Abstract
Background
Robotic systems combined with Functional Electrical Stimulation (FES) showed promising results on upper-limb motor recovery after stroke, but adequately-sized randomized controlled trials (RCTs) are still missing.
Objective
To evaluate whether arm training supported by RETRAINER, a passive exoskeleton integrated with electromyograph-triggered functional electrical stimulation, is superior to advanced conventional therapy (ACT) of equal intensity in the recovery of arm functions, dexterity, strength, activities of daily living, and quality of life after stroke.
Methods
A single-blind RCT recruiting 72 patients was conducted. Patients, randomly allocated to 2 groups, were trained for 9 weeks, 3 times per week: the experimental group performed task-oriented exercises assisted by RETRAINER for 30 minutes plus ACT (60 minutes), whereas the control group performed only ACT (90 minutes). Patients were assessed before, soon after, and 1 month after the end of the intervention. Outcome measures were as follows: Action Research Arm Test (ARAT), Motricity Index, Motor Activity Log, Box and Blocks Test (BBT), Stroke Specific Quality of Life Scale (SSQoL), and Muscle Research Council.
Results
All outcomes but SSQoL significantly improved over time in both groups (
Conclusions
Hybrid robotic systems, allowing to perform personalized, intensive, and task-oriented training, with an enriched sensory feedback, was superior to ACT in improving arm functions and dexterity after stroke.
Keywords
Introduction
The need to identify the best treatment options for arm recovery is included in the top 10 priorities relating to life after stroke. 1 Task-oriented and high-intensity training performed in a multidisciplinary environment with patient’s active participation have shown to facilitate cortical reorganizations and motor recovery after stroke.2,3
Robotic devices offer the opportunity to deliver high-intensity and task-oriented training in a safe environment, even to severely impaired patients. 4 Several randomized controlled trials (RCTs) were conducted to evaluate the effects of robot-assisted training on arm functions and activities of daily living (ADL) after stroke. A Cochrane review (45 trials; 1619 participants) showed that robot-assisted training improves ADL, arm function, and strength. 5 Another review (44 RCTs; 1362 participants) found instead small effects on motor function of the joints specifically targeted by the robot and no effect on arm capacity and ADL. 6 A recent multicenter RCT, enrolling 770 participants with moderate to severe arm impairment, compared 3 treatments of the same frequency and duration: robot-assisted arm training, enhanced upper limb therapy, and usual care. 7 Robot-assisted arm training, based on an end-effector traditional robotic therapy with visual targets, showed significant improvements with respect to usual care on arm impairment, i.e. Body Functions and Structure domain of the International Classification of Functioning, Disability, and Health (ICF), but these improvements did not translate to functions or ADL, i.e. Activity Domain of the ICF. Enhanced upper limb therapy, based on personalized training specifically focused on ADL, led to improvements in Body Functions and Structure and Activity domains of the ICF compared to usual care. When robotic training was combined with occupational therapy, promising results were found in acute stroke survivors (n = 12). 8 Training in lifelike scenarios, which entails the interaction with real objects, may also optimize the assimilation of motor gains, promoting the transfer of motor function improvements from robot-assisted therapy to ADL. 9 Overall, this supports the need to advance traditional robotics training, based on end-effector robots and video-target reaching tasks, toward robot-assisted personalized, task-oriented, and ADL training.
Functional electrical stimulation (FES) was proposed as a training modality for arm recovery after stroke more than 20 years ago, 10 and it is currently recommended for patients with minimal volitional movement within the first months after stroke. 11 A systematic review (8 trials; 192 participants) showed that FES improves arm activity with respect to no training or conventional therapy. 12 A more recent review (20 RCTs, 431 participants) found no benefit of FES for ADL, unless FES was initiated in the acute phase (<2 months since stroke). 13 In 2019, Yang et al 14 found that FES-treated patients achieved higher improvements in Body Functions and Structure and Activity domains (48 RCTs, 1712 patients). 14 When FES-induced afferent feedback is synchronized with subject’s volitional intention through the use of electromyograph (EMG)-controlled FES systems, neuroplasticity is further enhanced,15,16 so as to improve motor recovery. In a recent systematic review (26 studies, 782 participants), EMG-controlled FES showed a short-term effect on arm impairment with respect to no or other treatments. 17
To make robot-assisted arm training more functionally oriented, robots can be combined with FES, giving birth to the so-called “hybrid robotic systems.”18,19 Shoulder and elbow movements trained with a robotic arm combined with sub–motor threshold electrical stimulation showed significant improvements of the range of motion with respect to robotic training alone in subacute stroke patients (n = 30). 20 A novel arm rehabilitative system, combining antigravity robotic support, FES, and voluntary effort, showed improvements of arm impairment in 5 chronic patients. 21 More recently, an EMG-driven FES-robotic system assisting elbow, wrist, and finger movements showed significant improvements on arm impairment and functions in chronic patients (n = 11). 22 When the same system was used in subacute patients, the FES-robot group achieved higher motor outcomes at the distal joints and more effective release of muscle tone than the conventional therapy group (n = 24). 23 Starting from these promising results, the effects of hybrid robotic systems on arm recovery after stroke need to be further investigated through adequately sized RCTs.
This multicenter RCT aimed to evaluate whether arm training assisted by a hybrid robotic system, combining personalized EMG-triggered FES with a light-weight, passive exoskeleton during task-oriented exercises with real objects, 24 improves arm functions, strength, dexterity, ADL, and quality of life compared with advanced conventional therapy (ACT) of the same frequency and duration in a population of stroke survivors.
Methods
A single-blinded multicenter RCT, following the CONSORT recommendations, 25 was conducted at 2 rehabilitation facilities (Asklepios Neurologische Klinik Falkenstein in Germany and Villa Beretta Rehabilitation Center in Italy). The study was approved by the local ethical committees and was registered in ClinicalTrials.gov (NCT03171649). Patients were recruited between December 2016 and September 2018. First patients were recruited with the purpose of therapists training, system refinement, and protocol fine-tuning. After study registration (May 2017), recruited patients were actually included in the analysis.
Participants
Inclusion criteria were the following: first stroke (ischemic or hemorrhagic) 2 weeks up to 9 months before enrollment, major unilateral functional impairment (Motricity Index [MI], arm-subscale, >80 and <80 for the unaffected and affected sides, respectively), Muscle Research Council (MRC) of arm and shoulder muscles of the affected side ≥1, low level of spasticity of arm muscles (Modified Ashworth Scale < 2), Mini-Mental State Examination >20. Patients were excluded if they had severe aphasia preventing communication, previous neurological or psychiatric disorders, major visual deficits, cardiac pacemakers, allergy to electrodes, or inability to tolerate FES.
Participants, either inpatient or outpatient, were evaluated by a physiatrist and a physiotherapist, and eligible patients were asked to give their written informed consent; finally, patients who agreed were randomized to 1 of the 2 interventions. The list of treatment codes was previously generated through a permuted-block randomization procedure by the Bioengineering Department of Villa Beretta (MRo). He kept the randomization sequence hidden and was responsible for assigning participants to interventions.
Assessors, collecting the outcome measures, were blinded to the treatment allocation, whereas physiotherapists delivering the intervention and participants were not.
Intervention
Participants of both groups received 90 minutes of arm training 3 times a week for 9 weeks. Patients allocated to the experimental group received 60 minutes of advance conventional therapy (ACT) and 30 minutes of training with RETRAINER, whereas patients allocated to the control group received 90 minutes of ACT of the same type of the experimental group.
Four physical therapists (2 for each clinical center), specifically trained on upper-limb stroke rehabilitation, were responsible for ACT in both training groups. Four different physical therapists (2 for each clinical center) received a 1-week training on the use of the RETRAINER system and were responsible for the RETRAINER-assisted intervention.
In both groups, ACT was tailored on each single patient and could include the following: upper-limb passive and/or active motion, arm cycle-ergometer without FES, FES of forearm muscles, virtual reality–augmented arm exercises, repetitive task training, and mirror therapy.
The RETRAINER system (Figure 1) consisted of a passive arm exoskeleton with 4 degrees of freedom (trunk inclination, shoulder rotation and elevation, and elbow flex-extension) to relieve arm weight. Two versions of the exoskeleton were developed to fit the left and right sides. The exoskeleton was equipped with electromagnetic brakes at each degree of freedom to keep antigravity positions and integrated with a current-controlled stimulator (RehamovePro, Hasomed GmbH), with 2 stimulation and 2 EMG recording channels. FES was provided to up to 2 arm muscles selected among biceps, triceps, anterior, medial, and posterior deltoid based on the patient’s needs. FES was triggered when the volitional EMG of the same stimulated muscle overcame a patient-specific threshold set during each session within an initial automatic calibration procedure. Once triggered, biphasic pulses with an increasing current amplitude up to a predefined maximal value were delivered; stimulation was stopped once the target position was reached. When FES was active, EMG activity was continuously monitored in order to provide a visual feedback about the participant’s volitional activity (happy/sad emoji if the activity was considered active/passive). 26 The stimulation frequency and the pulse width were fixed to 25 Hz and 300 µs, respectively, whereas the stimulation intensity was subject specific and above the motor threshold. A set of interactive objects exploiting radio-frequency identification (RFID) technology completed the system. These objects consisted of either 3D-printed cylindrical objects of daily life (eg, bottles, cups) or target positions to be reached during the exercises. Each object was equipped with passive tags, and an antenna was mounted on the exoskeleton at the hand level. The 3D position of the arm was monitored by combining the information coming from the goniometer angle sensors and the RFID reader. RETRAINER allows to perform 7 exercises: anterior reaching (planar or in the space), moving objects (on a plane or in the space), lateral elevation of the extended arm, and hand to mouth movements with or without grasping an object. The training parameters—that is, type of exercises, number of repetitions, target position, gravity compensation level, duration of pauses, stimulated muscles, and current amplitude—were subject specific, defined on the basis of clinical experience, and could change throughout the intervention. At the beginning of each training session, about 10 to 15 minutes were dedicated to configuring and donning the system. Afterward, the 30 minutes of training started. For more technical details on the RETRAINER system, on the control algorithm, or on the exercises, please refer to our previous work. 24
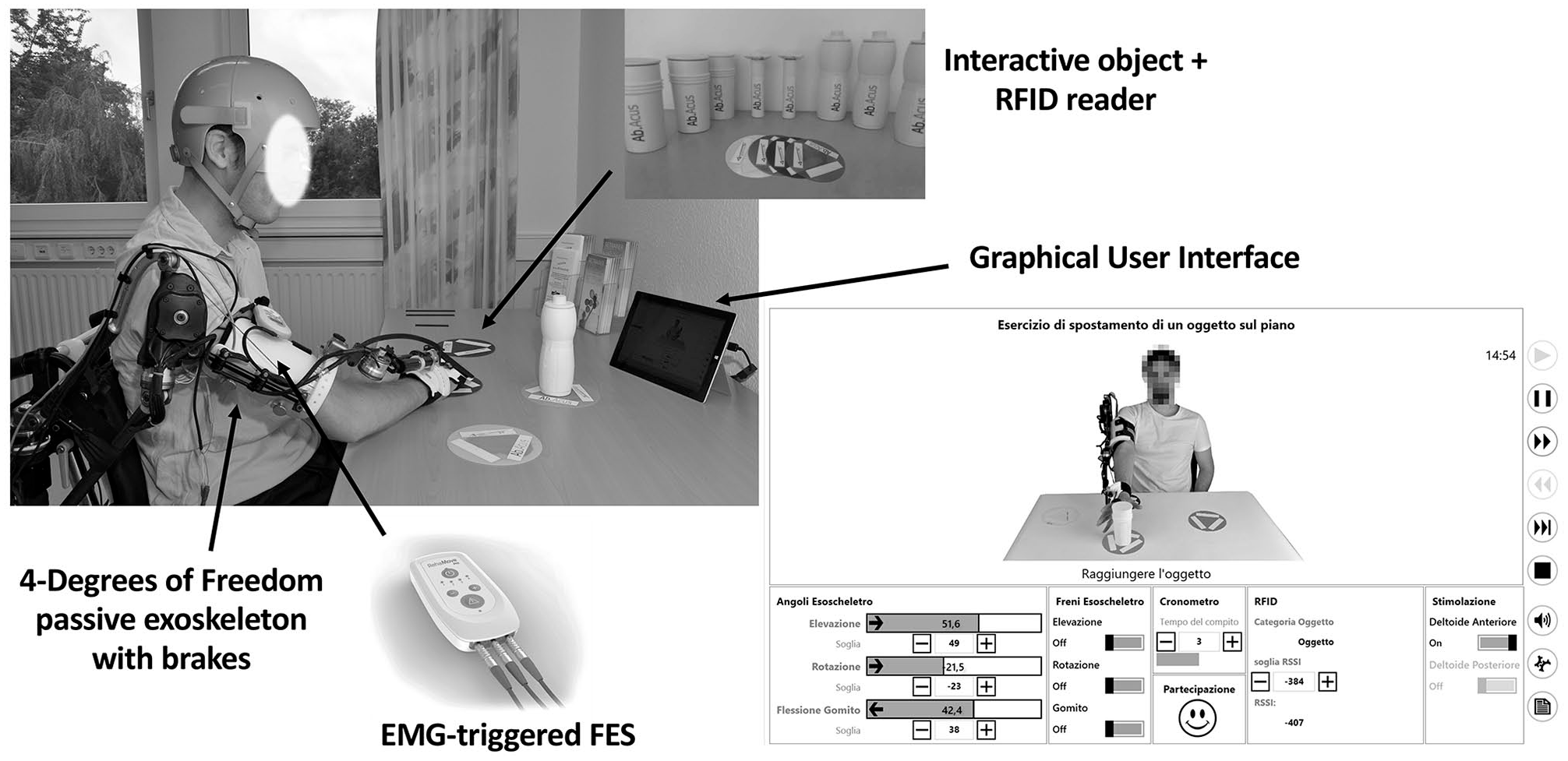
The RETRAINER system.
Patients in both groups did not receive any other interventions for the upper limb, apart from the ones foreseen by the protocol.
Outcome Measures
Patients were assessed before (T0), at the end (T1), and 1 month after the end of the intervention (T2). The primary outcome was the Action Research Arm Test (ARAT), a 19-item scale divided into 4 subtests (grasp, grip, pinch, and gross arm movement), which evaluates arm and hand functions, from 0 to 3. 27 To separate the effects of the intervention on proximal and distal functions of the upper arm, the subtests relative to gross arm movement and hand functions were analyzed separately.
Secondary outcome measures were as follows: MI arm subscale, evaluating arm motor impairment in terms of pinch grip, elbow flexion, and shoulder abduction (as for ARAT, proximal and distal segments were analyzed separately) 28 ; Motor Activity Log (MAL), a semistructured interview to assess quality and amount of movement during 30 ADLs 29 ; Box and Blocks Test (BBT), a test evaluating unilateral gross manual dexterity based on the number of blocks carried from one compartment of a box to another of equal size, within 60 s 30 ; Stroke Specific Quality of Life Scale (SSQoL), a 49-item questionnaire assessing health-related quality of life 31 ; and MRC of biceps, triceps, anterior, medial, and posterior deltoid to evaluate muscle strength.
The usability of the RETRAINER system was evaluated through the System Usability Scale (SUS), a 10-item questionnaire ranging between 0 (
Finally, using a specific form, patients were asked to report any serious and/or distressing symptoms they experienced during training.
Statistics
Minimally important change (MIC) for ARAT was estimated for early stroke patients (average of 9.5 days poststroke) in a range of 12 to 17 points. 34 Because our participants had a stroke up to 9 months before study enrollment, we considered the lower limit of the range (12 points; SD of 16.5), 34 corresponding to about 20% of the 57-point scale. Considering these values, a power of 80% and a significance level of 0.05, a sample size of 60 patients was derived. To allow a 20% dropout rate, the sample size was set to 72.
For all outcomes but MRC, linear mixed-model analyses for repeated measures were carried out, with group and time entered as fixed effects, time by group as the interaction term, age and time since stroke as covariates, patients as random factor, and the outcome as dependent variable. In case of a significant time effect, a post hoc analysis considering the groups separately was performed to estimate T1-T0 and T2-T1 changes. In case of a significant interaction effect, the between-group changes at T1 and T2, with T0, age, and time since stroke as covariates, were also estimated.
For MRC, because of its categorical nature, a nonparametric statistical analysis was carried out. Friedman test was performed separately for each group to evaluate the time effect. If a significant effect was found, Wilcoxon test with Bonferroni correction was also applied as post hoc analysis. Finally, the T1-T0 and T2-T1 changes were compared between groups using the Mann-Whitney
Percentages of clinically improved patients at T1 and T2 were finally computed: clinical improvement was defined as a change of ARAT score from baseline of >12 points. 34 The Pearson χ2 test was used to compare percentages of improved subjects between groups. Three separate subgroup analyses were performed for early subacute patients (time since stroke < 3 months 35 ), older patients (>65 years), and right/left hemiparesis. The statistical analysis was performed using SPSS v25.
Results
A total of 2190 patients were assessed for eligibility and 72 (39 in Germany and 33 in Italy) were recruited, randomized, and included in the analysis, as shown in Figure 2. The dropout rate was 11% at T1 and 14% at T2.
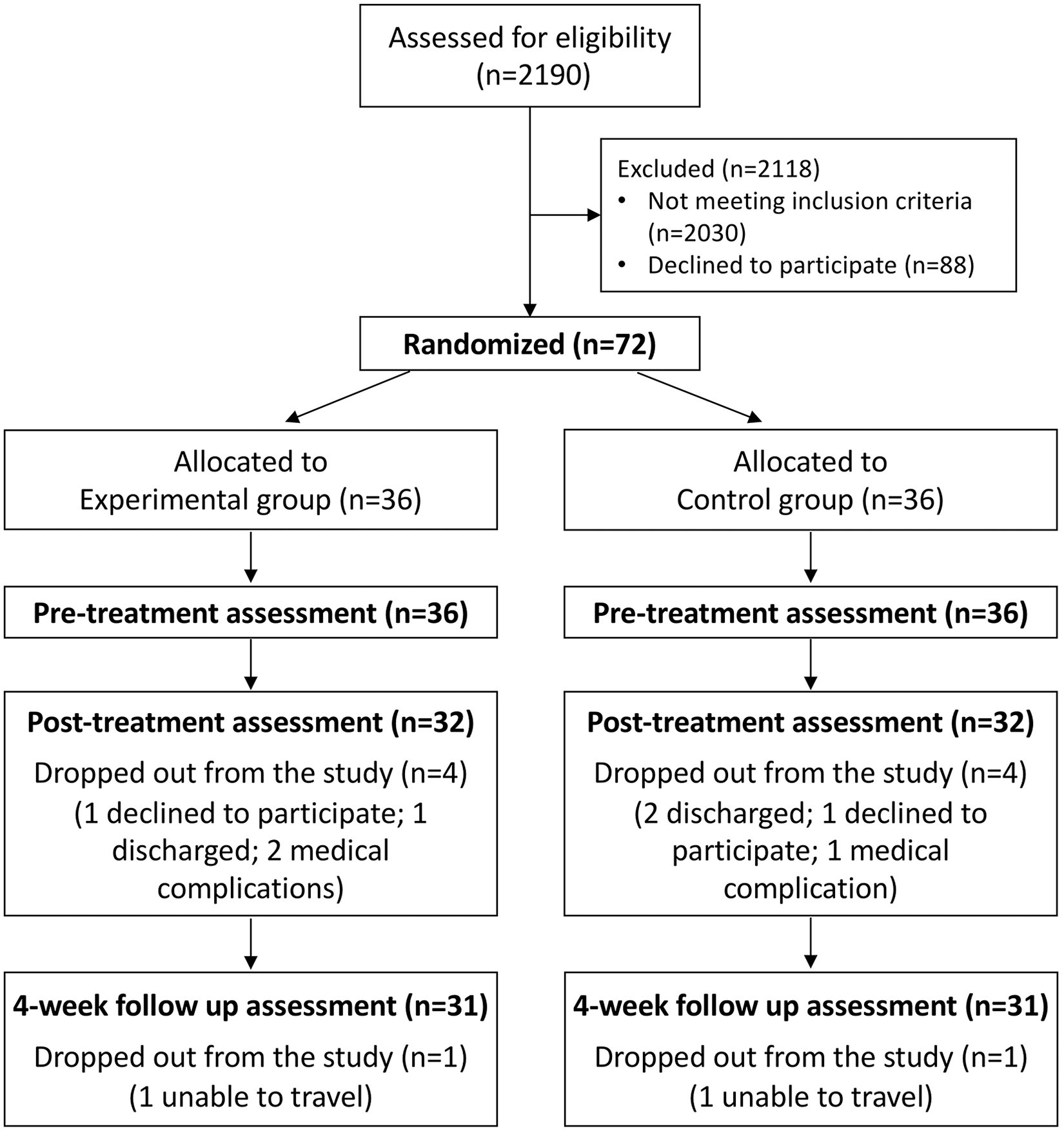
Participant CONSORT flowchart.
Table 1 shows the groups comparison at baseline: the groups were similar, with an average time since stroke of about 2 months and a severe impairment of arm functions (ARAT < 27 36 ); only age was significantly different between groups, with the experimental group having 7 years less than the control group.
Demographic Characteristics and Outcome Measures at Baseline (n = 72). a
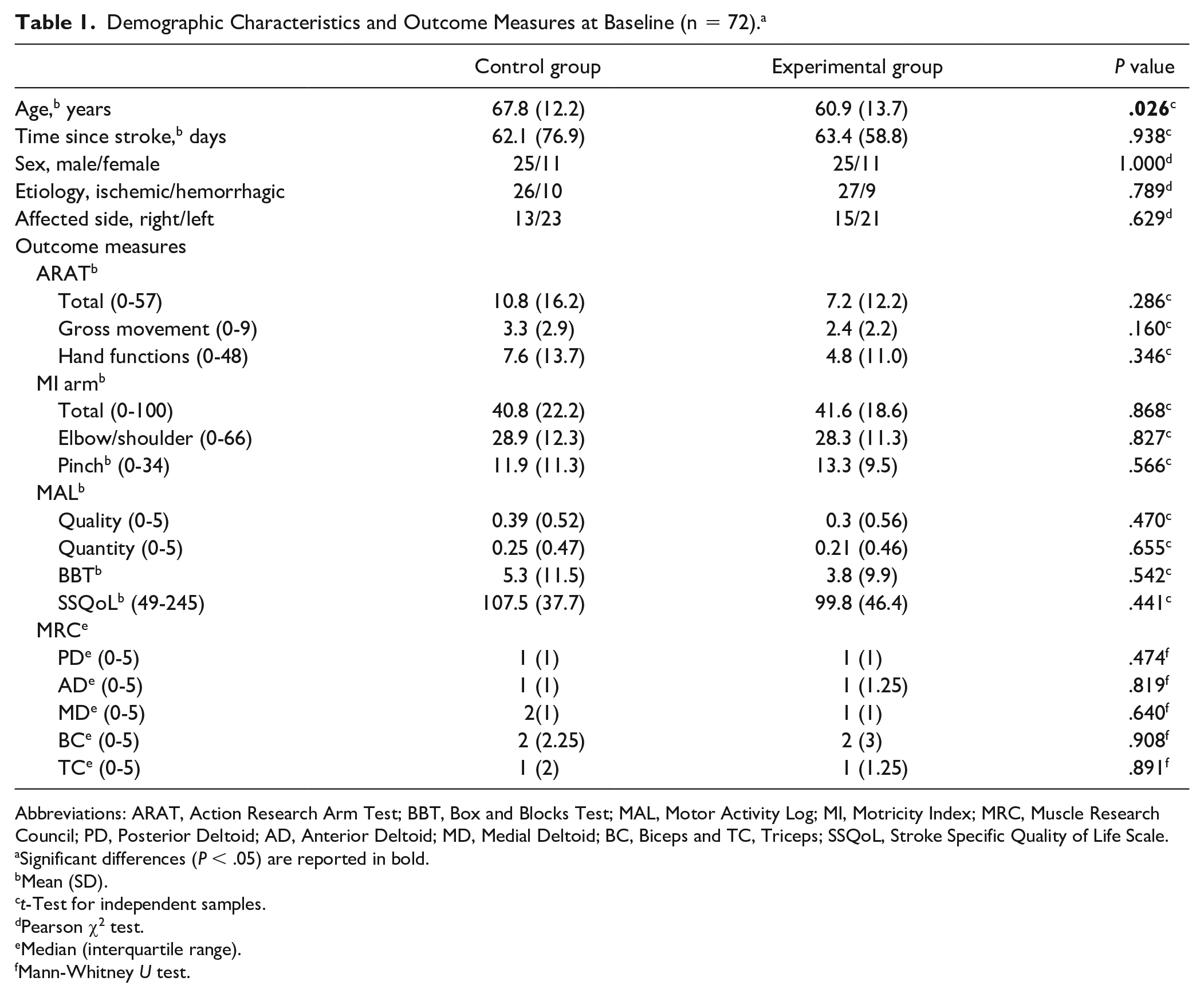
Abbreviations: ARAT, Action Research Arm Test; BBT, Box and Blocks Test; MAL, Motor Activity Log; MI, Motricity Index; MRC, Muscle Research Council; PD, Posterior Deltoid; AD, Anterior Deltoid; MD, Medial Deltoid; BC, Biceps and TC, Triceps; SSQoL, Stroke Specific Quality of Life Scale.
Significant differences (
Mean (SD).
Pearson χ2 test.
Median (interquartile range).
Mann-Whitney
Among ACT, the most representative types of therapy were repetitive task training and FES of forearm muscles for both groups (Figure 3). However, there were differences in the 2 groups relative to the type of ACT. In particular, about 20% more patients in the experimental group received FES of the forearm muscles, and about 40% of the control group received arm cycle-ergometer with respect to none in the experimental group.
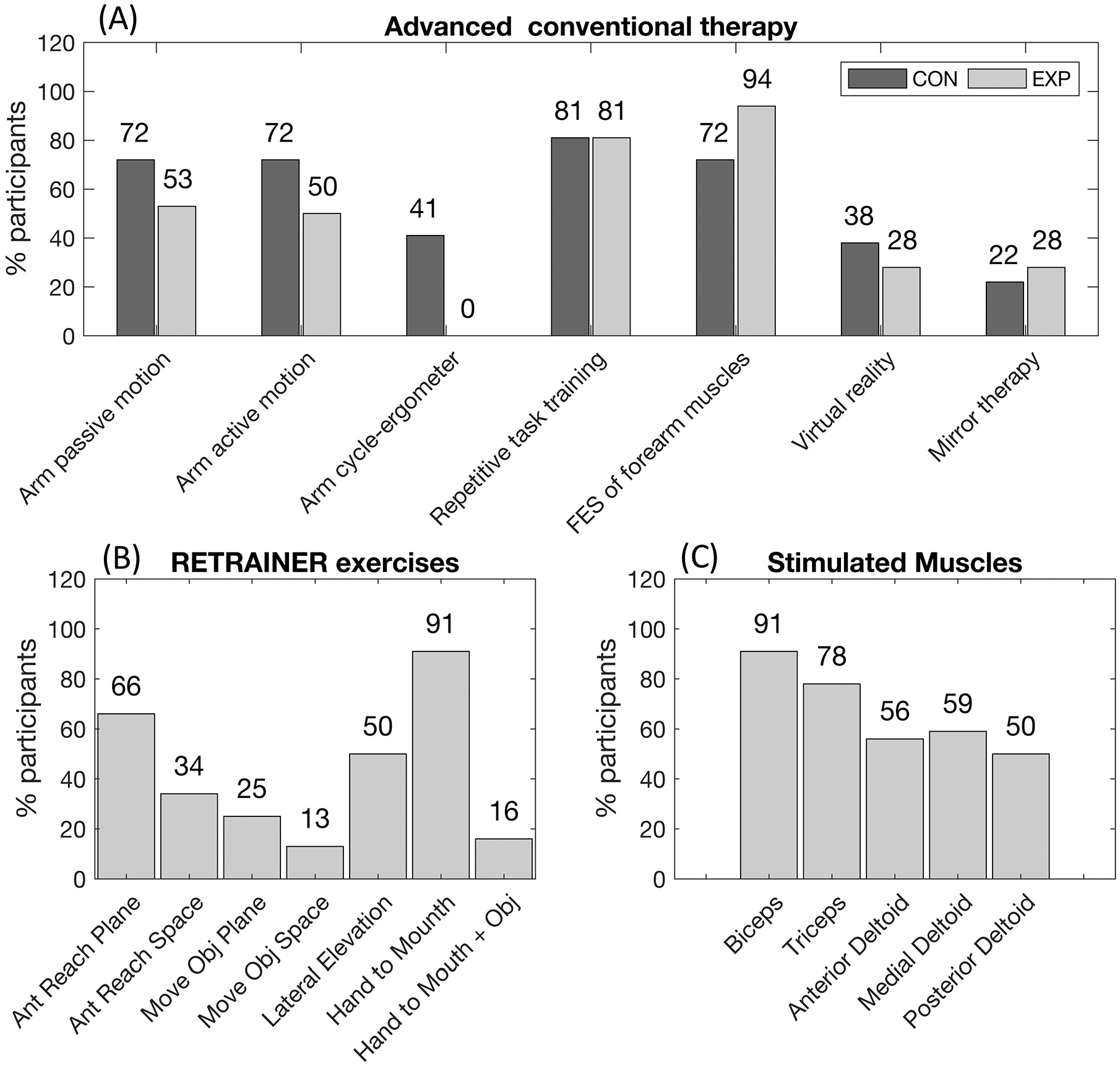
Training performed by the 2 groups: advanced conventional therapy (panel A); RETRAINER exercises (panel B); and stimulated muscles (panel C) for the experimental group.
The most selected RETRAINER exercises were “hand to mouth” and “anterior reaching on a plane” because of their simplicity. In 91% of patients, the biceps muscle was stimulated. Patients changed exercises and stimulated muscles over sessions. During each session, 1 to 3 exercises were performed; the mean training time was 29 (6) minutes, of which 12 (4) minutes involved active intense training (without pauses).
Table 2 reports the results for the primary and main secondary outcome measures. Overall, all outcomes but SSQoL showed a significant time effect (
Changes Over Time and Between Group for Primary and Main Secondary Outcome Measures (n = 72). a
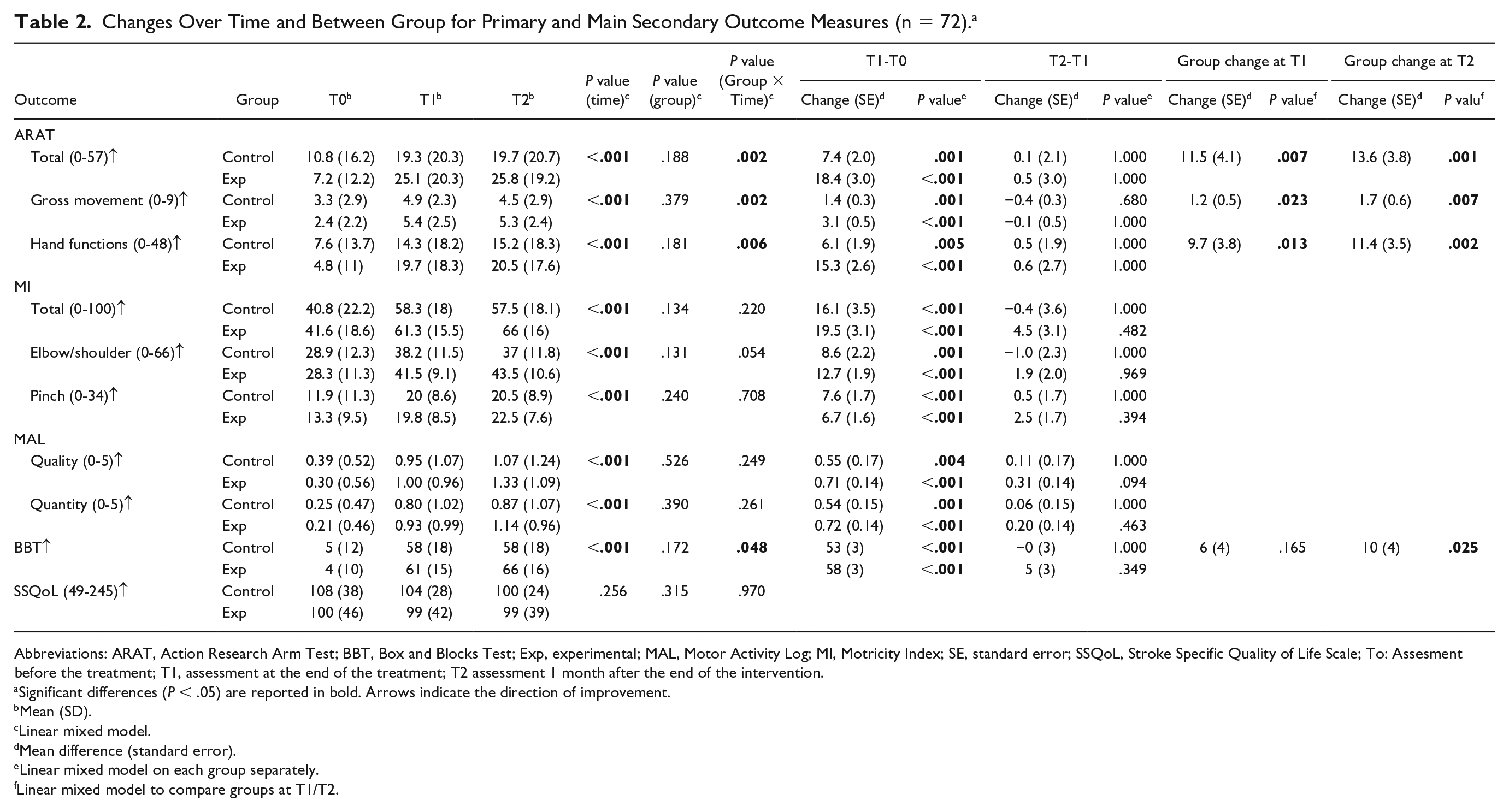
Abbreviations: ARAT, Action Research Arm Test; BBT, Box and Blocks Test; Exp, experimental; MAL, Motor Activity Log; MI, Motricity Index; SE, standard error; SSQoL, Stroke Specific Quality of Life Scale; To: Assesment before the treatment; T1, assessment at the end of the treatment; T2 assessment 1 month after the end of the intervention.
Significant differences (
Mean (SD).
Linear mixed model.
Mean difference (standard error).
Linear mixed model on each group separately.
Linear mixed model to compare groups at T1/T2.
Supplementary Table 1 reports MRC scores: both groups showed significant improvements for all muscles at T1, which were maintained at T2. No difference in terms of strength was highlighted between groups.
Figure 4 shows the proportion of clinically improved patients: 47% (15 out of 32) in the experimental group, and 28% (9 out of 32) in the control group improved arm functions at T1 (Figures 4A), but this difference was not significant; a significant difference (
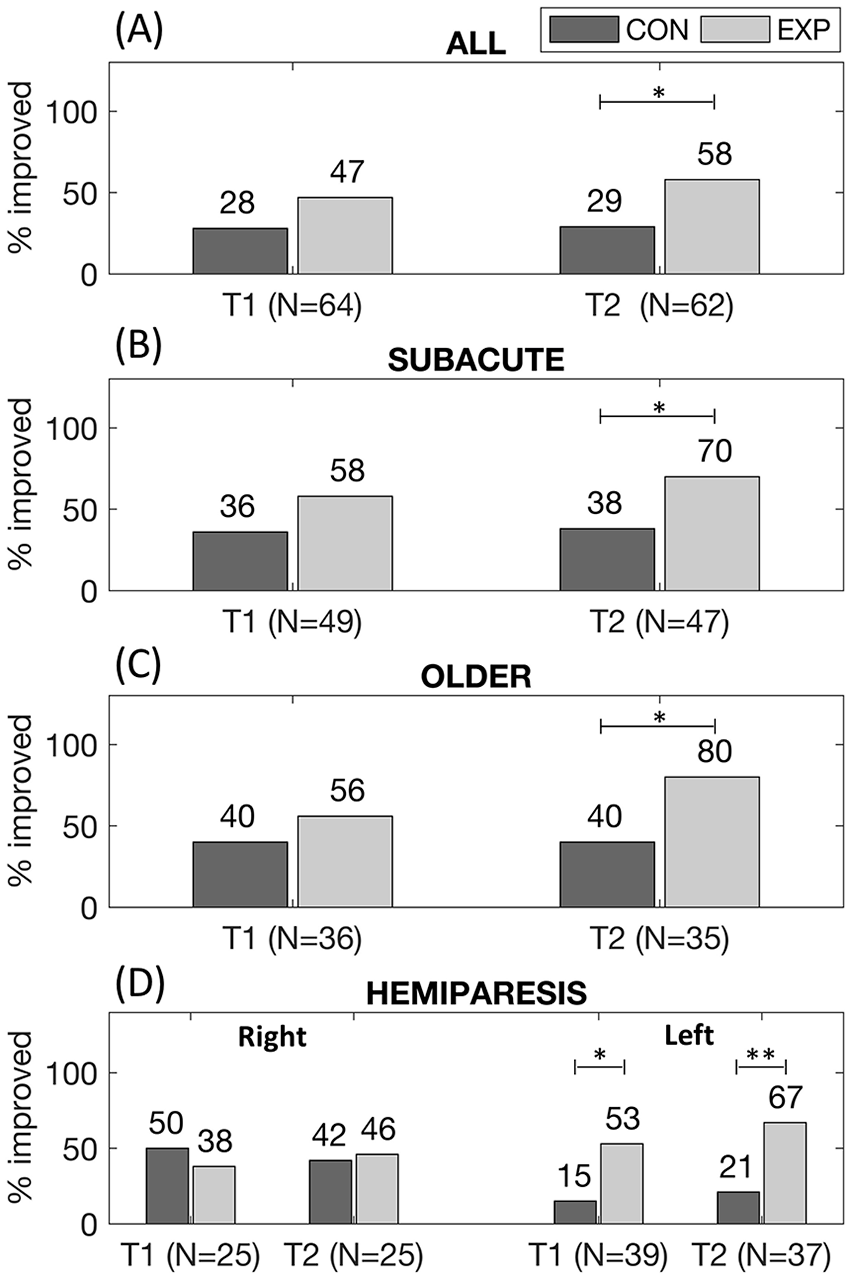
Proportion of improved patients (Action Research Arm Test change > 12 points) for all participants (panel A), for early subacute patients (time since stroke < 3 months; panel B), for older patients (age > 65 years; panel C), for patients with right (panel D, left) and left hemiparesis (panel D, right).
Compliance with the intervention was very high: patients of both groups received on average 26 (2) sessions, with minimal values of 21 and 22 in the control and experimental groups, respectively. Neither of the interventions caused serious adverse events.
Finally, patients considered RETRAINER moderately usable (mean value of SUS 61.5 ± 22.8): higher values were found for patients trained in Italy (72.8 ± 22.3) with respect to patients trained in Germany (50.8 ± 18.0). The results of the TAM questionnaire highlighted that RETRAINER was considered useful (76% ± 24%) and easy to use (76% ± 27%); however, it was moderately perceived as externally controlled (55%±35%) and no fun (53% ± 35%).
Discussion
This RCT showed the superiority of arm training assisted by an EMG-triggered hybrid robotic system with respect to equally intensive ACT for the recovery of arm functions (ARAT) and dexterity (BBT) in the Activity domain of ICF. Similar improvements were instead found for arm motor impairment (MI) and strength (MRC) in the Body Functions and Structure domain and ADL (MAL) in the Participation domain. Finally, neither of the interventions improved self-perceived quality of life (SSQoL).
Several aspects of the RETRAINER system might explain the higher improvements in arm functions achieved by the experimental group. First, RETRAINER allows to practice personalized and task-oriented exercises, which are associated with greater changes in brain activations, 37 correlated to motor and functional recovery. 3 Second, the experimental group performed active intensive training for 12 minutes out of the total 30-minute training, which means high-intensity training. Third, RETRAINER might have enhanced the sensory input, which plays a fundamental role in the motor recovery process. 38 Indeed, RETRAINER provides multisensorial afferent feedbacks in a 3D workspace: the FES-enhanced proprioception, which stimulates reorganization at the cortical level 10 ; the support of the exoskeleton, which helps the accomplishment of the task and provides the perception of an “almost” physiological motor experience 39 despite the muscular weakness; and the visual feedback in the form of happy/sad emoji, depending on the degree of active participation, which increases patient’s awareness and control of movements. Finally, RETRAINER exploits the use of EMG-triggered FES and visual feedback to favor the synchronization between the FES-induced afference and the volitional-evoked motor command, which has been shown to enhance cortical reorganization.15,16
The length of the intervention (9 weeks), longer than the majority of RCTs, which stop training after 4 to 6 weeks, might have also played a role in unveiling functional improvements of the arm, which is known to have a longer recovery time because functions of the upper extremities require finer motor control. 40
Although specifically focused on training proximal segments (ie, shoulder and elbow), RETRAINER showed greater improvements on arm functions both at the distal and proximal levels when compared with ACT. However, besides evaluating hand grasping and finger gripping, ARAT evaluates arm positioning. Indeed, participants are asked to lift objects on a shelf or move objects on a table. As previously observed, 41 these evaluation items are strongly related to the motor function of the proximal segments. RETRAINER could interfere with muscular synergies of the arm during training, improving multijoint coordinated movements, such those required for successfully completing the ARAT subtests.
Similar considerations could be drawn for BBT, which also showed the superiority of RETRAINER. Indeed, BBT provides information not only on manual dexterity, but also on the ability to organize and carry out the motor tasks and to coordinate eye-hand movements. 42
It is noteworthy to highlight that the higher improvements of arm functions did not result from a pure higher strength or reduced motor impairment (as measured by Body Functions and Structure outcomes), which instead showed similar changes in the 2 groups. These results are in contrast to those achieved in a recent RCT on upper-limb robotic therapy 7 : RATULS observes higher improvements in Body Functions and Structure outcomes in the robotic group but no between-group differences in Activity domain outcomes 7 ; on the contrary, the RETRAINER group showed significant improvements in Activity domain outcomes but similar changes in Body Functions and Structure outcomes. The hallmarks of the RETRAINER system, for example, the combination with FES, the integration of the volitional component, and the task-oriented approach, which includes also the interaction with real objects, well explain these different results. These features are well-grounded on the neurophysiological basis and go well beyond the traditional use of robots for simple imitation of therapist-mediated actions.43,44
However, the higher improvements of arm functions observed in the RETRAINER group translated neither to a better self-perceived capability of performing ADLs, which exhibited similar improvements for the 2 groups, nor to an enhanced self-perceived quality of life, which did not improve after training in either of the 2 groups. This confirms the overall limitation in transferring functional improvements from robot-assisted training to ADL performance, previously observed.7,45 One possible explanation could be the reduced functional use of objects allowed by RETRAINER. Indeed, the RETRAINER system presented here does not provide any support to hand functions, so object interaction was limited to users who had residual grasping capability. Actually, within the RETRAINER project, a second module for hand training was also designed, but its efficacy was tested in a separate RCT. 46 The arm and hand module of RETRAINER, when combined, will be able to propose a holistic approach to upper-limb training that includes the functional use of objects, recreating a more realistic training environment, potentially further impacting on the better self-perceived capability of performing ADLs.
The improvements achieved at the end of the intervention were maintained for at least 1 month in both groups, and an amplification effect could be observed in the experimental group, showing that when/if the patients get a more effective arm performance in the early intensive rehabilitation period, then they tend to exploit the better residual capacity in daily tasks, continuously increasing the improvements, hence triggering a virtuous loop.
When patients were stratified according to time since stroke (time since stroke <3 months; Figure 4B), higher proportions of improved patients were found in the early subacute subgroup confirming that early intervention is more beneficial. Clinically improved rates using RETRAINER, especially at T2, were impressively high (70%) and significantly higher than the control group37,47 Percentages of improved subjects increased also in the elderly subgroup (age >65 years; Figure 4C), indicating that technology familiarity does not influence the exploitation of RETRAINER-induced benefits and that the complexity of tasks is naturally understood for the similarity with ADLs.
Finally, a greater efficacy of the RETRAINER system was observed in patients with left hemiparesis (Figure 4D), whereas in patients with right hemiparesis, no difference in the proportion of improved subjects was found. Similarly, in a previous study, 48 patients with right brain injury were shown to respond better to virtual reality games. Left hemiparetic patients are often affected by perception disturbances of the personal and peripersonal space, which can be associated with an impaired awareness of the contralesional space/body, a negative prognostic factor for functional recovery. 49 Sensorimotor functions allowing successful interactions with the external world are linked to body representations. 50 RETRAINER allows to explore the peripersonal space with intensive multisensory augmented stimulations, and this might have increased its effect on left hemiparetic patients. Conversely, this treatment modality might not have such a crucial role in right hemiparetic patients, who generally show reduced body/spatial perception disorders. This consideration should be taken with caution because no specific assessment of cognitive-perceptual deficits was performed on the patients. Further investigation is needed to fully comprehend this point.
Overall, patients considered RETRAINER moderately usable (mean SUS = 62, with 68 being considered the threshold for a good usability level 32 ). Higher scores were registered in Villa Beretta, where therapists were involved earlier, since the start of system development. At this stage, RETRAINER is still a small-series prototype. After this RCT, important indications for design improvements emerged, especially about robustness, donning, and calibration time. For example, the graphical interface was unique, encompassing both information relevant for the therapist and instructions and feedback for the patient, resulting in a crowded window that was difficult to follow. A 2-screen solution, one for the therapist and another for the patient, might be an effective and easy solution to implement.
This study has some limitations. The groups showed a significant difference in terms of age at baseline. Although age was included as covariate in the statistical model, this imbalance might have biased the results. The MIC value used in the sample size calculation was derived from a study recruiting early stroke patients, 34 and this might have influenced the statistical power. ACT was tailored on each single patient in order to be his/her best treatment option; similarly, RETRAINER exercises were selected by the therapist on single-user necessity. Nevertheless, the variety of therapies that characterized ACT and RETRAINER might be a confounding factor for the results of this RCT but deals with the essential ethical requirement of proposing to each patient the best treatment. The range of the time since stroke event of the participants was quite broad, from 2 weeks to 9 months. To mitigate this confounding factor, the time since stroke was included as covariate in the statistical model, and a secondary analysis only on the subgroup of early subacute patients was performed. The length of the treatment (9 weeks) could have played an important role in the positive results, but it is difficult to match with typical hospitalization times. This recalls the importance of developing new devices that allow neurophysiology-based training, such as RETRAINER, usable in outpatient settings or even at home. The follow-up period of 1 month is too short to evaluate medium- and long-term effects. Future studies should include longer follow-up periods. Perceptive disorders were not assessed, and this might have prevented better identifying patients who could benefit the most from the system. Assessment of psychological symptoms was not included, although poststroke depression strongly influences the recovery process. Ultimately, comparisons of the effects for right and left hemiparesis are limited by 2 factors: first, we did not collect dominant side; second, we did not assess perceptual deficits.
Conclusions
This RCT compared the efficacy of 9-week arm training assisted by a robotic system, integrated with EMG triggered FES, to the best evidence for arm physiotherapy in stroke survivors. Both groups showed significant improvements in arm functions, dexterity, strength, and ADL, but patients trained with RETRAINER achieved a significantly better improvement of arm functions and dexterity. This result could be associated with the possibility to perform personalized, intensive, and task-oriented training within an enriched multimodal sensory environment (visual, tactile, and proprioceptive). However, the limited transferring of functional improvements to ADL suggests that an additional effort is needed in order to create more realistic and lifelike environments.
Overall, this RCT supports the use of hybrid robotic systems that are beyond the just autonomous imitator of therapist-mediated therapy and that are designed leveraging on a deep understanding of the neurorehabilitative mechanisms to improve the clinical effectiveness of neurotechnology. Eventually, the simultaneous use of several easy-to-use systems might allow to perform a personalized therapy in group sessions, enabling more intensive therapy without increasing personal resources, usable in outpatient settings or even at home.
Supplemental Material
sj-docx-1-nnr-10.1177_1545968321997769 – Supplemental material for A Robotic System with EMG-Triggered Functional Eletrical Stimulation for Restoring Arm Functions in Stroke Survivors
Supplemental material, sj-docx-1-nnr-10.1177_1545968321997769 for A Robotic System with EMG-Triggered Functional Eletrical Stimulation for Restoring Arm Functions in Stroke Survivors by Emilia Ambrosini, Giulio Gasperini, Johannes Zajc, Nancy Immick, Andreas Augsten, Mauro Rossini, Roberto Ballarati, Micheal Russold, Simona Ferrante, Giancarlo Ferrigno, Maria Bulgheroni, Walter Baccinelli, Thomas Schauer, Constantin Wiesener, Margit Gfoehler, Markus Puchinger, Mathias Weber, Sebastian Weber, Alessandra Pedrocchi, Franco Molteni and Karsten Krakow in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
The authors would like to acknowledge the patients for participating in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by EU’s Horizon 2020 Research and Innovation program (RETRAINER project; GA:644721).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
