was not sufficient to differentiate between BMI categories 33 and was not considered clinically significant. These variables were therefore considered balanced in the analyses.
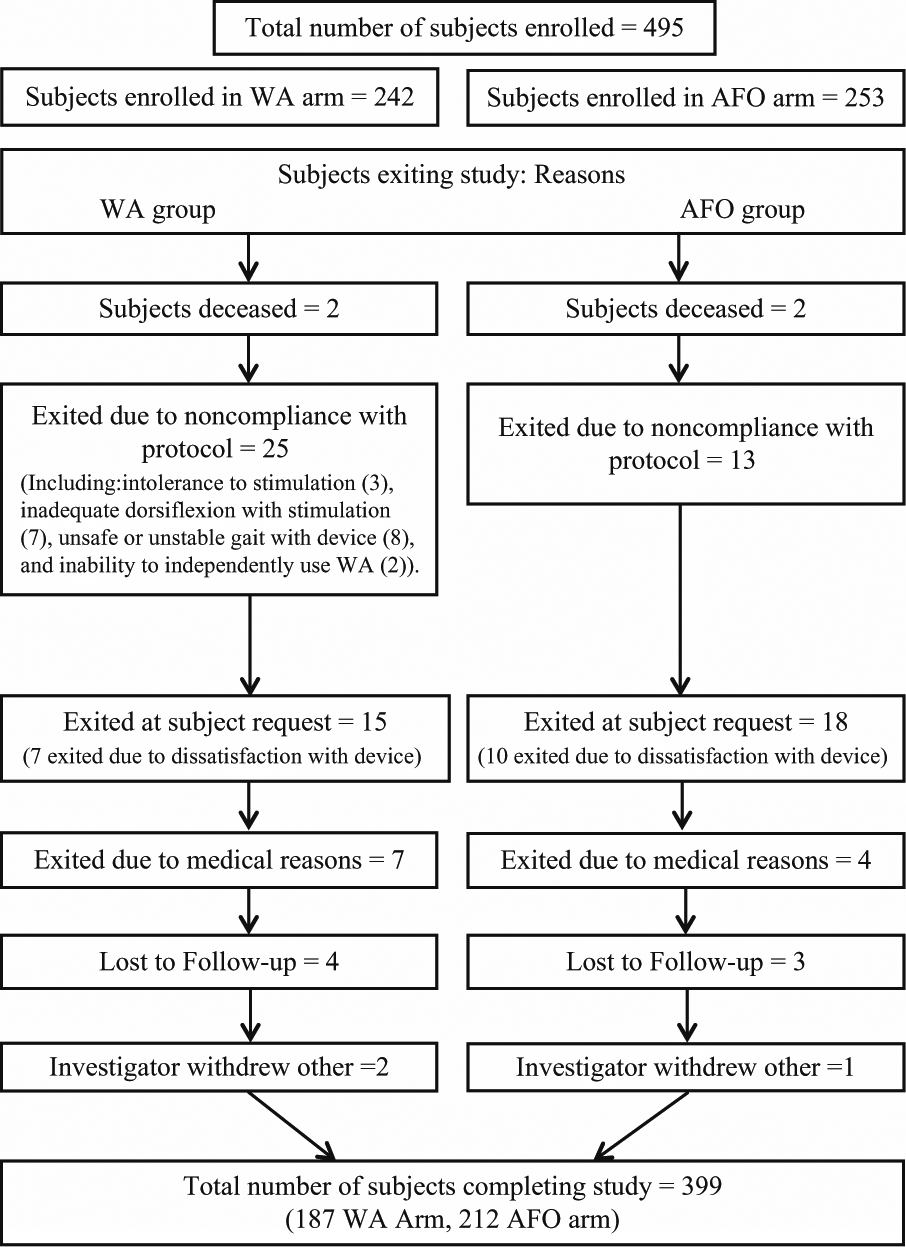
Participant flow diagram.
Subject Demographics and Characteristics at Screening.
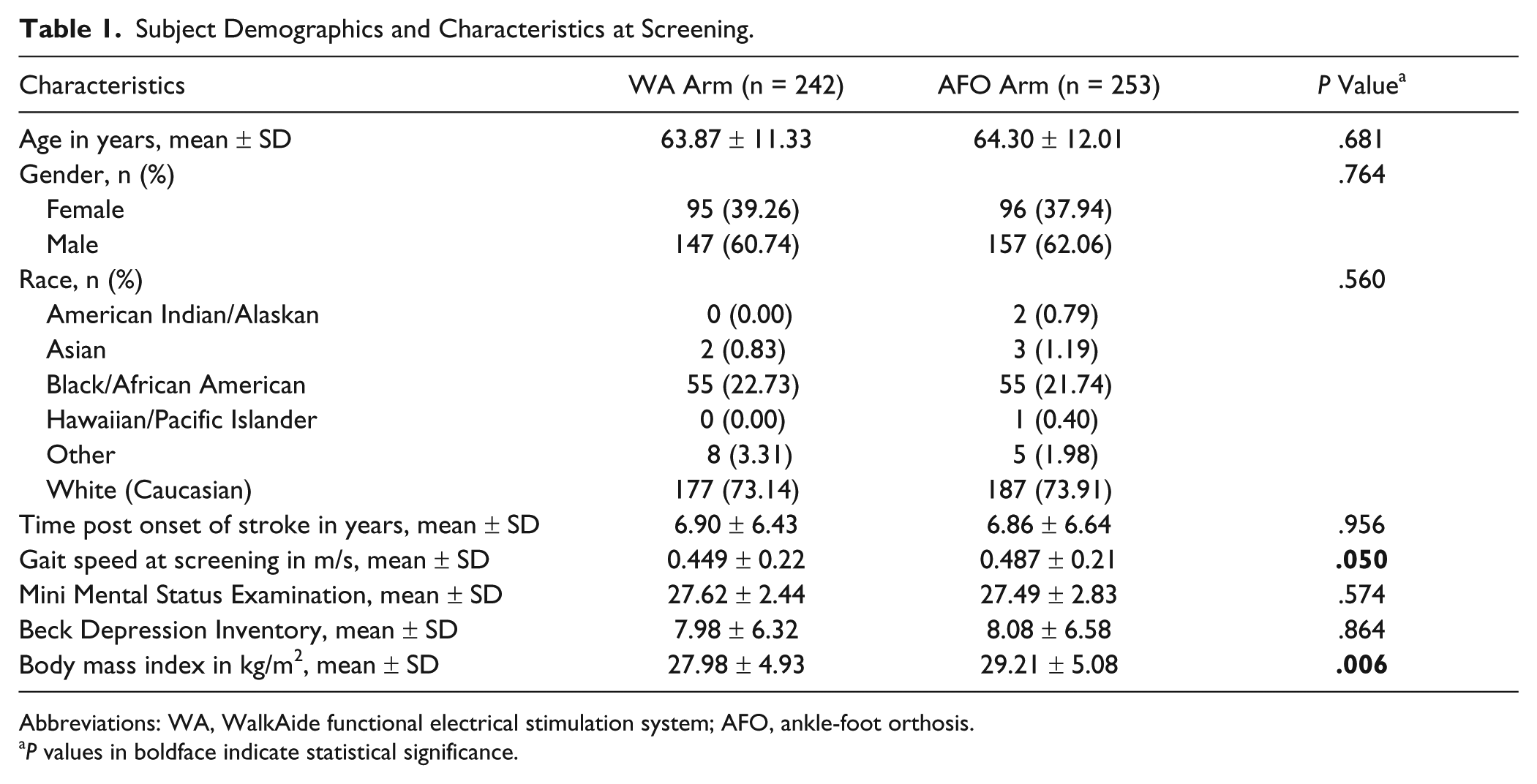
Abbreviations: WA, WalkAide functional electrical stimulation system; AFO, ankle-foot orthosis.
P values in boldface indicate statistical significance.
Primary Endpoints
The results of the primary efficacy endpoints (gait velocity and SIS composite score) and primary safety endpoint (device-related SAE rate) analyses show the WA to be noninferior to the AFO (values presented in Table 2). No statistically significant between-group differences were observed for gait velocity or SIS composite score. Both WA and AFO groups demonstrated statistically significant improvement in gait velocity from screening to 6 months (P < .001). The between-group difference in mean change in gait velocity was 0.009 m/s (95% confidence interval [CI] = −0.04 to 0.06). Therefore, the WA group was not inferior to the AFO group because the lower CI margin (−0.04) was larger than the margin set for noninferiority (−0.2 m/s). The WA group demonstrated statistically significant improvement from baseline to 6 months on the SIS composite score (P = .05). The between-group difference in mean change in SIS composite score was 1.1 points (95% CI = −1.62 to −0.58). Noninferiority was met for the WA group since the lower CI margin (−1.62) is greater than the noninferiority margin set for this variable (−15 points). Only 2 device-related SAEs were reported in the AFO and none in the WA group; therefore, device-related SAE rates were not computed (Table 3 summarizes all AEs for the trial with a frequency ≥2%).
Primary Endpoint Analysis.
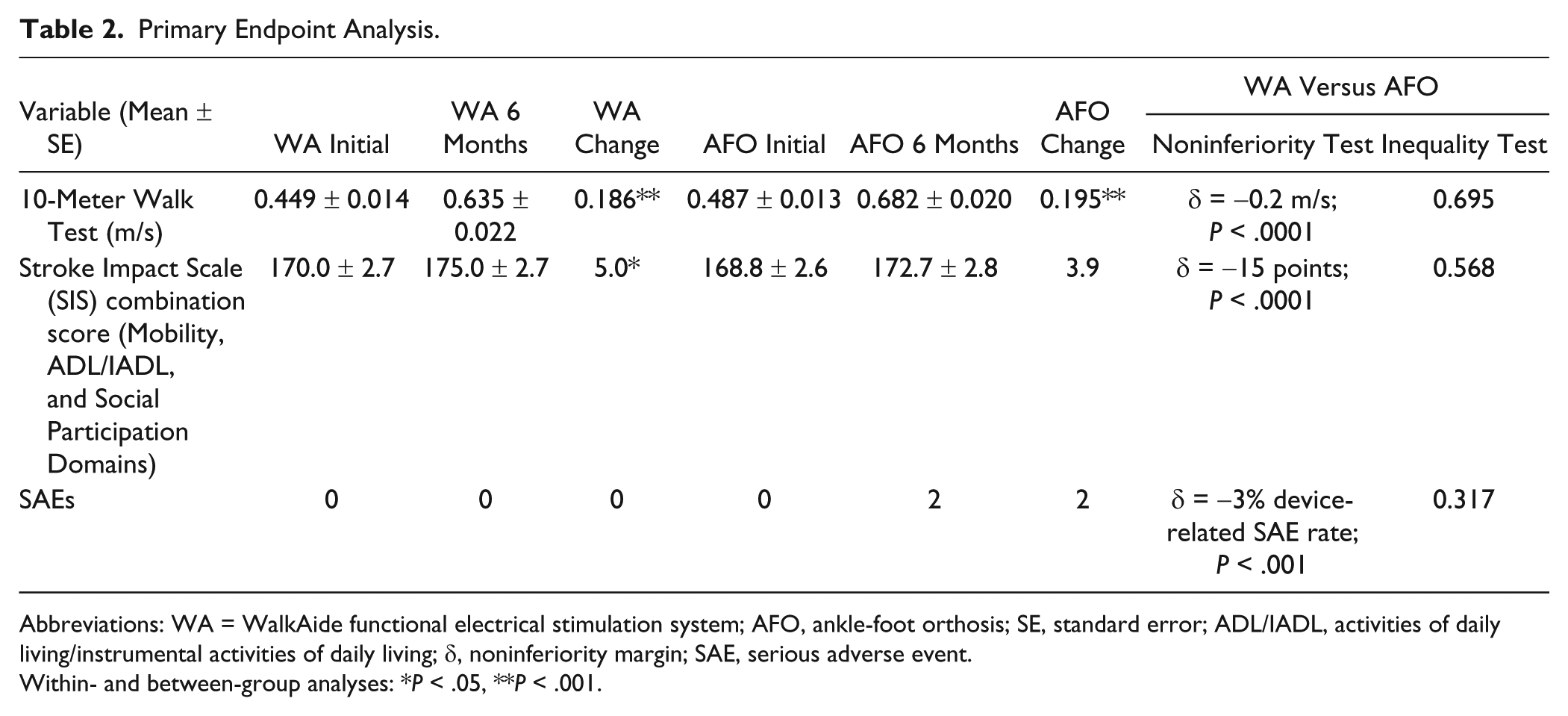
Abbreviations: WA = WalkAide functional electrical stimulation system; AFO, ankle-foot orthosis; SE, standard error; ADL/IADL, activities of daily living/instrumental activities of daily living; δ, noninferiority margin; SAE, serious adverse event.
Within- and between-group analyses: *P < .05, **P < .001.
Summary of Adverse Events.
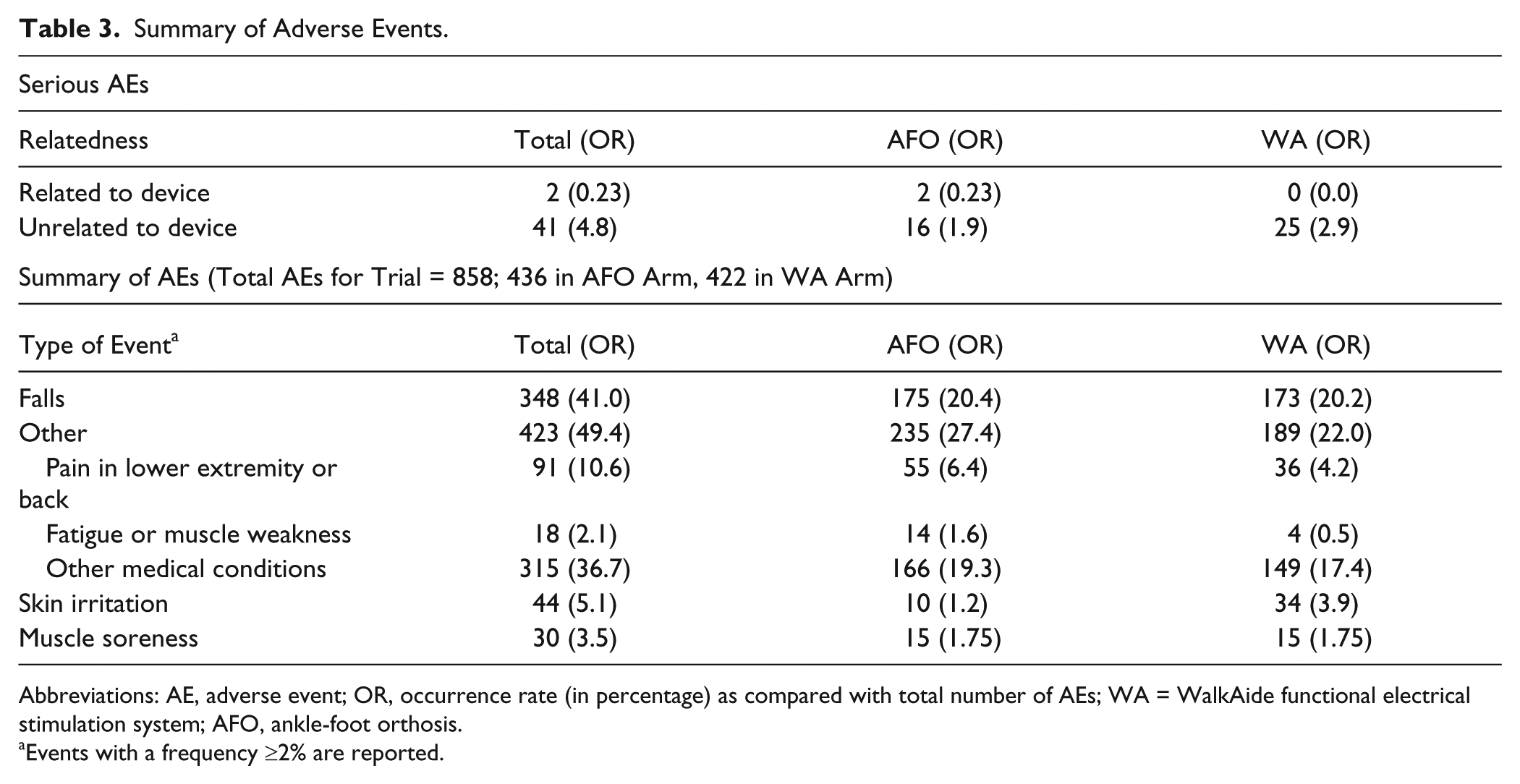
Abbreviations: AE, adverse event; OR, occurrence rate (in percentage) as compared with total number of AEs; WA = WalkAide functional electrical stimulation system; AFO, ankle-foot orthosis.
Events with a frequency ≥2% are reported.
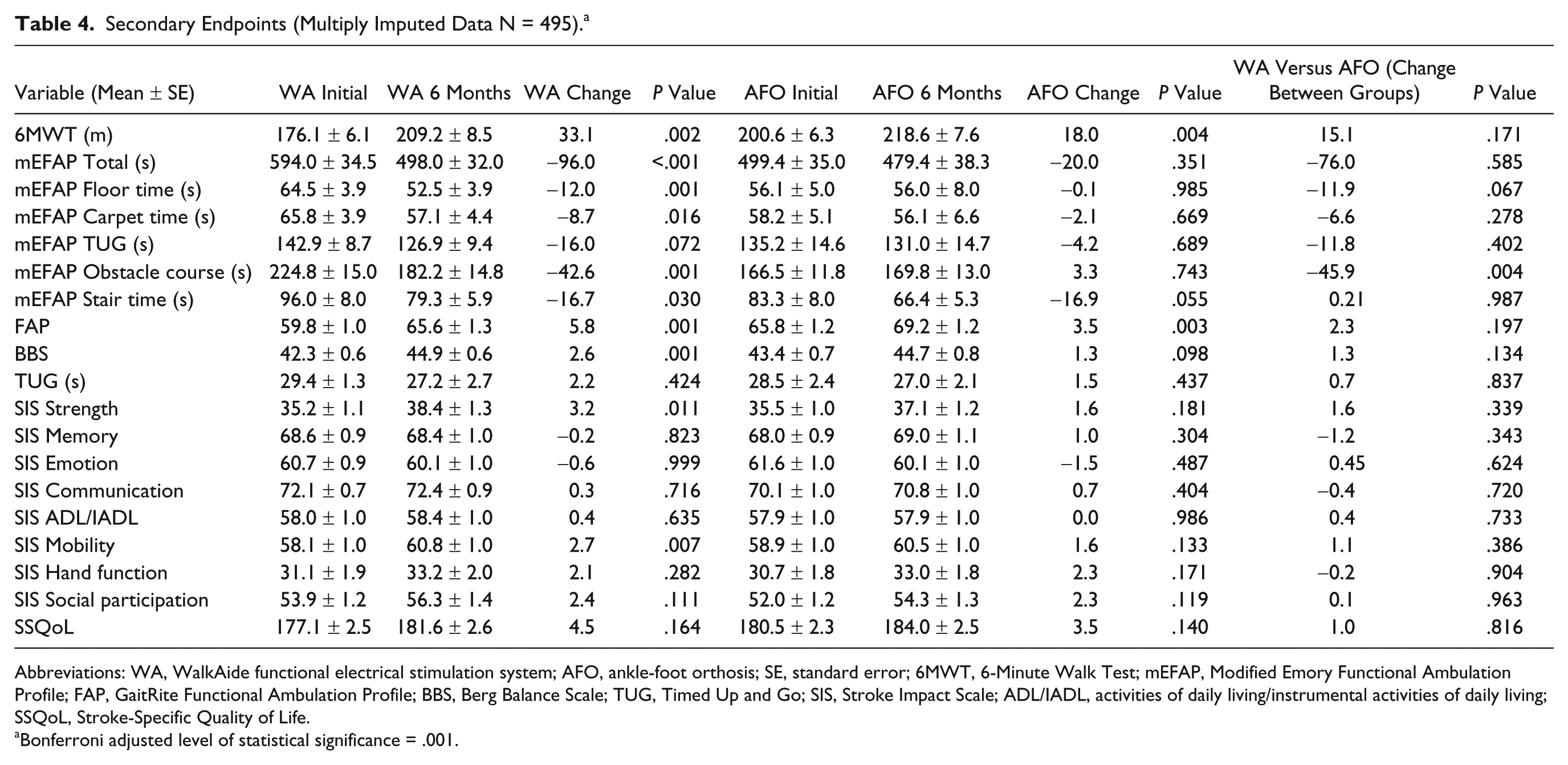
Secondary Endpoints: Walking Performance
Multiple endpoints were measured to determine the impact of the WA and AFO on walking performance (values presented in Table 4); differences were determined to be significant if P ≤ .001 after Bonferroni correction. Significant within-group differences between baseline and 6 months were noted in the WA group for GaitRite FAP score (P = .001), total mEFAP time (P < .001), and the mEFAP subtasks of Floor time (P = .001) and Obstacle Course time (P = .001). No significant between-group differences were found for these variables.
Secondary Endpoints (Multiply Imputed Data N = 495). a
Abbreviations: WA, WalkAide functional electrical stimulation system; AFO, ankle-foot orthosis; SE, standard error; 6MWT, 6-Minute Walk Test; mEFAP, Modified Emory Functional Ambulation Profile; FAP, GaitRite Functional Ambulation Profile; BBS, Berg Balance Scale; TUG, Timed Up and Go; SIS, Stroke Impact Scale; ADL/IADL, activities of daily living/instrumental activities of daily living; SSQoL, Stroke-Specific Quality of Life.
Bonferroni adjusted level of statistical significance = .001.
To demonstrate the effects of changes in gait speed on functional ambulation, the subjects were grouped into functional ambulation categories 30 by gait velocity (Supplementary Table 3 [available online at http://nnr.sagepub.com/content/by/supplemental-data] details subject categorization). A McNemar’s test showed statistically significant improvement in ambulation category between screening and 6 months for both groups (P < .001).
Secondary Endpoints: Balance
The impact of the WA and AFO on balance was measured using the BBS and TUG (Table 4). Statistically significant improvement on BBS score was noted for the WA group (P = .001). Neither the WA group nor AFO group exhibited statistically significant improvement in time to complete the TUG. No significant between groups differences were found for these variables.
Secondary Endpoints: Quality of Life
The individual SIS domains and the SSQoL test were used as measures of QoL (Table 4). Neither group demonstrated any significant within group improvement for the individual domains of the SIS or for SSQoL score from baseline to 6-month follow-up. No significant between-group differences were found for these variables.
Discussion
The results of this RCT confirm that the WA is noninferior to the standard of care AFO for each primary endpoint: (a) gait velocity, (b) SIS composite score, and (c) safety. The occurrence of device-related SAEs was extremely low (2 for AFO and none for WA group), demonstrating that both devices are safe and pose negligible risk to the user.
Gait velocity is an important indication of function and level of disability, 32 and the primary means of classifying ambulation status after stroke. 34 Ambulation categories have also been linked to the impact of stroke on QoL. Schmid et al 34 showed that individuals who transition from one ambulation category to another exhibited meaningful changes in SIS domain scores. MCIDs for gait speed have been reported in the stroke population and range from 0.1 m/s 31 to 0.16 m/s. 32 Both the WA and AFO groups in this study showed increases in gait speed above the established MCID (0.186 and 0.195 m/s, respectively). The results of this study show increases in gait speed slightly above those noted in the literature. The percentage changes in gait speed were 41.4% for the WA and 40.0% for the AFO group. Hausdorff and Ring 9 studied the effects of peroneal nerve FES in 24 subjects with chronic hemiparesis and noted a change in gait speed from baseline to 8 weeks of 34%. Studies by Stein et al7,12 looking at a population of subjects with nonprogressive (cardiovascular accident) and progressive (multiple sclerosis) disorders reported percent increases in gait speed with FES ranging from 32% at 6 months 12 to 37.8% at 11 months. 7 The changes observed in this study are also slightly above those of Kluding et al 17 who found a change in gait speed of 0.14 m/s in the FES group and 0.15 m/s in the AFO group.
The Perry ambulation categories use gait speed to classify ambulation status from physiologic to full community ambulation, with categories separated by a maximum change of 0.2 m/s. 30 The mean gait speed of both groups fell within the category of most-limited community ambulation at screening. The change in both groups (~0.2 m/s) was sufficient to move these means into the least-limited community ambulation category, representing a significant improvement in community mobility and function. These results indicate that the WA is equivalent to the AFO in facilitating clinically significant improvements in gait speed, and as such should be considered an appropriate alternative to bracing for individuals with foot drop secondary to chronic stroke.
The WA group demonstrated a statistically significant change in the SIS composite score (ADL/IADL, Mobility and Social Participation domains; 5.0 points, P = .050). Although the improvement in the composite score combining these domains reached statistical significance, they did not meet any established MCID for clinical significance. 35 Kluding et al 17 also reported greater improvement in the SIS mobility score in the FES group compared with the AFO group, changes in score did not meet the MCID for either group. These results suggest that, while FES may have a more favorable effect than an AFO on the impact of stroke on the QOL measured by these domains, the devices are equivalent in terms of producing a noticeable clinical impact.
No statistically significant between group differences were observed for secondary outcomes. The mEFAP was used to measure ambulatory ability in functional, real-world environments common to household and community mobility. The WA group demonstrated a statistically significant decrease in total time to complete the mEFAP, reducing completion time by 16.2% compared with a 4.0% decrease for the AFO group. The WA also showed improvement for 2 of the 5 subtasks: the Floor Time and Obstacle Course tasks. The time to complete the Floor Time subtask decreased by 18.6% for the WA compared with a 0.2% decrease for the AFO group. For the Obstacle Course subtask, the WA group decreased their time by 19.0% while the AFO group actually increased time to complete by 2.0%. Our findings contrast with those from 2 other studies that used the mEFAP to compare performance between FES and AFO. Sheffler et al 36 looked at single trial differences while a recent RCT by Sheffler et al 18 looked at differences between the devices after 12 weeks. Neither of these studies demonstrated significant differences on the total or subtask scores of the mEFAP.18,36
Some of the differences in performance noted between the WA and the AFO likely stem from differences in the way they correct foot drop. The WA device functions by generating active dorsiflexion during the swing phase of gait while the AFO passively fixes the ankle joint in a given position. Though both devices alleviate foot drop, the active dorsiflexion generated by FES has been shown in the literature to improve gait speed7,9,12,13 and to decrease walking asymmetry and variability noted in individuals with stroke.9,10,15 FES promotes ankle and foot flexibility, 12 and in doing so may improve ambulation over architectural barriers and obstacles. The use of FES does, however, require that the patient exhibit an adequate dorsiflexion response and the ability to tolerate the stimulus during fitting. These requirements explain the higher number of subjects who dropped out due to noncompliance with the protocol in the WA group (Figure 1), and reflect the fact that, in the clinical setting, not all stroke patients with foot drop will be appropriate for FES. The results from this study support the improvement in gait quality noted previously in the literature and demonstrate that subjects wearing the WA show statistically significant improvement for functional ambulation tasks in common gait environments measured by the mEFAP.
The results of the BBS indicate statistically significant improvement in balance during functional tasks for the WA group. However the amount of change, 2.6 points, failed to meet the smallest real difference level established in the literature and therefore was not clinically significant. 37 These results are similar to those of Kluding et al 17 who noted statistically but not clinically significant increases in BBS score for both the FES and AFO groups.
This study has a number of limitations. The subjects were not stratified by gait speed. Close to 20% of randomized patients exited the study before completion for a variety of reasons (Figure 1), which could introduce selection bias into the effect estimates and effectively break randomization. To mitigate the risk of these biases, we conducted an ITT analysis using multiple imputations to account for missing data, which serves to preserve randomization and reduce confounding related to patient dropout. There were 30 sites participating in this trial, and while data collection, management and processing were standardized as rigorously as possible, confounding effects due to multiple sites are possible. The time horizon for this study was limited to 6 months, which may not capture the long-term device impact on a patient population certain to have chronic morbidity for the rest of their lives. However, we are continuing to collect posttrial data up to the 12-month point (the stroke literature cites several studies reporting continued beneficial effects of FES up to 12 months).7,8,15,38 Compliance with device wear was not systematically tracked, so overall patterns of wear are unknown. Potential changes in spasticity medications over the course of the study were not tracked, and these may affect walking performance. However, our patients were at the chronic phase of stroke, during which changes in symptomatic medications are less likely to occur. Subjects who needed to start physical therapy after randomization were exited from the study. Finally, the trial was designed to collect the secondary endpoints beginning after device fitting, which prevents us from evaluating an “orthotic effect” in the classical sense. However, this trial design did enable us to measure the magnitude of post–device fitting improvement over 6 months as it pertains to ambulation ability, balance, and quality of life.
Conclusion
The results of this study show the WA to be equivalent to the AFO for improvements in gait velocity, SIS composite score and safety. The WA group also demonstrated statistically significant improvements on tasks of functional gait as measured by the mEFAP while the AFO group did not, though the between-group difference was not statistically significant after adjusting for multiple comparisons. The WA produces physiological dorsiflexion, with all the motor and sensory benefits inherent in active muscle contraction, including improved gait speed and improved overall quality of gait for individuals poststroke. Results from this study support the fact that use of the WA produces clinically relevant improvement in functional ambulation and thus should be considered a viable alternative to conventional bracing for individuals with foot drop secondary to chronic stroke. Because these results were reported in a population of individuals averaging 6.9 years from onset of stroke, this study demonstrates that functional improvements can be obtained in the chronic phase of stroke with appropriate intervention, and that continued recovery of function should remain a goal throughout the lifetime of a person with stroke.
Footnotes
Acknowledgements
We thank the following Primary Investigators for their participation: Ajay Arora, MD, Morton Plant Healthcare System, Clearwater, Florida; Everett Heinze, MD, St David’s Medical Center, Austin, Texas; David Patterson, MD, Casa Colina Centers for Rehabilitation, Pomona, California; Aamir Rasheed, MD, United Health Services, Johnson City, New York; Bruce Solomon, DO, FirstHealth of the Carolinas, Pinehurst, North Carolina; and Robert Taylor, MD, Cardinal Hill Rehabilitation, Lexington, Kentucky.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article : Francois Bethoux was a consultant with Innovative Neurotronics for the INSTRIDE trial (honoraria paid to his institution), and Helen L. Rogers is an employee of Innovative Neurotronics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Innovative Neurotronics.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
