Abstract
Introduction
Constraint-induced movement therapy (CIMT) is designed to improve upper-extremity motor function after stroke and consists of 3 key components: (1) repetitive, task-oriented training; (2) adherence-enhancing behavioral strategies (transfer package); and (3) constraining the use of the less-affected arm, usually by wearing a mitt.1,2 The original protocol was developed for patients with chronic stroke and included 10 days of therapy for 6 h/d and constraining the less-affected arm during 90% of the time awake. 3
There is a critical window for neuroplasticity and ability to relearn impaired activities within the first weeks after stroke.4,5 Modified forms of CIMT have already been tested in the early stages of stroke rehabilitation (<10 weeks). 5 In 2000, Dromerick et al 6 published the results from a small-scale trial of 23 patients and reported that a 2-hour/10-day CIMT program was associated with less arm impairment at the end of treatment. Another study of 23 patients by Boake et al 7 reported trends favoring a 3-hour/10-day CIMT program over standard therapy of equal duration. However, their only significant finding was improved Fugl-Meyer assessment scores immediately after treatment; no long-term effects were found. In the VECTORS study, 8 3 groups of stroke patients received 10 days of treatment within 4 weeks after stroke. The low-intensity CIMT group (2 h/d, 6-hour constraint) had significantly better improvement in the Action Research Arm test (ARAT), whereas the high-intensity CIMT group (3 h/d, 90% constraint) scored significantly worse than the control group. Yet another modification of CIMT was reported by Page et al, 9 who reported increased use of the affected arm and improved motor impairment after 0.5 hours of training, 3 d/wk for 10 weeks of CIMT in a sample of 10 patients. In summary, the existing evidence on the effect of CIMT in the early stages of rehabilitation is limited to 5 trials that included 64 CIMT patients and 41 controls. 10 These 5 trials included at least 3 different protocols and reported wide confidence intervals and large variations in treatment effects. Of particular concern are the negative results of the VECTORS trial. 8 Because of the limited data and diversity of the results between previous trials, the effect of CIMT in the early stroke rehabilitation is still uncertain.
The aim of the present study was to assess the effect of a modified CIMT protocol in the early phase of rehabilitation after stroke. The primary hypothesis was that patients who completed a modified CIMT protocol in the early phase after stroke would have better arm motor function measured with the Wolf Motor Function test (WMFT) 6 months after the intervention compared with patients who received the usual care. The secondary aims were to evaluate the effect of CIMT on arm motor impairment, dexterity, arm use in daily activities, and overall health status after stroke.
Methods
Design and Participants
The Norwegian CI therapy multisite trial (NORCIMT) is a single-blinded, multicenter, randomized controlled trial that recruited participants from 5 Norwegian hospitals: The University Hospital of North Norway, The Trondheim University Hospital, Oslo University Hospital, Vestfold Hospital, and Telemark Hospital. Patients were recruited from October 2008 until June 2012. The inclusion criteria were (1) first-ever or recurrent stroke at >5 and <26 days prior, (2) modified Rankin Scale between 0 and 2 prior to the stroke, (3) persistent unilateral arm or hand paresis (Scandinavian Stroke Scale items: arm function 2-5 or hand motor function 2-4), (4) able to lift 2 fingers with the forearm pronated on the table or able to extend the wrist at least 10° from a fully flexed position, (5) able to follow a 2-step command, and (6) had Mini Mental State Examination score >20 (or >16 in combination with expressive aphasia). Patients were diagnosed according to the World Health Organization (WHO) definition 11 of stroke. Patients with recurrent stroke were included if they had no detectable arm weakness after the first stroke. Exclusion criteria were (1) Modified Rankin Scale >4, (2) unable to give informed consent, (3) >2 cm deviation on the line bisection test, (4) life expectancy <1 year, (5) injury or condition in the affected upper extremity that limited use prior to the stroke, or (6) other neurological conditions affecting motor function. After providing consent, the patients were allocated to either the CIMT or the control group by a computer-generated block scheme for randomization. The randomization procedure was stratified in 3 groups according to treatment center only. Two treatment centers were collated because of a low planned number of participants. The Regional Committee of Medical Ethics and the Commission of privacy rights at the University Hospital of North Norway reviewed and approved the study (REK NORD 39/2008).
Interventions
CIMT group participants were admitted to the rehabilitation department of 1 of the 4 treatment sites as soon as they were enrolled in the study. They received CIMT for 10 consecutive workdays. The CIMT focused on repetitive task-oriented training (2.0 and 0.5 hours of shaping and task practice, respectively, per treatment day) and adherence-enhancing behavioral strategies (0.5 hours per treatment day). The therapists recorded the treatment time and the time spent on each activity. In addition, the patients were encouraged to wear a constraining mitt on the more affected arm for up to 90% of their waking hours. The behavioral strategies included a treatment contract and daily use of the motor activity log, home diary, and home skill assignments. 2 The home skill assignments were prescribed for 0.5 h/d. The therapists completed a 4-day training program on the study procedures before they could administer the treatment.
The control group was treated according to the Norwegian guidelines for stroke patient treatment. 12 All patients took part in the multidisciplinary treatment approaches in their respective stroke units. Patients suitable for inpatient rehabilitation service were admitted to units. The rest were discharged home or to community-based rehabilitation.
Outcome Measures
All participants were examined by a blinded examiner at their respective treatment site after inclusion but before randomization, after the 2-week treatment period, and after 6 months. The primary outcome measure, the WMFT, is a 17-item test of arm motor function. It consists of 15 movement tasks and 2 strength measures. Each movement task was filmed, and the time to complete a task was determined by analyzing each frame in the video. The maximum time to complete an item was 120 s. If a trial was not completed, the trial result was recorded as 121 s. The median time for all 15 tasks was used for analysis.13,14 In addition, the movement quality was rated from the videos using a 6-point functional ability scale (0 =
Secondary outcome measures were the Fugl-Meyer upper-extremity motor assessment (FMA upper-extremity),
20
the Nine-Hole Peg test (NHPT), the arm use ratio,
21
and the Stroke Impact Scale. The FMA was used to measure motor impairment.
20
The upper-extremity motor part includes 33 items scored on a 3-point ordinal scale (0 =
The NHPT is a dexterity measure for the upper extremities. 23 We used a wooden, 9-hole pegboard (Weston Home Health/Medical Equipment, West Sussex, UK) and previously described procedures.24,25 A stopwatch was used to measure the time taken to place the 9 pegs. The maximum time allowed for each trial was increased from 50 to 120 s to reduce the floor effect. 25 The number of pegs per second for the more affected arm was calculated and used in the analysis. Adequate to excellent reliability of the NHPT has been reported in acute stroke patients. 24
Arm use/arm activity in daily activities was measured using ActiGraph GT1M or GT3X Accelerometers (ActiGraph Inc, Pensacola, FL). The patients were asked to wear 1 accelerometer on each arm for 24 hours. The arm use ratio calculated between the durations of movement in the more-affected and the least-affected arm was used as to measure actual arm use. Several investigations have validated this method in chronic phases and subacute phases (3-9 months) after stroke.21,26,27 The measurement was adapted for acute stroke by using a 1-s epoch time. 28 The arm use ratio correlates well with the FMA and self-care dependence in acute stroke patients. 29
The Stroke Impact Scale is a self-report questionnaire with 64 items divided into 8 domains. In the present study, the domains of hand function (5 items), activities of daily living/instrumental activities of daily living (ADL/IADL, 10 items), participation/role function (8 items), and global perception of recovery (1 item) were reported because they were considered relevant for upper-extremity function. Each item is rated on a 5-point Likert scale. The scores in each domain are generated from an algorithm. Possible scores range from 0 to 100. The global perception of recovery was reported on a continuous scale from 0 to 100. The reliability of the included domains is reported as adequate to excellent, and the measures are valid and sensitive to change in stroke patients. 30
Statistical Analysis
SPSS for Windows 19.0 (SPSS Inc, Chicago, IL) was used for all statistical analyses. Means and standard deviations (SDs) were calculated for pretreatment, posttreatment, and the 6-month follow-up for each group. Changes within each group were evaluated with paired
Power Analysis
Pretrial power calculations were performed to determine the likelihood of detecting a true difference between the treatment and control groups on the primary outcome measures at the 6-month examination. We used the SD (logWMFT = 1.02) treatment effects (logWMFT = 0.55) from the EXCITE study treatment group (P. A. Thompson and S. L. Wolf, Means and standard deviations from the EXCITE trial, personal communication, 2008) 32 for the power calculations. A power of 0.8 would require 53 participants in each group. After adding a fourth treatment center and extending the trial for 2.5 years, the study was halted before the required 106 patients were included. To evaluate the effect of the low inclusion rate, we calculated the observed power of the ANCOVA analyses in the actual sample using the standard SPSS subcommand.
Results
In total, 47 participants (11 women and 36 men) took part in this study. Overall, 96% had experienced an ischemic stroke, and 22% had had a prior stroke. The CIMT group included 24 participants, and 23 were allocated to the control group. Table 1 shows the participants’ characteristics. There were no significant differences between the groups for any of the variables at baseline. Figure 1 shows the CONSORT (Consolidated Standards of Reporting Trials) diagram of participant recruitment. One of the patients in the CIMT group withdrew from the study after treatment day 2. Another patient declined further participation after 8 treatment days. As shown in Figure 1, a total of 7 of the randomized patients—4 from the CIMT group and 3 controls—dropped out of the study before the 6-month follow-up assessment. One patient did not complete 10 of the WMFT items on the 6-month follow-up examination because of shoulder pain, and we were unable to analyze the video from another patient. The drop-out rates were 17% and 13% in the CIMT and control groups, respectively.
Baseline Characteristics of Study Participants.
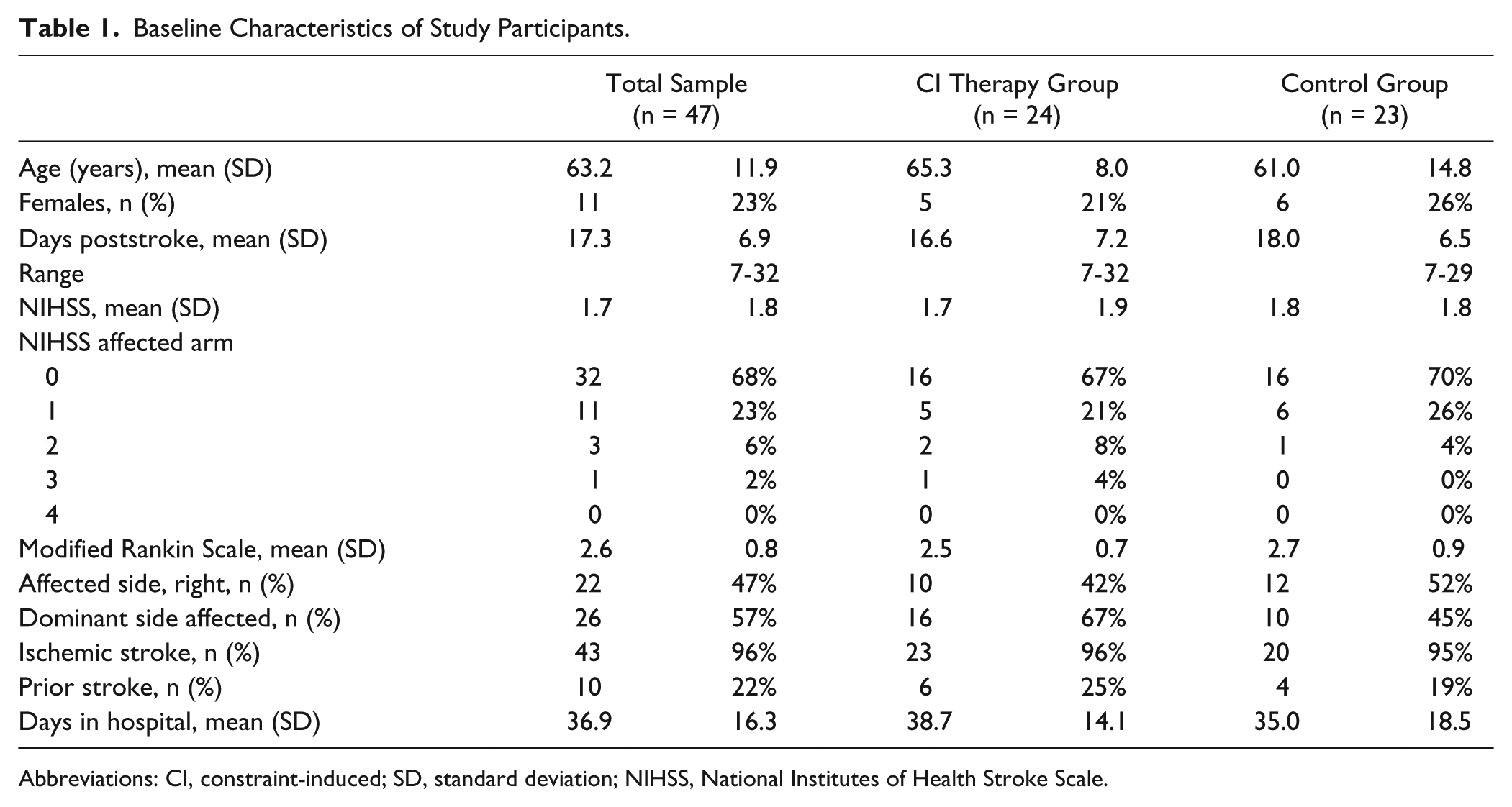
Abbreviations: CI, constraint-induced; SD, standard deviation; NIHSS, National Institutes of Health Stroke Scale.
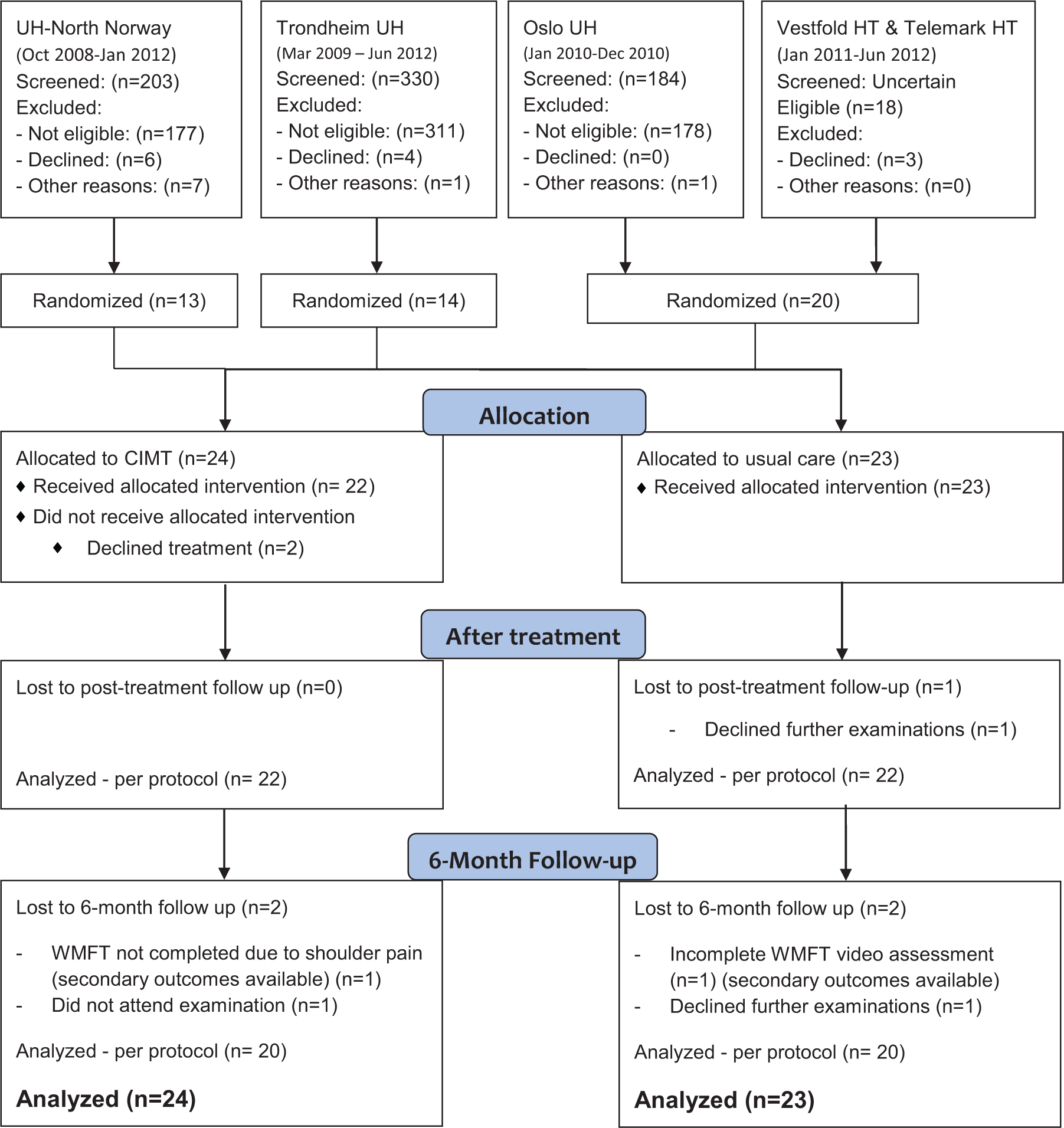
Flow diagram of eligibility and the inclusion in the Norwegian CI-Therapy multisite trial.
Of the 22 CIMT participants available for posttreatment assessment, 19 completed all 10 treatment days, 1 missed 3 days of treatment, and 2 participants missed 1 day of treatment. The mean time in CIMT treatment was 27.0 hours (SD = 2.8 hours). Among the participants, 20 (91%) were inpatients between the inclusion and posttreatment assessments. In comparison, 15 (68%) of the participants in the control group were inpatients between the pretreatment and posttreatment assessments (
Per Protocol Analysis of Primary and Secondary Motor Outcomes. a
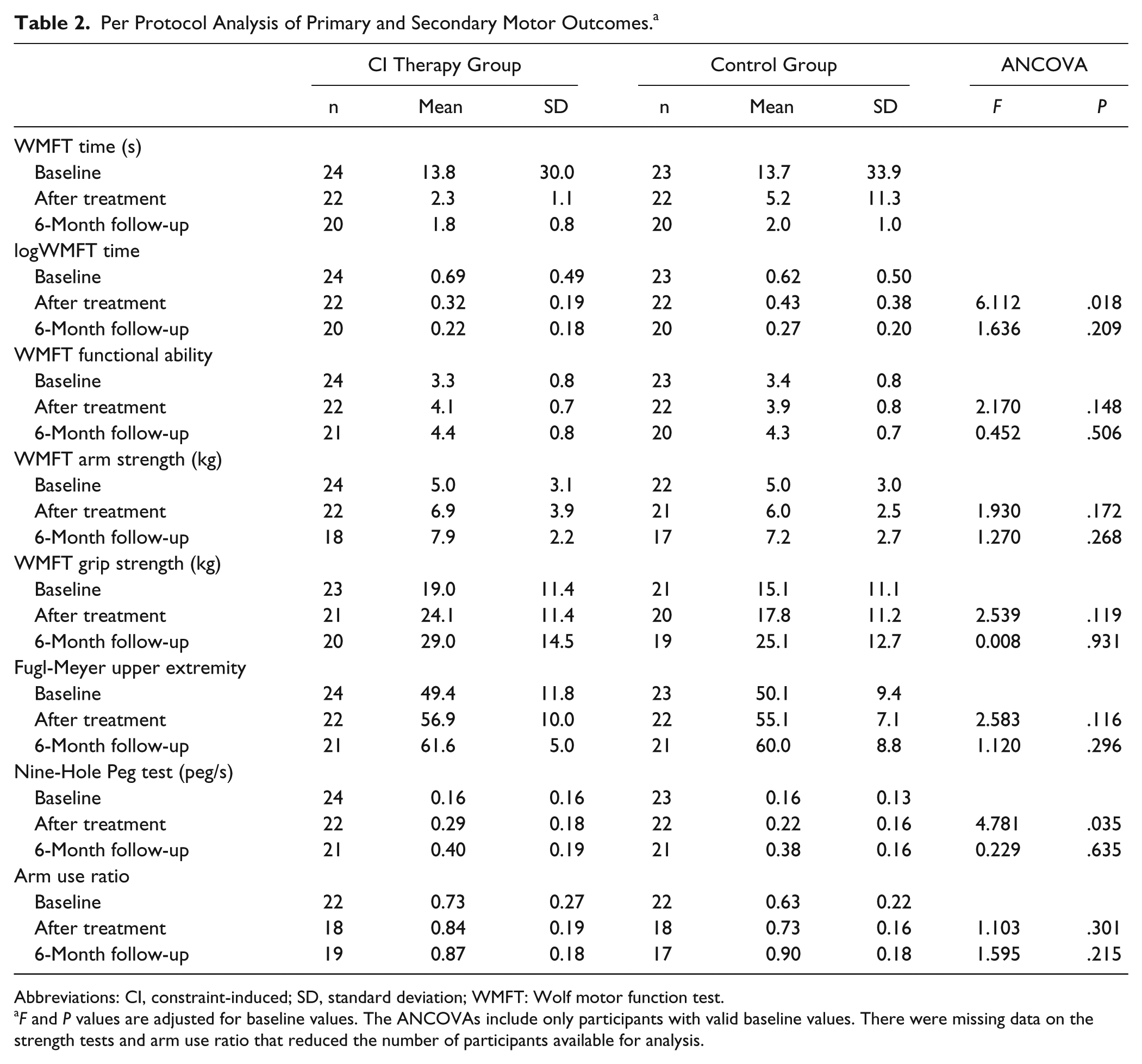
Abbreviations: CI, constraint-induced; SD, standard deviation; WMFT: Wolf motor function test.
Among the secondary outcomes, all motor variables improved significantly in both groups from baseline to posttreatment and from posttreatment to the 6-month follow-up. There was a significant posttreatment difference for the NHPT (partial η
2
= 0.104;
Stroke Impact Scale (SIS) at the 6-Month Follow-Up.
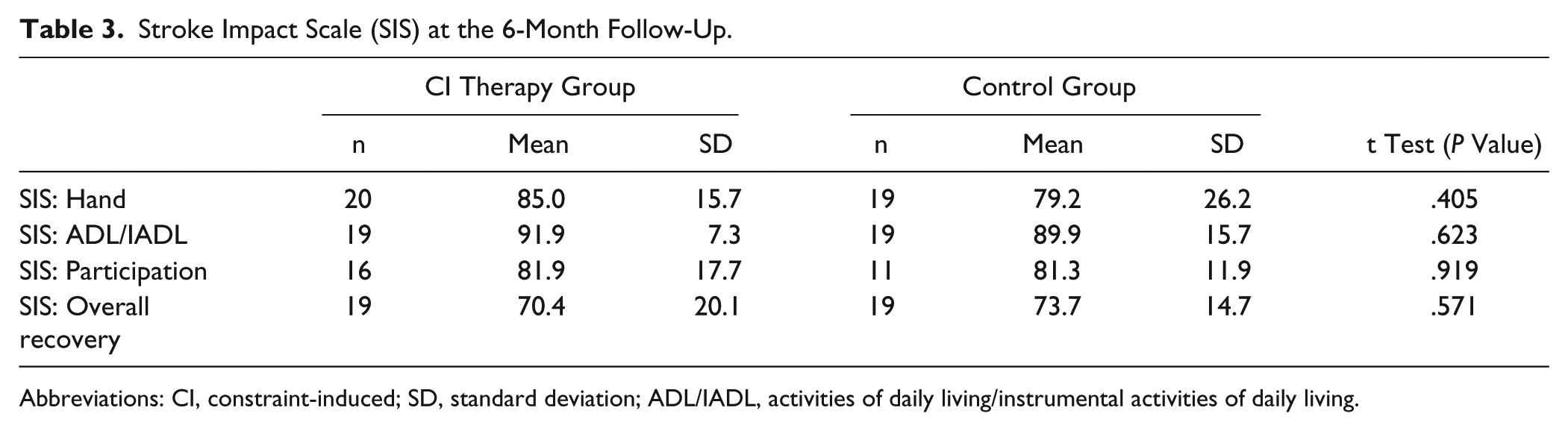
Abbreviations: CI, constraint-induced; SD, standard deviation; ADL/IADL, activities of daily living/instrumental activities of daily living.
Discussion
The main result from the present trial was that CIMT initiated within 1 month after stroke was safe and feasible but did not improve long-term motor function. However, that we observed significantly better WMFT scores in the CIMT group posttreatment indicates that CIMT might promote faster recovery compared with standard care. The 2.9-s difference in WMFT and the 0.7-peg/s difference in NHPT indicates a moderate short-term effect of CIMT on arm motor function and dexterity. There were no differences between the groups in the ability to reduce arm motor impairment or increase arm use.
An obvious limitation of this study is that it was halted early with far fewer participants than recommended from the pretrial power calculations. Despite the low power, the differences between the groups were rather small, and a larger sample might not reveal results of clinical significance. The sizes of the treatment arms in the present study are also larger than in comparable studies,6-8 and the trial, therefore, provides important new data on CIMT in early stroke rehabilitation. Another limitation is that there was no periodic reevaluation or training of the examiners. The trial was extended for 2.5 years more than planned, and this might have introduced bias in the measurements. The drop-out rate was also relatively high, and this represents a risk of an effect-dependent bias in the results. The nature of the control group has to be considered when the effects are interpreted. The control group in this trial was treated as usual and did not follow a predefined protocol. All control participants were treated in multidisciplinary stroke units, and the majority of both groups were inpatients in the time between inclusion and the first posttreatment assessment. However, we were not able to get reliable data about the actual time spent in active upper-limb training in the control group. Therefore, we are not able to control the effects for dosage of therapy. A large proportion of the included participants had mild to moderate strokes. This caused a ceiling effect in most of the outcome measures that might have influenced the results, especially at the 6-month follow-up. The trial included mostly men. Although sex did not appear to affect the outcome, the results might not be applicable to women. In the CIMT group, 16 (67%) of the participants were affected on the dominant side, and the results might be less relevant for stroke patients with an affected nondominant arm. Unfortunately, technical problems with accelerometer logistics reduced the data for this secondary outcome.
The findings of the present study do not demonstrate the same trend toward less impairment after CIMT as shown by Ro et al 34 and Boake et al. 7 On the other hand, the results are in line with those described in a meta-analysis by Nijland et al, 10 who concluded that there was no significant improvement following high-intensity CIMT initiated within 1 month after stroke. Another important finding was that, despite 2 cases of shoulder pain, there was no unfavorable development in motor function in the CIMT group compared with the usual care group. This is in contrast with the results of the VECTORS study, 8 where the high-intensity CIMT group (3 h/d, 10 days) scored significantly worse on arm motor function (ARAT) than the low-intensity CIMT and ADL practice/bilateral training groups. The ARAT and FMA are strongly correlated,35,36 and the difference is probably not attributable to the different measurements. Possible explanations could be that different components of the CIMT treatment, like greater levels of task practice in the VECTORS study, accounted for differences in component compliance. Task practice usually leads to more continuous work during the treatment sessions, with fewer breaks, compared with shaping exercises. 37 The time from stroke to inclusion was also lower in the VECTOR study (9.7 days) compared with ours (17.3 days). This difference might contribute significantly to the differences in results. In addition, our control intervention was not standardized, whereas Dromerick et al 8 studied an experimental control group that received standardized treatment. The 2-hour ADL practice and bilateral training used in the VECTORS study might have been more effective than the protocol used here. Our results showed a considerably weaker effect than the study published by Page et al, 9 which might be attributable to the different CIMT protocol in their study (0.5 h/d, 30 days over 10 weeks). The difference might also be related to the fact that learned nonuse is developed over time and that the lower-intensity, longer-duration treatment is more effective than the 2-week program used in our trial in the early rehabilitation. Another possible reason is that Page et al 9 only included patients with a motor activity log score <2.5. The motor activity log is highly sensitive to nonuse and including patients with low scores and equal motor impairment would result in a sample of participants with more pronounced nonuse that might respond better to the treatment. The differences between our study and others might also be attributed to another difference in inclusion criteria. Previous trials7,8,34 excluded participants without detectable shoulder weakness (NIHSS arm score <1). Among our participants, 32 had a NIHSS arm motor score of 0, and the FMA values of our participants indicate less impairment than that of participants included in the other studies.7,8,34 The effects of CIMT might change according to the level of upper-extremity impairment.
Implications
The CIMT protocol used in this trial can be safely applied within 28 days poststroke and might increase movement speed in the early stage. Future research should focus on the variability between treatment protocols, optimal therapy time, and the reasons for the large variations in results within 4 weeks poststroke. At the moment, application of CIMT in early stroke rehabilitation is not warranted because of limited evidence of lasting effects.
Footnotes
Acknowledgements
We would like to thank Martin Vatshaug for collating and ensuring the quality of data, Gunn Marte Vadset and Eli Langørgen for taking part in the design of the treatment protocol, and Grete Jakobsen for teaching the study protocol and acting as a site administrator. We would also like to thank Paul A. Thompson and Steven Wolf for providing the data for the power calculation and Tom Wilsgaard for statistical advice. The study was supported by the Northern Norway Regional Health Authority (SAT 544-06). The authors declare no conflict of interests.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
