Abstract
Background. Transcranial direct current stimulation (tDCS) of the motor cortex can enhance the performance of a paretic upper extremity after stroke. Reported effects on lower limb (LL) function are sparse. Objective. The authors examined whether tDCS can increase the force production of the paretic quadriceps. Methods. In this double-blind, crossover, sham-controlled experimental design, 8 participants with chronic subcortical stroke performed knee extension using their hemiparetic leg before, during, and after anodal or sham tDCS of the LL motor cortex representation in the affected hemisphere. Affected hand-grip force was also recorded. Results. The maximal knee-extension force increased by 21 N (13.2%, P < .01) during anodal tDCS compared with baseline and sham stimulation. The increase persisted less than 30 minutes. Maximal hand-grip force did not change. Conclusions. Anodal tDCS transiently enhanced knee extensor strength. The modest increase was specific to the LL. Thus, tDCS might augment the rehabilitation of stroke patients when combined with lower extremity strengthening or functional training.
Keywords
Introduction
Transcranial direct current stimulation (tDCS) of the brain is a noninvasive technique that modulates cortical excitability in humans. 1 Recent studies have indicated that tDCS can facilitate upper limb (UL) motor performance in stroke patients. 2-4 Although the effects of tDCS on lower limb (LL) function in stroke patients have been rarely tested, previous studies have demonstrated that anodal tDCS can transiently enhance the maximum force (MF) of the lower extremities and the cortical excitability of LL representation in healthy subjects. 5-7 We used the MF for knee extension as the outcome measure, because previous studies showed that the strength of knee extension correlated with performance of LL functional activity, including sit-to-stand, gait, and stair ascent in stroke patients. 8 We tested the hypothesis that anodal tDCS facilitates the MF of the paretic quadriceps in stroke patients.
Methods
Patients
Eight patients with chronic subcortical stroke (4 female; mean age = 59.6 years) participated in the study (Table 1). The sample size was based on a power analysis, 9 where the effect size (Cohen d), α, and β error probability were set to 1.02, .05, and .20, respectively. The effect size was determined based on a previous study that examined the effect of tDCS on the muscle strength of the LL in healthy subjects. 6 All participants gave written, informed consent before the experiments, which were approved by the local ethics committee of Tokyo Bay Rehabilitation Hospital.
Patient Information
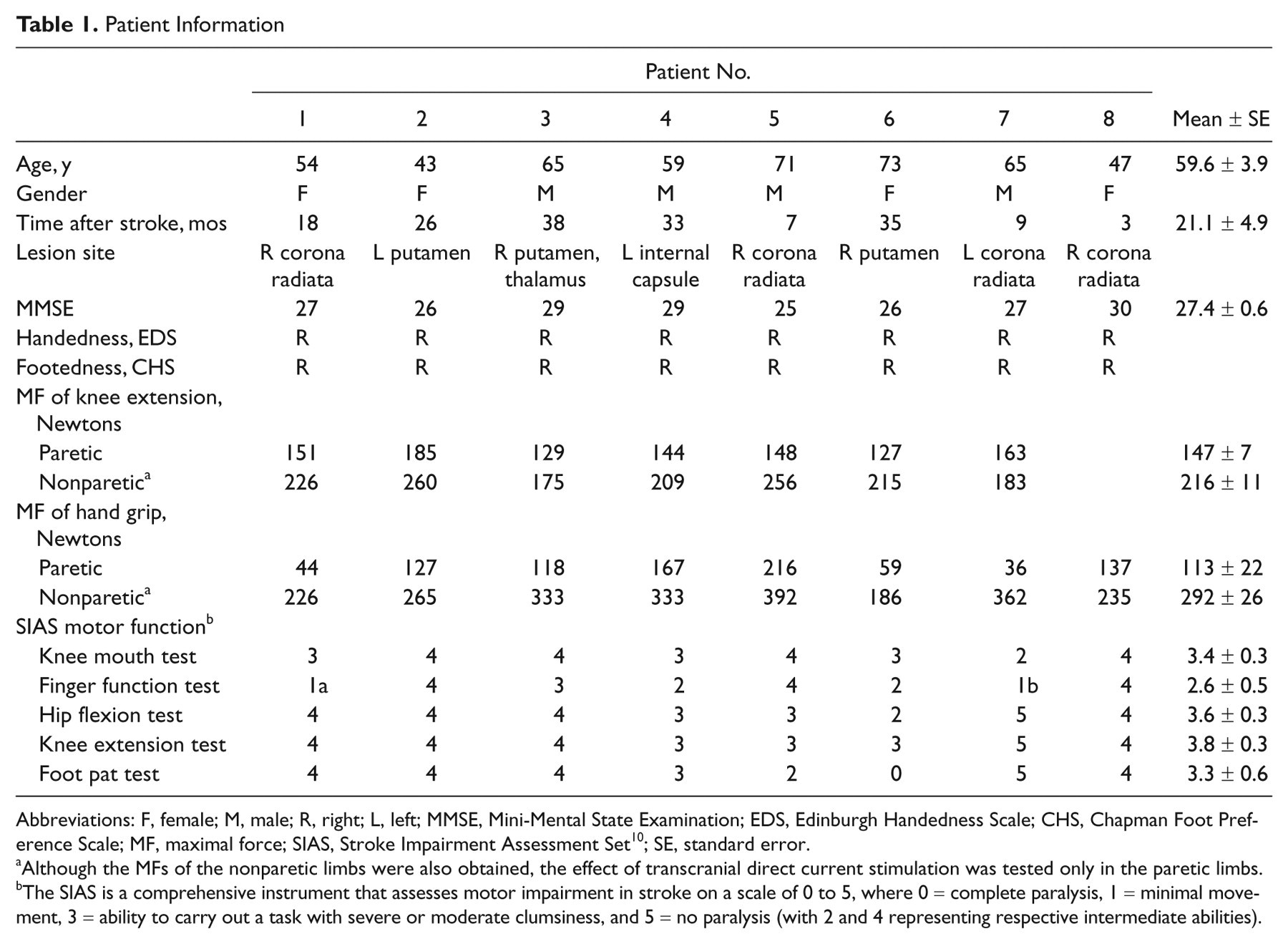
Abbreviations: F, female; M, male; R, right; L, left; MMSE, Mini-Mental State Examination; EDS, Edinburgh Handedness Scale; CHS, Chapman Foot Preference Scale; MF, maximal force; SIAS, Stroke Impairment Assessment Set 10 ; SE, standard error.
Although the MFs of the nonparetic limbs were also obtained, the effect of transcranial direct current stimulation was tested only in the paretic limbs.
The SIAS is a comprehensive instrument that assesses motor impairment in stroke on a scale of 0 to 5, where 0 = complete paralysis, 1 = minimal movement, 3 = ability to carry out a task with severe or moderate clumsiness, and 5 = no paralysis (with 2 and 4 representing respective intermediate abilities).
Experimental Procedure
The study employed a double-blind, crossover, sham-controlled experimental design. 3,4 We examined the effect of tDCS over the LL motor cortex (M1) in the affected hemisphere on the MF of knee extension and hand grip of the paretic limbs. All participants underwent 2 sessions (anodal tDCS and sham stimulation) separated by at least 1 week. The session order was counterbalanced among the participants. The experimenter who measured the MF of knee extension and the patients did not know which session was real and which a sham stimulation. Before starting each session, the subjects were familiarized with the tasks. Each session consisted of 3 task blocks (before, during, and 30 minutes after each intervention). The order of the 2 tasks was counterbalanced across the subjects. Visual analogue scale (VAS) scores of patients’ attention, fatigue, pain, and discomfort levels were obtained immediately after each intervention (Table 2).
Visual Analogue Scale Scores After Each Intervention a
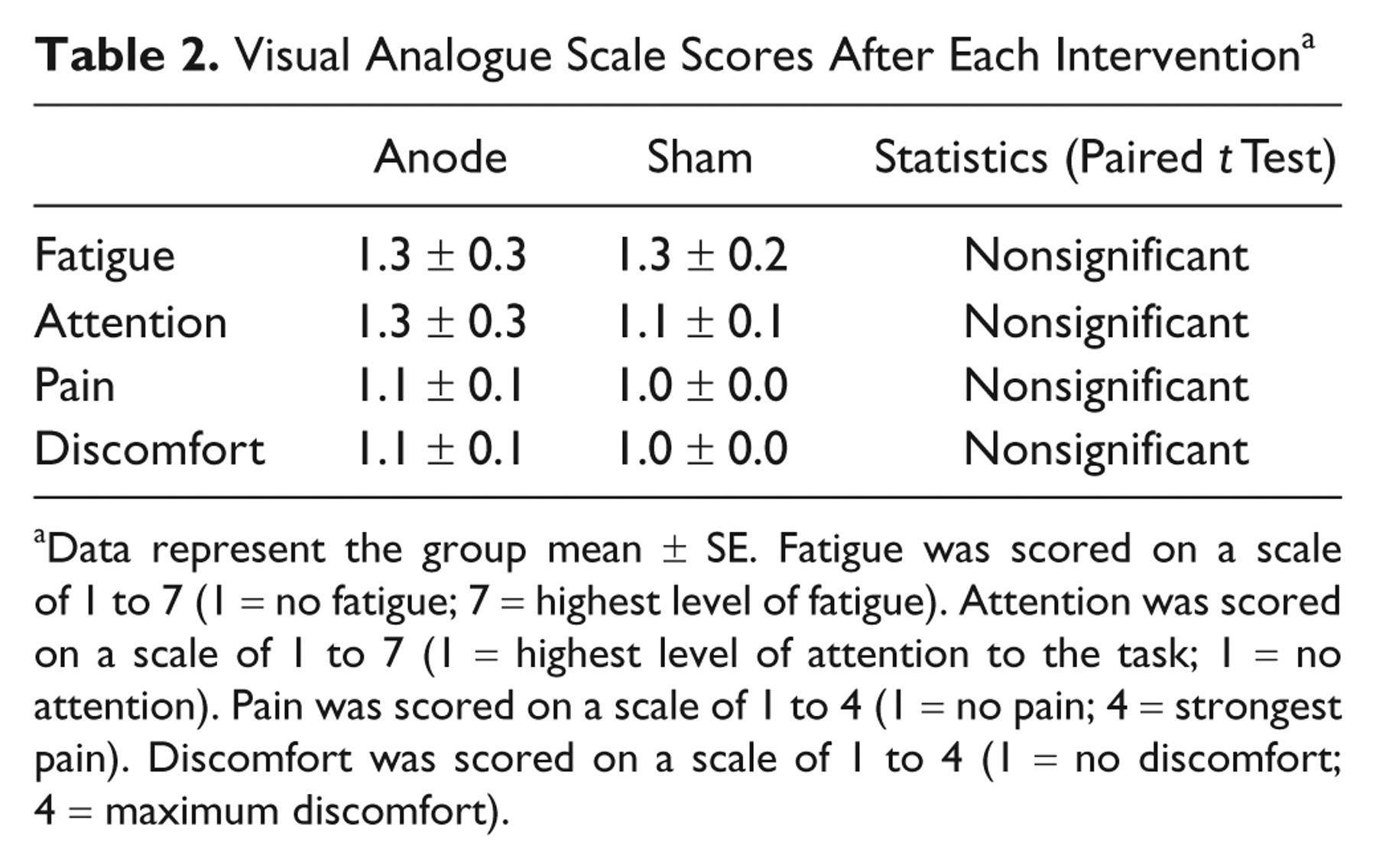
Data represent the group mean ± SE. Fatigue was scored on a scale of 1 to 7 (1 = no fatigue; 7 = highest level of fatigue). Attention was scored on a scale of 1 to 7 (1 = highest level of attention to the task; 1 = no attention). Pain was scored on a scale of 1 to 4 (1 = no pain; 4 = strongest pain). Discomfort was scored on a scale of 1 to 4 (1 = no discomfort; 4 = maximum discomfort).
Tasks
The MF of knee extension was assessed using a handheld dynamometer (Power Track; JTECH Medical Industry, Salt Lake City, Utah). During MF measurement, each subject was seated on an armchair with both the hips and the knees flexed (90°). The dynamometer was placed over the tibia just proximal to the ankle. Each subject was asked to extend the knee against the dynamometer as hard as possible for 3 seconds. For each measurement, the MF was defined as the maximum power during the 3-second task period. This method has high test–retest reliability. 11 The measurement was repeated 4 times, and the data were averaged to yield the mean MF per block.
The MF of the hand grip was measured using a standardized method. 12 Each subject gripped the arm of a dynamometer (GRIP-A; Takei Scientific Instruments, Niigata, Japan) with his or her hand and squeezed it as hard as possible for 3 seconds. The measurement was repeated 3 times, and the mean MF was calculated for each block.
Transcranial Direct Current Stimulation
The DC Stimulator Plus (NeuroConn, Ilmenau, Germany) was used to deliver direct current through 2 sponge surface electrodes soaked with sodium chloride. One electrode (surface area 35 cm2) was positioned over the LL M1, where transcranial magnetic stimulation elicited twitches in the tibialis anterior (TA) of the limb. The other, cathode electrode (surface area 50 cm2) was placed on the forehead above the contralateral orbit. tDCS at an intensity of 2 mA was applied for 10 minutes. These parameters were used in previous LL tDCS studies with no reported side effects. 5-7 For the sham stimulation, the same procedure was used, but the current was delivered only for the initial 15 seconds.
Results
Transcranial Direct Current Stimulation
There was significant correlation of the individual MF between the anodal and sham prestimulation blocks in either the LL (r = .91, P < .01) or the UL (r = .96, P < .001) task. Therefore, the reproducibility of the force measurement was high in this study.
In the prestimulation blocks, there were no significant differences in the mean MF between the anodal and sham tDCS conditions in either the LL (paired t test, t (7) = 1.19, not significant (n.s.); mean MF, anode = 149.4 N, sham = 157.4 N) or UL (t (7) = 0.95, n.s.; mean MF, anode = 111.6 N, sham = 105.0 N) task. The data were normalized with respect to the preintervention performance for each subject (Figure 1A).
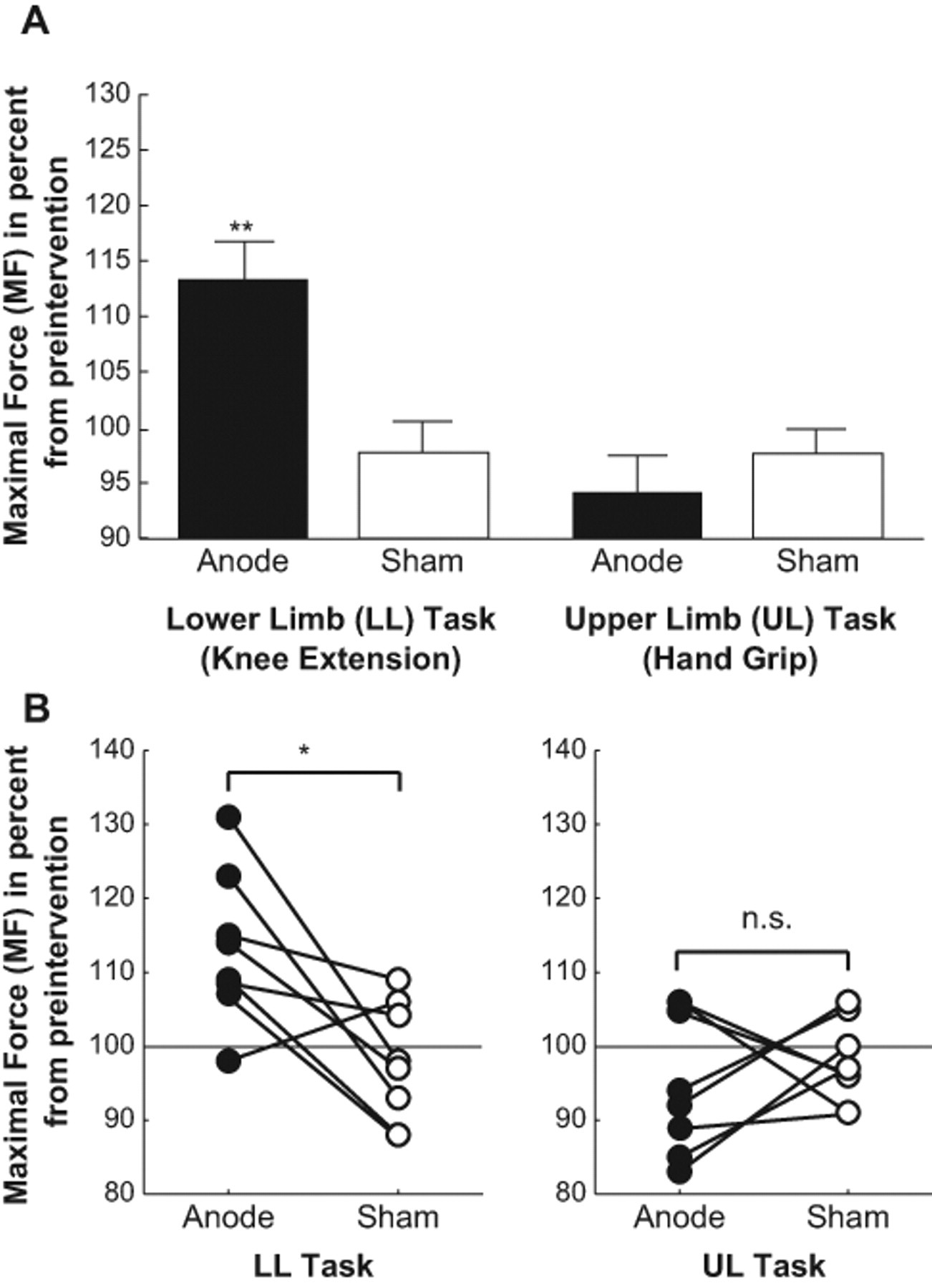
Results of online effect. A, Group results showing the effects of transcranial direct current stimulation (tDCS) on the lower limb/upper limb (LL/UL) tasks. Data are presented as the mean maximal force (MF; n = 8) with bars indicating the standard error (SE). The black and white boxes indicate the data from the anodal and sham conditions, respectively. Data were normalized with respect to the baseline values prior to intervention. Anodal tDCS significantly affected the MF only in the LL task (post hoc one-sample t test, t (7) = 3.67, P < .01), and there were no significant changes in the other 3 conditions. For details of the other statistical analyses, see the main text. **P < .01. B, Individual results for the MF in the LL/UL tasks. Each plotted value is an individual MF value during the anodal or sham stimulation conditions. MF scores were normalized to the preintervention conditions. The normalized MF in the LL task was larger during anodal tDCS (black circles) than during sham stimulation (white circles) in all except patient 6, whereas there was no consistent change in the MF of the UL task across subjects. *P < .05.
The effects of the online tDCS on the MF of the LL/UL tasks were evaluated by performing a 2-way repeated-measures analysis of variance (ANOVA) with INTERVENTION (anodal or sham) and TASK (LL or UL) as factors. The main effects of TASK (F (1,7) = 13.79, P < .01) and the INTERVENTION × TASK interaction (F (1,7) = 15.81, P < .01) were both significant, whereas the main effect of INTERVENTION (F (1,7) = 2.37, n.s.) was not. Consistent with our hypothesis, planned comparison revealed that the MF in the LL task was significantly higher during the anodal tDCS than in the sham condition (one-tailed paired t test with Bonferroni correction, t (7) = 3.16, P < .05). The effect size (Cohen d) was 1.11. However, the MF in the UL task did not significantly differ between the 2 conditions (t (7) = 0.82, n.s.). This finding indicated that the beneficial effect of the anodal tDCS was effector specific. The data for individual patients (Figure 1B) revealed that the improvement of the MF in the LL task was larger for the anodal tDCS compared with the sham condition in all but one case (patient 6). There was no consistent change in the MF in the UL task across patients.
To examine the after-effects of the tDCS, the MF on the LL and UL tasks was reevaluated 30 minutes after the offset of the current flow. None of the interventions significantly affected the MF in either the LL or the UL task: INTERVENTION (F (1,7) = 0.01, n.s.), TASK (F (1,7) = 1.25, n.s.), interaction (F (1,7) = 0.80, n.s.). These results indicated that the after-effects of anodal tDCS were minimal.
Psychological Data
None of the subjects reported side effects. The VAS scores recorded immediately after each intervention revealed that the INTERVENTION (anodal or sham) did not significantly influence the patients’ attention, fatigue, pain, or discomfort (Table 2). Thus, we expect that the confounding effects of these factors are minimal in this study.
Discussion
We demonstrated that anodal tDCS over the LL M1 in the affected hemisphere temporarily facilitated the MF of knee extension in chronic subcortical stoke patients. This effect was not observed for the MF in the hand-grip task. The effector specificity of the modulation indicated that the results were not caused by general effects, such as changes in attention, fatigue, or pain/discomfort. The VAS scores supported this notion.
Single-cell recordings from the primate M1 have shown a direct relationship between the discharge rate of cortical neurons and force generation. 13 Thus, one possible mechanism underlying the observed result is an increase of corticospinal excitability by anodal tDCS over the LL M1. 1,5,7
A previous tDCS study reported that MF of the UL was improved by anodal tDCS in patients with stroke. 4 The degree of the MF improvement of UL in their study (15.7%) was comparable with that of LL in our study (13.2%). However, the current was 1 mA in their study whereas it was 2 mA in our study, in which the target LL M1 was located deeper in the inter-hemispheric fissure and less excitable than the UL M1. Therefore, the effects on behavioral performance might be comparable between 1-mA anodal tDCS over the UL M1 and 2-mA anodal tDCS over the LL M1.
Because of its low spatial resolution, it is likely that tDCS could affect not only the representation of the quadriceps but also those of the other LL muscle groups such as the TA and hamstring muscles (HS) in the M1. In fact, a recent study demonstrated that anodal tDCS over the LL M1 enhanced dorsiflexion–plantarflexion movements of the ankle. 14 This finding suggests that tDCS could also influence the cortical excitability of the muscles controlling ankle movements. It will be important to examine whether anodal tDCS could facilitate the strength of other muscle groups such as TA or HS because selective control of these muscles is also a major problem for the recovery of gait function in stroke patients.
We did not measure the effect of tDCS on knee extension force in the nonparetic LL. However, a recent study reported that anodal tDCS over the LL M1 increased the cortical excitability in the target hemisphere but decreased it in the nontarget contralateral hemisphere. 7 This study suggested that tDCS could modulate the excitability of the LL M1 in the target hemisphere with a reasonable spatial resolution, and also raise the possibility that tDCS might modulate motor function on nonparetic LL on the opposite direction via interhemispheric connection.
Previous studies have reported the correlation of the strength of knee extension with performance of LL functional activity in patients with stroke. 8 Therefore, the increase in knee extension force by anodal tDCS shown in this study might be relevant to the future application of cortical stimulation for neurorehabilitation of LL dysfunctions in stroke patients. To test this hypothesis, future studies are required to examine whether an improvement of knee extension force by anodal tDCS would facilitate functional recovery of LL functions in stroke patients.
Very recently, Madhavan et al 14 reported that anodal tDCS enhanced LL functions, especially fine control of ankle movement affected after hemiparetic stroke. 14 In contrast, our study demonstrated for the first time that anodal tDCS enhanced force production of knee extension in subcortical stroke patients in a more carefully controlled experiment with a double-blind, crossover, sham-controlled design. Together with the finding by Madhavan et al, 14 the present result provides evidence for improvement of LL functions after chronic subcortical stroke by tDCS, although the small number of subjects may limit the strength of the conclusion. These findings raise the possibility that repeated applications of tDCS combined with rehabilitation training may improve mobility, 15,16 as shown with upper extremity motor training. 17
Footnotes
TH and KW contributed equally to this work.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by grants from the Grants-in-Aid for Scientific Research (KAKENHI) to ST (22700442), TH (20033030), and MH (22240049), CREST to MH and KW, a JSPS Research Fellowship to KK and SRBPS, and MEXT to KT and RO.
