Abstract
Cancer of the reproductive tract includes diseases with higher prevalence in the female population. This investigation examined whether an anthocyanin-enriched extract of Aristotelia chilensis, commonly known as “maqui,” could affect some hallmarks of endometrial cancer. Cultures of the human endometrial cancer cell line Ishikawa were treated with a hydroethanolic maqui extract at 1, 3, 10, 30, 100, 300, or 1000 µg/mL to determine the effect on cell viability by MTT assay. Then, we used the 50% Effective Concentration (EC50) to evaluate whether the effect of the maqui extract is mediated via an arrest of the cell cycle or induction of apoptosis using flow cytometry or Annexin V-FITC assays, respectively. The effects of sublethal doses of the maqui extract on migration and invasiveness of Ishikawa cells were also evaluated by the wound healing and Boyden Chamber assay, respectively. Our results show that the hydroethanolic maqui extract inhibits the cell viability with an EC50 of 472.3 µg/mL via increased apoptosis, and that reduces the invasive capacity but not migration of Ishikawa cells. These findings suggest that the hydroethanolic maqui extract has antineoplastic properties for endometrial cancer and merits further studies to corroborate its efficiency as anticancer therapy in reproductive organs.
Introduction
Cancer of the reproductive tract accounts for 12% of all female neoplasia, and 15% of mortalities are associated with this pathology; furthermore, endometrial cancer is the most common and with high indices of mortality in women.1,2 Risk factors for endometrial cancer include obesity, diabetes mellitus, breast cancer, use of tamoxifen, nulliparity, late menopause, exposure to high levels of estrogen, and old age. 1 The use of classical antineoplastic drugs is associated with side effects that affect the quality of life of patients. Thus, it is imperative to find new effective therapies for endometrial malignancy with minimal toxicity risks to normal cells.
The traits that describe how a normal cell slowly transforms into a precancerous and ultimately into a malignant cell are called the hallmarks of cancer and reflects the characteristics that distinguish cancer cells from normal cells. Most of the healthy cells when reaching their differentiation state become quiescent to control proliferation and cell number. However, cancer cells can evade these anti-growth signals allowing uncontrollable proliferation and can survive.3,4 Metastatic capacity is acquired via changes in the expression of cellular adhesion proteins and the overexpression of extracellular matrix-digestingenzymes such as matrix metalloproteinases.3,4 The ability to proliferate uncontrollably and invade healthy tissues are interesting therapeutic targets for developing new antineoplastic drugs.
Aristotelia chilensis (Molina) Stuntz (Elaeocarpaceae), known as maqui, is a Chilean endemic bush whose fruit is a small purple berry rich in anthocyanins, a type of water-soluble polyphenol. It has been used in ethnomedicine as therapeutics for sore throat, diarrhea, and dysenteries. 5 Anthocyanin molecules are a group of flavonoids which are responsible for the colors of many vegetables, flowers, and fruits, and have attracted considerable attention due to their medicinal properties such as vasoactive, anticancer and anti-inflammatory effects.6-10 The main anthocyanins present in the maqui fruit are delphinidin derivatives (delphinidin-3-sambubiosid-5-glucoside; delphinidin-3,5-diglucosideee; delphinidin-3-glucosid; delphinidin3-sambubiosid) and cyanidin derivatives (cyanidin-3-sambubiosid-5-glucoside; cyanidin-3,5-diglucoside; cyanidin-3-glucoside; cyanidin-3-sambubioside).11-13 It has previously been observed that the anthocyanins delphinidin and cyanidin-3-glucoside reduced the proliferation of SKOV3 (ovarian cancer) and HaCaT (epidermal cancer) cells,14,15 while cyanidin-3-sambubioside reduces cell migration and invasion in the breast cancer cell line MDA-MB-231. 16 Interestingly, maqui extract also reduces inflammation markers of the colorectal adenocarcinoma cells Caco-2 and the macrophage cell line, Raw 264.7.12,17-19 Altogether, these studies show that anthocyanins present in the maqui extract could have antiproliferative properties in a variety of cancer types.11,12
Oancea et al 20 have shown that the optimal conditions to extract the enriched-anthocyanins fraction from a Vaccinium corymbosum L. (Ericaceae) fruit is to make a hydroethanolic extraction with an ethanol:water mixture (1:1). Thus, we used the hydroethanolic maqui extract to determine whether the maqui fruit can impair some hallmarks of endometrium cancer. As an experimental model, we used the Ishikawa cell line, which is a well differentiated human endometrial adenocarcinoma expressing estrogen and progesterone receptors and is widely utilized to test new compounds with therapeutic effects for reproductive cancers. 21 Herein, we analyzed the effect of the maqui extract on the cellular viability, migration, and invasive capacity of the Ishikawa cells.
Material and methods
Preparation of the hydroethanolic maqui extract
We used the seedless fruit pulp of A. chilensis which was collected in a local market of Santiago, Chile. In the markets of Chile, maqui fruits are freely sold without brand name and no preservatives added and the whole plants from which the fruits were harvested were not available. This is a limitation of our work. The hydroethanolic extract was obtained according to Oancea et al 20 with some modifications. Briefly, the pulp (3% p/v) was pulverized in liquid nitrogen, and then it was macerated with ethanol:water in a relation 1:1 for 72 hours at room temperature. Then, the macerated extract was dried overnight at 37°C and a stock solution was prepared adding 100 mg of the extract in 1 mL of dimethylsulfoxide (DMSO).
Quantification of total phenolic compounds by Folin-Ciocalteu
The total phenols were determined using the Folin-Ciocalteu assay as described by Singleton and Rossi. 22 The absorbance of the samples was measured at 740 nm in a spectrophotometer Tecan Infinite F50 (Tecan Trading AG, Switzerland). Gallic acid was used as reference standard and the results were expressed as gallic acid equivalents (mg Ac. Equ/mL).
Cell culture
Ishikawa cells were maintained in DMEM medium with 4.0 mM L-Glutamine and 4.500 mg/L Glucose (Thermo Fisher Scientific, Waltham, USA) supplemented with 10% (vol/vol) of Fetal Bovine Serum (FBS; Thermo Fisher Scientific), sodium pyruvate 1 mM, penicillin 100 UI/mL and streptomycin 100 µg/mL (Sigma Chemical, St. Louis, USA). Initial cultures contained 100 000 to 150 000 cells/well.
Cell viability assay
Ishikawa cells were seeded at a confluence of 70% and treated with the maqui extract at 1 µg/mL, 3 µg/mL, 10 µg/mL, 30 µg/mL, 100 µg/mL, 300 µg/mL, and 1000 µg/mL, DMSO at 0.001%, 0.003%, 0.01%, 0.03%, 0.1%, 0.3% and 1% as vehicle, or untreated as control for 72 hours. Then, cells were incubated with thiazolyl blue tetrazolium bromide (MTT; Abcam Inc., Cambridge, MA, USA) 500 µg/mL in serum free-DMEM for 2 hours at 37°C according to Mosmann. 23 Finally, the formazan crystals formed in the bottom of the plate were dissolved with DMSO and the absorbance at 550 nm was measured in spectrophotometer Tecan Infinite F50. Cell viability was calculated according to the equation below24,25
The 50% effective concentration (EC50) value was determined using GraphPad Prism (version 6.01, GraphPad Software, Inc.), subtracting the effect of the vehicle on cell viability.
Cell cycle analysis by flow cytometry
Ishikawa cells were seeded at a confluence of 70% and treated with the hydroethanolic extract 500 µg/mL (approximate to EC50) dissolved in DMSO 0.5% as vehicle during 24 hours. The cells were then washed, pelleted, resuspended in phosphate buffered saline (PBS) and fixed with cold ethanol 75%, resuspended in PBS and stained with a solution of propidium iodide (PI; Sigma-Aldrich. Saint Louis, MO, USA) 20 µg/mL and RNase 200 µg/mL for 30 minutes at room temperature in the dark. Finally, the content of DNA was measured in the flow Cytometer BD FACS Canto II (BD Biosciences, San Jose, CA, USA.) in PI channel. 2-methoxiestradiol (2ME) 5 µM was used as positive control26,27 and untreated cells were used as control. The percentages for each cell cycle stage were analyzed by the ModFit LT program (v5.0.9, Verity Software House), and the subG1 population was excluded from the histogram because the purpose of this assay was the analysis of cell cycle.
Annexin V apoptosis assay
Ishikawa cells were seeded at a confluence of 70% and measured after 24 hours of treatment with 500 µg/mL (value close to EC50) of the extract, or DMSO 0.5% as vehicle in DMEM media at 37°C. Treated cells were stained with Annexin V-FITC reagent (Biolegend, Inc.San Diego, CA, USA.) and Propidium Iodide (PI) as described by the manufacturer. Stained cells were run in flow cytometer BD Accuri C6 (BD Life Sciences, San Jose, CA, USA.) to analyze viable (double negative), early apoptotic (Annexin V positive), late apoptotic (Annexin V positive, PI positive), and necrotic (PI positive) populations in BD Accuri C6 software (version 1.0.264.21, Accuri® cytometers, Inc.). As positive control we used 2ME 5 µM26,27 and untreated cells were used as control.
Boyden Chamber cell invasion assay
Ishikawa cells were seeded at a confluence of 20% to 30% in order to avoid the contact inhibition of migration and treated for 24 hours at 37°C and 5% of CO2 with the hydroethanolic extract 10 µg/mL (a dose that not affect cell viability because cell death could cause a false positive), or with DMSO 0.01% as vehicle, and untreated cells were used as controls. Cells were harvested with trypsin-EDTA 0.25% (Biological Industries, Beit HaEmek, Israel), washed and then 20 000 cells were resuspended in serum-free medium and seeded in an upper chamber of a Transwell insert (Corning Incorporated, Lowel, MA, USA.), coated with Matrigel (Corning Incorporated, Bedford, MA, USA.) 1:30 in DMEM. Additionally, the lower chamber was filled with DMEM supplemented with FSB 10% as chemoattractant (or serum free-DMEM as control without chemoattractant). Then, cells were incubated for 24 hours at 37°C and 5% of CO2. Later, the cells were fixed with absolute methanol at −20°C for 20 minutes and stained with crystal violet 0.01% for 30 minutes at 37°C. The cells in the upper side of the membrane of the Transwell insert were removed using a cotton swab, and finally, the lower side of the Transwell insert was observed in an inverted microscope to count invading cells.
Wound healing assay
Ishikawa cells were seeded at a confluence of 70%; a wound was made on a monolayer of Ishikawa cells with a micropipette tip with the purpose of enabling cell migration. Then, the cells were washed with PBS 3 times, and treated with 10 µg/mL of maqui extract or with DMSO 0.01% as vehicle, with untreated cells as control for 24 hours at 37°C and 5% of CO2. Wounds in the cell monolayer were microphotographed at 0 and 24 hours. Then the wound healing was analyzed in ImageJ program (version 1.52a, National Institute of Health, USA).
Statistical analysis
The data are represented as mean ± standard error of the mean (SEM) and analyzed with the Stata/SE program (version 14.0, StataCorp LP, USA) or GraphPad Prism (version 6.01, GraphPad Software, Inc.) software to find statistical differences using Kruskall-Wallis test and Dunn’s post-test. Statistical differences were considered with a P < .05.
Results
Anthocyanin profile of the hydroethanolic maqui extract
The results of the Folin-Ciocalteu assay showed that 100 mg/mL of the maqui extract had a total phenol content of 2.68 ± 0.17 of mg Ac. Equ/mL (n = 5).
Hydroethanolic maqui extract reduced cell viability of Ishikawa cells
This experiment was performed to determine whether the hydroethanolic maqui extract affects the viability of Ishikawa cells. Figure 1 shows that the maqui extract reduced in a dose-response manner cell viability of Ishikawa cells from 30 µg/mL, reaching the highest response between 300 and 1000 µg/mL (percentage of cell viability: 1 µg/mL: 89.13 ± 3.79%; 3 µg/mL: 84.47 ± 4.42%; 10 µg/mL: 74.08 ± 6.60%; 30 µg/mL: 59.45 ± 6.20%; 100 µg/mL: 51.48 ± 6.83%; 300 µg/mL: 26.28 ± 3.85%; 1000 µg/mL: 14.32 ± 2.67%). On the other hand, the vehicle had a stable effect on cell viability between 0.001% and 0.1%, however, DMSO at 0.3% and 1% reduced viability by 35% and 40% respectively (percentage of cell viability in cells treated with DMSO: 0.001%: 84.19 ± 3.45%; 0.003%: 86.75 ± 3.16%; 0.01%: 85.54 ± 6.89%; 0.03%: 83.95 ± 5.78%; 0.1%: 84.23 ± 5.65%; 0.3%: 76.15 ± 4.03%; 1%: 60.62 ± 5.61%). The EC50 of the maqui extract on cell viability was 472.30 µg/mL, which was calculated by subtracting the effect of the vehicle on cell viability.

Effect of maqui extract on cell viability of Ishikawa cells. Dose response curve of the maqui extract on the cell viability in Ishikawa cells. Cells were treated with maqui extract (1, 3, 10, 30, 100, 300, and 1000 μg/mL) or DMSO (vehicle, 0.001%, 0.003%, 0.01%, 0.03%, 0.1%, 0.3%, and 1%) for 72 hours and then were analyzed by MTT assay. Each point represents the cell viability normalized respect untreated cells (control). Pointed line represents control. *P < .05 respect to vehicle according to a Kruskall-Wallis test with a Dunn’s post-test, n = 8.
Hydroethanolic maqui extract did not arrest the cell cycle in Ishikawa cells
This experiment was designed to determine the effect of maqui extract on the cell cycle in Ishikawa cells. Figure 2A shows the representative histograms obtained by flow cytometry while Figure 2B shows the quantification of the cell cycle stages for each treatment. Contrary to 2ME, which arrested cell cycle in the G2/M phase, treatment with maqui extract had no effect on the cell cycle of Ishikawa cells (percentage of cells for each cell cycle phase: Vehicle: G1/G0: 59.46 ± 1.95%; S: 14.17 ± 2.39%; G2/M: 26.37 ± 2.40%; Maqui 500 µg/mL: G1/G0: 49.29 ± 4.83%; S: 22.25 ± 5.36%; G2/M: 28.46 ± 6.08%; 2ME 5 µM (C+): G1/G0: 12.76 ± 1.80%; S: 20.24 ± 3.16%; G2/M: 69.5 ± 3.80%). In these graphs, we did not show the subG1 population because the purpose of this experiment was to determine changes in the cell cycle and not to measure apoptosis.

Effect of maqui extract on cell cycle in Ishikawa cells. Ishikawa cells were treated with DMSO 0.5% (vehicle), maqui 500 μg/mL, or 2ME 5 μM (C+) for 24 hours and then were fixed and stained with Propidium Iodide to analyze changes in the cell cycle by flow cytometry. (A) Representative flow cytometry histograms shows cell populations in the stages G1/G0, S, and G2/M of the cell cycle. (B) Populations of each stage of the cell cycle (G1/G0 in black, S in white, and G2/M in grey) were quantified for each treatment (vehicle, maqui 500 μg/mL, or 2ME 5 μM). *P < .05 respect to vehicle according to a Kruskall-Wallis test with a Dunn’s post-test, n = 8.
Hydroethanolic maqui extract induced apoptosis in Ishikawa cells
This experiment was designed to determine the effect of hydroethanolic maqui extract on apoptosis in Ishikawa cells. Figure 3A illustrates representative dot plots showing 3 cell populations: viable cells in green (double negative), early apoptotic cells in blue (Annexin-V positive) and late apoptotic (end-stage) cells in red (Annexin-V positive, PI positive). Figure 3B quantifies the percentages of early and late apoptotic cells. For early apoptosis, the maqui extract induces an increase in the percentage of apoptotic cells compared to vehicle (percentage of early apoptotic cells: Vehicle: 83.43 ± 9.87%; Maqui 500 µg/mL: 146.30 ± 8.84%; 2ME 5 µM: 200.70 ± 44.21%) while the percentage of late apoptosis is increased with maqui extract treatment in comparison with vehicle (percentage of late apoptotic cells: Vehicle: 113.00 ± 11.25%; Maqui 500 µg/mL: 411.10 ± 91.99%; 2ME 5 µM: 543.00 ± 27.22%).
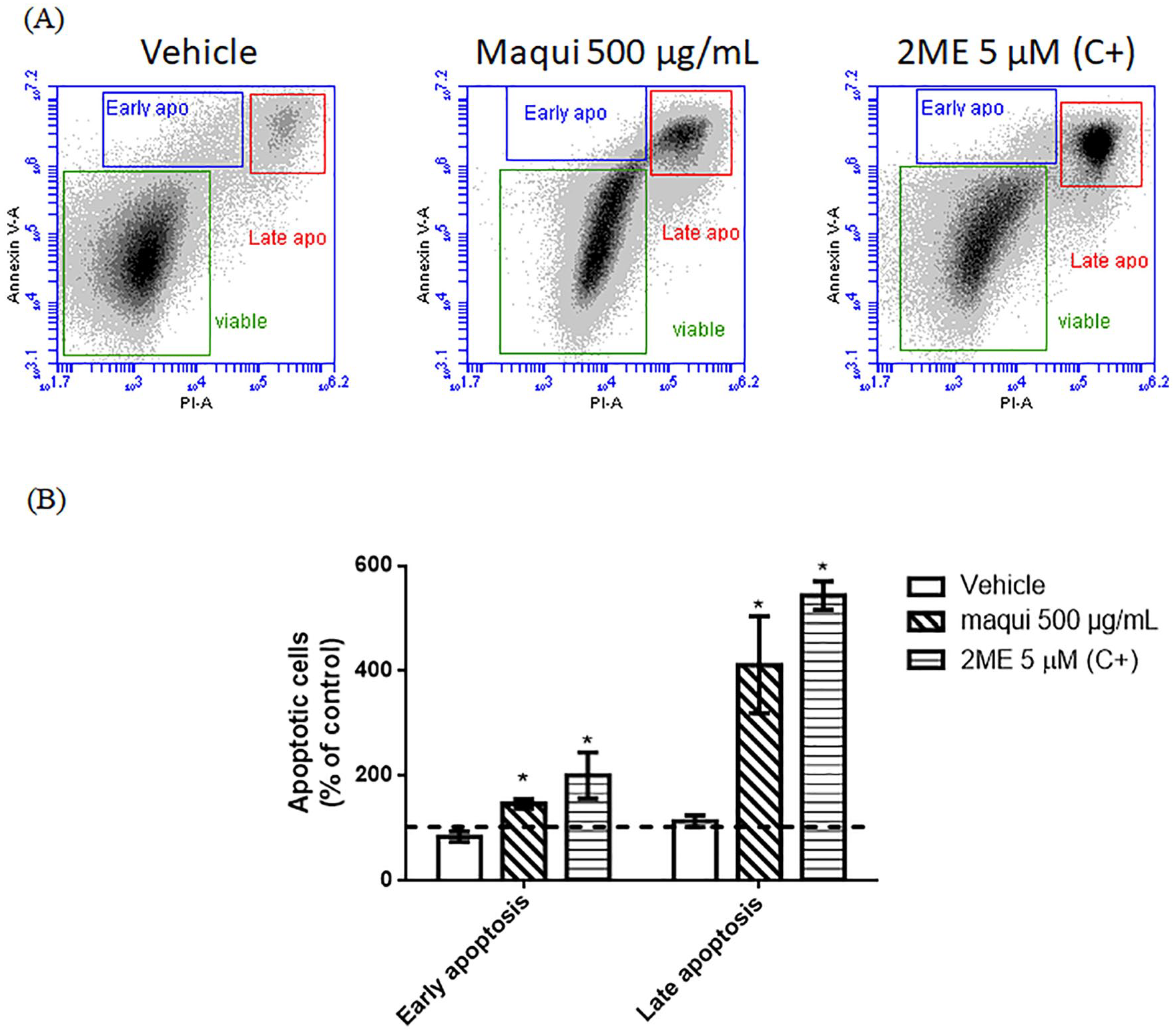
Effect of maqui extract on induction of apoptosis in Ishikawa cells. Ishikawa cells were treated with DMSO 0.5% (vehicle), maqui 500 μg/mL, or 2ME 5 μM (C+) for 24 hours and then were stained with Annexin-V-FITC and Propidium Iodide to analyze by flow cytometry induction of apoptosis. (A) Representative dot plots of flow cytometry shows populations of viable cells in green (double negative), early apoptotic cells in blue (Annexin-V positive), and late apoptotic cells in red (Annexin-V positive, PI positive). (B) Quantification of early and late apoptotic cells population for each treatment in a histogram. Data represents the percentage of cells relative to untreated cells (control). Pointed line represents control. *P < .05 respect to vehicle according to a Kruskall Wallis test with a Dunn’s post-test, n = 4.
Hydroethanolic maqui extract reduced invasiveness of Ishikawa cells
This experiment was designed to determine the effect of the hydroethanolic maqui extract on the invasive capacity of Ishikawa cells. Figure 4A illustrates the representative photomicrographs of the invasive cells in the lower side of a Transwell insert. In the vehicle group, cells invaded the lower side of the Transwell insert, while few invasive cells are observed when treated with maqui 10 µg/mL or in the control without chemoattractant. These results were quantified in Figure 4B showing that the treatment with maqui extract reduces the Ishikawa cell invasiveness (Percentages of invasive cells: Vehicle: 95.25 ± 19.76%; Maqui 10 µg/mL: 28.81 ± 10.23%; control without chemoattractant: 14.21 ± 13.78%).

Effect of maqui extract on invasion ability of Ishikawa cells. (A) Matrigel coated-Transwell invasion assay was performed to determine if maqui extract reduces invasiveness of Ishikawa cells. Cells were treated with DMSO 0.01% (vehicle), maqui 10 μg/mL, or untreated (control). Lower chamber was filled with DMEM plus FBS 10% as chemoattractant (or serum free medium as without chemoattractant control). After 24 hours, the lower face of transwell insert was microphotographed to count invader cells. In (A) are shown representative microphotographs of lower face of transwell insert. (B) Quantification of invasive cells in transwell insert after 24 hours. Data was normalized respect to untreated cells (control), pointed line represents control. *P < .05 respect to vehicle, n = 4.
Hydroethanolic maqui extract did not affect migration of Ishikawa cells
This experiment was performed to determine whether the hydroethanolic maqui extract affects migration in Ishikawa cells. No differences in the wound area between the vehicle and maqui treatments were found (percentage of wound area relative to time 0: Vehicle: 75.07 ± 3.36%; Maqui 10 µg/mL: 72.10 ± 1.70%; n = 4).
Discussion
Polyphenols are organic compounds found in plant foods like fruits, vegetables, cereals, and beverages. These molecules are secondary metabolites responsible for defending the plant against pathogens or ultraviolet radiation. In human health, polyphenols have antioxidant and anti-inflammatory properties and protect against cancer and cardiovascular diseases, among others. 28 A subgroup of polyphenols are the anthocyanins, which have a potent anticancer effect. These molecules can be found in grapes, cherries, or berries like maqui.29,30
The consumption of berries in the diet brings health benefits, because the compounds of these fruits can regulate signaling pathways that are deregulated in the carcinogenesis process, such as nuclear factor-κB (NF-κB), cyclooxygenase 2 (Cox2), phosphoinositide-3-kinase (PI3K/AKT), p38, vascular endothelial growth factor (VEGF) and apoptosis regulatory proteins such as Bcl-2, Bax, and caspase 3. 31 Additionally, the consumption of berries can reestablish the redox balance. This is beneficial because the redox imbalance allows carcinogenesis and the development of cancer. 32
There are few reports about the composition of maqui fruit, however all of these agree that the major compounds are derivatives of mono or diglycosylated delphinidin and cyanidin, with delphinidin-3-sambubioside-5-glucoside being the most abundant polyphenol in maqui fruit.11-13,33,34 Other polyphenols that have also been found in the maqui fruit are: quercetin, myricetin, and kaempferol of the flavonoid subgroup; gallic acid, protocatechuic acid, ellagic acid, and p-coumaric acid.35,36 In our hydroethanolic extract, at a concentration of 100 mg/mL, the total phenol content was 2.678 ± 0.17 mg Ac. Equ/mL. Therefore, if we consider that the maqui fruit contains 23.4 to 31.9 mg of gallic acid equivalent/g of dry weight, 36 then our extraction method allowed extraction of a large amount of polyphenols.
In this work, we established that the maqui extract reduced the cell viability in Ishikawa cells, with an EC50 of 472.3 µg/mL. In order to compare the potency of our extract with others of similar composition, we carry out a bibliographic review and found that Wang et al 37 treated murine melanoma B16-F10 cells with an extract of blueberries. Among vegetables, blueberries, like maqui, are considered the fruits with the highest anthocyanin content. In their experiment, they treated B16-F10 cells with an extract of low-bush wild blueberry (Vaccinium uliginosum L. (Ericaceae)) enriched in anthocyanins. The blueberry extract reduces cell viability by 50% at a concentration close to 200 µg/mL, similar to the effect of our maqui extract on the viability of Ishikawa cells. On the other hand, many studies have established the EC50 of delphinidin, cyanidin, and its derivatives on cell viability in different cell lines. According to these reports, it can be established that the EC50 range of these anthocyanins has values between approximately 10 to 40 µg/mL.38-40 If we consider that the anthocyanin content of the maqui fruit is approximately 211.9 mg per 100 g of dry weight, 11 it is very likely that anthocyanins in maqui extract have a lower EC50 compared to pure molecules. This phenomenon was described in Kausar et al. 38 They compared the effect of pure anthocyanidins versus an equimolar mixture on cell viability and established that the EC50 of the molecules increases synergistically when mixed.
Our results show that the hydroethanolic maqui extract had no effect on the cell cycle stages suggesting that the phenolic compounds from this extract can reduce cell viability without exerting cytostatic effects in Ishikawa cells. Previous reports have shown that anthocyanins from Hibiscus sabdariffa L. (Malvaceae), mainly delphinidin 3-sambubioside and cyanidin 3-sambubioside, reduce the cell viability arresting the cell cycle in the G2/M phase in leukemia cells HL-60. 41 This could be explained because in the maqui fruit, delphinidin is usually found as 3-glucoside, 3.5-diglucoside, 3-sambubioside, and 3-sambubioside-5-glucoside, but not in its glycosylated-free form. 11 The same occurs with other phenolic compounds such as kaempferol or quercetin, which also stop the cell cycle in the G2/M phase.35,42,43 Therefore, it is possible that saccharides conjugated to polyphenols inhibit its effect on the cell cycle. Another explanation could be associated with the nature of the extract, because it is a complex mixture of compounds and there could be molecules that counteract the cytostatic effect of the maqui phenolic compounds, but this remains to be tested.
Although the hydroethanolic maqui extract did not arrest cell cycle, its increased apoptosis in Ishikawa cells suggests that the effect of maqui on the viability of Ishikawa cells is by activation of the apoptotic pathways. In this sense, it has been previously reported that different constituents of the maqui fruit can induce apoptosis. For example, the anthocyanidin delphinidin, which is the basis of the most abundant anthocyanins of A. chilensis, increases apoptosis through the decrease of the antiapoptotic protein Bcl-2, the increase of the proapoptotic protein Bax and increasing the activation of caspases 3 and 9 in non-small-cell lung cancer cells. 44 Additionally, it has been shown that delphinidin, cyanidin-3-glucoside, and an anthocyanin enriched maqui extract, reduces the expression of Cox2 in HaCaT cells (human keratinocyte), Raw 264.7 (murine macrophage) and Caco-2 cells (human colorectal adenocarcinoma), which has antiapoptotic effects in endometrial cancer.14,18,19,45 Additionally, other polyphenols present in maqui fruit, such as quercetin, kaempferol, and myricetin, have also been shown to induce apoptosis in some cancer cell lines.46-48
To our knowledge, this the first report showing that a maqui extract rich in polyphenols increases apoptosis in endometrial cancer cells; however, further studies are necessary to elucidate the apoptotic pathways involved in this maqui extract effect.
We also found that the hydroethanolic maqui extract reduced by approximately 70% the invasion capacity of Ishikawa cells in the matrigel-coated Transwell invasion assay. Thus, besides reducing cell viability, the maqui extract reduces metastatic properties in endometrial cancer. These results agree with those described by Chen et al 49 who reports that an extract of black rice (Oryza sativa L. (Poaceae)) rich in cyanidin-3-glucoside reduces the cell invasion in SKHep-1 and Huh-7 cells from human hepatocarcinoma, HeLa cells from human cervical carcinoma and SCC-4 cells from human squamous cell carcinoma. This finding is consistent with another report that indicates that cyanidin-3-sambubioside extracted from Acanthopanax sessiliflorus (Rupr. and Maxim.) Seem. (Araliaceae) fruit reduces the expression of metalloproteinases MMP-2 and 9 in breast cancer cell line MDA-MB-231. 16 Other polyphenols present in the maqui fruit have also been implicated in the reduction of cell invasion through a reduction of matrix metalloproteinases. Myricetin inhibits invasion in hepatocellular carcinoma cells 50 and kaempferol reduces invasion in breast cancer cells and reduces lung metastasis of B16 mice melanoma cells. 51
Although the results showed that the maqui extract reduces the invasion capacity of the Ishikawa cells, it had no effect on the migration capacity. Lee et al 16 established that the extract of A. sessiliflorus, enriched in cyanidin-3-sambubioside, dose-dependently reduces cell migration capacity in MDA-MB-231 cells in a wound-healing assay. Moreover, it has been established that pure or mixed anthocyanidins reduce the migration capacity of H1299 cells from human non-small cell lung cancer. 38 A possible explanation for this inconsistency could be that the effect of the polyphenols on cancer cell migration could be dependent on the kind of cancer. For example, cyanidin-3-glucoside extracted from black rice has been reported to reduce motility of Huh-7 cells but does not affect HeLa or SSC-4 cells. 49 Therefore, it would be interesting to establish whether the maqui extract affects the motility of other cell lines. Additionally, the concentration of the compounds could also be an important factor in the inhibition of cell motility. In this context, cyanidin-3-sambubioside and cyanidin-3-glucoside exert their effect on cell migration at 30 and 50 µg/mL respectively. Furthermore, small reductions in the concentration of these compounds drastically decrease their effect on cell motility.16,49 Therefore, a limitation of our study is that we do not know the concentrations for each phenolic compound in the hydroethanolic maqui extract used in this work.
We concluded that a hydroethanolic maqui extract affects two important cancer hallmarks as apoptosis evasion and invasive capacity in the Ishikawa cells, allowing us to propose this this Chilean traditional plant as a potential source of therapeutic agents for endometrial cancer.
Footnotes
Author Contributions
J.M. participated in the design of the study, in directing and completing experimental analysis and in writing the manuscript. E.E. collaborated in the preparation of the extract of A. chilensis fruit and in its characterization. F.E-G. collaborated in carrying out the cell viability test and reviewing the manuscript. P.O. and H.C. participated in planning the experiments and contributed to drafting the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received financial support from grants DICYT Asociativo 021743OD_DAS, Basal Program for Centers of Excellence, CEDENNA-Grant AFB180001, Vicerrectoria de Investigación USACH (Beca Ayudante de Investigación USA1899) and National Doctorate Scholarship 21151146, CONICYT.

