Abstract
Background. Recovery of motor function after stroke may depend on a balance of activity in the neural network involving the affected and the unaffected motor cortices. Objective. To assess whether transcranial direct current stimulation (tDCS) can increase the training-induced recovery of motor functions. Methods. In an exploratory study, 14 patients with chronic stroke and mean Fugl-Meyer Upper Extremity Motor Assessment of 29 (range = 8-50) entered a double-blind sham-controlled study, aimed to investigate neurophysiological and behavioral effects of bihemispheric tDCS (cathodal stimulation of the unaffected motor cortex and anodal stimulation of the affected motor cortex), combined with constraint-induced movement therapy (CIMT). Results. Patients in both groups demonstrated gains on primary outcome measures, that is, Jebsen Taylor Hand Function Test, Handgrip Strength, Motor Activity Log Scale, and Fugl-Meyer Motor Score. Gains were larger in the active tDCS group. Neurophysiological measurements showed a reduction in transcallosal inhibition from the intact to the affected hemisphere and increased corticospinal excitability in the affected hemisphere only in the active tDCS/CIMT group. Such neurophysiological changes correlated with the magnitude of the behavioral gains. Both groups showed a reduction in corticospinal excitability of the unaffected hemisphere. Conclusions. CIMT alone appears effective in modulating local excitability but not in removing the imbalance in transcallosal inhibition. Bihemispheric tDCS may achieve this goal and foster greater functional recovery.
Keywords
Introduction
After stroke, intensive motor practice is essential to promote recovery from motor impairments. 1 It is worth noting that skill learning through physical therapy seems necessary to induce cortical plasticity, relearning of lost capacities, and long-term retention.2,3 As far as hemispheric stroke is concerned, although behavioral motor training is known to promote contralesional and ipsilesional plastic changes, 3 functional outcomes are often of limited practical significance. 4 In this context, noninvasive brain stimulation appears an interesting option as an add-on intervention to standard physical therapies. Particularly, transcranial direct current stimulation (tDCS) might be used for priming cortical excitability during motor training. Indeed, by delivering a weak direct current over the scalp, tDCS induces long-lasting alterations in cortical excitability through the depolarization or hyperpolarization of neuronal populations. 5 Different tDCS strategies have been tested to promote motor recovery, following a model of interhemispheric rivalry between the damaged and the intact hemispheres. The model assumes that a motor deficit may arise from both a reduced output from the damaged hemisphere and a disproportionate (ie, uncontrasted) inhibition from the unaffected hemisphere.6,7 Hence, motor recovery might be facilitated by upregulating the excitability of the lesioned motor cortex through anodal tDCS or by downregulating the excitability of the intact motor cortex through cathodal tDCS.6-8
So far, in patients with stroke the effects of tDCS have been mostly examined without coupling it with any specific behavioral therapy. These studies show only limited functional benefits. 9 This is likely to be a suboptimal approach, given that tDCS activates a bulk of neural circuits in a nonspecific way. Instead, motor training, if coupled with tDCS, might steer plastic processes toward a functional outcome,10-13 a time-honored concept already proposed for neuroplastic drugs. 14 Beyond contrasting the potentially maladaptive recovery fostered by interhemispheric inhibition, tDCS might also induce some focal beneficial effects by activating neural circuitries in the damaged hemisphere.15-17
In this article, we investigated the behavioral effects of bihemispheric motor stimulation (ie, cathode electrode on the unaffected motor cortex and anode electrode on the affected motor cortex) associated with constraint-induced movement therapy (CIMT).18,19 This therapy combines intensive physical practice using the affected upper limb with restricted use of the unaffected upper limb, in order to foster intrinsic recovery and overcome “learned nonuse.” 18 CIMT was chosen as it shares some similarities with the mechanisms of action of bihemispheric tDCS, namely, it aims at decreasing activity in the unaffected motor cortex and increasing it in the affected motor cortex.20,21 The adjuvant use of bihemispheric tDCS with CIMT might potentiate its individual modulatory effects on the motor neural network, thus augmenting clinical gains. This combined treatment was compared with the benefit achievable with CIMT alone (plus sham tDCS). We also explored local and bihemispheric neurophysiological changes by means of transcranial magnetic stimulation (TMS).
Materials and Method
Participants
Fourteen patients suffering from hemiparesis following hemispheric stroke were recruited from the outpatient population of 3 neurological research units. Demographic and clinical data are reported in Table 1. Patients gave their informed consent to the protocol approved by the ethical committee in each research center.
Summary of the Clinical Data
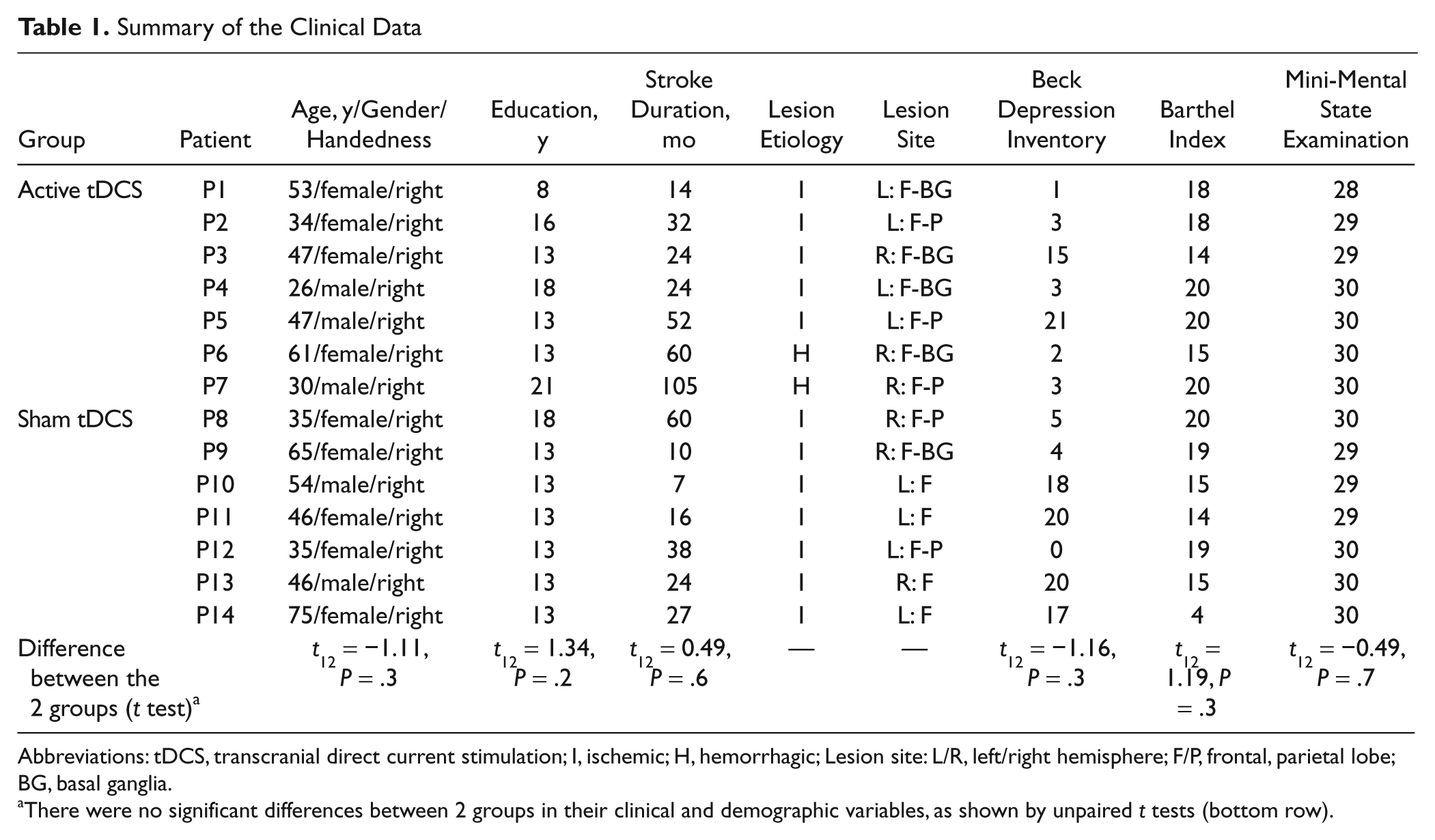
Abbreviations: tDCS, transcranial direct current stimulation; I, ischemic; H, hemorrhagic; Lesion site: L/R, left/right hemisphere; F/P, frontal, parietal lobe; BG, basal ganglia.
There were no significant differences between 2 groups in their clinical and demographic variables, as shown by unpaired t tests (bottom row).
Entry criteria were ischemic or hemorrhagic first-ever stroke, stroke onset >6 months prior to study enrollment, and functional inclusion criteria defined by the EXCITE trial. 22 Exclusion criteria were prestroke motor impairments affecting the upper limbs, moderate to severe major depression, having already received CIMT and/or tDCS treatment, and contraindications to TMS or tDCS.5,23
Patients met either higher functioning or lower functioning motor criteria derived from Taub et al 24 and showed moderate to severe hemiparesis, as indexed by Upper Extremity Fugl-Meyer (mean score= 26; range = 8-50).
Procedure
The assessment of motor functions and the administration of the functional scales and questionnaires were performed by a trained staff, blinded to group assignment.
The following instruments for motor function assessment were used. The Jebsen Taylor Hand Function Test (JHFT) 25 measures hand function in real-life activities, by evaluating the time required to perform different tasks. Handgrip Strength (HS) 26 measures the maximum voluntary isometric strength of the affected hand, through a hydraulic hand dynamometer (the mean of 3 trials was taken as result). Motor Activity Log Rating Scale (MAL) 27 evaluates the amount and quality of daily life activities of the paretic arm. Upper Extremity Fugl-Meyer Motor Score (FMM) 28 assesses selective movement. These measures were obtained at baseline, day 1 (D1), day 5 (D5), at the end of the treatment (D10), and at 2 and 4 weeks follow-up (Fu1 and Fu2, respectively). The FMM was not recorded at D1 and D5. Five patients (2 in the active group and 3 in the sham group) did not complete the JHFT. Two patients (1 in the active group and 1 in the sham group) did not complete the HS task.
Functional scales and questionnaire included the Barthel Index Score, 29 the Beck Depression Inventory, 30 and Mini-Mental State Examination 31 scored at baseline only (see Table 1). Two visual analogue scales for anxiety and pain/comfort were administered daily, before and after each treatment session. A questionnaire for the adverse effects of tDCS was administered at the end of each tDCS session.
Assessment of cortical excitability
TMS was used to measure corticospinal excitability (motor evoked potential [MEP]) and transcallosal inhibition (TI) in both the affected and the unaffected motor cortices. 32 The evaluation was performed by a trained staff, blinded to group assignment.
Focal TMS to the primary motor cortex (M1) was delivered using a 70-mm figure-of-eight coil, held tangential to the skull and aligned in the parasagittal plane with the handle rotated 45° lateral. Two coils were integrated using a Bistim device (The Magstim Company, Dyfed, UK) to deliver paired-pulse TMS. Online monitoring of the electromyographic (EMG) activity in response to TMS was performed. EMG signals were band-pass filtered (20-2000 Hz), digitized, and stored on a computer for offline analysis (Powerlab/Scope; AD Instruments, Colorado Springs, Colorado; XCalibur EMG System, Optima Medical Ltd, London, UK). MEPs were recorded from the left and the right first interosseous (FDI) muscles. Pairs of silver/silver chloride surface electrodes were placed over the muscle belly (active electrode) and over the associated joint of the muscle (reference electrode).
For each hemisphere, first the optimal scalp position for the TMS induction of an MEP from the contralateral FDI was determined. The coil was then held in place by a mechanical device throughout the evaluation.
The resting motor threshold (MT = the lowest stimulation intensity inducing 5 out of 10 MEPs, with peak-to-peak amplitude of at least 50 µV) was determined in both hemispheres. The 2 groups were similar with respect to their MT, and no change of MT occurred after the training (see Analysis of Motor Threshold in the Supplementary Material). Cortical excitability was tested by delivering 10 TMS pulses to M1 of each hemisphere (TMS intensity of 20% above the individual MT to induce MEPs of 1 mV). The same intensity was kept constant at all assessments. Muscle activity was monitored by real-time EMG to confirm the relaxed status before the stimulation.
TI was evaluated by using paired-pulse TMS: a suprathreshold stimulus (using the same intensity as for MEP) was applied to M1 of 1 hemisphere, and 10 ms later a suprathreshold stimulus was delivered to the contralateral M1 through the twin coil. Ten MEPs were recorded for each hemisphere, and their peak-to-peak amplitude was averaged. Evaluation order was randomly balanced between the 2 hemispheres (ABBA design; in each hemisphere, the measurement of cortical excitability was followed by TI). The assessment was performed at the baseline and on D10, at least 5 hours after the end of the training.
Constraint-induced movement therapy
Each subject underwent a 14-day CIMT, administered by a trained therapist, who was not involved in the pre–post evaluations. Participants had to wear, on the nonparetic hand, a resting splint secured in a sling, which hindered hand and finger activity (Skil-Care Rigid Palm Padded Mitt; AliMed, Inc, Dedham, Massachusetts; Figure 1). The splint had to be worn for at least 90% of waking hours. During the 10 weekdays of the treatment period, all patients received up to 4 hours per day of training of the affected arm, only in the laboratory. 33 Training tasks were designed according to a behavioral “shaping” technique and were designed to force an intensive use of the paretic extremity, while requiring a progressive improvement of the quality of movement. 34 Nine different shaping tasks were used during the 4-hour training period (the list of the tasks is available at ptjournal.apta.org; see Ref. 13). On D1, the majority of patients completed a minimum of 6 out of 9 tasks. On D10, all patients were able to complete, on average, 1.5 repetitions of the 9 tasks, showing an improvement of their performance.
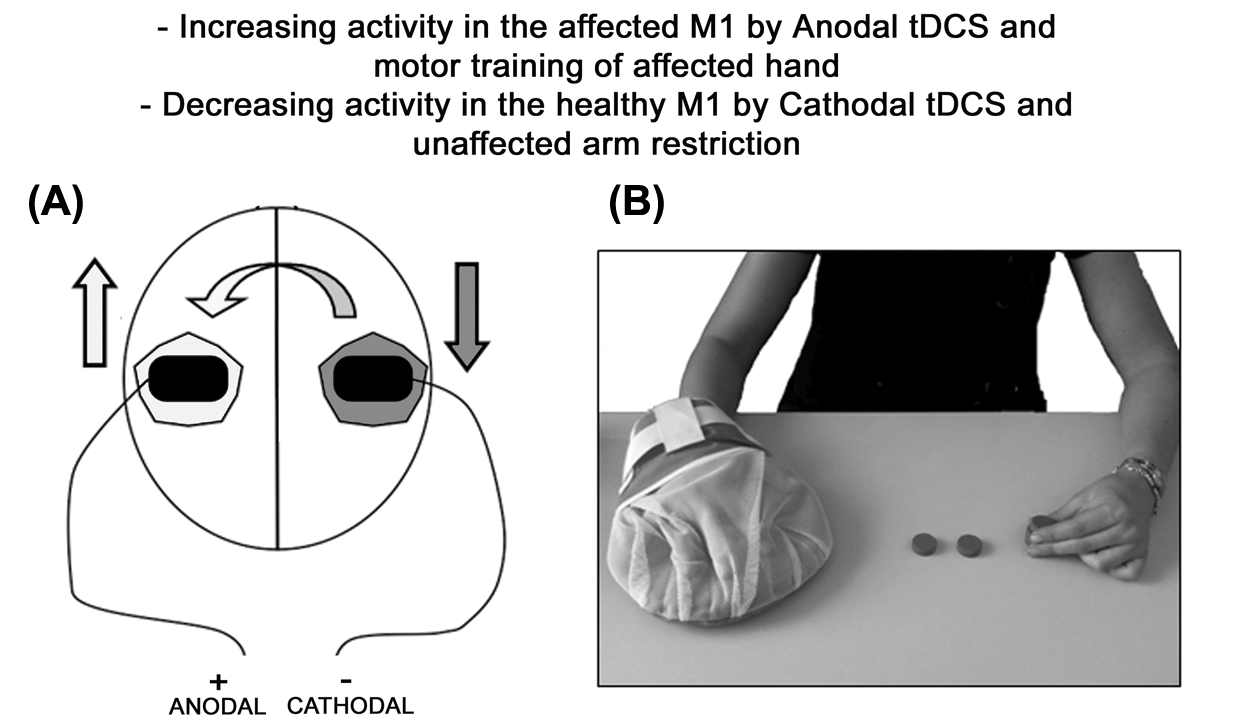
(A) Transcranial direct current stimulation (tDCS) stimulation. Electrode montage: the unaffected hemisphere (grey area) is inhibited (down arrow) by the excitability-diminishing cathode electrode; at the same time, the affected hemisphere (white area) is stimulated (up arrow) by the excitability-enhancing anode electrode. The curving arrow represents transcallosal inhibition from intact to affected M1. (B) The CIMT therapy combines intensive physical practice using the affected upper limb with restricted use of the unaffected upper limb by using a resting hand splint.
Transcranial direct current stimulation
Direct current was transferred by a saline-soaked pair of surface sponge electrodes (7 × 5 cm, 35 cm2) and delivered by a battery-driven, constant current stimulator (Eldith Ltd, Illmenau, Germany). The device can be set in advance to deliver either the active or the sham stimulation, thus keeping both the patient and the therapist masked. 35 The 2 treatment modalities were the following:
Active tDCS (plus CIMT): The anode electrode was placed over the affected M1 (C3/C4, according to the 10/20 EEG system). The cathode electrode was placed over the contralateral (unaffected) M1 (Figure 1). At the beginning of each training session, a constant current of 2 mA intensity (current density of 0.57 A/m2) was applied for 40 minutes (total charge of 4.8 C; fade-in/fade-out phases = 10 seconds). 36
Sham tDCS (plus CIMT): The same site and parameters of stimulation were employed, but the stimulator was turned off after 30 seconds of stimulation. This ensured that patients could feel the initial itching sensation at the beginning of tDCS, a requisite for successful masking. 35
Patients were randomly assigned to 1 of the 2 groups (7 patients in each group). With respect to the clinical and demographic data, no significant differences between the 2 groups were found, as assessed by unpaired t tests (see Table 1).
Statistical Analyses
In this pilot study, our a priori primary outcome was the motor performance as indexed by the JHFT, HS, FMM, and MAL, while cortical excitability parameters were used to identify neurophysiological correlates of the treatment. The data were analyzed using Statistica for Windows (release 6.0, StatSoft). For each test, repeated-measures analyses of variance (ANOVA) were performed, with a between-subjects factor Group (active, sham tDCS) and a within-subjects factor Time (baseline, D1, D5, posttreatment assessment on D10, 2 and 4 weeks follow-up, ie, Fu1 and Fu2). For the FMM, the assessment was not performed on D1 and D5. Outcomes of the ANOVAs are reported in Table 2. Post hoc analyses were performed by using the Bonferroni correction.
F, P-Level, and pη2 Values From ANOVAs (Between-Subjects Factor: Group, Within-Subjects Factor: Time) of the Output of Each Test Assessing Motor Functions

Abbreviations: ANOVA, analysis of variance; JHFT, Jebsen Hand Function Test; HS, Handgrip Strength; FMM, Upper Extremity Fugl-Meyer Motor Score; MAL, Motor Activity Log Rating Scale.
The effect of treatment on cortical excitability was assessed using the same ANOVA model. Finally, changes in the percentages of TI were assessed by paired t tests. For every analysis, the significance level was set at P < .05. Effects were evaluated also according to their standardized effect size index, that is, partial eta-squared (pη2). 37
A power analysis was also run, which is described in the Supplementary Material.
Results
None of the patients reported any discomfort.
Motor Function
Jebsen Taylor Hand Function Test
The analysis of the total latency scores (in seconds) showed a greater improvement in patients who received active tDCS (Group by Time interaction; see Table 2) at the end of the treatment, which remained stable at the follow-up (baseline = 102 seconds, P < .01, vs D10 = 68 seconds, Fu1 = 70 seconds, Fu2 = 72 seconds). Conversely, no differences across Time emerged in the sham group (P = 1, for all comparisons). Baseline performance was comparable in the 2 groups (P = 1; Figure 2A).
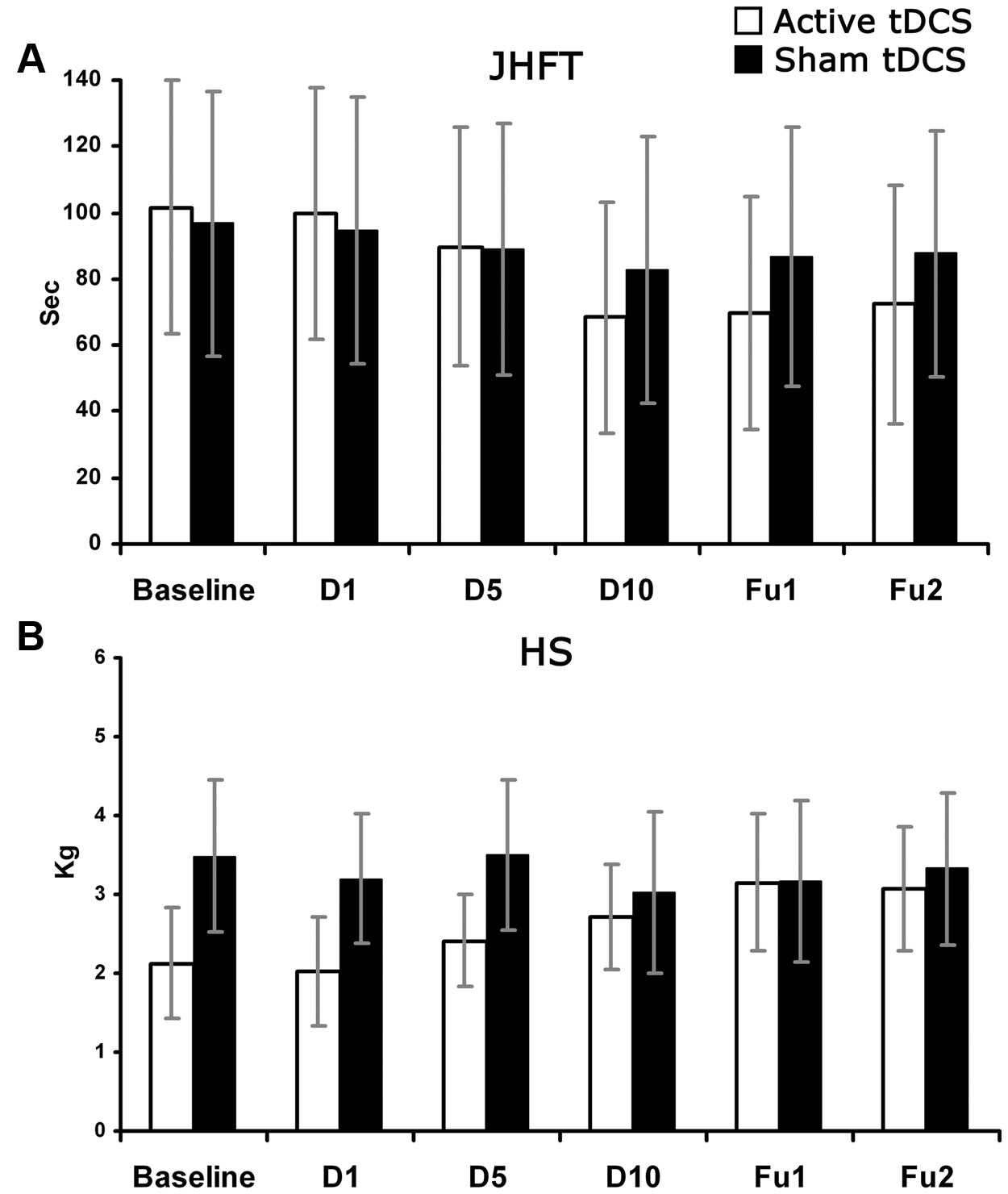
Patients’ average performance scores (SE) in the baseline and in the posttreatment evaluations: D1, 1st day of treatment; D5, 5th day of treatment; D10, 10th day of treatment; Fu1, follow-up 2 weeks after the end of treatment; Fu2, follow-up 4 weeks after the end of treatment. (A) JHFT = Jebsen Hand Function Test: RTs (in seconds); (B) HS = Handgrip strength (in kg). White columns = Active transcranial direct current stimulation (tDCS) group; black columns = Sham tDCS group. Error bars = SE.
Hand Strength
The Group by Time interaction (Table 2) showed that only patients who received active tDCS improved after the treatment (baseline = 2.1 kg, vs Fu1 = 3.2 kg, Fu2 = 3.1 kg, P < .05). No change emerged in the sham group (P = 1, for all comparisons). Baseline performance was comparable in the 2 groups (P = 1; Figure 2B).
Upper Extremity Fugl-Meyer Motor Score
The Group by Time interaction showed an improvement at the end of the treatment in the active group only (baseline = 25.4, vs D10 = 31.7, Fu1 = 32.9, Fu2 = 33.3, P < .01); instead, in the sham group no differences were found among the evaluations (P = 1). The 2 groups did not differ at baseline (P = 1; Table 2 and Figure 3A).

Motor Activity Log Rating Scale
With respect to the amount of use, the effect of Time showed an improvement in both groups at the 4-week follow-up (Fu2 = 2.6, vs baseline = 2, P < .05). As for the quality of movement, the effect of Time showed in both groups an improvement at the end of the treatment (baseline = 1.5, vs D10 = 2.5, P < .05), which then remained stable (Fu1 = 2.4, Fu2 = 2.4, P < .05). The 2 groups did not differ at baseline (Table 2 and Figure 3B).
Cortical Excitability Measures
Single-pulse TMS
For the damaged hemisphere, the Group by Time interaction (F 1,12 = 5.22, P < .04, pη2 = .31) showed that only in the active group the mean peak-to-peak amplitude of MEPs increased after the treatment (baseline = 98 µV vs D10 = 119 µV, P < .05), whereas in the sham group no change in the cortical excitability of the damaged motor cortex emerged (baseline = 231 µV vs D10 = 210 µV, P = 1). No difference was found between the 2 groups at baseline (P = 1). The effects of Group (F1,12 = 0.44, P = .5, pη2 = .04) and of Time (F1,12 = 0.006, P = .9, pη2 = .001) were not significant.
For the intact hemisphere, the effect of Time (F1,12 = 12.37, P < .01, pη2 = .5) showed in both groups a decrement of MEP amplitude after the treatment (1057 µV vs baseline = 1262 µV, P < .05). The main effect of Group (F1,12 = 0.51, P = .5, pη2 = .04) and the Group by Time interaction (F1,12 = 1.96, P = .1, pη2 = .2) were not significant (see Figure 4A).
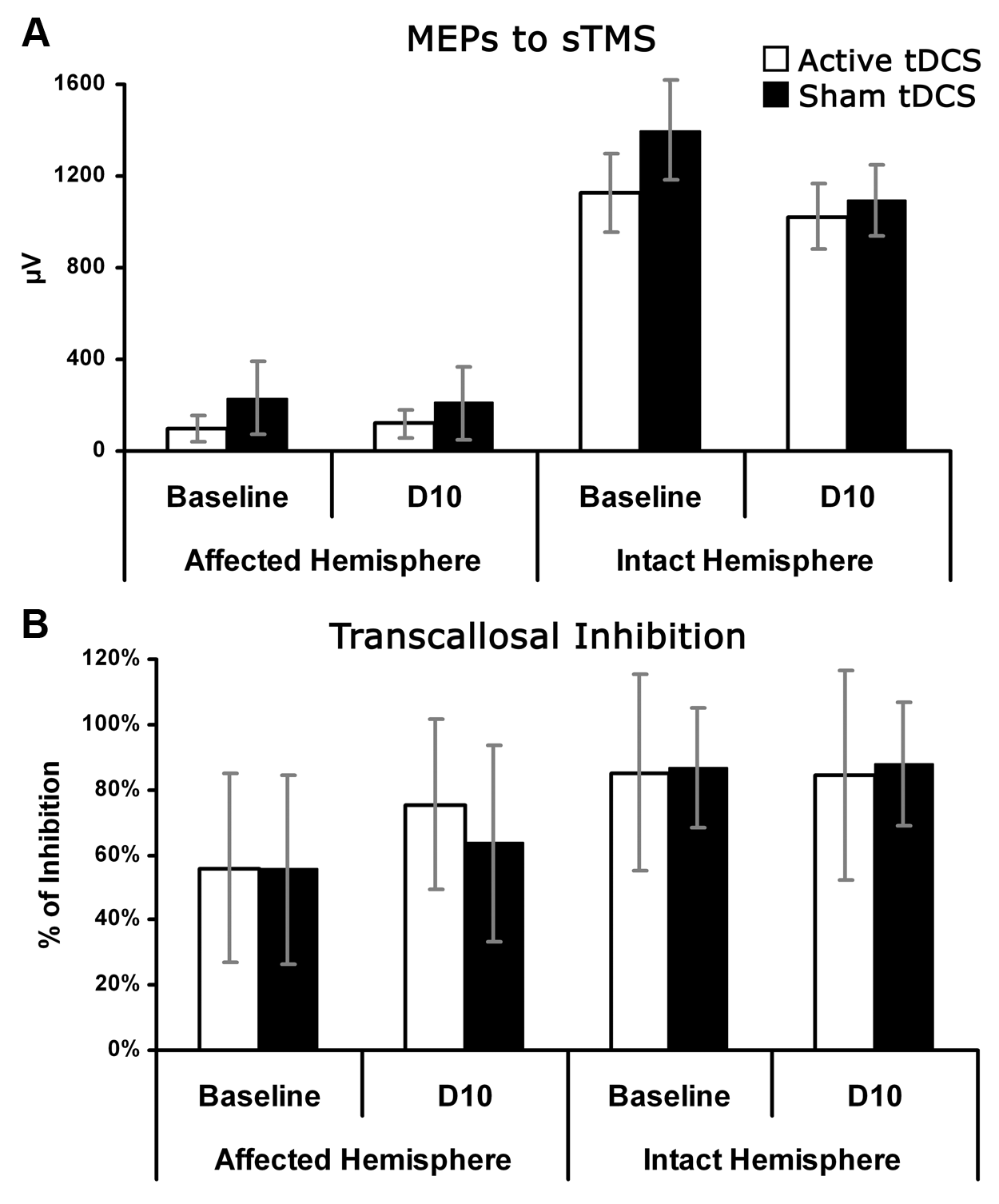
(A) Motor evoked potentials’ (MEPs’) peak-to-peak amplitude (µV) to single-pulse transcranial magnetic stimulation (TMS) in the baseline and after treatment (D10) for the affected and the intact motor cortices. (B) Mean percentage transcallosal inhibition, assessed by paired-pulse TMS, at the baseline and at the posttreatment evaluation (D10) for the affected and unaffected motor cortices. tDCS, transcranial direct current stimulation. Columns and error bars as in Figure 2.
Transcallosal inhibition
The percentage of TI was calculated by dividing the single-pulse MEPs’ amplitude by the MEPs’ amplitude modulated by a previous pulse in the contralateral hemisphere × 100. Values <100% indicated the degree of TI. In each group, and for each hemisphere, the mean percentages of TI before and after the therapy were compared by paired t tests. In the active group, a significant reduction of TI by the intact motor cortex on the damaged motor cortex after treatment was found (baseline = 56%, D10 = 75%, t 6 = −3.32, P < .04), whereas in the sham group no significant change was found (baseline = 56%, D10 = 64%, t6 = −0.75, P = .5). As for the damaged-to-intact TI, the treatment did not induce any significant change in both groups: Active group, 85% versus 85%, t6 = 0.1, P = .9; Sham group, 87% versus 88%, t6 = −0.38, P = .7 (see Figure 4A). Interestingly, at baseline, there was a significant difference in TI when comparing the 2 hemispheres: the unaffected hemisphere had a stronger inhibitory influence on the affected one, compared with the unaffected- to-affected TI (t6 = −2.9, P < .02). The 2 groups did not differ at baseline with respect to their TI (t6 = 0.08, P = .9; see Figure 4B).
Association Between Motor Improvement and Cortical Excitability Changes
To explore whether changes in cortical excitability correlated with changes in motor functions, Pearson correlation analyses were performed between the change in motor performance and the change in MEPs’ amplitude and in TI, both given as percentage difference between the baseline and the posttreatment. For the MEPs’ amplitude, a positive correlation was found between cortical excitability in the affected hemisphere and the FMM score (r = .67, P < .01). Conversely, there were negative correlations between intact-to-damaged TI and the FMM score (r = −.81, P < .01) and the JHFT score (r = −.55, P < .02). Other correlations did not reach significance (P > .1).
Baseline Performance and Treatment Effects
In our study, the patients’ enrollment was based on the random assignment to 1 of 2 groups. The 2 groups may not have been matched for functional severity, as we did not conduct stratified randomization. Although the previous analyses did not show any difference at baseline between the 2 groups, we further controlled for possible effects of the baseline level of performance on the outcomes of the treatment, by running repeated-measures analyses of covariance (ANCOVA), with Group as a between-subject factor, Time as a within-subject factor, and with the baseline score as a linear and interactive covariate. 38 Overall, these analyses confirmed the results, as shown in Table 3. For the JHFT, the Time by Group interaction showed a greater improvement in the active group. For the HS, no significant effect was found, whereas for the MAL, the effect of Time showed an improvement in both groups. For the FMM, the effect of Group showed a greater improvement in the active group. Crucially, the absence of significant interactions with the baseline indicates that the observed improvements were unrelated to the baseline score.
Repeated-Measures ANCOVA, With Group and Time Main Factors (see Table 2) and With Baseline as a Linear and Interactive Covariate
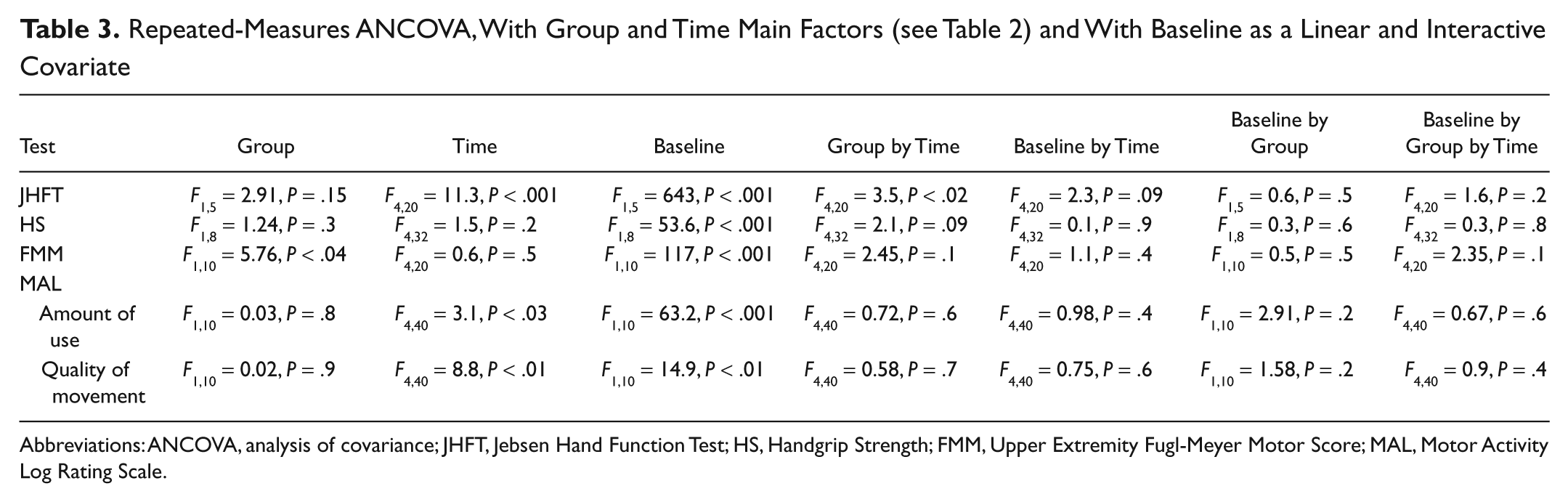
Abbreviations: ANCOVA, analysis of covariance; JHFT, Jebsen Hand Function Test; HS, Handgrip Strength; FMM, Upper Extremity Fugl-Meyer Motor Score; MAL, Motor Activity Log Rating Scale.
Discussion
Bihemispheric tDCS increased the gain of motor function induced by CIMT, 33 as assessed by the JHFT, the HS, and the FFM. This is the main novel finding of this study. Besides being larger, the motor improvement in the active group was mediated by a different mechanism of action, characterized by a reduction of the intact-to-affected TI and an increase in the amplitude of MEPs obtained by stimulating the damaged hemisphere. Accordingly, adding tDCS brings about a more favorable interhemispheric balance, which is associated with a greater motor improvement. Since spontaneous recovery is unlikely in the chronic phase of stroke (>6 months), 39 the observed improvements may be ascribed to the intervention. The tDCS benefit remained stable up to 4 weeks after treatment and was quite consistent across tests (see pη2 in Table 2), notwithstanding the small size of the groups.
Regarding the neurophysiological effects, in the sham tDCS group only a decrease of the cortical excitability in the intact M1 was found after treatment. Instead, despite the CIMT training, cortical excitability in the affected M1 remained largely unchanged, 20 as well as TI recorded from both hemispheres. It is worth noting that the resting splint used here neither prevents any elbow activity nor completely rules out finger movements inside the splint itself. A cast that completely prevents arm movements could have a greater impact on cortical excitability. Conversely, the active tDCS group showed a significant reduction of cortical excitability in the intact hemisphere, along with an increase of excitability in the damaged hemisphere and a reduced TI from the intact to the damaged hemisphere. This latter finding is in line with evidence showing that tDCS influences motor cortical excitability. 40 Moreover, ipsilesional cortical excitability changes (ie, increased MEP amplitude, decreased intact-to-damaged TI) correlate with a decreased time to complete the JHFT with the paretic hand, as well as with the improvement in the FMM score, which is in agreement with previous evidence.41,42 It should be noted that performance in the JHFT is mostly mediated through fast corticospinal projections 43 originating in the M1 area. 44 This is consistent with the upregulation of excitability of the damaged motor cortex found in this study. Because we did not measure cortical excitability in the follow-up evaluations, it was not possible to assess whether the long-lasting behavioral changes were also associated with sustained physiological alterations.
Similar results have been obtained in previous studies in healthy subjects.9,13,45 In patients with stroke, the evidence is controversial. For instance, in patients with subacute stroke, 30 sessions of 7 minutes of anodal tDCS (1.5 mA) integrated into 20 minutes of robot-assisted arm training did not bring about additional benefits from tDCS. 46 On the other hand, in patients with chronic stroke, anodal tDCS (1 mA for 20 minutes) given before a robotic wrist training (1 hour) raised corticospinal excitability and reduced cortical inhibition. 47 Finally, 5 sessions of bihemispheric tDCS and simultaneous physical/occupational therapy improved motor function and increased the activation of ipsilesional motor regions during paced movements of the affected limb, as assessed by functional magnetic resonance imaging. 8 Several factors may influence the outcome of the combined treatment approach, such as the temporal window of tDCS delivery and the type of training to be combined. In this context, an important issue that needs to be taken into account for the successful implementation of tDCS as an adjuvant strategy to physical therapy is related to the concept of homeostatic plasticity,10,48 namely, the human brain’s ability to regulate changes in synaptic plasticity in response to functionally nonspecific changes in network activity, such as those probably induced by brain stimulation.
In conclusion, in patients with chronic stroke and moderate to severe hemiparesis, CIMT-dependent motor recovery might be limited by an imbalanced TI, hindering the motor cortex of the affected hemisphere. The proper polarization of the motor cortices through bihemispheric tDCS can remove the abnormal interhemispheric competition, thus increasing ipsilesional cortical excitability. The possibility of enhancing plasticity by synchronous bihemispheric tDCS to the motor areas engaged in CIMT is consistent with the Hebbian principle that potentiation of synaptic efficacy occurs when its presynaptic and postsynaptic elements are simultaneously active. 49 Our approach is also consistent with more general rehabilitation principles. The “plastic” response to brain damage may also include maladaptive changes that contribute to functional disability. 3 With this in mind, an optimal therapy should include the inhibition of mechanisms leading to maladaptations.
On a broader perspective, our study provides novel evidence supporting the efficacy of the combined treatment approach, ie, modulating brain activity in ipsilesional and contralesional motor cortices with a simultaneous behavioral therapy, which is based broadly on the same mechanisms of action as bihemispheric tDCS. This leads to functional reorganization of interhemispheric interactions and patterns of cortical excitability, and, in turn, enhances clinical gains.
Our research was designed as an exploratory study, including a small number of patients and uncontrolled differences between groups with respect to the localization and size of the hemispheric lesions. This may be a critical issue, as shown by a previous study using a different technique of brain stimulation, rTMS. 50 Future studies including a larger number of patients, matched for site and size of the neurological lesion, may further elucidate the present results.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Heart Association (0735535T) (FF), University of Milano-Bicocca (NB, GV), IRCCS Istituto Auxologico Italiano (NB, GV, LT), Regione Lombardia-Ricerca Finalizzata 2009 (LT, CC).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
