Abstract
Introduction:
There are vast differences in the size, scope, and needs of institutions that conduct research involving biohazardous materials, thus resulting in vast differences among Institutional Biosafety Committees (IBCs) and biosafety programs.
Methods:
A benchmarking survey of IBC and biosafety programs was conducted in an effort to identify common practices in the field and compare this information with that of the other institutional bioethics committees, namely, Institutional Animal Care and Use Committees (IACUCs) and Institutional Review Boards (IRBs).
Objectives:
The primary objectives of the survey were to assess the organizational structure of IBC and biosafety programs, determine the scope of IBC review, and compare the size of IBC and biosafety programs with that of IACUCs and IRBs.
Results:
The survey results showed that IBCs most commonly reside under the same administrative unit as the IACUC and IRB, while the majority of institutions’ biosafety officers report to a different unit. The majority of respondents indicated their IBC reviews research utilizing biological hazards beyond what is required by the National Institutes of Health Guidelines. The survey data suggest that IBCs have fewer support staff than the other bioethics committees; 57% of institutions report one or more full-time employee (FTE) dedicated to support the IBC, compared to 86%, 85%, and 83% of institutions that reported one or more FTE to support the IACUC, the IRB, and the biosafety program, respectively.
Conclusion:
Data from the survey identified common practices among IBCs and provides institutions a tool to compare their program with others.
Keywords
Introduction
In 1976, the National Institutes of Health (NIH) published the first version of the
The flexibility of the NIH Guidelines allows institutions to develop administrative practices that meet their own programmatic needs, resulting in a wide array of options for implementation of recombinant or synthetic nucleic acid research oversight. This can also be challenging since there is no “one size fits all” IBC or biosafety program; institutions must determine the most effective way to carry out the requirements of the NIH Guidelines and often borrow practices and procedures from their animal care and use committee (IACUC) or Institutional Review Board (IRB). The number of IBCs has grown significantly since the inception of the program. At the onset of the NIH Guidelines in 1976, there were only 57 IBCs registered with NIH. 4 This number has steadily increased over time, 5 and today, there are nearly 1200 registered IBCs, 6 many with unique needs and programs. This further illustrates a need to collect, analyze, and report current practices toward building community around effective administration of IBC programs.
Benchmarking is a method of discovering common practices in a field and comparing an organization’s program to industry standards. The information gained from a benchmarking exercise can then be used to identify gaps in an organization’s processes or as a guide for future program development. We wanted to conduct a benchmarking survey of IBCs registered with the NIH Office of Science Policy (OSP) to determine if certain administrative practices rise to the top among the myriad of possibilities. The primary objectives of this survey included (a) assessing of the organizational structure of IBC and biosafety programs in terms of which administrative unit IBCs and biosafety officers report to; (b) determining the scope of IBC review in terms of what type of biohazardous materials IBCs are reviewing and how the review process is managed, and (c) comparing the size of IBC and biosafety programs based on the number of IBC protocols/registrations and staffing for IBC and biosafety programs. The purpose of the benchmarking survey was to determine if best practices could be identified among IBC and biosafety programs and compare this information with IACUCs and IRBs, with the intent of providing institutions a tool to compare the organizational structure, scope, and size of their own IBC and biosafety program to other programs.
Methods
Survey
The survey consisted of 24 questions (Supplemental Document 1) regarding the organizational structure of the institutional bioethics committees (IBC, IACUC, and IRB) and biosafety officers, the scope of IBC review, how IBCs conduct their reviews (i.e., principal investigator [PI]/program vs individual projects), what events trigger a laboratory audit, the number of protocols reviewed by each of the bioethics committees (IBC, IACUC, and IRB), and the number of full-time employees (FTE) dedicated to support each of the bioethics committees and biosafety program. The survey also asked demographic questions such as the type of institution (academic, government, hospital, private, or other) as well as total research expenditures (in 2016) for the institution.
Administration of the Survey
Contact information for IBCs registered with the NIH OSP was obtained through a Freedom of Information Act request to NIH OSP (August 2017). An invitation to participate was emailed to IBC contacts and posted on the ABSA International listserv, which included a link to the web-based survey (created in Google Forms). The survey was open for approximately 2.5 weeks, and email reminders were sent weekly to increase the response. Responses were kept confidential, and the survey results are reported only in aggregate, as the population of institutions that responded to the survey. Survey results were exported into Microsoft Excel, data were analyzed using Excel statistical functions, and summary statistics reported.
Results
Of the 1197 IBC contacts received from NIH OSP, 40 of the contact email addresses were invalid and the survey email was unable to be delivered. Upon closing the survey, 157 of 1157 completed or partially completed survey responses were received, resulting in a 13.6% response rate. An additional 6 surveys were submitted completely empty and were removed from the analysis. As shown in Table 1, the majority of respondents were from academic institutions. The size of the institutions that responded to the survey, based on the level of research expenditures (from 2016), ranged from very small (under $1 million) to very large (over $1 billion), with the majority (60%) of respondents being between $11 and $500 million (Table 2).
Type of Research Institution.
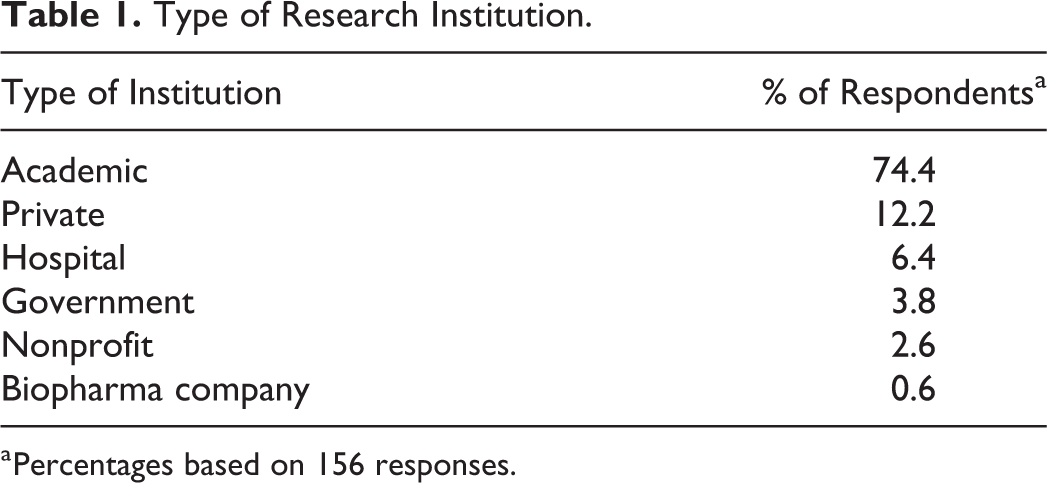
Percentages based on 156 responses.
Total Research Expenditures in 2016.
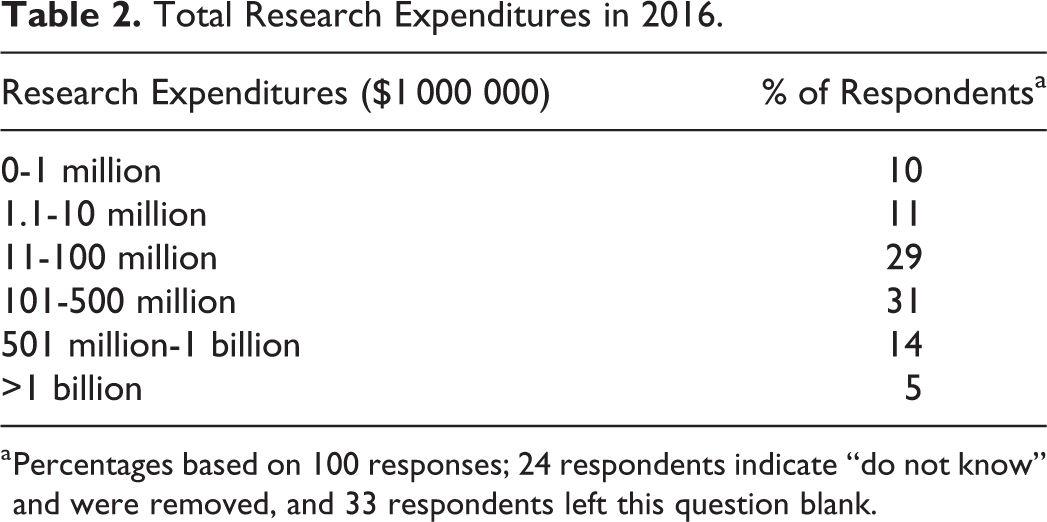
Percentages based on 100 responses; 24 respondents indicate “do not know” and were removed, and 33 respondents left this question blank.
Organizational Structure of IBCs and Biosafety Programs
As shown in Table 3, IBCs most frequently reported to a vice president (or vice chancellor) for research, whereas biosafety officers (BSOs) most commonly reported to environmental health and safety departments. IACUCs and IRBs shared a nearly identical reporting structure, with the majority reporting to a vice president/vice chancellor for research (VPR/VCR). When the reporting structure of the IBC, BSO, IACUC, and IRB were assessed in relation to each other at the same institution, we see that approximately half of respondents indicated that the IBC resides within the same administrative unit as the IACUC and/or IRB; 20.0% of respondents indicated that IBC, BSO, IACUC, and IRB all report to the same administrative unit; and 16.8% of respondents indicated that their IBC and BSOs report to the same administrative unit (Table 4). There were 58 different reporting combinations; however, the 3 most commonly reported combinations, as shown in Table 5, placed the IBC under the same administrative unit as the IACUC and/or IRB (either under VPR/VCR or research integrity/compliance) and the BSOs either in EHS or with the IBC in VPR/VCR.
IBC, BSO, IACUC, and IRB Reporting Structure.a
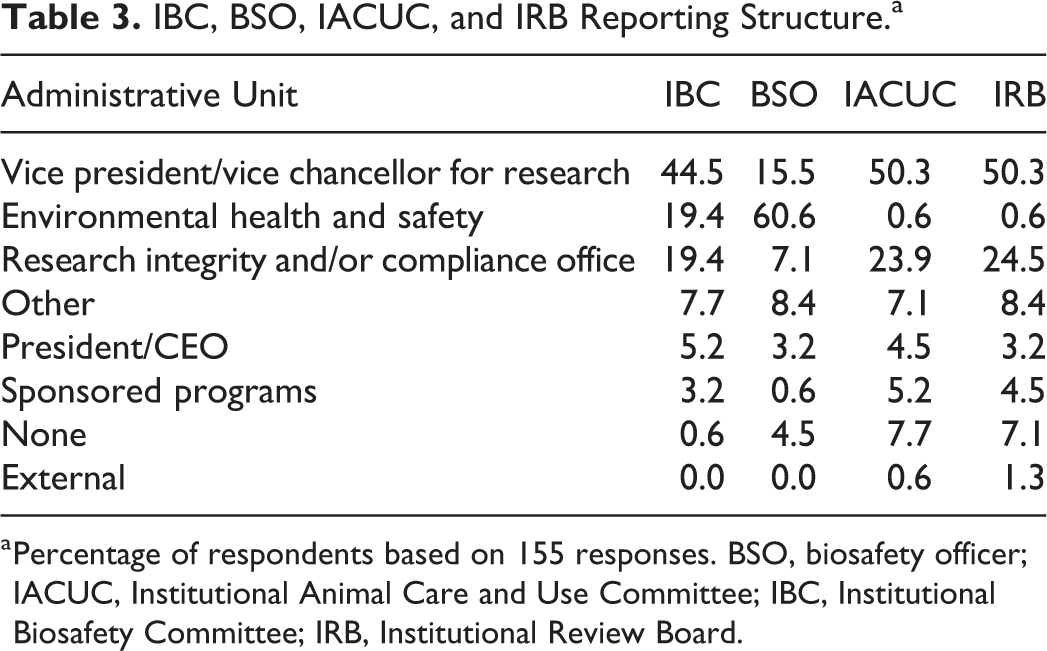
Percentage of respondents based on 155 responses. BSO, biosafety officer; IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee; IRB, Institutional Review Board.
IBC Reporting Structure in Relation to the BSO, IACUC, and/or IRB.

Abbreviations: BSO, biosafety officer; IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee; IRB, Institutional Review Board.
Three Most Common Reporting Structure Combinations.a
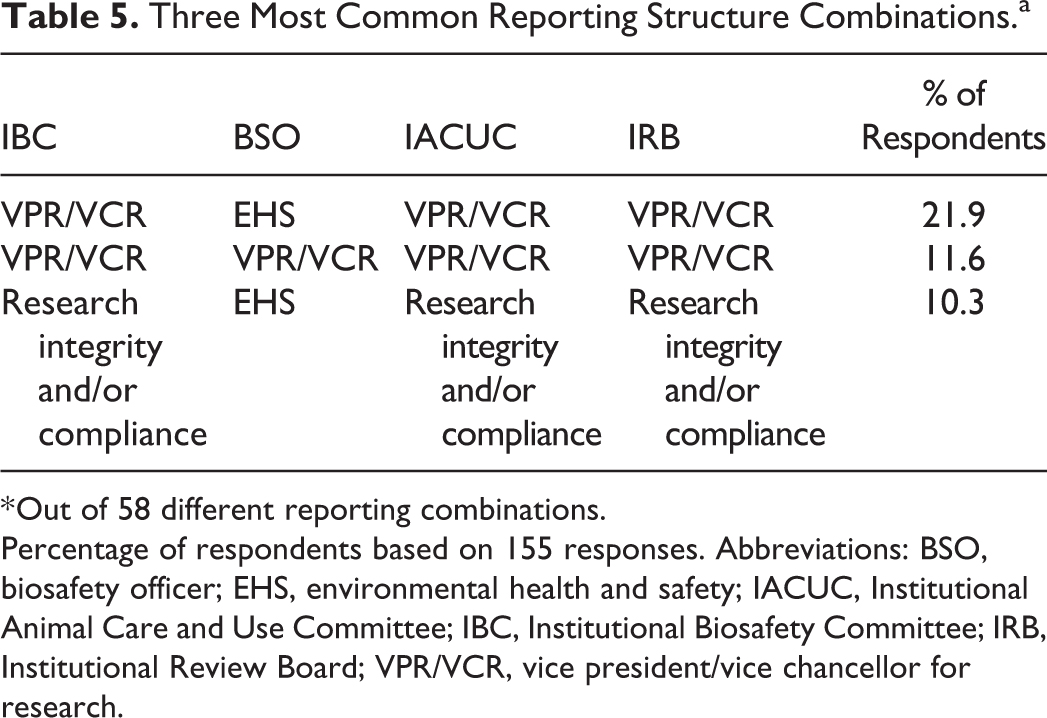
* Out of 58 different reporting combinations.
Percentage of respondents based on 155 responses. Abbreviations: BSO, biosafety officer; EHS, environmental health and safety; IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee; IRB, Institutional Review Board; VPR/VCR, vice president/vice chancellor for research.
When the organizational structure was analyzed based on institutional size (as determined by research expenditures), smaller institutions deviate from the aforementioned trend when it comes to IBC reporting structure. Specifically, in institutions that reported less than $10 million in research expenditures, the IBC most commonly reported to environmental health and safety (24%), followed by research integrity and/or compliance office (19%) or president/CEO (19%). Likewise, the reporting structure of biosafety officers at small institutions was evenly split among environmental health and safety (19%), research integrity and/or compliance office (19%), or “other” type of office (19%). IACUCs and IRBs share a similar reporting structure at the smaller institutions and most commonly reported to a research integrity and/or compliance office (both at 29%; Supplemental Spreadsheet S1).
The Scope of IBC Review
When asked about what IBCs are reviewing, 8% of respondents reported that their IBCs only reviewed research involving recombinant or synthetic nucleic acid molecules as directed by NIH Guidelines, while 90% of respondents indicated reviewing research with other biohazards in addition to recombinant or synthetic nucleic acid molecules (Figure 1). The additional biohazards reviewed by IBCs included (but were not limited to) human, animal, and plant pathogens; human samples/blood-borne pathogens; select agents; biological toxins; and nanoparticles (Figure 2). Additionally, the majority of respondents (75%) also indicated their IBCs require some type of registration or approval for exempt recombinant or synthetic nucleic acid molecules research (ie, research that is exempt from the NIH Guidelines). To determine the extent that IBC review overlaps with the other bioethics committees, the survey also asked how many IBC protocols require review by multiple committees. Eighty-four percent of respondents indicated having IBC protocols that require review by IACUC, 70% indicated IBC protocols requiring review by IRB, and 42% of respondents reported having protocols that require review from all three committees.
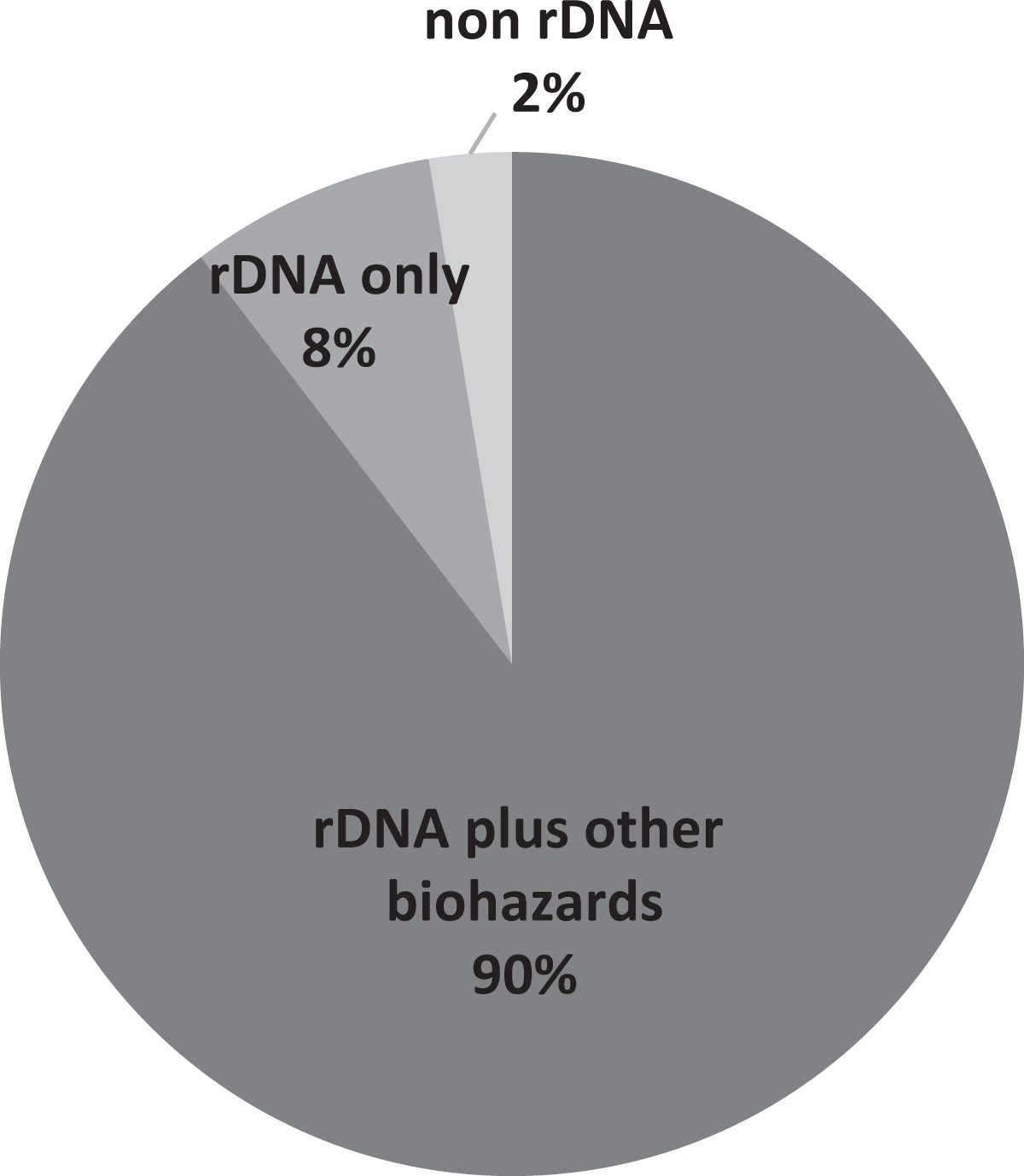
Types of research reviewed by IBCs. rDNA only (only rDNA research reviewed as articulated by the NIH Guidelines); non-rDNA (no rDNA research reviewed, only other biohazards); rDNA plus other biohazards (rDNA research reviewed as articulated by the NIH Guidelines and other biohazards).
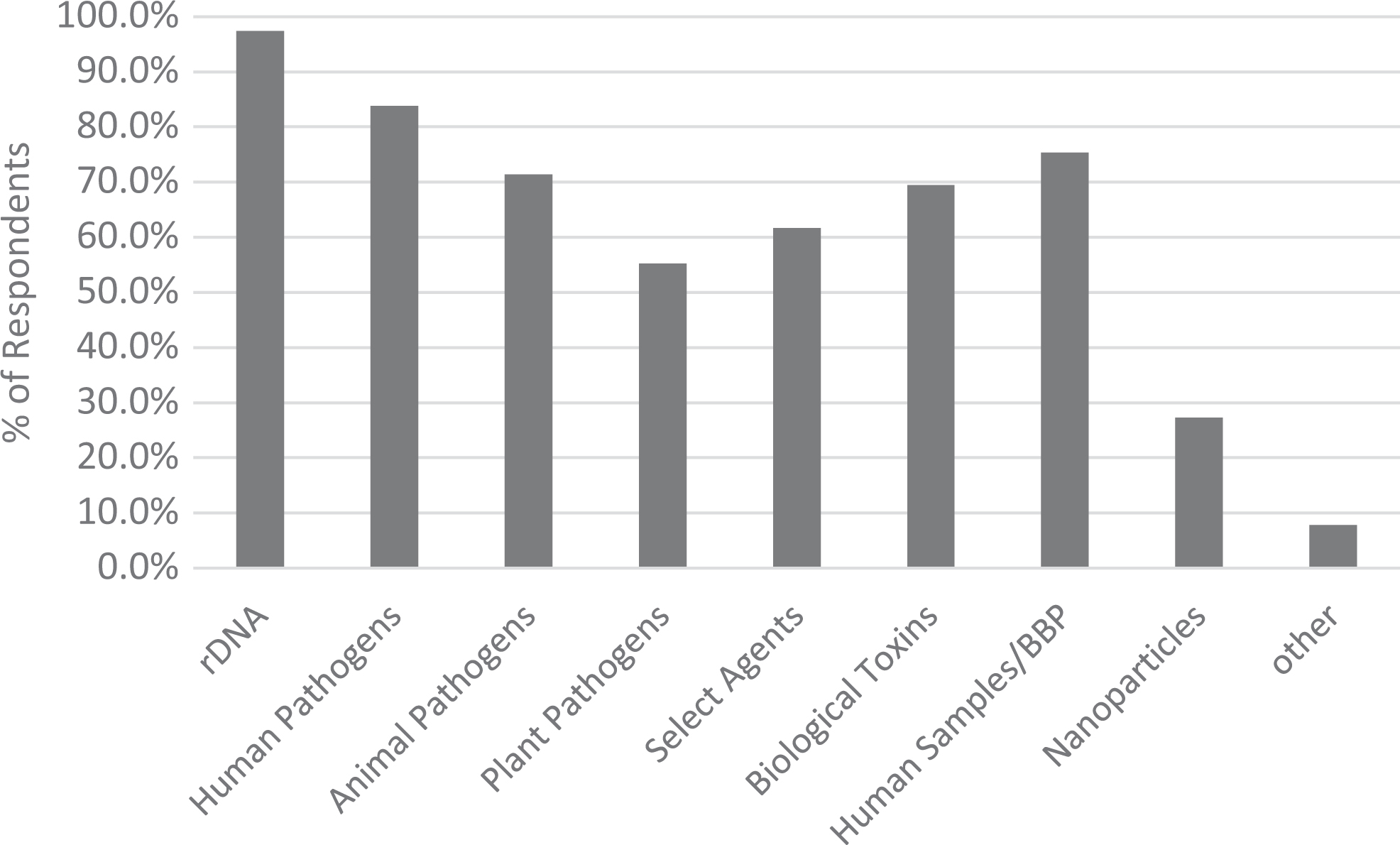
Types of biohazards reviewed by IBCs.
Management of the IBC review and approval process was conducted on an individual project basis by the majority of the respondents, whereas a lower percentage indicated a PI or program-based review and approval system, and some indicated using both (Table 6). The majority of respondents indicated that laboratory audits were conducted on a routine basis. In addition, respondents indicated that lab audits were triggered by a new protocol submission, in responses to a laboratory incident, or some combination of these 3 events (Table 7).
Program- Versus Project-Based IBC Protocol Review and Approval.a

Program based general indicates a whole lab/program receive 1 IBC approval, whereas individual project-based suggests that the investigator receives IBC approval for each individual project under their supervision. Percentage of respondents based on 153 responses. IBC, Institutional Biosafety Committee.
Primary Events Triggering a Lab Audit.

Percentage of respondents based on 151 responses.
The question of who serves as the responsible official (RO) was asked of those institutions that reported working with select agents under the federal select agent program. The BSO most commonly (43%) fulfilled this role, while 29% of respondents indicated these duties were handled at a director level (usually environmental health and safety) and 14% at a senior leadership level (eg, VP or assistant VP).
Size and Staffing of IBC and Biosafety Programs
The number of protocols reviewed by each of the bioethics committees (IBC, IACUC, and IRB) ranged from less than 10 to several hundred for each committee, with a few respondents (2% for IBC, 4% for IACUC, 24% for IRB) reporting protocol numbers in the thousands (Figure 3). Many respondents reported that committees have dedicated support staff (administrators or coordinators) that perform administrative functions of the committee. Of the 3 committee types, IBCs appeared to have fewer support staff than the other bioethics committees, with 22% of respondents indicating having no dedicated staff to support the IBC and 60% of respondents indicating having 1 or less (ie, part-time) FTE for IBC support (Table 8). In contrast, 60% of respondents reported having 1 to 2 FTE IACUC support staff, and IRBs appeared to have the highest level of staffing, with 49% of respondents indicating having greater than 2 FTE for IRB support staff. When the number of FTE was normalized to the number of protocols (ie, number of protocols/number of FTE), we see that on average, each IBC FTE manages 207 IBC protocols, whereas each IRB FTE manages 284 IRB protocols, and each IACUC FTE manages 150 IACUC protocols.
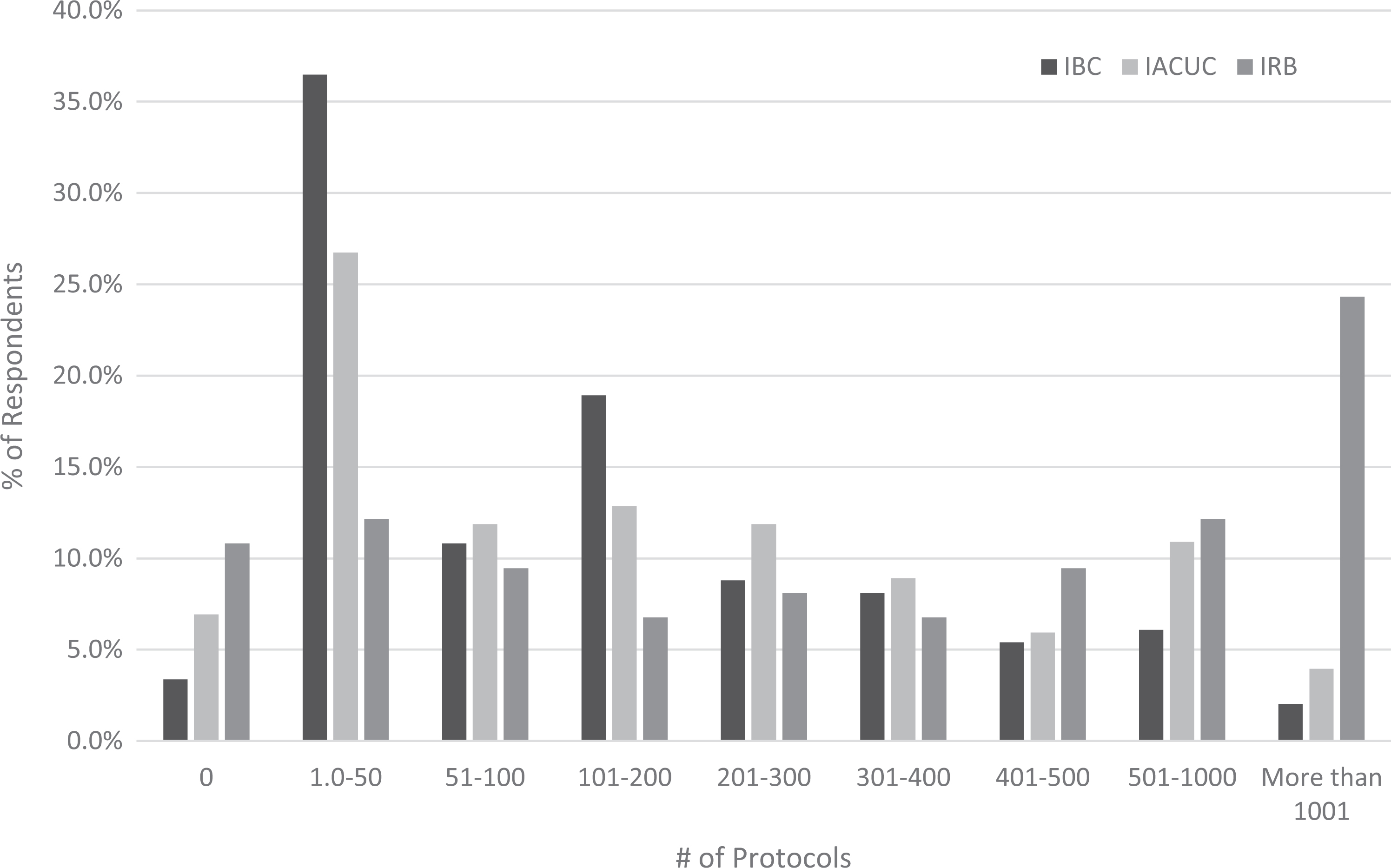
Percentage of respondents reporting protocol numbers for IBC, IACUC, and IRB within the ranges indicated.
Number of FTE Dedicated to Support IBC, BSO, IACUC, and IRB.a
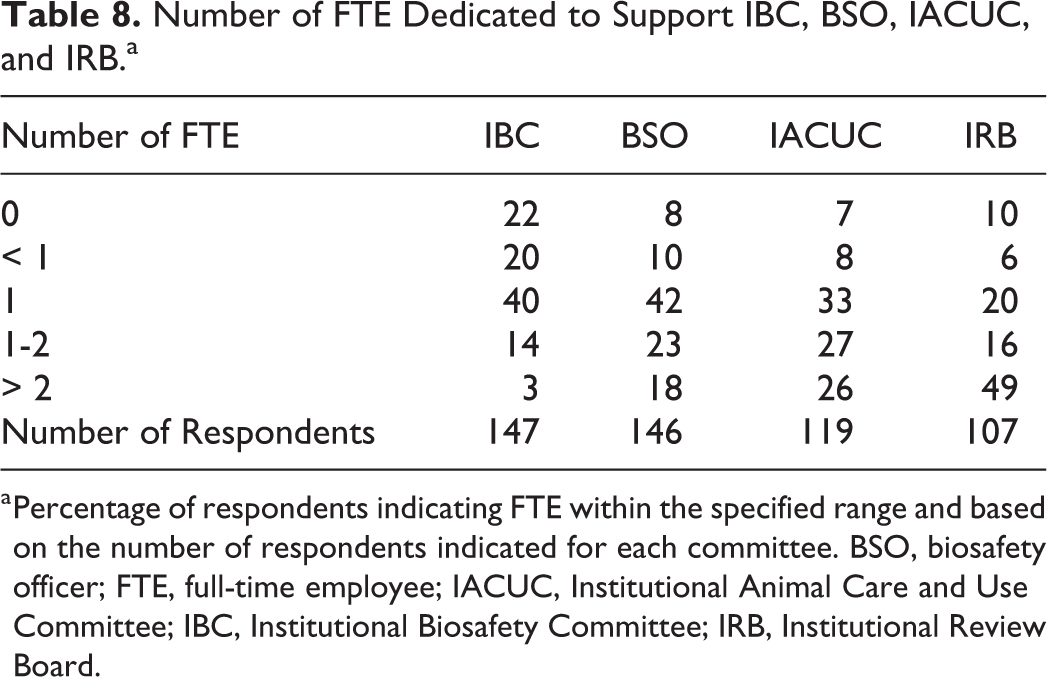
Percentage of respondents indicating FTE within the specified range and based on the number of respondents indicated for each committee. BSO, biosafety officer; FTE, full-time employee; IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee; IRB, Institutional Review Board.
Conversely, dedicated biosafety staff (biosafety officers and assistant biosafety officers) was more consistent across institutions. Specifically, most respondents (65%) indicated 1 to 2 FTE serving as BSO. Additionally, when the number of BSO FTE was normalized to the number of IBC protocols, there was roughly 1 BSO for every 129 IBC protocols.
Discussion
While the survey results further demonstrated the diversity among IBCs and biosafety programs, there were several commonalities that emerged. In assessing the organizational structure of IBCs and biosafety programs, the survey data revealed that despite the 58 different reporting combinations identified, IBCs most commonly reside under the same administrative unit (vice president/chancellor for research) as their sister research compliance committees (IACUC and IRB). The proximity of IBCs to the IACUC and IRB can be beneficial to those who manage the IBCs, aiding in the facilitation of a coordinated review process, particularly for protocols requiring review from more than 1 committee. It also allows IBCs the opportunity to share administrative support and resources and provides continuity to the researchers by harmonizing the various committee review processes.
In contrast, the majority of IBCs (63.2%) report to a different administrative unit than the biosafety officers (which most commonly reports to environmental health and safety). These findings were surprising, considering the overlapping purview of most IBCs and BSOs and that most IBCs are tasked with reviewing research that utilizes a wide range of biohazardous materials. It is the opinion of the authors that coordinating the efforts between the IBC and the BSO is essential to the overall success of a biosafety program. Findings from a recent National Academies of Science report support this opinion and suggest that the optimal regulatory framework is one that harmonizes all components of the system so that efforts and resources are not duplicated. 7 Based on these opinions, it could be argued that the ideal organizational structure for IBCs would be one that places them in the same administrative unit as the IACUC, the IRB, and the BSO. Historically, more IBCs had been managed under environmental health and safety and thus presumably would have been under the same administrative unit as the BSOs and not the other bioethics compliance committees. But as indicated by the Hackney et al 4 surveys, and further demonstrated here, there has been a decrease over time in the number of IBCs managed by an environmental health and safety office from 45% (in 2002), 33% (in 2010), and in this survey to 19% (in 2017). Some speculate 8 that the shift of IBC management is due to new and increasing research regulations such as dual use research of concern (DURC), which many IBCs are now responsible for. Anecdotally, our institute has also seen a shift in BSO management from environmental health and safety to research integrity/compliance and vice president/chancellor for research, although as evident by the data presented here, this is not occurring at the same pace as the IBC shift.
Our survey also uncovered several similarities in the scope of what IBCs are reviewing and how that review is managed. It has been well documented4,5 and further demonstrated by this survey that most IBCs have been tasked with far more oversight duties than originally prescribed by the NIH Guidelines, and as a result, many IBCs have become more of general biohazards committee. A large percentage of respondents (75%) also indicated requiring some type of approval or registration for research exempt from the NIH Guidelines even though this is not a requirement of the NIH Guidelines. 1 This finding was somewhat unexpected as review of exempt recombinant or synthetic nucleic acid research is one of the areas where the NIH Guidelines affords flexibility to institutions.1,3 Our survey did not address why some institutions choose to review exempt work and others choose not to, but all institutions should carefully consider the efforts put into reviewing exempt work versus the risk this type of work presents. The majority of respondents also indicated management of the IBC review/approval process was conducted on an individual project basis with investigators having multiple projects and IBC approvals and that laboratory audits were primarily conducted on a routine basis.
Given these added responsibilities, it is surprising to note that most institutions reported having fewer FTE dedicated to support their IBC (and for 22% none at all) compared to that of the IACUCs, IRBs, and BSOs. The Hackney et al 4 surveys showed that from 2002 to 2010, there was an increase in the number of FTE supporting IBCs, with the percentage of institutions reporting 1 or more FTE growing from 36% to 58%. Based on our survey results, it would appear that this upward trend has reached its limit as the percentage of IBCs with 1 or more FTE remains at 57%. This is in stark contrast to the number of institutions reporting 1 or more FTE dedicated to support IACUCs, IRBs, and biosafety programs. Our findings indicated that 86%, 85%, and 83% of in institutions employed 1 or more FTE to support the IACUC, the IRB, and the biosafety program, respectively.
It is unclear why this discrepancy in FTE exists. One plausible explanation may be that this is due to the higher average protocol numbers for IRBs (1137) and IACUCs (360) compared to IBCs (216), therefore necessitating more IRB and IACUC FTE. To control for this difference in protocol numbers, the number of FTE was normalized to the number of protocols (ie, number of protocols/number of FTE per each institution), and it was determined that on average, each of IBC FTE manages approximately 200 IBC protocols, whereas each IRB FTE manages about 250, and each IACUC FTE manages about 120. However, since many institutions (21%) manage their IBC review/approval process on a PI or program basis (often only having 1 protocol per PI) instead of an individual project basis, it is possible that the reported IBC protocol number is artificially low. Others have also speculated that perhaps IBCs tend to be less staffed than the IACUCs and IRBs because IBCs are not regulated by the same types of federal laws or rigorous accreditation organizations as the other bioethics committees 4 and also require less information for protocol review and compliance adherence. This conjecture is supported by the fact that IACUCs and IRBs generally need to comply with multiple federal agencies for compliance adherence, such as the United States Department of Agriculture and Office of Laboratory Animal Welfare, and Office for Human Research Protections and Food and Drug Administration, respectively, and additionally may require adherence to the Environmental Protection Agency and good practice standards; each of these regulatory bodies requires additional information and inspections for compliance. However, many IBCs are taking on more regulatory tasks, including management of dual use of research concern policy, and thus may want to reevaluate staffing resources allocated to support IBC programs.
Normalizing the number of protocols to the number of FTE per each institution also revealed that there was 1 BSO for roughly every 125 IBC protocols, suggesting a higher rate of BSOs than IBC support staff. To determine if the type of research conducted at an institution had an impact on this difference, we compared the number of BSO and IBC FTEs from institutions that reported conducting BSL-3 and/or select agent research to those that did not. It was no surprise to find institutions with BSL-3/select agent research had a greater number of BSO FTE, averaging 3 BSO FTE compared to 1 BSO FTE at institutions without these types of research. However, the average number of IBC FTE remained at 1 regardless of whether BSL-3/select agent research was conducted; this was a bit unexpected since we know that many IBCs also review research for DURC potential 5 and institutions that conduct BSL-3/select agent research tend to have a greater potential for DURC.
The study has several limitations. First, due to the high response rate from academic institutions, other types of institutions (eg, hospitals and nonprofits) were underrepresented. Over 70% of respondents were from academic institutions, and less than 10% of respondents were from hospitals; however, academic institutions only represent 38% of IBCs registered with NIH (July 2017), while hospitals and clinics comprise 32% of registered IBCs. 9 This would suggest the results of this study are more representative of academic IBCs than IBCs from other types of institutions. This limitation was also observed in surveys conducted by Hackney et al 4 and Jenkins, 5 who speculated that one of reasons for the lack of response from medical institutions was that many of them utilize commercial organizations to manage their IBCs. A second limitation is that several respondents lacked the ability to provide staffing and protocol numbers for their IACUC and/or IRB. The main reasons reported by respondents are that they do not have access to this information (either because they reside in a different administrative units or their institutions use external committees) or that they do not have the means to track this information. A third limitation is the relatively low response rate. While a response rate of 13.6% is within the range often seen for external surveys, it is lower than that reported by similar surveys.4,5 This could possibly be due to the complexity of the survey questions (requiring participants to look up information) and/or the somewhat short window of time (2.5 weeks) that the survey was open.
Conclusions
There were several successful outcomes resulting from the benchmarking survey. First, several similarities and shared practices among IBCs and biosafety programs were discovered. Second, the data collected can be used by institutions (particularly academic institutions) as a tool to compare the organizational structure, scope of review, and size and staffing of their program to that of other programs. Finally, this survey identified disparities in organizational structure, staffing, and IBC protocol review and thus provides opportunities to continue to explore the advantages and challenges among these differences.
Footnotes
Acknowledgment
The authors wish to thank the individuals who generously shared their time by participating in the survey.
Ethical Approval Statement
Not applicable to this research.
Statement of Human and Animal Rights
Not applicable to this research.
Statement of Informed Consent
Not applicable to this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prior Presentations
These data were presented in a poster format during the ABSA International Conference in Charleston, South Carolina, October 15-17, 2018. Preliminary data presented during PRIM&R IBC Boot Camp/Workshop in Denver, Colorado, September 18-19, 2017.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
