Abstract
Objective. We compared the long-term effect of anodal versus cathodal transcranial direct current stimulation (tDCS) on motor recovery in patients after subacute stroke. Methods. Forty patients with ischemic stroke undergoing rehabilitation were randomly assigned to 1 of 3 groups: Anodal, Cathodal (over-affected and unaffected hemisphere, respectively), and Sham. Each group received tDCS at an intensity of 2 mA for 25 minutes daily for 6 consecutive days over of the motor cortex hand area. Patients were assessed with the National Institutes of Health Stroke Scale (NIHSS), Orgogozo’s MCA scale (OMCASS), the Barthel index (BI), and the Medical Research Council (MRC) muscle strength scale at baseline, after the sixth tDCS session and then 1, 2, and 3 months later. Motor cortical excitability was measured with transcranial magnetic stimulation (TMS) at baseline and after the sixth session. Results. By the 3-month follow-up, all groups had improved on all scales with P values ranging from .01 to .0001. Improvement was equal in the Anodal and Cathodal groups. When these treated groups were combined and compared with Sham, significant interactions were seen for the OMCASS and BI scales of functional ability (P = .002 for each). There was increased cortical excitability of the affected hemisphere in all groups with the changes being greater in the real versus sham groups. There were borderline significant improvements in muscle strength. Conclusion. A brief course of 2 types of tDCS stimulation is superior to sham stimulation in enhancing the effect of rehabilitation training to improve motor recovery after stroke.
Introduction
Recent studies in people with chronic stroke have shown that transcranial direct current stimulation (tDCS) can facilitate upper limb motor performance.1,2 There are fewer descriptions of the effects of tDCS on lower limb function, 3 but published work does suggest that anodal tDCS can transiently enhance the maximum force of the lower extremities and cortical excitability of the lower limb representation in healthy subjects.4,5 This pilot trial evaluates the effect of 6 daily sessions of anodal versus cathodal tDCS on motor recovery during inpatient stroke rehabilitation and compares motor and functional changes to sham intervention. The study also assesses cortical excitability prestimulation and poststimulation aiming to understand the mechanism of action of tDCS.
Methods
All patients admitted to the Stroke Unit, Assiut University Hospital, from January through December 2011 were assessed for eligibility. Inclusion criteria were acute hemiparesis with single thromboembolic nonhemorrhagic infarction documented by magnetic resonance imaging. A lesion was classified as cortical if it involved cortical structures or subcortical if it involved only the corona radiata or internal capsule or basal ganglia. Exclusion criteria were extensive infarction (all territories of middle cerebral artery), severe flaccid hemiplegia, head injury, or neurological disease other than stroke, renal or hepatic impairment, previous administration of tranquilizer, and patients who were unable to give informed consent because of severe aphasia, anosognosia, or cognitive deficit. Patients with no motor-evoked response recorded from first dorsal interosseus (FDI) muscle of the affected hand were also excluded. Patients stayed up to 20 days after initial admission and were assessed for inclusion in the trial as soon as they were medically stable, usually within 10 days. Patients remained in hospital for the 6 days of treatment and were discharged 1 to 2 days later. The protocol was approved by the Assiut Medical School Ethical Review Board, and the participants gave written consent.
Clinical Intervention
All patients received the same primary stroke management as indicated in the hospital protocol (anticoagulant low-molecular-weight heparin plus acetyl salicylic acid and Piracetam 2000-4000 mg/day) in the first week followed by acetyl salicylic acid and Piracetam 800 mg twice daily for all patients.
As can be seen from the range of clinical scores in Table 1, all patients were classified as having a moderate range of movement disability, and all had some volitional control of the affected arm and leg. The rehabilitation program began with passive movement and range of motion exercises and gradually progressed to active resistive exercise as muscle tone and strength returned. Patients with severe leg weakness that prevented standing balance received additional electric faradic stimulation to proximal and distal leg muscles during standing exercise. Therapy sessions began within 1 hour after each tDCS/sham session and lasted 30 minutes. Inpatient therapy was given daily; after discharge, patients returned 3 times per week (on alternate days) for outpatient therapy sessions, which were maintained until the end of the follow-up period. All therapists were blinded to group allocation.
Demographics and Baseline Assessment for the 3 Groups.
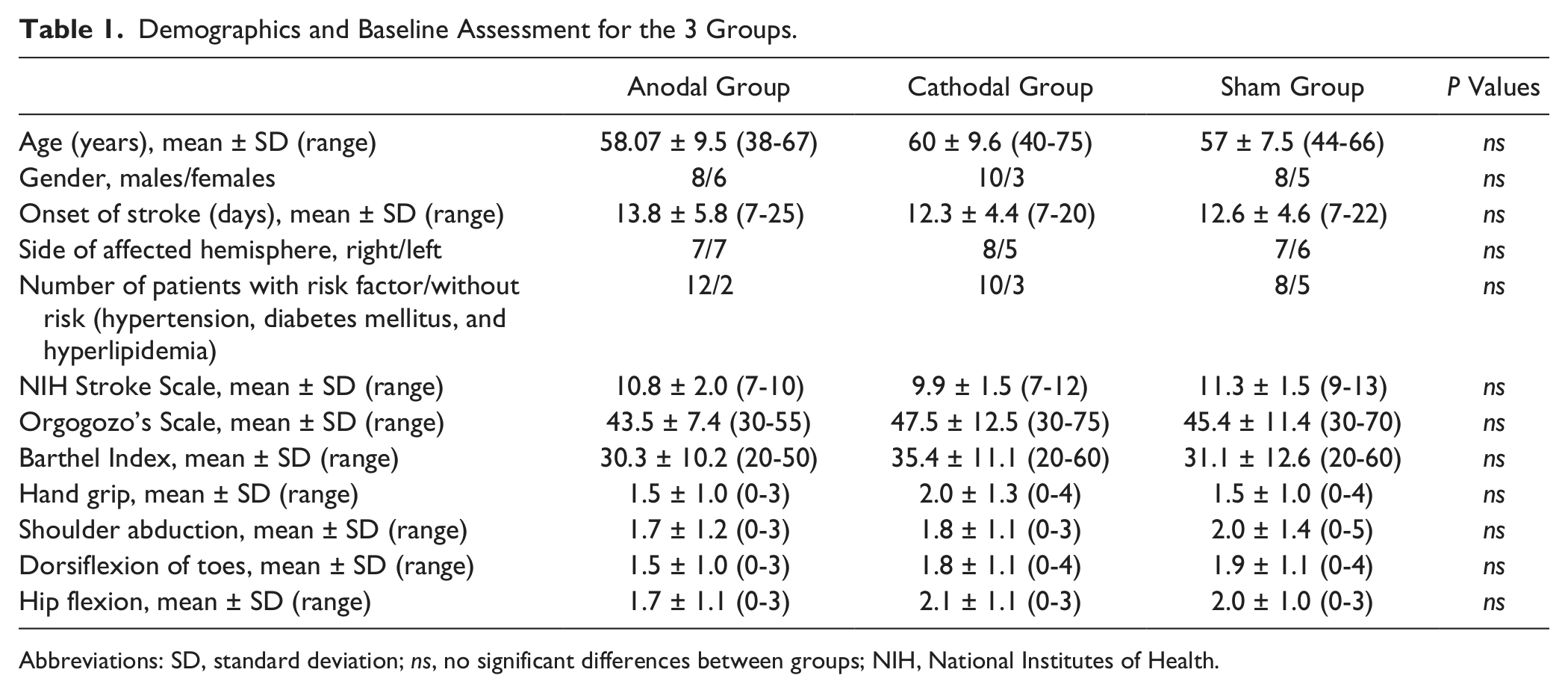
Abbreviations: SD, standard deviation; ns, no significant differences between groups; NIH, National Institutes of Health.
Assessments
Severity of stroke and functional ability were scored using the National Institutes of Health Stroke Scale (NIHSS), 6 Orgogozo MCA scale (OMCASS), 7 and the Barthel index, (BI). 8 Strength of hand grip, shoulder abduction, toes dorsiflexion, and hip flexion were measured on the hemiplegic side according to Medical Research Council (MRC) scale 9 (ranging from 0 to 5; 5 = normal power and 0 = no movement). Clinical assessments were carried out by a neurologist blinded to group allocation.
Cortical excitability was measured in terms of the resting (RMT) and active motor threshold (AMT) using a monophasic magnetic stimulator (Magstim Model 200; Magstim, Whitland, UK) connected to a 90-mm outer diameter figure-of-eight coil, which had a maximal output of 2.2 Tesla. We located the optimal scalp location in each hemisphere from which the largest transcranial magnetic stimulation (TMS) evoked motor-evoked potentials (MEPs) in the FDI muscle could be elicited by moving the figure-of-eight coil systematically in 1-cm steps. The coil was positioned tangentially to the scalp and oriented so that the induced electrical currents would flow approximately perpendicular to the central sulcus, at a 45° angle from the midsagittal line. Single-pulse TMS was then delivered at the optimal location starting at suprathreshold intensity and decreasing in steps of 1% of the stimulator output to determine motor thresholds as defined by Rothwell et al. 10 We used silver–silver chloride surface electrodes, using a muscle belly-tendon montage, with a 3 cm diameter circular ground electrode placed on the wrist. A Nihon Kohden Machine model 9400 (Tokyo, Japan) was used to amplify and record the signal. Electromyography (EMG) parameters included a bandpass of 20 to 1000 Hz and a recording time window of 200 ms. EMG was monitored on line for 20 seconds prior to stimulation to confirm muscle relaxation.
For the ipsilateral silent period (transcallosal inhibition) measurement, approximately 5 seconds prior to each stimulus, subjects were instructed to make an isometric (approximately 50% of maximum) contraction of the ipsilateral FDI and to maintain it for a similar period after the stimulus. Stimulation intensity was 150% RMT of the corresponding hemisphere. The onset and offset of the silent period were defined as the points where the EMG trace fell persistently below and where it returned persistently above the baseline. The duration is the difference between the offset and onset times. 11
Randomization
Group allocations (Anodal, Cathodal, or Sham) were placed in serially numbered, opaque closed envelopes. Each patient was given a serial number from a computer-generated randomization table and was placed in the appropriate group after opening the corresponding sealed envelope. The Anodal and Cathodal groups received real tDCS for 25 minutes at 2 mA daily for 6 consecutive days. Electrodes were saline-soaked pads (5 × 7 cm; CX-6650 Model TRCU-04A, Rolf Schneider Electronics, Gleichen, Germany) secured by headbands. The anode of the tDCS was placed over the M1 hand area of the affected hemisphere (Anodal group), or the cathode was placed over the same area of unaffected hemisphere (Cathodal group); the remaining electrode was over the contralateral supraorbital region. For Sham stimulation, the current was applied for 2 minutes over the affected M1 with a ramp time of 5 seconds and then turned off gradually at the start of the 25-minute period; at the end of the period, the current was ramped up slowly and then turned off more quickly. This procedure mimics the initial tingling sensation under the electrodes when a constant current is switched on or off; during real stimulation, the sensation gradually disappears after these initial transients. The experimenter responsible for delivering tDCS had no contact with the patients. Patients sat quietly during the stimulation and were given normal therapy within 1 hour of the tDCS session. Measurements of cortical excitability and transcallosal inhibition were performed by a neurologist blind to the treatment group before the first tDCS session and repeated after the sixth session. The patients also were blind to the type of stimulation they had been receiving.
Follow-Up
Patients were assessed at the end of the tDCS sessions and then monthly for 3 months. At each visit, patients were scored using the NIHSS, OMCASS, BI, and MRC clinical rating scales, and the rehabilitation program was modified according to the patient’s progress.
Statistical Methods
Power calculations to determine patient numbers in each group were assessed relative to the expected change in BI over the course of assessment. In our previous study, which is similar to the sham treated group here, the mean expected improvement is about 10 points, with a standard deviation (SD) of 7 points. A clinically significant difference in Barthel score was estimated to be 1.85 points on the 20-point Barthel scoring system by Hsieh et al. 12 On a 100 point scale, as used in the present work, this is just more than 9 points. A power calculation with P = .025 (because there are 2 comparisons: Cathodal vs Sham and Anodal vs Sham) and 80% power suggests that it would require at least 12 patients per group to detect whether such a difference is produced by real tDCS over sham.
Values are mean ± SD. At baseline, disability scales (OMCASS, NIHSS, and BI) between groups were compared using 1-way ANOVA for independent samples. Statistical analysis of the scores in each test was done with 2-way ANOVA with “time” point (pre- and postsessions, and first, second, and third months after the end of sessions) as within-subject factor, and “patient group” as between-subject measure. Analysis of the physiological data for AMT and RMT employed a 3-way ANOVA with between-subject factor of “group” (Sham, Anodal, Cathodal) and within-subject factors of “hemisphere” (stroke and nonstroke) and “time” (baseline and immediately posttreatment sessions). The Greenhouse–Geisser correction was applied to correct for nonsphericity of data. All distributions were confirmed to be normal using the Kolmogorov–Smirnoff test. Since there are a number of different methods available to correct for multiple comparisons, we have reported uncorrected P values in the text. The effect of correcting these is discussed in the appropriate points in the Results section.
Changes in rating scales for muscle strength were analyzed with nonparametric methods. The Friedman test was used to detect improvement in each group over time during follow-up. A Kruskal–Wallis test was performed to evaluate differences in improvement (pre to 3 month after sessions) between the 3 groups. Combined data from Anodal and Cathodal groups were then compared with sham using a Mann–Whitney test. The level of significance was set at P < .05.
Correlations between changes in clinical scores and electrophysiological measures were assessed as follows. Since the scores on clinical rating scales were highly correlated, we created a combined clinical score by converting each rating into a z-score. In each patient, we then averaged the z-score across all 3 rating scales to give a single overall score. Similarly, the AMT and RMT measures in each patient were also highly correlated so that we created a single threshold measure by taking the average of AMT and RMT. This threshold was then correlated with the combined clinical score.
Results
Seventy subacute ischemic stroke patients were assessed for participation (Figure 1). Thirty patients were excluded, leaving 26 men and 14 women. The mean interval from onset of stroke to the start of the intervention was 17.1 ± 3.6 days. There were no significant differences between the groups in any of the baseline NIHSS, OMCASS, BI scales or strength measures (hand grip, shoulder abduction, toes dorsiflexion, hip flexion; see Table 1). There was no significant difference between groups with regard to the site of infarction and the presence or absence of 1 or more risk factor for stroke (hypertension, diabetes mellitus, and hyperlipidemia; Table 2). Baseline ratings in the NIHSS indicated that all patients could be classified clinically as having moderate dysfunction (range = 7-13 points).
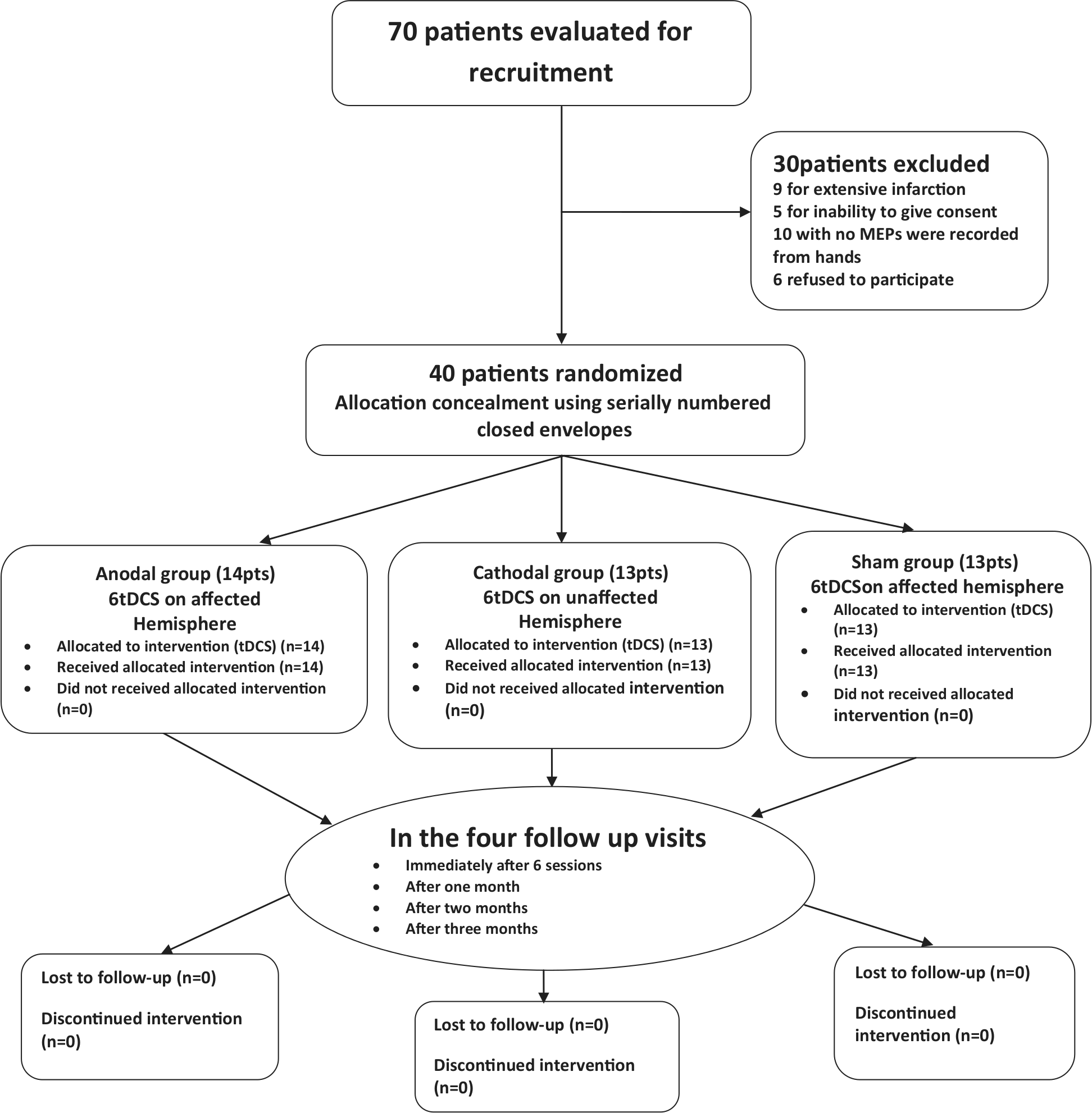
Study flow chart.
Site of Infarction in the Studied Groups.

All patients tolerated tDCS well without any adverse effects. Two-factor ANOVAs on the clinical outcome measures revealed significant “time” × “group” (real vs sham) interactions in the OMCASS (F = 4.58, df = 3.2[60], P = .005; partial η2 = .88) and BI (F = 4.2, df = 3.5[64], P = .006; partial η2 = .87) and a marginal interaction for the NIHSS (F = 2.2, df = 3.1[58], P = .09), which does not survive correction for multiple comparison (see Figure 2). Given the superiority of the OMCASS and BI for functional assessment of disability (as opposed to clinical severity as measured by the NIHSS), we conclude that improvement in function varied between the groups.
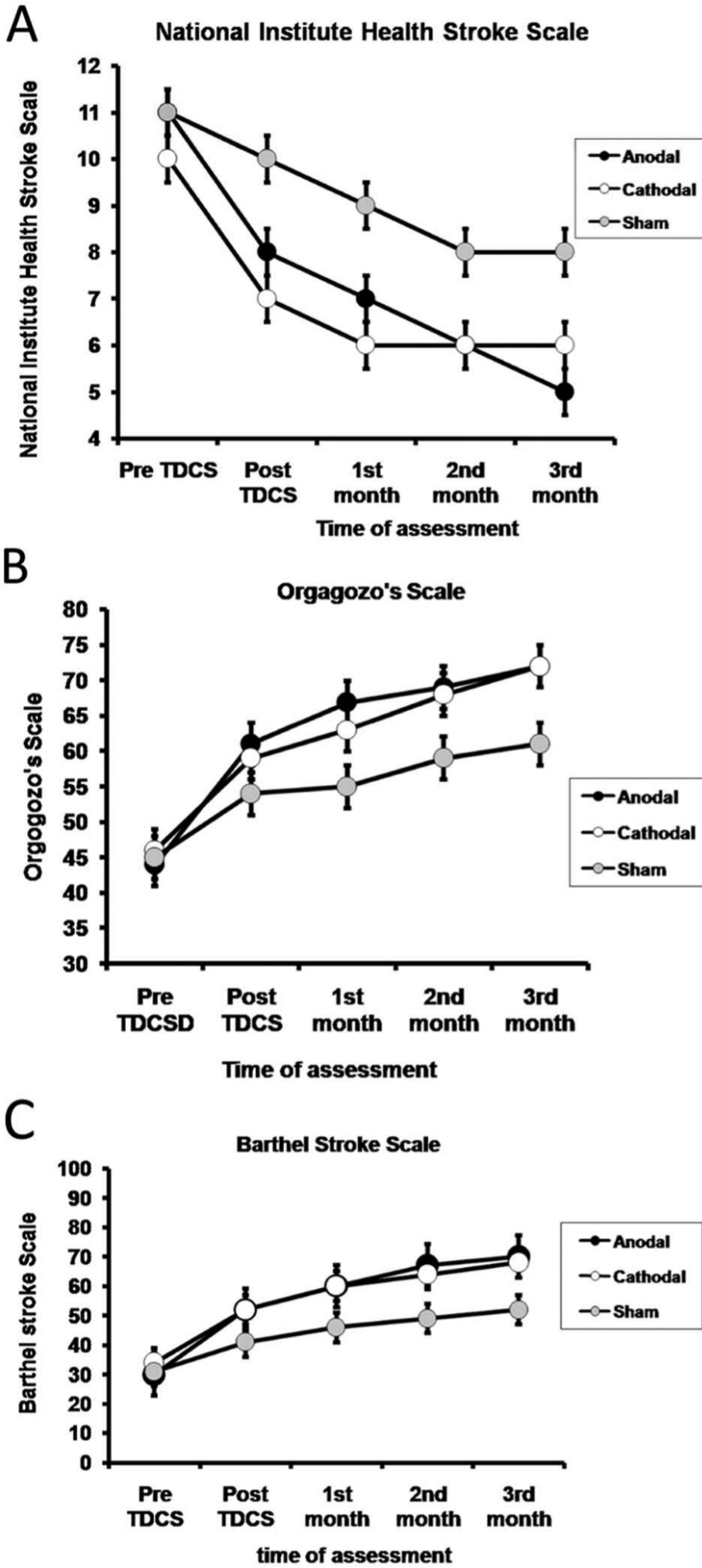
Changes in mean different scores of disability—National Institutes of Health Stroke Scale (A), Orgogozo’s Scale (B), and Barthel Index (C)—at the 6 assessment points for the 3 groups of patients. The first assessment was immediately prior to commencing tDCS treatment (Pre), the second (Post session) was immediately after the end of the sixth session of tDCS, and then monthly for 3 months. Data are expressed as mean ± SE.
In follow-up analyses, we first examined the performance of each group separately over time. Repeated measurement analysis using 1-factor ANOVAs for the NIHSS, OMCASS, and BI scales with “time” as the main factor (pre- and postsessions, and first, second, and third months after stroke) showed significant improvement in all scales for each group with P values ranging from .01 to .0001.
We then performed separate 2-factor ANOVAs to compare the Cathodal and Anodal groups with Sham. There was a significant “time” × “group” interaction for Anodal versus Sham (OMCASS: F = 10.2, df = 1.7[42], P < .001; BI: F = 7.5, df = 1.7[43], P = .002) and a marginally significant effect for Cathodal versus Sham (OMCASS: F = 4.2, df = 1.4[35], P = .033; BI: F = 4.8, df = 1.7[41], P = .017). This suggests that both forms of active tDCS have a different effect on the time course of functional recovery. To improve the statistical power of the analysis, we then compared directly the Anodal and Cathodal groups. There were no significant effects of “group” or “time” × “group” interactions, indicating that the results were similar for both types of real stimulation. In view of this we combined the data and made a final comparison between real (Anodal/Cathodal) and Sham stimulation. Two-factor ANOVAs revealed significant “time” × “group” (real vs sham) interactions in the OMCASS (F = 7.8, df = 1.7[63], P = .002) and BI (F = 7.5, df = 1.7[66], P = .002) scales, but not, if corrected for multiple comparisons, for the NIHSS (F = 3.5, df = 1.6[60], P = .046) scale. We conclude that functional ability as assessed by the OMCASS and BI improved more in the real stimulation groups than in the sham group.
Muscle Strength
Data for muscle strength were initially analyzed for each group and each muscle separately. There was a significant effect of time from before to after treatment in all groups (Friedman test: df = 4, P < .0001; Figure 3). The groups were then compared by taking the difference in muscle strength scores between baseline and 3 months in each patient. There were marginally significant differences between the groups that do not survive correction for multiple comparison in shoulder abduction, foot dorsiflexion, and hip flexion (Kruskal–Wallis: P = .017, .04, and .050, respectively) and no difference between groups for hand grip strength (P = .175). Finally, following the procedure used in the clinical scores, we combined data from Cathodal and Anodal groups and compared with the Sham group. There was slightly more improvement in shoulder abduction, foot dorsiflexion, and hip flexion in the combined group than in the Sham group (Mann–Whitney: P = .005, .014, and .016, respectively, uncorrected for multiple comparisons). There was no difference between groups in hand grip strength (P = .346).
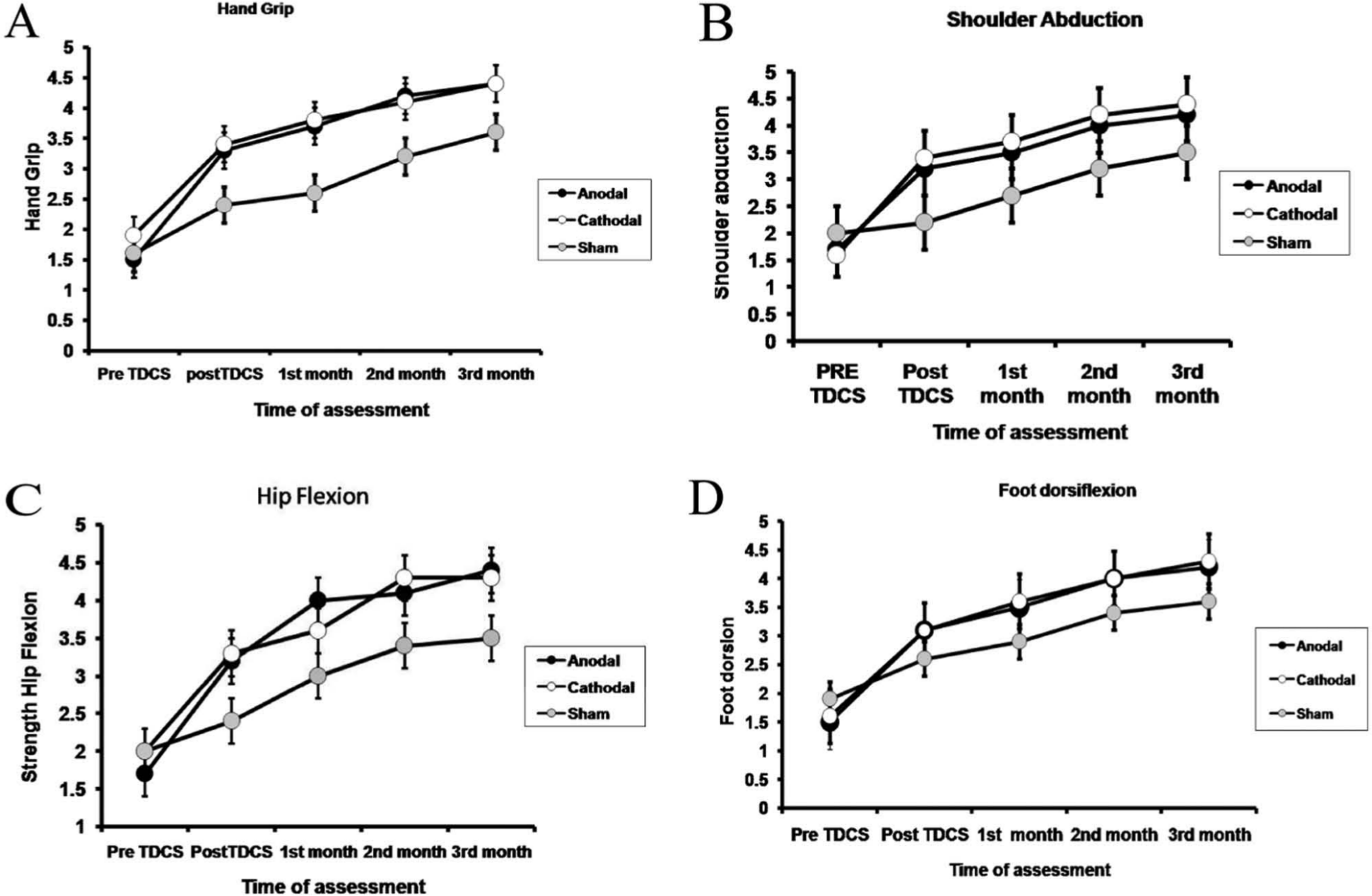
Changes in mean power rating scores of hand grip, shoulder abduction, hip flexion, and toes dorsiflexion at the 5 assessment points for the 3 groups of patients. The first assessment was immediately prior to commencing tDCS treatment (pre-tDCS), the second (post-tDCS) was immediately after the end of the 6 sessions of tDCS, and then monthly for 3 months.
Neurophysiological measures
Data from AMT and RMT were analyzed separately. A 3-way ANOVA for AMT and RMT revealed a significant “group” × “hemisphere” × “time” interaction in each case (RMT: F = 7.1, df = 2[37], P = .003; AMT: F = 5.0, df = 2[37], P = .012). This indicates that the effect of treatment was different in the 3 groups and between the 2 hemispheres. We therefore analyzed data from each hemisphere separately with 2-way ANOVAs employing the between-subject factor of “group” and between-subject factor of “time.” For the unaffected hemisphere, there were was no significant “group” × “time” interaction for either RMT or AMT (P > .8 in each case) showing that both sham and real treatments had the same effect. There was a marginal effect of “time” for RMT (F = 4.9, df = 1[37], P = .033), due to a slight decrease in thresholds over time, but no main effect for AMT. None of the post hoc paired t-tests comparing pre- and posttreatment in each group separately was significant (see Table 3).
Resting (RMT) and Active Motor Threshold (AMT) of the Unaffected and Affected Hemispheres (Pre–Post Sessions).
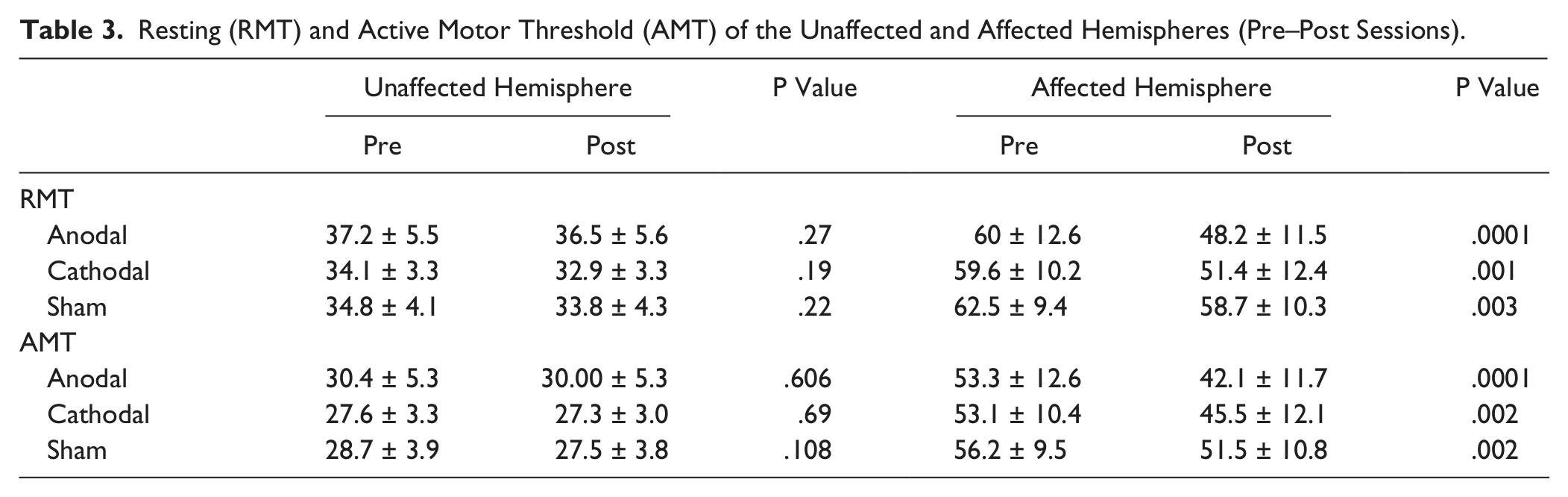
The situation was different for data from the stroke hemisphere. In this case, there were significant “group” × “time” interactions for both RMT and AMT (RMT: F = 8.6, df = 2[37], P = .001; AMT: F = 4.7, df = 2[37], P = .015), indicating that the thresholds were affected differently by treatment in each group. Post hoc pairwise comparisons in Table 3 show that there was a significant decline after treatment in all 3 groups. Finally, we combined the data from the Cathodal and Anodal stimulation groups and compared directly with the Sham group. A 2-way ANOVA with time as the main factor (pre–post) showed that there was a significant “group” × “time” interaction for both RMT and AMT (RMT: F = 12.9, df = 1[38], P = 0.001; AMT: F = 6.2, df = 1[38], P = .017 for AMT) indicating that thresholds declined more in the real groups than in the Sham group. We had also measured the durations of the ipsilateral silent periods on both sides. However, interpretation of these effects is difficult (see discussion) because the absolute intensity of the stimulus used to evoke the ISP (150% RMT) was smaller in the real tDCS groups after treatment due to the concurrent decrease in RMT.
Correlations Between TDCS Effects on Electrophysiology and Clinical Scores
Note that these correlations are calculated between clinical scores measured before and immediately after tDCS, corresponding to the time points at which thresholds were measured. Since the scores on the 3 main clinical rating scales (BI, NIHSS,OMCASS) were correlated, we created a single combined score to compare the pre- and post-tDCS treatment sessions (see Methods). Similarly, there was a close correlation between the values of RMT and AMT at baseline (r2 = .97), so that these were averaged to provide a single measure of threshold. There was a significant correlation between the change in TMS thresholds and improvement in clinical scores for all groups combined (r2 = .37, P ≤ .001), which was marginally significant for the Cathodal and Sham groups alone (r2 = .36 and .32; P = .03 and .04, respectively, uncorrected for multiple comparisons) but not for Anodal alone. We also tested for correlations between handgrip strength and clinical scores. There was a relationship between the change in threshold and the increase in grip strength taking all groups combined (Spearman’s ρ = .61, P < .001), but this was not significant for either the Anodal or Cathodal groups separately and only borderline significant for the Sham group (r2 = .55, P = .05; Figure 4).
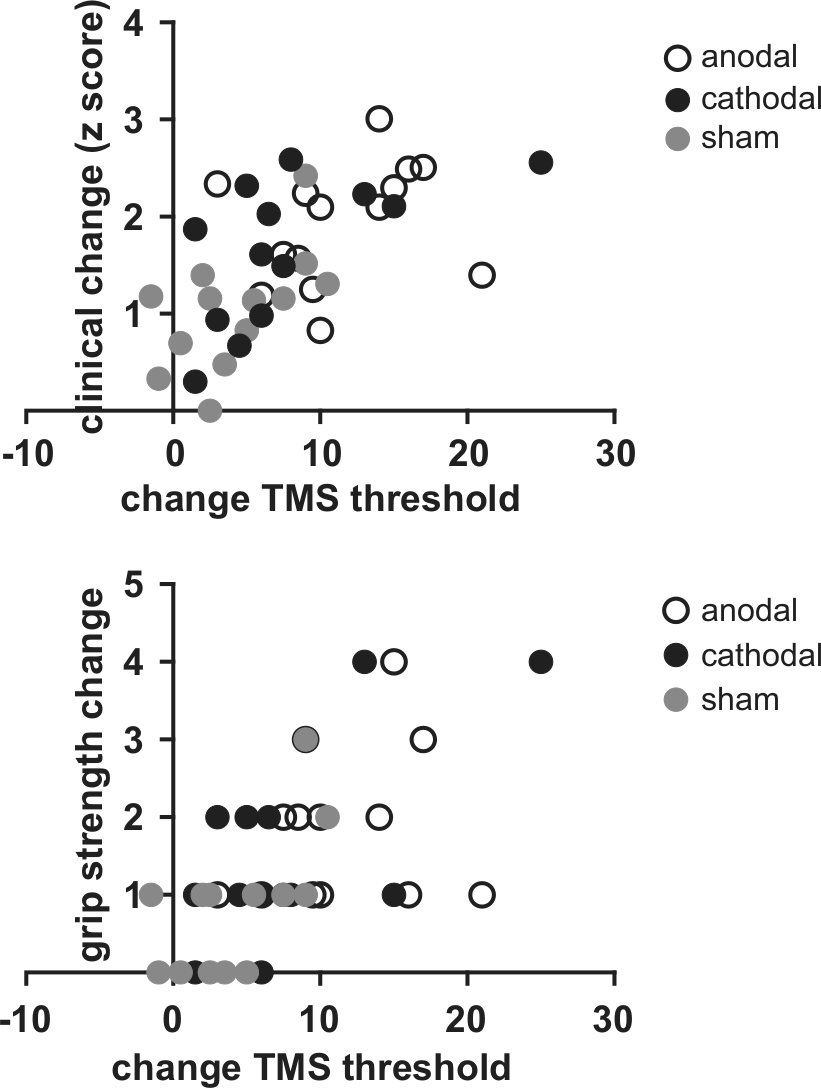
(Top) All clinical scores are combined to be a single score to compare the pre- and post-tDCS treatment sessions with the changes (pre- and post-tDCS) in the threshold (combined RMT and AMT). There was a significant correlation (r2 = .37, P ≤ .001) between the change in TMS thresholds and improvement in clinical scores (all groups combined). (Bottom) There was a relationship between the change in threshold and the increase in grip strength taking all groups combined (Spearman’s ρ = .61, P < .001).
Discussion
The present results showed that as expected in subacute stroke, patients in all 3 groups improved over the time course of treatment. There was no difference between the effects of Anodal and Cathodal stimulation on clinical scores, and both improved more than Sham in clinical measures of functional ability. The mean additional improvement produced by real tDCS over sham was 7.3 points on the OMCASS scale and 11 points on the BI, both of which are of the order of 10% of the total range of each scale and hence likely to be clinically significant. These effects were complemented by increased cortical excitability (ie, reduced stimulation thresholds) and marginally increased muscle strength in all groups, with the effects being larger in the combined Cathodal and Anodal groups than Sham. Importantly, improvement in this study was not only seen in the upper limb but also in performance of the lower limb on the affected side.
The results are more positive than in 4 previous studies that compared the effect of cathodal, anodal, and sham tDCS on motor recovery in patients with subacute subcortical ischemic stroke.12-16 The first small study by Kim and colleagues 13 only found an effect of cathodal tDCS. Five of the 18 patients who were given cathodal stimulation over the nonstroke hemisphere improved significantly more in their Fugl-Meyer scores (but not on the BI) than the 7 patients in the sham group. Surprisingly, the latter did not show any clinical improvement up to 6 months poststroke. The 2 more recent studies by Hesse and colleagues14,15 and by Rossi and colleagues 16 were both negative, in that tDCS had no additional effect over and above sham. Patients in the Hesse study received a combination of robotic arm training with simultaneous tDCS daily for 6 weeks. Although all improved function, there was no difference between the real and sham tDCS groups. The design of the Rossi study was similar to ours, but they examined only anodal tDCS to the affected hemisphere, again with a negative result.
Two main factors could contribute to the difference between previous data and those reported here: severity of stroke and time of recruitment after stroke. The Hesse study was conducted on 96 patients but they were more severely impaired (mean upper extremity Fugl-Meyer score of approximately 8 points [of 66 maximum]) than those in the present study (baseline Barthel index of 32 [of 100]); they were also recruited later than ours after their stroke (approximately 25 days vs 17 here). It is possible that the extent of the damage in severely affected patients reduces their capacity to respond to tDCS. Indeed, in a previous rTMS study in subacute stroke, we found that more severely affected patients did not benefit from additional rTMS. 17 Patients in the Rossi study had similar impairment to those in the present report. However, they were recruited much earlier (2 days). It could be that this is too early to intervene, potentially leading to adverse effects by promoting excess excitatory glutamate release in the early part of treatment. If correct, it will be very important in subsequent studies to define the optimal timing of the tDCS intervention.
Previous studies have demonstrated a beneficial effect of tDCS in chronic stroke patients. The initial trials, like the present one, applied anodal tDCS before motor therapy of the upper limb and showed that there was a significant functional improvement of the paretic hand compared with therapy alone.1,18 A more recent study has combined brain stimulation with simultaneous peripheral stimulation in a single-session design to enhance the effect on a performance of a finger sequence task. 19 Peripheral stimulation alone or tDCS alone both improved performance, but the combination was superior to either. Another recent study employed two 5-day intervention periods of bihemispheric tDCS together with simultaneous occupational/physical therapy in a group of 10 chronic stroke patients. They found that bihemispheric tDCS in combination with peripheral sensorimotor stimulation increased upper-extremity Fugl-Meyer scores. Furthermore, they demonstrate that the effects of multiple treatment sessions in chronic stroke patients may not necessarily have a linear response function. 20
Two features of the present results deserve comment. First, although tDCS was applied over the hand/arm area of cortex, improvement above sham was seen in overall clinical scores that examine whole body function. Indeed, we found that the increase in strength in ankle dorsiflexion as well as hip flexion also exceeded that seen in the sham group. Kim et al 13 previously reported that cathodal tDCS over hand area improved a global clinical rating. There are several possible reasons why tDCS could have effects on parts of the body somatotopically distant from the site of motor cortex stimulation. tDCS could have a primary effect on arm function that might encourage patients to increase use of their lower limb and trunk and effectively increase overall body training to promote recovery. A second possibility relates to the lack of focality of the tDCS stimulus itself. The stimulus is applied over a large surface area, and both modeling 21 and positron emission tomography studies 22 have shown that there is likely to be widespread activation of cortex between the 2 stimulating electrodes. Madhavan et al 5 have shown improvements in leg function in chronic stroke after tDCS over the motor leg area. Thus, it is possible that the stimulus activated large areas of motor cortex in addition to the arm/hand area and that this was responsible for the widespread effects on function that were achieved.
A second feature of the results was the demonstration that cathodal tDCS over the nonstroke hemisphere had the same beneficial effect as anodal tDCS over the stroke hemisphere. Bradnam et al 23 had previously reported that in patients with greater corticospinal damage in the stroke hemisphere, cathodal tDCS of the nonstroke M1 worsened proximal paretic arm control. They commented that suppressing M1 in the nonstroke hemisphere may be deleterious in patients with more damage to the motor pathways in the stroke hemisphere. The present study may have avoided this by excluding patients in whom it was not possible to recruit MEPs in the paretic FDI.
The electrophysiological results confirmed that real tDCS had additional effects over and above those seen as part of the natural process of recovery: RMT and AMT declined more over the week of treatment than was observed in the sham group. Others have reported that thresholds decline in the subacute phase of recovery, 24 and we speculate that tDCS enhanced this natural process. However, the electrophysiological measures were only weakly related to changes in clinical scores: changes in threshold correlated with clinical changes when data from all groups were combined but not within each group separately. A similar result was seen for the correlation between threshold change and increase in hand grip. Functional recovery presumably reflects a combination of many of such basic physiological effects.
A final point concerns the measures of ipsilateral silent period, which are known to be reduced after stroke. We had wanted to test whether these would lengthen during recovery, but we found that the duration of the ipsilateral silent period on the paretic side was correlated with the motor threshold on that side making it impossible to comment on any changes that may have occurred. Thresholds change during recovery so that any change in ipsilateral silent period could be secondary to this rather than a change in the excitability of interhemispheric interactions.
Although the results of this study are promising, a number of questions and limitations arise that should be addressed in future studies. As noted above, given the negative results of Rossi et al, who applied tDCS unsuccessfully in patients much earlier after stroke, more work is needed to test the optimal time to incorporate tDCS therapy. In addition, more information is needed to test whether these results in moderately affected individuals will also apply to patients with more severe symptoms and signs. We also limited the dose of tDCS to 25 minutes at 2 mA for 6 days, and applied the tDCS prior to therapy; at the present time we do not know whether better results might be obtained with different dosages perhaps delivered during therapy. Finally, in spite of the regular 3-month follow-up period and the rehabilitation guidelines given to all participants, individual variability in adhering to this exercise program cannot be guaranteed. Nevertheless, the results do serve as further evidence that brain stimulation may be a successful additional therapy to enhance recovery from stroke.
In conclusion, anodal tDCS over the affected hemisphere may be as effective as cathodal tDCS on the unaffected hemisphere to enhance recovery after subacute ischemic stroke. Both types of stimulation are superior to the sham stimulation. Combined rehabilitation training may improve mobility in the upper and lower extremities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
