Abstract
Background. Muscle vibration modifies corticomotor excitability in healthy subjects and reduces muscle tonus in stroke patients. Objective. This study examined whether repeated muscle vibration (rMV) applied over the flexor carpi radialis (FCR) and biceps brachii (BB) can induce long-lasting changes, using transcranial magnetic stimulation (TMS), in patients with chronic stroke. Methods. Thirty hemiparetic patients who offered at least minimal wrist and elbow isometric voluntary contractions were randomly assigned to either an experimental group, which received rMV in addition to physiotherapy (rMV + PT), or a control group that underwent PT alone. The following parameters of the FCR, BB, and extensor digitorum communis (EDC) were measured through TMS before, and 1 hour, 1 week, and 2 weeks after the end of intervention: resting motor threshold (RMT), map area, map volume, short-interval intracortical inhibition (SICI), and intracortical facilitation (ICF). Muscle tonus and motor function were assessed on the same day as TMS. Results. Pre–post analysis revealed a reduction in RMT and an increase in motor map areas occurred in the vibrated muscles only in the rMV + PT group, with an increase in map volumes of all muscles. Moreover, SICI increased in the flexors and decreased in the extensor. These neurophysiological changes lasted for at least 2 weeks after the end of rMV + PT and paralleled the reduction in spasticity and increase in motor function. A significant correlation was found between the degree of spasticity and the amount of intracortical inhibition. Conclusion. rMV with PT may be used as a nonpharmacological intervention in the neurorehabilitation of mild to moderate hemiparesis.
Introduction
Functional recovery following stroke is usually incomplete and most stroke survivors experience long-term disabilities. 1 Although poststroke motor functional recovery is a complex process, one of the most effective modulators of cortical structure and function is repeated sensory input. 2,3 The direct manipulation of sensory experience can modify brain plasticity and enhance the effects of motor training in chronic stroke patients. 4,5
Muscle vibration is a strong proprioceptive stimulus, which, at low amplitudes, preferentially produces Ia afferent input and reaches both the SI and M1 directly. 6-8 The specific pattern of direct connections linking SI and M1 cortices 9,10 may provide the anatomical substrate for the role played by muscle vibration in reorganizing the motor and somatosensory cortices. Long-term potentiation 11 from changes in the strength of the aforementioned corticocortical connections, ascribable to a Hebbian-like mechanism of synaptic plasticity, might be neurophysiological correlate underlying vibration-induced cortical reorganization. Numerous transcranial magnetic stimulation (TMS) studies performed in healthy subjects have shown that low-amplitude muscle vibration induces different changes in the corticomotor excitability of the vibrated muscle if compared with the nonvibrated muscle. 12-21 Recently, we demonstrated that a repeated muscle vibration (rMV) intervention, applied during a voluntary contraction of the target muscle (ie, flexor carpi radialis or FCR), induced a long-lasting increase in intracortical inhibition of the FCR and an inverse pattern in its functional antagonist. 22 These findings raised the hypothesis that muscle vibration might, by eliciting motor cortical reorganization, influence functional recovery in stroke patients. This hypothesis is supported by the results of a recent study in which the application of vibratory stimuli to the spastic muscles of poststroke patients resulted in a significant and persistent (up to 30 minutes) reduction in muscle tonus. 23 The aim of the present study was to use TMS measurements to test rMV-induced modifications in a population of chronic stroke patients to gain an insight into the possible neurophysiological mechanisms underlying these effects.
Materials and Methods
Patients
A total of 30 patients with hemiparetic stroke (13 women and 17 men; mean age, 65.0 ± 9.4 years) were enrolled in the study. The clinical characteristics of the patients are summarized in Table 1. All were right-handed. 24 Inclusion criteria were: (a) stroke experienced more than 12 months prior to enrollment; (b) single lesion detected by brain computed tomography (CT) or magnetic resonance imaging (MRI); (c) motor deficit ranging from 2 to 4 on the Medical Research Council Scale (MRC); (d) presence of spasticity, ranging from 1 to 3 on the Modified Ashworth Scale (MAS); (e) ability to perform at least a minimal wrist and elbow isometric voluntary contraction; (f) age >18 and <80 years; and (g) absence of cognitive or severe language impairment by standard neuropsychological examinations. Exclusion criteria were: other neurological disorders, central nervous system (CNS)-active drug intake or botulin toxin treatment in the previous 12 months, epilepsy, metal implants, pacemaker, peripheral neuropathies, systemic vasculopathies, and presence of cognitive deficits. The local ethics committee approved the experimental procedure in accordance with the Declaration of Helsinki; informed consent was obtained from all of the participants.
Stroke Patient Characteristics
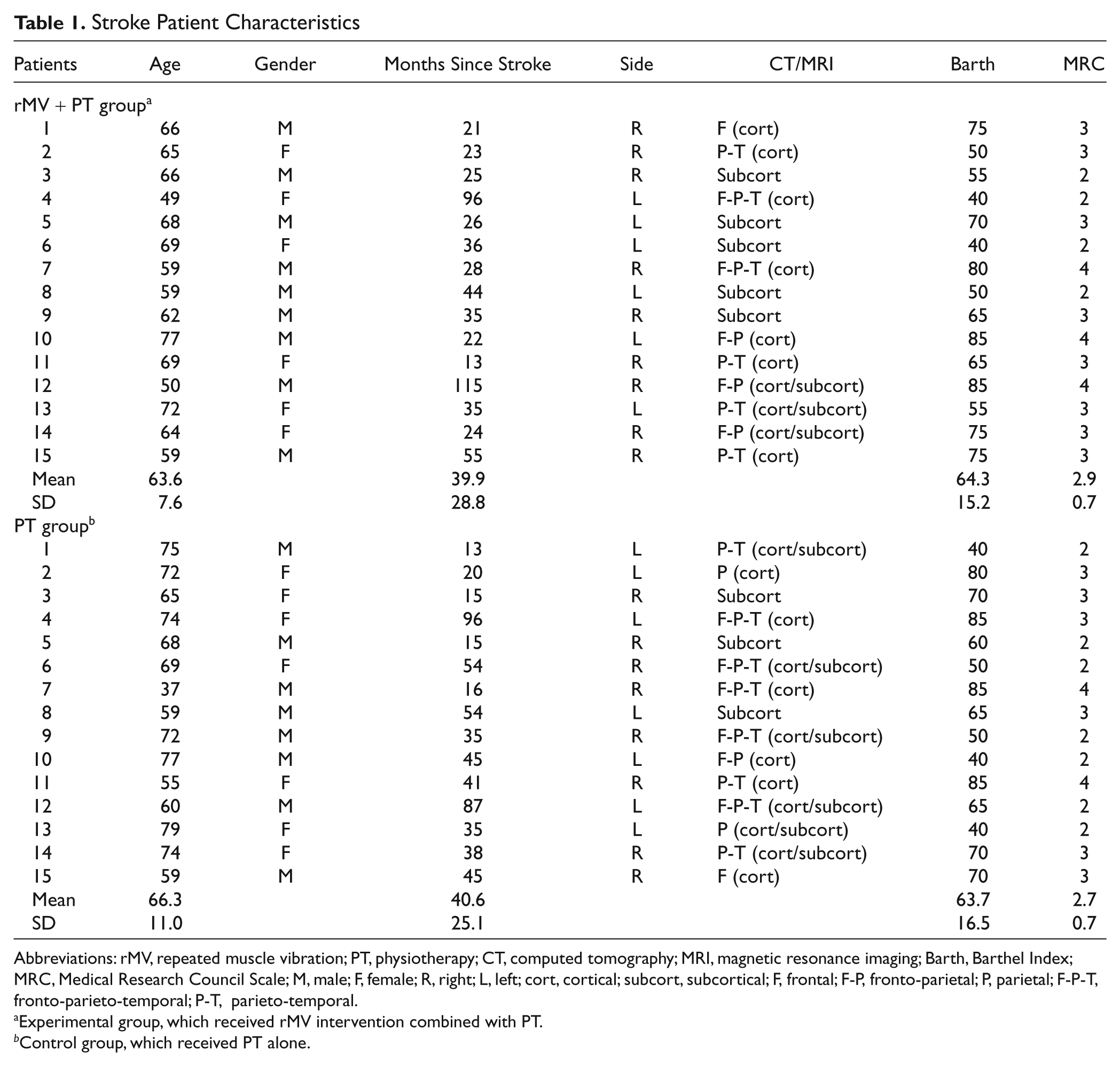
Abbreviations: rMV, repeated muscle vibration; PT, physiotherapy; CT, computed tomography; MRI, magnetic resonance imaging; Barth, Barthel Index; MRC, Medical Research Council Scale; M, male; F, female; R, right; L, left; cort, cortical; subcort, subcortical; F, frontal; F-P, fronto-parietal; P, parietal; F-P-T, fronto-parieto-temporal; P-T, parieto-temporal.
Experimental group, which received rMV intervention combined with PT.
Control group, which received PT alone.
Procedures and Interventions
Electromyography and TMS setup
Surface electromyography (EMG) was recorded from the FCR, biceps brachii (BB), and extensor digitorum communis (EDC) muscles with silver–silver chloride electrodes taped in a belly-tendon montage. EMG signals were amplified with gain set at 3000 (Digitimer D360 amplifier; Digitimer Ltd, Herts, UK), band-pass filtered (30 Hz to 2 kHz), then recorded by a computer using SIGNAL software (Cambridge Electronic Design, Cambridge, UK) with a sampling rate of 5 kHz per channel. An audio-feedback system ensured that total relaxation was maintained; any trials with voluntary EMG activity were excluded from the analysis.
TMS was performed using 2 Magstim 200 stimulators (Magstim, Dyfed, UK) connected to a figure-of-8-shaped coil (7 cm internal diameter). An adherent, inelastic cap was placed over the participant’s head, and the reference to an anatomical landmark (intersection of the interaural line and the nasion–inion connection, Cz, in the 10-20 International System) was taken. The coil was held with the handle pointing backward and sideways, approximately 45° to the midline, to evoke anteriorly directed current in the brain. The optimal position (hot spot) for eliciting motor-evoked potentials (MEPs) from the FCR was identified in each individual; the motor threshold at rest (RMT) was then assessed in a step-wise fashion and defined as the lowest stimulation intensity required to evoke MEPs larger than 50 µV in at least 50% of the trials. 25 To map out the muscle representation, a 49-position grid (10.5 × 10.5 cm) was fixed on the subject’s head and centered on the hot spot (Figure 1). For each scalp position, we recorded the muscular responses evoked by 5 stimulations at 120% of the RMT. The mean peak-to-peak MEP amplitude recorded from each excitable scalp site was calculated, as were the area (number of scalp positions from which the MEPs were elicited) and volume (the sum of the averaged MEP amplitudes for each excitable scalp site) for all of the cortical maps. A site was defined as excitable when TMS elicited at least 2 reproducible MEPs with an amplitude of at least 50 µV. Once mapping of the FCR muscle representation was completed, hot spot, RMT, and motor map representations were also identified for the BB and EDC muscles with the same experimental procedures as those used for the FCR. The center of gravity (CoG) was calculated for each muscle map according to Wassermann et al 26 : The lateral coordinate (x-coordinate) of the CoG was computed by multiplying the lateral coordinate at each position by its amplitude-weight and summing over all positions. The anteroposterior coordinate (y-coordinate) was computed using the same method. Intracortical excitability was investigated using the paired-pulse protocol 27 with a suprathreshold test stimulus preceded by a subthreshold conditioning magnetic stimulus. Two different interstimulus intervals (ISIs) were tested: 3-ms ISIs were measured to study short-interval intracortical inhibition (SICI), whereas 10-ms ISIs were measured to study intracortical facilitation (ICF). The intensity of the conditioning stimulus (CS) was set at 70% of the RMT. In each experimental session, the intensity of the test stimulus (TS) was adjusted to evoke an MEP amplitude of ~0.7 mV. SICI and ICF were investigated in blocks of trials consisting of randomly intermixed conditions (TS either alone or preceded by the CS at different ISIs), each presented 10 times. The time between 2 consecutive trials was always 5 ± 20% seconds.
Single- and double-pulse TMS measurements were performed across sessions using the same RMT values as those found at the preintervention time.
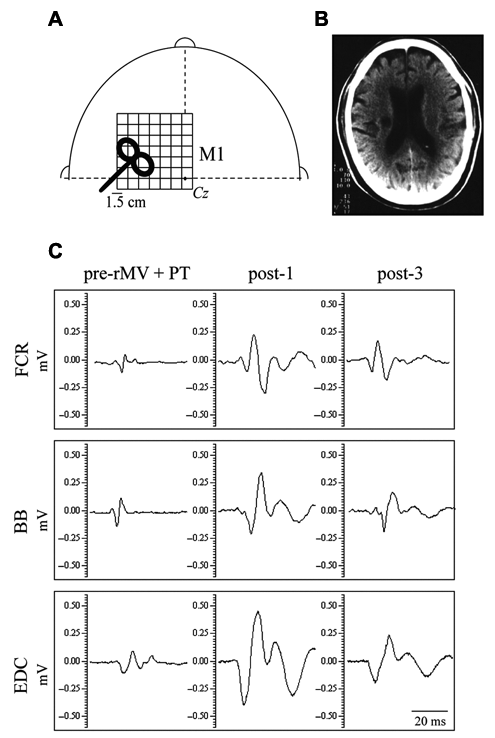
(A) Schematic head with a grid showing the stimulated scalp sites. Cz represents the intersection of the interaural line and the nasion–inion connection. (B) Magnetic resonance imaging of a stroke patient (no. 9 in Table 1) with an ischemic capsular lesion in the right hemisphere. (C) Original motor-evoked potentials obtained from the same patient acquired before the intervention (pre-rMV + PT), and 1 hour (post-1) and 2 weeks (post-3) after the end of the intervention. Given the similarity of the pattern displayed by the muscles between post-3 and post-2, the latter was not shown
Physiotherapy
All subjects underwent 3 sessions (60 minutes’ duration) of physiotherapy (PT) administered on 3 consecutive days, which included passive and active movements and mobilization, proprioceptive neuromuscular facilitation, and stretching of the affected upper limb. Before the start of the experimental phase, all of the physical therapists recruited for this study received specific instructions to ensure uniformity in the treatment procedures (ie, duration, frequency, and content of therapy). They were also instructed not to talk to the patients about the goal of the study.
Repeated Muscle Vibration
A subgroup of patients (n = 15) received the rMV intervention in addition to PT. Low-amplitude rMV (frequency 100 Hz; amplitude range 0.2-0.5 mm) was applied over the FCR and the BB by means of a specific commercial device (CroSystem, NEMOCO srl, Italy). The instrument consists of an electromechanical transducer, a specific mechanical support and an electronic control device. 22
The rMV intervention was applied for 3 consecutive days, 3 times a day, with each application lasting 10 minutes. During the rMV, subjects were required to make a voluntary isometric contraction of both flexor muscles.
Since the patients’ ability to execute a maximal voluntary contraction (MVC) varied, between 10% and 20% of the MVC was calculated for each of them. This range was shown on the computer screen as 2 lines and used as visual feedback to help the patients keep the contraction level constant during the intervention.
Experimental Design
Patients were randomly assigned to 1 of 2 groups using a computer-generated random numbers table: (a) an experimental group (n =15), which received rMV in addition to physiotherapy (rMV + PT), and (b) a control group (n =15), which received physiotherapy alone (PT). The experimental group received PT on the same day as rMV, approximately 30 minutes later.
All of the patients were neurophysiologically and clinically tested immediately before rMV + PT or PT alone (pre) and at the following time points after the last rMV + PT or PT alone: 1 hour (post-1), 1 week (post-2), and 2 weeks (post-3). Spasticity was assessed by means of the MAS, whereas motor function was assessed by the Motricity Index for the upper limbs (MI-ul), 28 and the Wolf Motor Function Test–Functional Ability (WMFT-FA). 29
Both the clinicians and researchers were blinded to whether the patient was in the rMV + PT or in the PT group. Only the neurophysiological data relative to the affected hemisphere (AH) were analyzed and are presented in this study.
Data Analysis
For RMT, map areas and volumes, the effects of the rMV were quantified by measuring the peak-to-peak MEP amplitudes to single pulse TMS. To rule out any systematic changes in background activity that might confound the interpretation of the MEP data, we calculated prestimulation EMG activity as the area under the curve (mVms) of the rectified EMG during the 100 ms preceding the TMS stimuli. When determining SICI and ICF the mean MEP amplitude was calculated for each state, and this MEP was then expressed as a percentage of that resulting from a single pulse (conditioned/unconditioned).
We applied analysis of variance (ANOVA) for repeated measures as the main statistical procedure, in which group (rMV + PT vs PT) was considered as between-subject factor, whereas muscle and time were considered as within-subject factors. Moreover, we performed other ANOVAs to assess whether rMV-induced changes varied depending on the stroke lesion location (factor group: “right” vs “left” and “cortical” vs “subcortical”). Finally, separate ANOVAs for each muscle were performed to assess any significant differences between the CoG maps.
A nonparametric analysis (Wilcoxon test) was used to analyze the MAS scores. Moreover, to facilitate the data analysis of the MAS scores, a value of 1.5 was assigned to the score of 1+.
We investigated the relationship between parameters of corticospinal excitability and intracortical inhibition, and clinical outcomes, by correlating RMT, area, volume, and SICI with the MAS, MI-ul, and WMFT-FA clinical scores obtained at each time point. To assess any association, Spearman’s rank order correlation test was used.
For all of the ANOVAs undertaken, post hoc comparisons were performed when the interaction was statistically significant. The assumption of sphericity, which was checked by means of Mauchly’s test, was not significant; no correction was applied to the degrees of freedom. Student’s t test was used when 2 means were compared.
The P value level of significance throughout the statistical analysis was set at .05, considering Bonferroni correction.
Results
Persisting Effects Induced by rMV on Corticospinal Excitability and Intracortical Inhibition
The amount of background EMG activity at baseline was 2.18 ± 0.1 mVms. The ANOVA performed (group × muscle × time) to detect differences in the level of background EMG activity showed that, following removal of the invalid trials (~10% of the total number), the significance was not reached neither for the main factors nor for their interactions (all Ps >.05).
The ANOVA performed on the RMT of the FCR, BB, and EDC muscles revealed that the main factors group, F(1, 28) = 10.04, P < .05; muscle, F(2, 56) = 8.86, P < .001; and time, F(3, 84) = 19.24, P < .001; as well as their interactions group × muscle, F(2, 56) = 8.86, P < .001 and group × time, F(3, 84) = 49.74, P < .001, were significant. Post hoc comparisons revealed that the RMT of the FCR and BB muscles was significantly reduced in the rMV + PT group at all of the time points postintervention, when compared with the values of preintervention (all Ps <.05), whereas the RMT of the EDC was slightly, but not significantly, reduced (Figure 2A). The ANOVA performed on map areas revealed that the main factors group, F(1, 28) = 38.83, P < .001; muscle, F(2, 56) = 13.47, P < .001; and time, F(3, 84) = 18.40, P < .001; as well as their interactions group × muscle, F(2, 56) = 12.54, P < .001; group × time, F(3, 84) = 10.61, P < .001; muscle × time, F(6, 168) = 15.13, P < .001; and group muscle × time, F(6, 168) = 14.51, P < .001, were all significant. Post hoc comparisons showed that map areas significantly increased in the FCR and BB muscles in the rMV + PT group at all time points postintervention, when compared with the preintervention values (all Ps <.05; Figure 2B). A slight, though not significant, increase was observed in the map areas of the EDC muscle. The ANOVA performed on map volumes showed that the main factors group, F(1, 28) = 526.45, P < .001; muscle, F(2, 56) = 5.15, P < .05]; and time, F(3, 84) = 103.42, P < .001; as well as their interactions group × muscle, F(2, 56) = 6.02, P < .05; group × time, F(3, 84) = 101.29, P < .001; muscle × time, F(6, 168) = 5.54, P < .001; and group muscle × time, F(6, 168) = 5.73, P < .001, were all significant. Post hoc comparisons revealed that map volumes significantly increased for all muscles in the rMV + PT group at all time points postintervention, when compared with preintervention (all Ps <.001), with the exception of the BB map volume, which did not reach significance at post-3 (Figure 2C). The rMV-induced changes in map areas and volumes observed in the rMV + PT group are visible in the 2-dimensional (2D) maps shown in Figure 3. No modifications in the RMT, map areas, and volumes were observed in the PT group (Figure 2). Moreover, in the rMV + PT group we found a backward shift in the y (anteroposterior) coordinate of the CoG in all the muscle maps (as can be seen in the 2D maps shown in Figure 3), though this change did not reach significance.
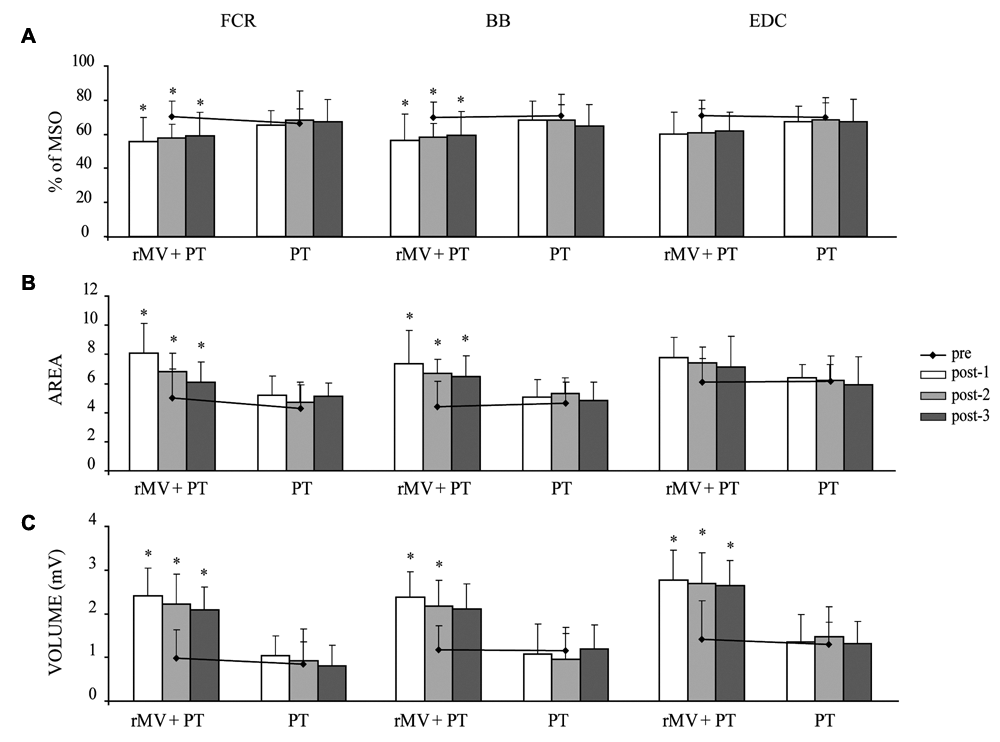
(A) RMT, (B) map areas, and (C) map volumes of FCR, BB, and EDC obtained from the rMV + PT and PT groups at all the time points
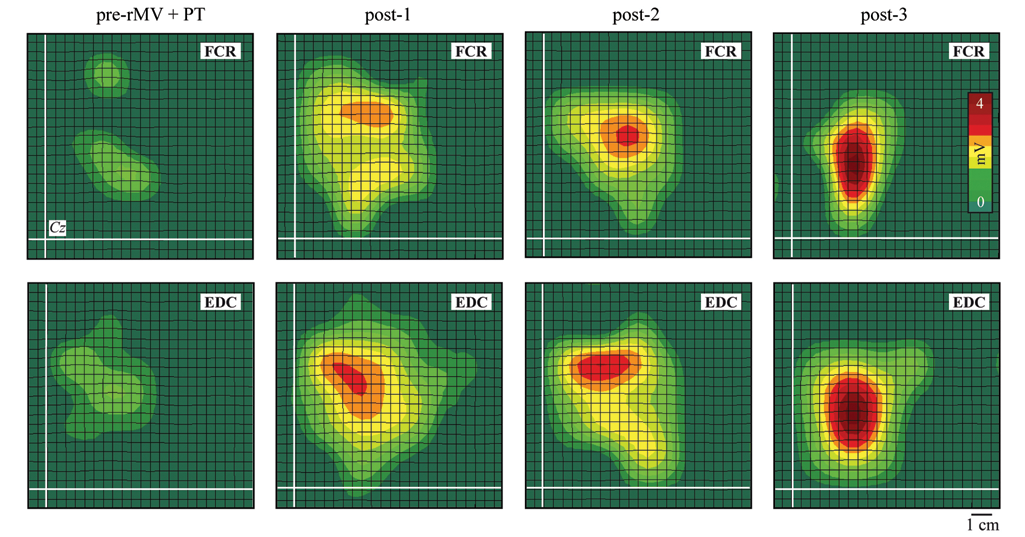
Two-dimensional (2D) maps of FCR and EDC (averaged data of 9 patients with right-hemisphere lesion)
The 3-way ANOVA (group × muscle × time) performed to assess differences in SICI (3-ms ISI) of the FCR, BB, and EDC muscles after rMV showed that the main factors group, F(1, 28) = 7.20, P < .05; muscle, F(2, 56) = 40.56, P < .001; and time, F(3, 84) = 8.17, P < .001; as well as the interaction group × ISI × time, F(6, 168) = 4.68, P < .05, were significant. Post hoc comparisons showed that SICI for the FCR and BB muscles significantly increased in the rMV + PT group at all time points postintervention, when compared with the values of preintervention (all Ps <.001; Figures 4A and 4B). Conversely, SICI for the EDC significantly decreased in the rMV + PT group at all the time points post (P < .05; Figure 4C). The ANOVA performed to assess differences in ICF (10-ms ISI) of all muscles failed to detect significant changes in either group at any of the time points.
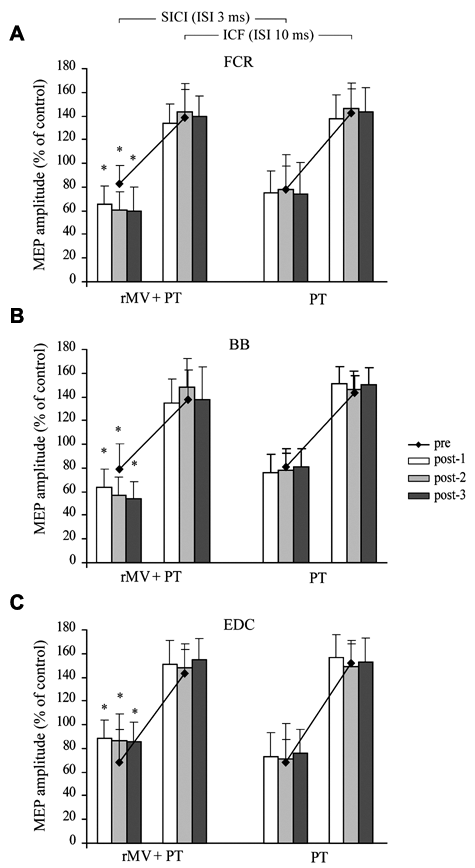
SICI (ISI 3 ms) and ICF (ISI 10 ms) for (A) FCR, (B), BB, and (C) EDC, obtained from the rMV + PT and PT groups at all time points.
Persisting Effects Induced by rMV on Muscle Tonus and Motor Function
The analyses revealed that the MAS scores obtained at wrist and elbow joints significantly decreased (Friedman ANOVA χ2 = 94.58, P < .001) in the rMV + PT group at all the time points post, when compared with the pre values (P < .05, at Wilcoxon test; Figure 5A). Moreover, the ANOVAs revealed that the MI-ul and WMFT-FA scores significantly increased in the rMV + PT at all time points post, when compared with pre (all Ps <.05; Figures 5B and 5C). Neither a reduction in muscle tonus nor an increase in motor function was observed in the PT group (Figure 5).
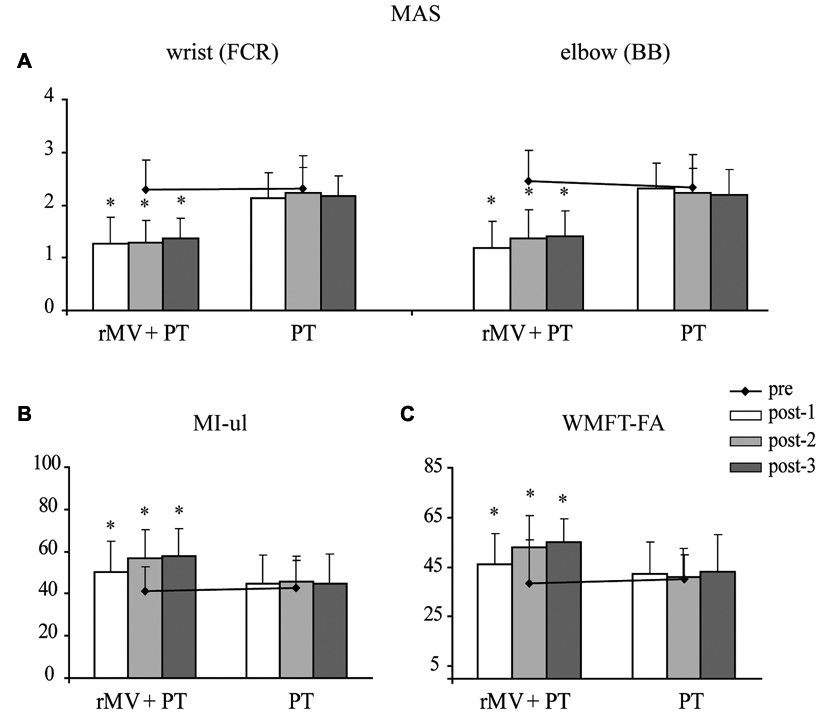
(A) MAS scores for FCR and BB; (B) MI-ul scores; and (C) WMFT-FA scores in the rMV + PT and PT groups at all time points
rMV-Induced Effects in Relation to the Stroke Lesion Location: Neurophysiological and Clinical Data
ANOVAs did not reveal any difference in the rMV-induced neurophysiological and clinical changes in respect to the side affected by stroke (ie, right vs left). However, ANOVAs did detect significant differences when the patients in the rMV + PT group were divided according to whether the stroke was “cortical” or “subcortical,” after patients with mixed lesions (ie, cortical plus subcortical) had been excluded from the analysis. Both the neurophysiological and clinical results are summarized in Table 2.
TMS and Clinical Outcomes in (A) Cortical and (B) Subcortical Patients in rMV + PT Group
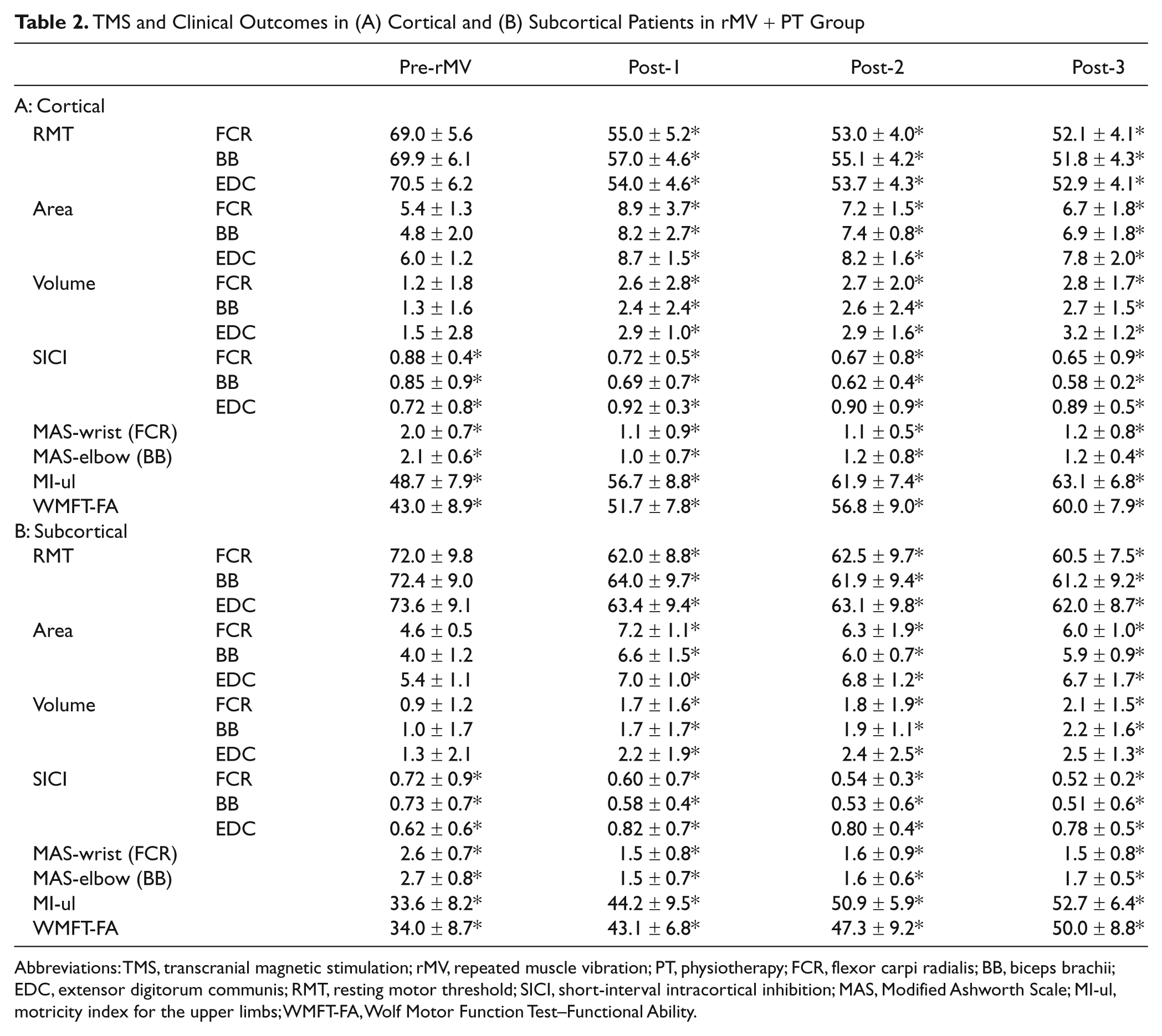
Abbreviations: TMS, transcranial magnetic stimulation; rMV, repeated muscle vibration; PT, physiotherapy; FCR, flexor carpi radialis; BB, biceps brachii; EDC, extensor digitorum communis; RMT, resting motor threshold; SICI, short-interval intracortical inhibition; MAS, Modified Ashworth Scale; MI-ul, motricity index for the upper limbs; WMFT-FA, Wolf Motor Function Test–Functional Ability.
Relationships Between TMS Outcomes and Clinical Findings
The results of the correlation analysis are shown in Table 3. The amount of intracortical inhibition (expressed in terms of percentage of the conditioned/unconditioned MEP amplitude ratio) of the flexor muscles correlated with the degree of spasticity (MAS scores), showing that a higher level of intracortical inhibition was associated with a lower degree of spasticity. In addition, the amount of intracortical inhibition of the same muscles correlated with functional improvement (MI-ul and WMFT-FA scores; all Ps <.05), revealing that a higher level of intracortical inhibition was associated to higher functional scores.
Furthermore, all of the TMS outcomes reflecting corticospinal excitability (eg, motor threshold and map volume) significantly correlated with the scores obtained on the MI-ul and WMFT-FA scales (all Ps <.05).
Correlation Between TMS and Clinical Outcomes

Abbreviations: FCR, flexor carpi radialis; BB, biceps brachii; EDC, extensor digitorum communis; RMT, resting motor threshold; SICI, short-interval intracortical inhibition; MAS, Modified Ashworth Scale; MI-ul, motricity index for the upper limbs; WMFT-FA, Wolf Motor Function Test–Functional Ability.
Discussion
Our data demonstrated that the rMV intervention, combined with physiotherapy, induces long-lasting changes in a cohort of poststroke chronic patients. These changes consisted in a reduction in muscle tonus and an increase in motor function coupled with a reduction in motor thresholds and an increase in motor map size of both flexors; an increase in motor map volumes of all muscles; an increase in SICI for the flexor muscles, as well as a reduction in SICI for the extensor muscle. This is consistent with previous TMS studies that demonstrated a decrease in the motor threshold and an increase in MEP amplitudes and map sizes in response to rehabilitation, 30 and following therapeutic interventions in chronic stroke patients. 31-35
Since the motor threshold and MEP amplitudes are influenced by different mechanisms, 36,37 we may speculate that the rMV-induced modifications are very likely due to at least 2 forms of plasticity: (a) a form of nonsynaptic plasticity that induces changes in the intrinsic properties of neural membranes, which may explain the lowering of the motor threshold, and (b) a Hebbian-like mechanism of synaptic plasticity, which may account for the functional restoration of inactivated, though preserved, motor pathways and/or rearrangements of motor cortical maps. Changes in the strength of the spared sensory-motor corticocortical connections 9,10 might provide the anatomical substrate for the rMV-driven widespread cortical reactivation. Indeed, our protocol, which combines the long exposure time of vibratory stimulation with the maintenance of a contraction during the intervention, shares features with the associative long-term potentiation of synaptic plasticity. We also found that rMV induces a significant, long-lasting increase in the SICI of the target muscles (FCR and BB) and a decrease in the SICI of its functional antagonist (EDC). The intracortical inhibitory system in stroke patients has been shown to be suppressed. 38 This phenomenon may represent an adaptive response that promotes access to the residual motor output system to facilitate structural changes and ultimately to promote motor recovery. 39,40 However, this disinhibition may be required less in poststroke chronic stages of motor recovery, when an alternative motor network becomes better established. 41 The impairment of inhibitory properties within the motor cortex might be one of the mechanisms through which spasticity may be evoked and maintained. 35,42 Furthermore, studies have demonstrated the role of intracortical inhibitory processes in the prevention of co-activation of separate motor cortical points in animals 43 and of unwanted muscle activation in healthy humans. 44 In view of these previous findings, we hypothesize that the rMV-induced parallel upregulation and downregulation of GABAergic circuits in cortical areas controlling the pair of antagonists might underlie the reduction in spasticity and prevention of unwanted agonist/antagonist co-contraction.
We believe that the aforementioned rMV-induced modifications may reflect 2 different needs in the surviving motor system: (a) the increase in corticospinal excitability may be necessary to generate output to the spinal motoneurons as efficiently as possible, and (b) the upregulation in the intracortical inhibitory circuits may help remodel the remaining connections in the motor network and thereby maximize motor control. We consider these neurophysiological changes as potential functional substrates for the reduction in spasticity and improvement in motor function.
We observed strong correlations between corticospinal and intracortical excitability and the level of motor function and muscle tonus. The correlation between the decrease in motor threshold and increase in map volume and the increase in motor function is not surprising since net excitation results in corticomotor outflow, which is the critical requisite for motor unit recruitment. 45 Moreover, the correlation between the increase in intracortical inhibition of the flexor muscles and the reduction in spasticity and increase in motor function is in keeping with the hypotheses made by Liepert et al. 35,42 However, since the correlation analysis is affected by certain biases (eg, the clinical scales we used measure muscle tonus and motor function broadly, whereas TMS values are derived from specific muscles), these intriguing findings need to be interpreted with some caution.
The rMV-induced effects varied depending on whether the stroke was cortical or subcortical. However, several neurophysiological and clinical differences were found in our cohort of poststroke chronic patients at the baseline. Indeed, patients with a cortical lesion had a lower motor threshold, larger map sizes and volumes, stronger disinhibition, less severe spasticity, and better functional outcome than patients with a subcortical lesion. Interestingly, this pattern persisted after the rMV intervention. These results are in agreement with previous TMS studies, which demonstrated that the site of the lesion determines the changes in motor cortex excitability. 46-49 Although the majority of these studies were performed on stroke patients in the acute and/or subacute phase, other studies have demonstrated that some of these location-related changes persist in chronic stroke patients. 35,50 In particular, the reduction in intracortical inhibition is more pronounced in cortical than in subcortical-stroke patients. 35 This finding is not surprising since SICI is a GABAergically mediated intracortical process, 51,52 and subcortical lesions affecting the corticospinal tract do not consequently affect SICI to any great extent. Moreover, these observations are in keeping with what is currently known about redundant cortical motor representation relative to the convergence of corticofugal motor efferents as they approach the internal capsule. 53 In this regard, we suggest that the extent of the rMV-induced effects might depend on the inhibitory/excitatory state within the motor system, which reflects the site of lesion.
Limitations of the Study
The present study has some limitations, including a possible bias in our patient selection: (a) all of the patients had regained some ability to perform wrist and elbow flexion movements with the affected arm, and (b) MEPs could be elicited from the affected side. We cannot, therefore, exclude that the rMV-induced changes not only occur when M1 is preserved structurally but also when it is also capable of enhanced workload, that is, it is not completely disconnected.
The short follow-up (2 weeks) is a limitation of our study and some of the changes in the TMS values may be attributable to changes in spinal excitability. Furthermore, although recent studies on humans have highlighted the role played by somatosensory afferents combined with intrinsic motor activity in inducing plasticity in M1, 11,37 we cannot exclude that the benefits we observed could be induced by repeated periods of voluntary contraction alone, since we did not perform a specific control experiment.
In addition, single- and double-pulse TMS measurements were performed across sessions using the same RMT values as those found at the preintervention time. Although rMV induced an effective reduction of RMT, we cannot rule out that part of the changes in map area and volume, as well as those in SICI, could be because of the higher stimulation intensity. A stimulus–response curve might have yielded more information on the corticomotor excitability changes that we observed to discriminate the rMV-induced effects on threshold from those on cortical reorganization. Finally, although the choice of applying rMV before PT has allowed us to exploit the antispastic effects of the vibratory stimulation during the PT sessions, we cannot exclude that a different temporal relationship between the two can be equally or more effective in the rehabilitation of motor disorders. Such hypothesis may represent and intriguing topic for further investigation.
Conclusions
We show that muscle vibration administered in addition to conventional physiotherapy can reduce certain abnormalities of corticospinal excitability and intracortical inhibitory systems in poststroke chronic patients. We believe that rMV may be used as a complementary nonpharmacological therapy to promote neural plasticity and motor recovery, even a long time after stroke.
Footnotes
Acknowledgements
We are grateful to Professor Alexandra Battaglia-Mayer and Dr Andrea Cherubini for their helpful statistical suggestions.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article:
This work was supported by Regione Lazio REG/09 and Baroni Foundation grants.
