Abstract
Background. In animal stroke models, peri-infarct cortical stimulation (CS) combined with rehabilitative reach training (RT) enhances motor functional outcome and cortical reorganization, compared with RT alone. It was unknown whether the effects of CS + RT (a) persist long after treatment, (b) can be enhanced by forcing greater use of the paretic limb, and (C) vary with treatment onset time. Objective. To test the endurance, time sensitivity, and the potential for augmentation by forced forelimb use of CS + RT treatment effects following ischemic stroke. Methods. Adult rats that were proficient in skilled reaching received unilateral ischemic motor cortical lesions. RT was delivered for 3 weeks alone or concurrently with 100-Hz cathodal epidural CS, delivered at 50% of movement thresholds. In study 1, this treatment was initiated at 14 days postinfarct, with some subgroups receiving an overlapping period of continuous constraint of the nonparetic forelimb to force use of the paretic limb. The function of the paretic limb was assessed weekly for 9 to 10 months posttreatment. In study 2, rats underwent CS, RT, and the combination during the chronic postinfarct period. Results. Early onset CS + RT resulted in greater functional improvements than RT alone. The CS-related gains persisted for 9 to 10 months posttreatment and were not significantly influenced by forced use of the paretic limb. When treatment onset was delayed until 3 months post-infarct, RT alone improved function, but CS + RT was no more effective than RT alone. Conclusion. CS can enhance the persistence, as well as the magnitude of RT-driven functional improvements, but its effectiveness in doing so may vary with time postinfarct.
Keywords
Introduction
Stroke is a leading cause of long-term disability worldwide. 1 Rehabilitative training (RT) reduces impairments but is often insufficient to restore normal functionality. Cortical stimulation (CS) combined with RT (CS + RT) is promising for facilitating RT effects. In rats and monkeys, high-frequency bipolar, cathodal, or anodal CS delivered via epidural or subdural electrodes over peri-infarct motor cortex (MC) enhances RT-induced performance improvements on skilled reaching tasks compared with RT alone2-8 and increases dendritic and synaptic densities,3,4 forelimb movement representation area6,7 and motor cortical evoked potentials 8 in peri-infarct MC. This CS approach appears to be particularly beneficial for improving behaviors that are practiced during its delivery.2-4 Noninvasive stimulation approaches, for example, repetitive transcranial magnetic stimulation (rTMS) and transcranial direct current stimulation (tDCS), also enhance motor learning in healthy individuals9,10 and improve behavioral outcomes and modulate brain reorganization after stroke.11-13 Despite differences in effective delivery parameters and polarities between clinical transcranial CS approaches and the subcranial CS tested in animal studies, they converge in supporting that modulation of cortical activity can yield at least short-lasting improvements in poststroke motor function.
The persistence of, and potential time-dependency in, CS + RT effects have not been examined, and this was the focus of the present study. We also investigated whether CS + RT effects are enhanced by forced use of the nonparetic limb outside of RT sessions, to model aspects of constraint-induced movement therapy (CIMT), a clinical treatment combining forced use of the paretic upper extremity with intense RT. 14 In study 1, we investigated whether CS+RT initiated 14 days after MC infarcts, with or without forced use, yields enduring improvements in skilled motor function as assessed for 9 to 10 months posttreatment. In study 2, CS + RT was initiated ~3 months postinfarct to test whether delayed treatment promotes the functional improvements and dendritic plasticity observed after earlier treatment.2-4
Materials and Methods
Design Overview
Study 1 tested the persistence of functional improvements resulting from early onset CS + RT and the effects of combining it with forced use of the paretic (contralesional) forelimb (Figure 1A). On postlesion day 9, rats were placed into vests that either constrained the nonparetic forelimb (forced-use vests) or permitted unrestricted use of both forelimbs (control vests) for 14 days. This timing avoids an early period of vulnerability to excessive forced use 15 and enabled rats to become accustomed to the vests prior to RT onset (day 14). The 2-week duration in the vests is one that is safe and well tolerated. 16 RT was performed in 19 sessions over 24 days.
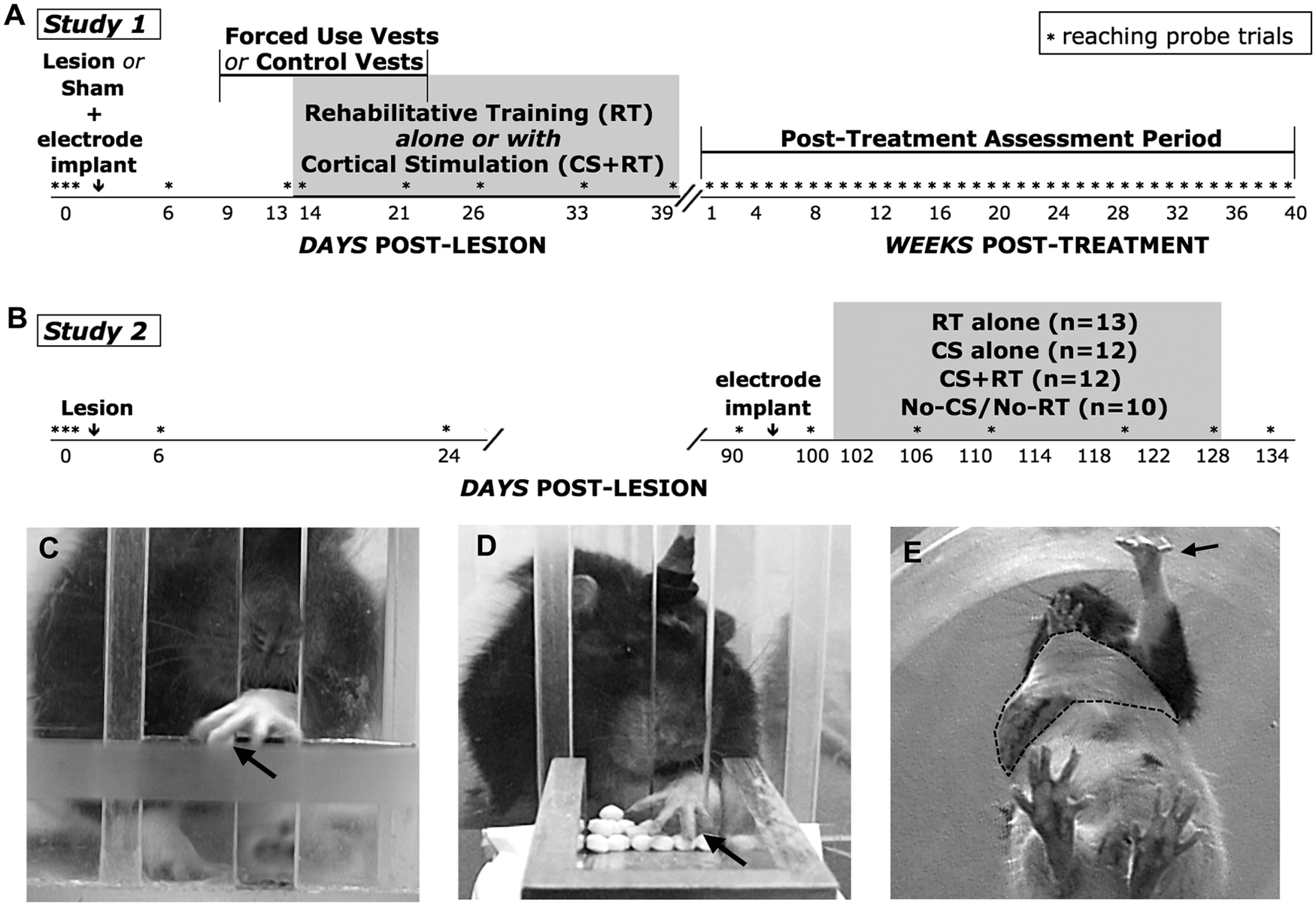
Study overviews. Both studies tested the effects of combining cortical stimulation (CS) with rehabilitative reach training (RT). (A) Study 1 tested the long-term persistence of CS + RT effects when treatment was initiated early (2 weeks) after ischemic lesions. Lesion groups received RT or CS + RT with or without forced use of the paretic limb (4 groups, n = 12-13). Sham-operates (no ischemia) received the same reach training procedures with or without CS (2 groups, n = 5-6). (B) Study 2 tested the effects of treatment that was delayed in onset postlesion (~3 months). Histological endpoints were at the end of each study time line, with the exception of a subset of study 1 lesions sampled in post-RT week 1 (n = 4). Note the changes in time scales. In both studies, the single-pellet retrieval task (SPR) (C) was learned by all rats preoperatively and used postoperatively to measure lesion and treatment effects on reaching function. The apparatus design enforces the use of the designated forelimb for reaching. The arrow points to a pellet. The tray reaching task (D) was used to provide RT. The arrow points to the impaired limb. CS treated groups received current delivery concurrent with performance of the RT task or no-training control procedures. All other groups were connected to stimulating cables during the sessions, but received no current delivery. (E) Constraint vest (dashed outline) used in study 1 to force use of the paretic limb (arrow).
Study 2 tested effects of delayed CS + RT treatment compared with RT alone (Figure 1B). Electrodes were implanted 10 days prior to RT (to ensure their viability). No-treatment and CS-alone groups were included to determine the effects of delayed RT, which can wane in efficacy over time after injury.17,18 A slightly greater quantity of RT was used (24 sessions over 24 days) to probe for late emergence of treatment effects. Finally, immunolabeling with microtubule-associated protein 2 (MAP2) was used to visualize and quantify cortical dendrites to test whether, as with early onset CS + RT, 4 delayed CS + RT increases dendritic densities in peri-infarct MC.
In both studies, a highly sensitive measure of fine motor function, the single-pellet retrieval task 19 (SPR; Figure 1C) was the primary behavioral measure. Rats were preoperatively trained to proficiency on the SPR with their preferred-for-reaching limb to maximize sensitivity in detecting lesion and treatment effects on this skill. The similarity of postoperative impairment levels across conditions and effectiveness of the forced-use procedure were also verified with other sensorimotor tests (see Supplementary Material). Lesions and electrode implantations were contralateral to the preoperatively trained forelimb. RT consisted of daily practice with the paretic limb on a tray-reaching task (Figure 1D). This task provides a less sensitive measure of reaching function than the SPR, but encourages practice by requiring less precise reach-to-grasp movements. 20 During the RT period, rats received 1 session per day of RT or no-RT control procedures with or without concurrent CS.
Animals
Male Long-Evans rats (n = 102, Charles Rivers Laboratories, Wilmington, MA) were housed in pairs or triplets on a 12:12-hour light:dark cycle with water ad libitum, in cages with standardized supplements (PVC and cardboard tubes, wooden objects and a complex food mixture). Rats were tamed by handling and maintained on scheduled feeding (15 g chow per day, gradually increased to permit normal age-related weight gain). Lesion and sham surgeries were performed at age 3.5 to 4 months. Animals were assigned randomly to treatment conditions with the exception that they were carefully matched for initial severity of lesion-induced impairments (Supplementary Table 2). In study 1, the first batch of rats examined at the 12- to 13-month postoperative histological endpoint were found to have pockets of infection surrounding the electrodes, with evident morphological shifts of underlying cortex (n = 2-4 per lesion group, 1 per sham group, 11 total). This was linked with a single electrode batch and was not found subsequently. The infections appeared to be fully encapsulated by skin and dura, did not invade brain tissue, there was no indication of poor health, and all behavioral measures were similar to other animals in the same conditions. Therefore, these animals were kept in the behavioral analyses but were omitted from anatomical analyses. All animal procedures were approved by the University of Texas Animal Care and Use Committee.
Surgical Procedures
Focal ischemia of the caudal forelimb area of MC was created with endothelin-1 (ET-1), a vasoconstricting peptide21,22 (see Supplementary Methods). Sham operates received all procedures except ET-1 application.
Electrodes consisted of two 0.4 × 2 mm platinum epidural contacts, a skull mounted connector pedestal (9-pin ABS plug, Ginder Scientific, Inc, Ottawa, Ontario, Canada), and a 3-mm diameter platinum disk return electrode inserted subcutaneously caudal to Lambda. For study 1, 10 minutes after ET-1 application (or skull opening in sham operates), the craniotomy was enlarged ~1 mm rostrally and medially to expose perilesion MC. The epidural contacts were positioned parallel to midline over remaining forelimb and surrounding regions of MC, a position that reliably elicits contralateral forelimb and/or face movements when current is delivered.2-4 Electrode implantation was identical in study 2 except that it was performed in a separate surgery (postinfarct day 92).
Single-Pellet Retrieval Task
The single-pellet retrieval task (SPR) has been described in detail previously.23-25 After shaping to determine limb preferences, animals were trained to reach with the preferred limb through a narrow window to retrieve a banana flavored food pellet (45 mg, Bio-Serve, Frenchtown, NJ) from a well 1 cm from the opening (Figure 1C). Per trial, up to 5 reach attempts were permitted to retrieve one pellet. Preoperatively, rats were trained to approximately the end of the “rapid acquisition” phase of task learning, after which time further performance improvements are typically subtle.26,27 (See Supplementary Methods.) This quantity of pretraining enables highly sensitive detection of postlesion impairments and treatment effects.2,3,20,28 Postlesion probes of reaching performance were analyzed as the change from prelesion in the percentage success (grasping, retrieving, and eating the pellet) per reach attempt. Frame-by-frame analysis of reaching movements was also performed using a quantitative adaptation of Whishaw’s movement analysis29-31 (Supplementary Material).
Cortical Stimulation and Rehabilitative Training
During RT sessions, rats reached with the paretic limb for 100 pellets, delivered 50 at a time in two 5-minute increments on a 25° inclined tray (Figure 1D). No-RT groups received 100 pellets on the chamber floor. CS consisted of 100-Hz cathodal current (2-second trains of 100-µs pulses at 50% movement threshold) delivered for the entire RT session or no-RT control procedure. Cathodal CS was used because it tends to yield greater effects than anodal CS in this model.2,6 All animals were connected to stimulator cables during RT, but no current was delivered to No-CS and RT-only groups. Movement thresholds were defined weekly by the minimum current needed to evoke forelimb or head/neck movement. Consistent with previous findings,2-8 movement thresholds declined over time, similarly across treatment conditions (Supplementary Table 1).
Forced Use
On postlesion day 9 of study 1, animals were anesthetized with ketamine (100 mg/kg) and xylazine (10 mg/kg) and fitted in customized vests that forced reliance on the paretic forelimb (Figure 1E) or allowed free movement of both forelimbs (Supplementary Methods). The forced-use vests allowed limited movements of the ipsilesional/nonparetic forelimb, while largely preventing its use for postural support. Vests were removed on postlesion day 25.
Histology
After pentobarbital overdose, rats were transcardially perfused with 0.1 M phosphate buffer and fixative (2.5% glutaradehyde and 2% paraformaldehyde in study 1; 4% paraformaldehyde in study 2). Coronal serial sections (50 µm) created with a vibratome were Nissl stained with toluidine blue for volume measures and lesion reconstructions. In study 2, an adjacent section series was used for MAP2 labeling. One RT (study 1) and one CS + RT (study 2) brain were excluded from the anatomical analyses because of sectioning errors. Anatomical samples were coded to blind for experimental condition.
Sensorimotor cortical volumes were measured using the Cavalieri method, 32 and lesion placements were reconstructed using cytoarchitectonics 33 (Supplementary Methods). Volume measures were expected to be sensitive to treatment effects on surviving cortex, rather than differences in initial infarct size. The use of volume measures to infer infarct size is complicated by other morphological changes, and these were likely in the present studies given the long time spans and the propensity of CS and reach training to promote neuronal growth in cortex,2-4 which can increase its volume. The potential for group differences in initial infarct size was minimized by the highly reproducible infarct induction method 21 and by matching for pretreatment behavioral impairment levels (Supplementary Table 2) on measures established to be sensitive to variations in sizes of these lesions.34-39 In study 1, we also used cytoarchitectonics to delineate and measure volumes of sensorimotor cortical subregions: the medial agranular (AGm), lateral agranular (AGl), and forelimb (FlOL) and hindlimb (HlOL) sensorimotor overlap zones (Supplementary Methods).
In study 2, sections were immunostained for the dendritic cytoskeletal protein, MAP2, using a free-floating section method 21 . The cycloid grid intersection method was used to quantify the surface density of MAP2-labeled dendrites in peri-infarct MC 21 (Supplementary Methods).
Statistical Analyses
Both studies were designed to use a priori planned comparisons for the primary analyses, using SPSS (SPSS, Inc.) or SAS (SAS Institute, Inc, Cary, NC) general linear models procedures for analysis of variance (ANOVA), with post hoc T tests as warranted by ANOVA. In study 1, repeated-measures ANOVAs tested whether RT effects were influenced by (a) CS (CS + RT vs RT alone) or (b) forced use (RT + Forced Use vs RT alone), and whether (c) forced use altered CS + RT effects (CS + RT + Forced-Use vs CS + RT). Additionally, we tested whether CS affected sham operates (Sham-CS + RT vs Sham-RT). Data from the RT period and chronic period were analyzed separately to investigate the persistence of CS effects. In study 2, planned comparisons tested effects of delayed-onset of (a) RT alone (RT alone vs No-CS/No-RT) or (b) CS alone (CS vs No-CS/No-RT), and whether (c) CS alters RT effects (CS + RT vs RT). Volume data were analyzed with ANOVA using the same planned comparisons. Secondary analyses compared cortical volumes of lesion groups with sham operates (Study 1), ipsi to contra hemisphere volumes within groups, and pre- versus postoperative reaching performance. Data are reported as means (M) and standard errors (SE).
Results
Behavior During the Early Treatment Period
Lesions impaired reaching performance, as probed on the SPR prior to RT (days 6 and 13; Figure 2A and B; see also Supplementary Figure 1). During treatment, CS + RT significantly improved reaching performance on the SPR compared with RT alone: Group × Day interaction, F(7, 140) = 2.80, P = .009. In post hoc analyses, groups were not significantly different on any individual probe day, although the CS + RT group tended to perform best near the end of treatment. Forced use did not further improve effects of CS + RT: versus CS + RT + Forced Use, Group × Day, F(7, 154) = 0.87, P = .54; nor RT alone: versus RT + Forced Use, Group × Day, F(7, 141) = 1.43, P = .18 (Figure 2B). In sham operates, CS did not significantly affect reaching performance: Group × Day, F(6, 54) = 0.95, P >.05 (Figure 2C), though performance transiently surpassed preoperative baseline performance (possibly because of motivational influences of the more rewarding RT task, as described in Supplementary Results). The modest transient declines in performance early postoperatively in sham operates may be due to craniectomies 40 and/or electrode implants. There were no significant main effects of Group in any of the planned comparisons (Ps > .05). All analyses indicated significant effects of Day (Fs = 12.5-31.5, Ps < .001), reflecting performance improvements in all groups over the RT period. In the RT (tray reaching) task, rats with sensorimotor cortex infarcts initially retrieved fewer pellets than did sham-operates, but by the ninth training day, all groups retrieved most pellets (Supplementary Figure 2A).
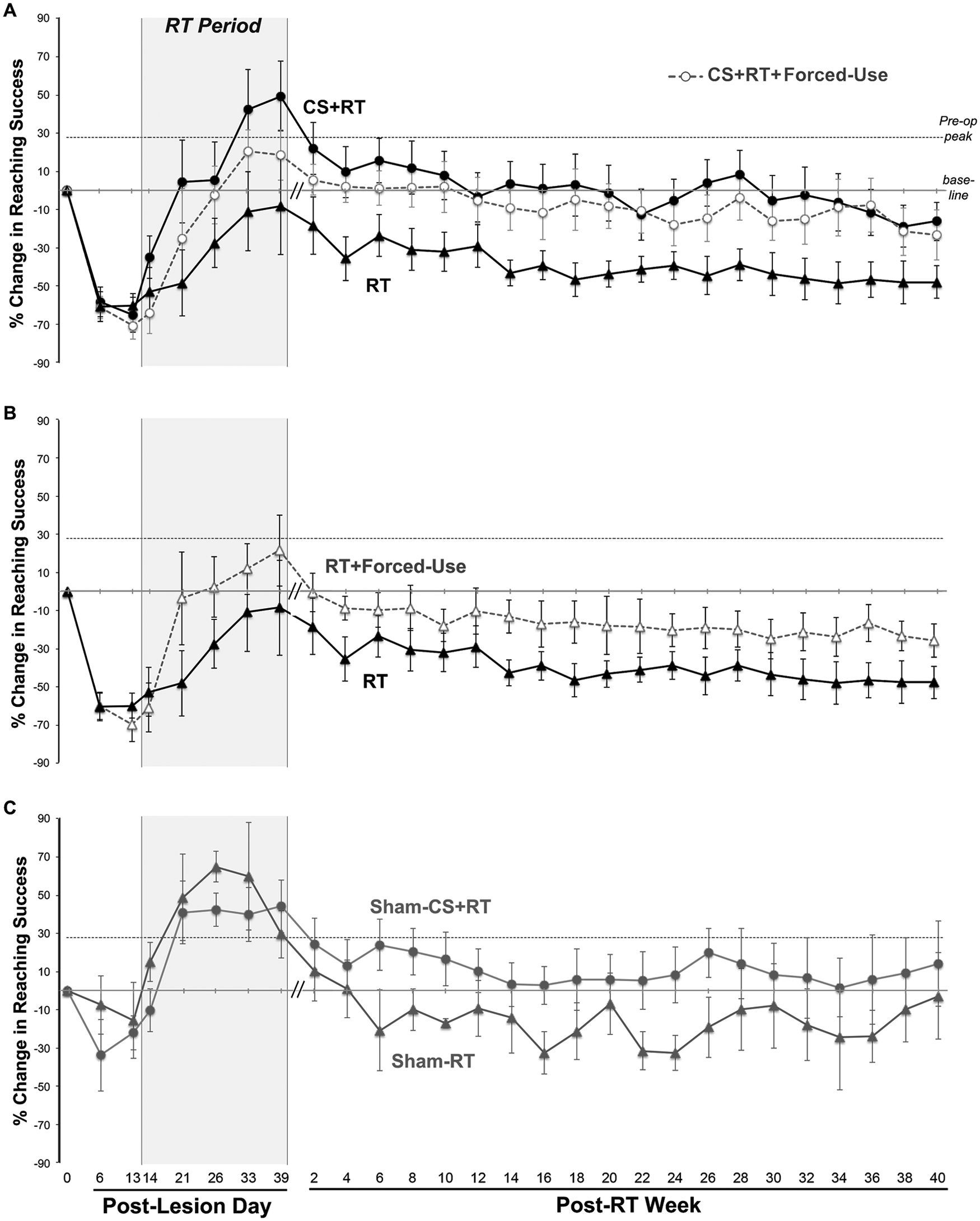
Reaching performance on the single-pellet retrieval task in study 1 (early treatment onset). (A) During the rehabilitative training (RT) period, animals receiving cortical stimulation combined with RT (CS + RT) had improved reaching performance compared to RT alone (P < .01). The improved relative performance was maintained over the following 9 to 10 months (P = .01). Forced use did not significantly alter CS + RT effects. (B) The effects of forced use without CS (RT + forced use) failed to reach significance compared with RT alone (P = .06 in the posttreatment period). The same data from the RT group are plotted in A and B to facilitate comparisons. (C) In the sham (no-lesion) groups reaching performance was similar with and without CS during the RT period and not significantly different in the posttreatment period (P = .13). Data are mean ± standard error (M ± SE) % change in reaching success rates relative to preoperative baseline [%(postoperative − preoperative)/preoperative]. Baseline is the M of the last 3 days of preoperative training, which was lower than the peak preoperative performance due to day-to-day variability after this proficiency level was achieved.
Behavior in the Chronic Period After Early Treatment
Over the 9 to 10 months after treatment, the CS + RT group maintained SPR performance improvements relative to RT alone (Figure 2A): Group, F(1, 20) = 8.06, P = .01. This effect did not significantly vary over time (Group × Week, P > .05) or with prior forced-use condition (CS + RT vs CS + RT + Forced Use): Group, F(1, 22) = 0.29, P = .59). Effects of RT + Forced Use compared with RT alone (Figure 2B) failed to reach significance: Group, F(1, 21) = 3.96, P = .060. In sham operates, there were no significant CS effects (Figure 2C): Group, F(1, 9) = 2.79, P = .13. All analyses indicated significant main effects of Day (Fs = 4.22-5.32, Ps < .001), reflecting declines in reaching performance in all groups with time (and age). However, reaching movement abnormalities also diminished over the first 12 weeks after RT in lesion versus sham groups (Supplementary Figure 3A).
Behavioral Effects of Delayed Treatment Onset
Prior to treatment onset in study 2, reaching performance in each group remained impaired compared with preinjury performance (days 0 vs 100: Ps < .05, Figure 3). RT beginning at ~3 months postlesion significantly improved SPR performance compared to No-CS/No-RT controls: Group × Day, F(8, 160) = 6.37, P < .001. In contrast to the earlier treatment, CS did not improve RT effects: CS + RT versus RT, F(8, 168) = 0.42, P = .92. CS alone also had no significant influence on reaching performance: CS versus No-CS/No-RT, F(8, 144) = 0.94, P = .48). There was also a nonsignificant (P > .05) drop in performance in the CS + RT and RT groups 1 week after CS and RT were discontinued. Groups performed similarly on the RT task (Supplementary Figure 2B).
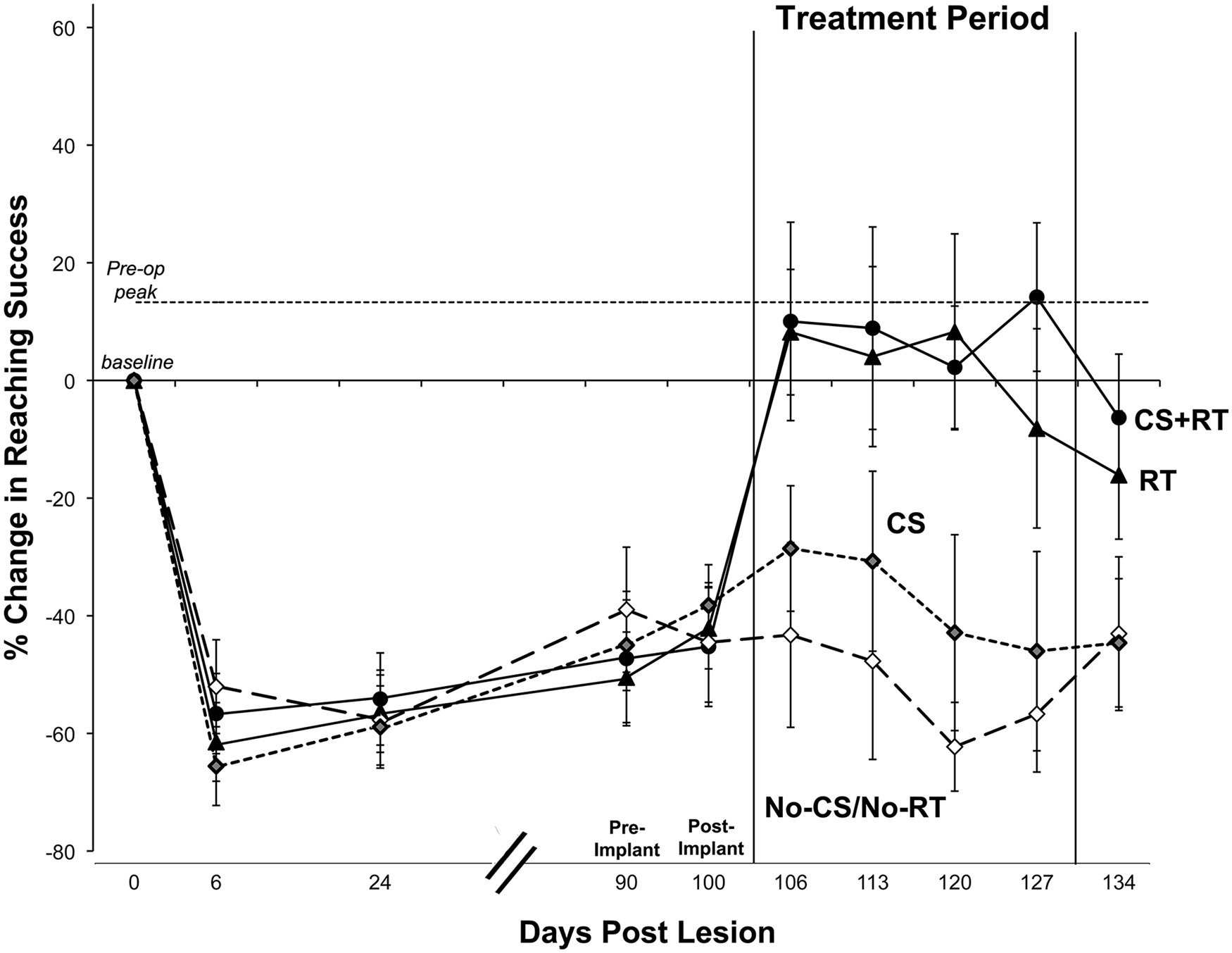
Performance on the single-pellet retrieval task in study 2 (delayed treatment onset). When initiated ~3 months after ischemic lesion induction, rehabilitative training (RT) alone (P < .05), but not cortical stimulation (CS) alone, improved performance compared to animals with neither treatment (No-CS/No-RT). In contrast to its effects earlier after the lesion, CS + RT did not significantly improve performance compared with RT alone. Data are mean ± standard error (M ± SE) % change for preoperative performance in reaching success rates.
Anatomical Results 9 to 10 Months After Early Treatment
Reconstruction of cortical damage relative to cytoarchitectural subregions revealed lesion placements that were similar between groups (Figure 4A and B). Damage was centered in the FlOL and extended into AGm and AGl subregions, with the HlOL relatively spared, as intended (Figure 4C). Most animals had superficial damage to the corpus callosum. Eight animals (from RT, RT + Forced-Use, and CS + RT + Forced-Use groups) had deeper white matter damage; however, direct damage to the striatum was not evident. Lesion placements were similar in the brains (n = 4) examined immediately after the RT period.
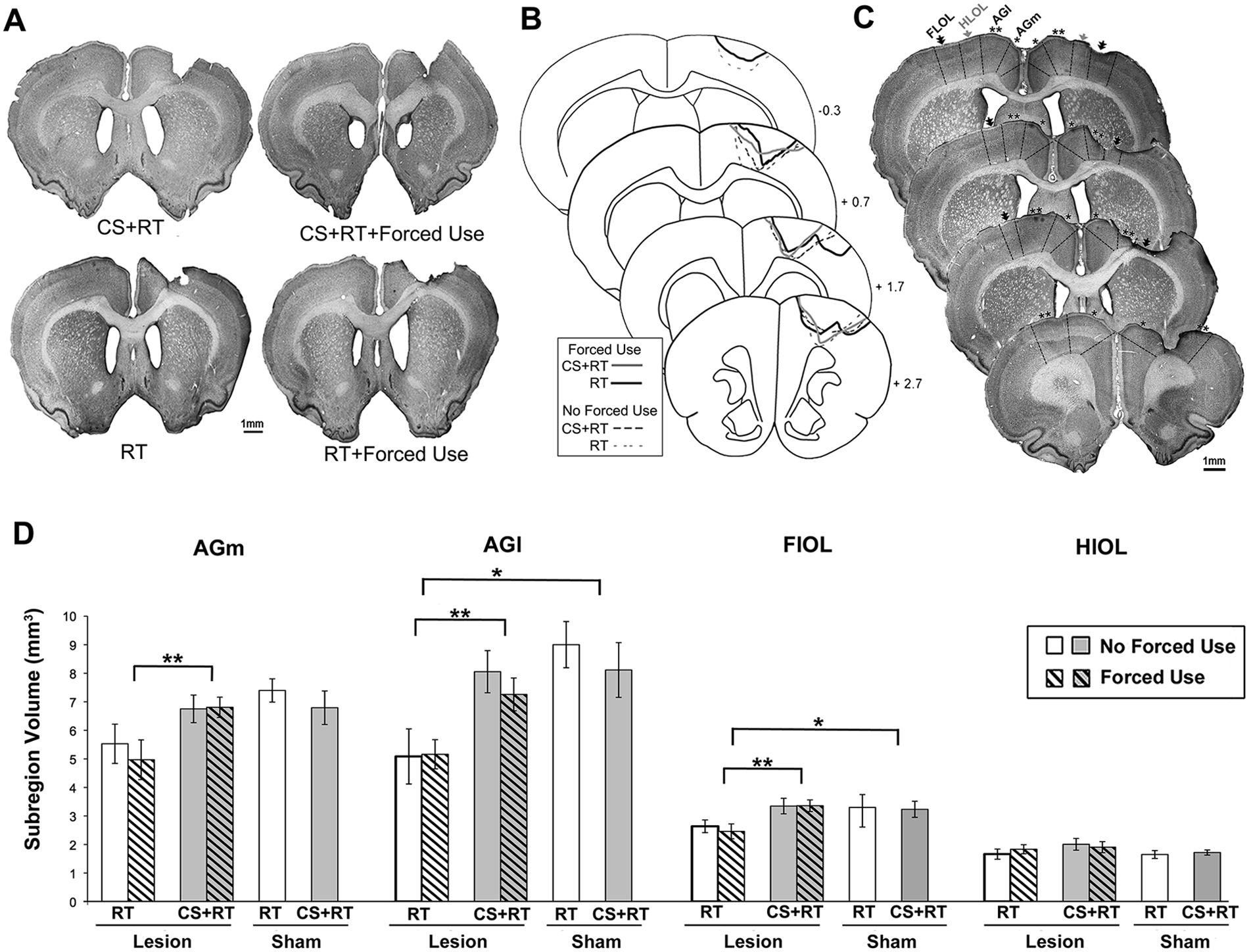
Lesion placement and cortical volume measures as assessed 9 to 10 months after the early treatments of study 1. (A-C) Representative coronal sections (A) and reconstructions (B) based on cytoarchitectonics (C) of the cortical damage in each of the lesion groups. The reconstructions indicated similar lesion extents across treatment conditions. (D) Volumes of sensorimotor cortical subregions. Compared with rehabilitative training (RT) groups, combined cortical stimulation and RT (CS + RT) groups had significantly greater volumes of cortical subregions surrounding the infarct (and near the focus of stimulation). This included the medial agranular cortex (AGm), lateral agranular cortex (AGl) and forelimb overlap zone (FlOL). The volumes of the same subregions were significantly reduced in RT, but not in CS + RT, lesion groups compared with Sham. There were no group differences in the hindlimb overlap zone (HlOL), which is posterior to the infarct and more remote from the focus of CS than the other regions. Data are mean ± standard error (M ± SE). *P < .02; **P < .005.
Cortical volumes differed neither between the 2 sham subgroups nor between the forced-use versus control vest subgroups (Ps > .05). These groups were therefore combined for subsequent analyses. As shown in Table 1, each lesion group had significantly reduced ipsilesional versus contralesional cortical volumes. The interhemispheric volume difference (contra minus ipsi) in the RT groups (−17.5 ± 3.1) was significantly different than Shams (−4.6 ± 2.8): F(1, 23) = 8.80, P = .007, as well as CS + RT (−6.3 ± 2.6): F(1, 34) = 8.02, P = .008. CS + RT was not different from Sham: F(1, 29) = 0.15, P = .69. As discussed below, these volume effects are unlikely to reflect differences in initial infarct size and are likely to reflect CS + RT treatment effects.
Sensorimotor Cortical Volumes (mm3) of the Injured (Ipsi) and Intact (Contra) Hemispheres. a
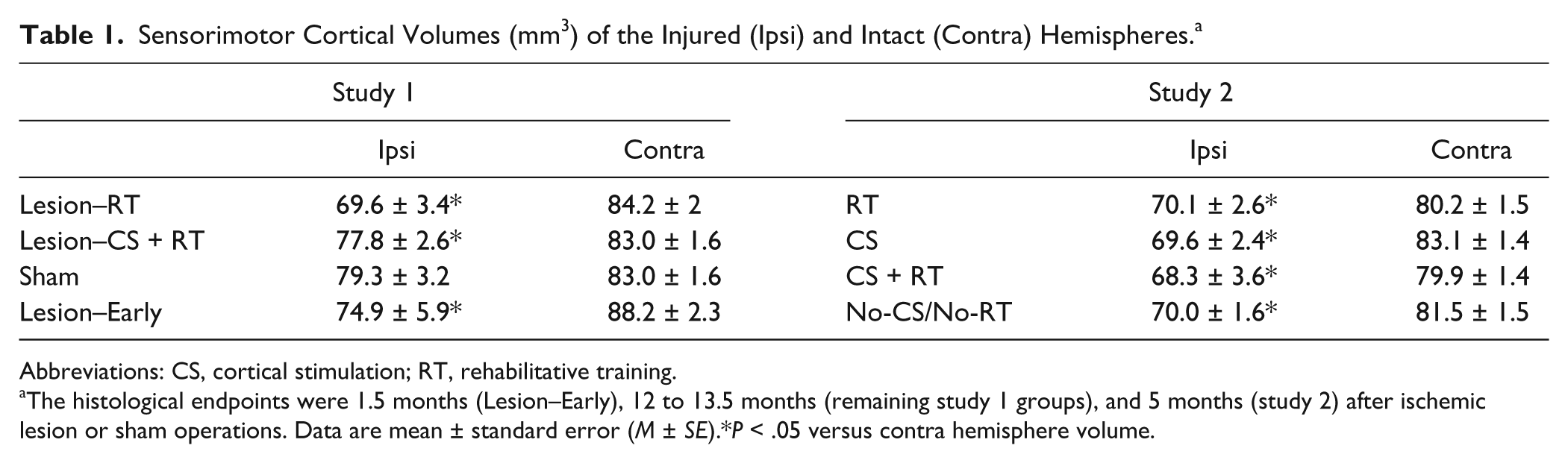
Abbreviations: CS, cortical stimulation; RT, rehabilitative training.
The histological endpoints were 1.5 months (Lesion–Early), 12 to 13.5 months (remaining study 1 groups), and 5 months (study 2) after ischemic lesion or sham operations. Data are mean ± standard error (M ± SE).*P < .05 versus contra hemisphere volume.
As shown in Figure 4D, CS + RT groups also had significantly greater volumes in the cytoarchitecturally defined AGl, AGm, and FlOL of peri-infarct cortex compared with RT. Volumes of these subregions were not significantly different between CS + RT and Shams. In contrast, RT groups had reduced volume compared with Sham in these same areas. In the subset of lesions examined 1 week post-RT, remaining ipsilesional cortical volume was more similar to CS + RT than to RT, consistent with continued atrophy of perilesion cortex over the longer postlesion period. HlOL volumes were not significantly different between groups (Ps > .05). In CS + RT, reaching performance during the RT period was significantly correlated with the volumes of AGl and AGm (combined: R2 = .33, P = .011), but not the other subregions (FlOL, P = .68; HlOL, P = .87), whereas these correlations were nonsignificant in the RT group (Ps = .31-.51).
Anatomical Results After Delayed Treatment
The placement of lesions in study 2 was generally similar to that described for study 1 (Figure 5A and B). In contrast to study 1, volume measurements of remaining sensorimotor cortex revealed no differences among conditions: Fs(1, 18-20) = 0.01 to 0.97, Ps = .97 to .43 (Table 1).
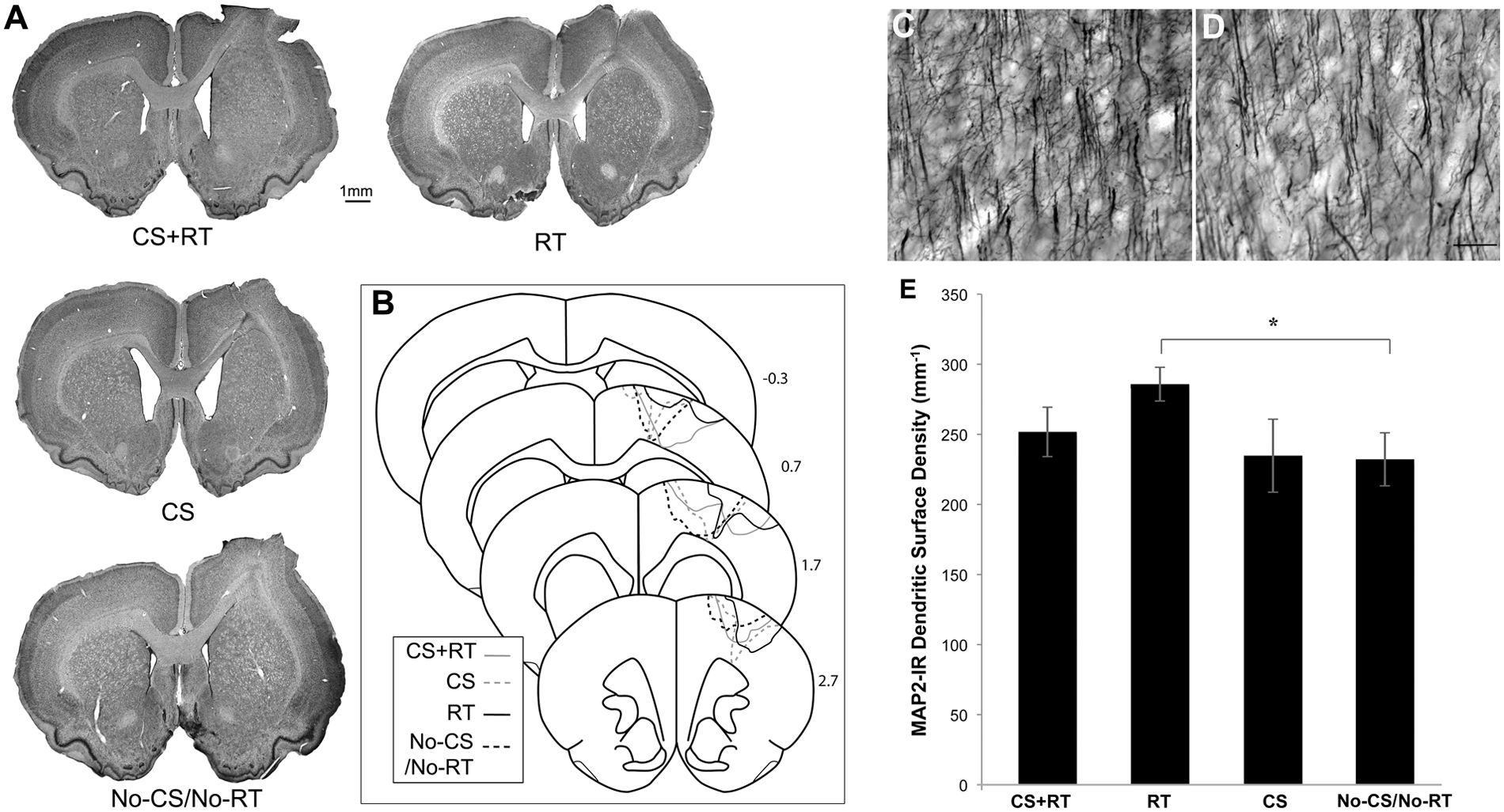
Lesion placement and microtubule-associated protein 2 (MAP2) analysis after delayed treatment in study 2. Representative coronal sections (A) and reconstructions (B) based on cytoarchitectonics in delayed treatment groups of study 2. Representative images of MAP2-labeled dendrites in peri-infarct layer V of rehabilitative training (RT) (C) and no cortical stimulation/No-RT (No-CS/No-RT) (D) groups. After the delayed onset of RT, there was a greater surface density of dendrites in layer V than found in No-CS/No-RT (E). Dendritic densities in CS + RT were not significantly different from those of RT (scale bar = 50 µm). Data are mean ± standard error (M ± SE). *P < .05.
As shown in Figure 5C to E, animals receiving delayed onset RT had significantly greater surface density of MAP2 labeled dendritic processes in layer V of peri-infarct cortex compared with No-CS/No-RT: F(1, 20) = 6.74, P = .017. Dendritic densities in CS + RT tended to be lower but were not significantly different from RT: F(1, 21) = 2.88, P = .11, in contrast to previous findings that early CS + RT increases dendritic densities relative to RT. 4 There were no differences between CS alone and No-CS/No-RT (P = .93). There also were no significant differences in layers II/III.
Discussion
During the early, but not late, phase of postinfarct recovery, delivery of epidural CS (CS + RT) during motor RT on a reaching task significantly and persistently improved reaching performance compared with RT alone. Rats receiving CS + RT beginning 2 weeks after the infarcts maintained improved performance relative to RT alone over the 9 to 10 months posttreatment period, although reaching performance declined in all groups with time. When treatment was delayed until 3 months postinfarct, RT alone improved performance, but there was no further improvement resulting from CS.
Cortical stimulation combined with RT enhances functional activity patterns and neuronal structural plasticity in peri-infarct MC and improves reaching performance of the paretic limb, as assessed during and shortly after treatment.2-8 This CS-facilitated plasticity likely contributed both to the long-term functional gains and the increased volume of MC subregions, as observed 9 to 10 months after CS + RT compared with RT alone. It is unlikely that the volume differences were because of differences in initial infarct sizes, as groups were matched for initial postoperative impairment levels on behavioral tests that are sensitive to lesion size34-39 and cytoarchitectural reconstructions indicated similar injury extents. Furthermore, previous studies have revealed no influence of CS + RT on injury extent or cortical volumes, as assessed shortly after early treatment.2-4 This is not surprising given that even the earliest treatment onsets of previous studies, as with the present, were well past sensitive windows for either neuroprotective 41 or neurotoxic15,42 effects on infarct evolution. Comparisons with a small subset of infarcts analyzed immediately after the early RT period suggest that there may be atrophy of peri-infarct cortex over the long posttreatment period, which was countered by CS + RT. 2 Although a thorough time-course analysis of cortical volume would be useful for verifying this, the possibility is consistent with findings of continued atrophy of surviving tissue long after the initial ischemic event in animals 43 and humans.44,45
It is also possible that early CS + RT promotes an expansion of residual tissue. In intact animals, neuronal growth and synapse addition in neocortex is often accompanied by expanded cortical volume. 46 Early CS + RT increases densities of dendrites and synapses in peri-infarct MC.3,4 This has not been linked with increased cortical volume as assessed shortly after the end of treatment, but it remains possible that these observations reflect a stage of structural plasticity that later contribute to volume increases. It is also possible that the neuronal structural plasticity protects against gradual atrophy of residual cortex in the posttreatment period.
We did not find that constraint of the nonparetic limb further improved CS + RT effects. In humans, CIMT improves motor function,47,48 enhances cortical activity49,50 and reduces gray matter atrophy. 51 In rats with striatal hemorrhage, forced use of the paretic forelimb combined with RT improves motor deficits and decreases tissue loss. 52 There was no indication of an additive effect of forced use and CS + RT in the present study. However, there was a nonsignificant (P = .06) tendency for forced use to improve the efficacy of RT without CS, raising the possibility that variations in the forced-use approach, including a more complete overlap in treatment periods, would have had a more robust effect. Furthermore, given that CS + RT and RT alone are less effective in animals with more severe initial impairments,3,36 forced-use of the paretic limb is potentially more effective in combination with CS + RT in more severely impaired animals. It should also be noted that, though the 2 weeks of forced use in this study was on par in duration with human CIMT studies, our manipulation is dissimilar from CIMT in numerous ways,48,53 for example, rats spend more time per day with the nonparetic forelimb constrained, but less in RT, and it seems reasonable to assume that the complexity of manual skill activities outside of RT is more limited in laboratory rats.
A striking behavioral pattern was the gradual decay in reaching performance over the chronic posttreatment period, which diminished performance improvements gained during RT in all groups. The decline may have been due to a lack of task practice, age, or both. The ~1-year period of observation spanned young adulthood to late-middle age in rats of study 1. Age-related declines in manual function are well established.27,54 However, reaching movement abnormalities also partially recovered in all lesion groups in the posttreatment period, which seems contrary to the idea that the performance decrements purely reflect age-related motor impairments. A similar tendency for performance to decline after RT was also seen in study 2. These data may indicate the need for skills that have been improved by RT to continue to be practiced after treatment ends. The rats had limited opportunity to do so outside of the testing environment. Even in the context of this decline, the early CS + RT group maintained relative performance improvements over RT alone. This probably reflects the promotion of more useful compensatory strategies in CS + RT than RT, as opposed to more true “recovery,” given that movement abnormalities recovered similarly in these groups. However, it is also possible for CS + RT to partially normalize movements when it is combined with task-specific training. 3
Another novel finding is that the effects of CS + RT vary with its timing after the injury. Because CS can facilitate skill acquisition even in healthy humans, 55 we predicted that CS might vary in potency but would nevertheless facilitate RT efficacy at delayed time points. Instead, we found no further improvements as a result of CS + RT versus RT alone. The sensitivity in detecting these effects may have been limited by the magnitude of RT-only effects and the minor degree of spontaneous recovery. Thus, it is possible that delayed CS + RT would be more effective in animals with more severe persisting impairments. However, delayed CS + RT also failed to increase dendritic densities in peri-infarct cortex compared with RT, in contrast to early CS + RT effects, 4 suggesting that the delay diminished its capacity to facilitate RT-driven plasticity.
We did find that the delayed onset of RT was very effective in improving reaching performance, and this was linked with greater densities of dendrites in peri-infarct MC. Studies 1 and 2 were not intended to be directly compared, but the pattern of results across them do not strongly suggest a major difference in RT efficacy with the delay. Previous studies indicate that RT is particularly potent when initiated early (<1 week) after stroke.56,57 The present results do not contradict this idea, because even our earlier treatment onset (2 weeks) was delayed compared with these prior studies.
The mechanisms of the functional improvements resulting from CS + RT are likely to involve its plasticity-promoting effects.3-6 Though it has yet to be demonstrated in our model, these effects may depend on upregulation of neurotropic factors. Direct current stimulation in rodent tissue slices is NMDA (N-methyl-
In conclusion, these findings support the usefulness of CS in improving both the short-term and enduring benefits of RT. They also suggest that the influence of CS on RT efficacy can diminish with time after stroke. Forced use of the paretic forelimb did not further improve early CS + RT effects, but it remains possible that an alternative combination of forced use with CS could do so. The present findings also support the continued efficacy of RT in enhancing functional outcome and promoting cortical plasticity in the chronic period after ischemic stroke. One timing issue that was not addressed is that of age. RT is effective after similar infarcts even in much older animals, 60 but the generalization of CS + RT effects after older-onset stroke remains to be tested.
Footnotes
Acknowledgements
We thank Dr R. J. Nudo for advice on the study designs.
Authors’ Note
AJO and DLA contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by NIH/NINDS R21NS063332, U54NS048126, and NS056839.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
