Abstract
Integrative medicine is an approach to health and healing that “makes use of all appropriate therapeutic approaches, health care professionals, and disciplines to achieve optimal health and healing.” A comprehensive integrative medicine intervention for cancer patients typically includes nutritional counseling, biobehavioral strategies, and promotion of physical activity, as well as dietary supplements including herbs, nutraceuticals, and phytochemicals. A broad-spectrum intervention of this type may contribute uniquely to improvement in cancer outcomes through its impact on a wide variety of relevant molecular targets, including effects on multiple cancer hallmarks. Hallmarks that may be particularly affected include genetic instability, tumor-promoting inflammation, deregulated metabolism, and immune system evasion. Because of their susceptibility to manipulation by diet, exercise, and supplementation, these may be characterized as metabolic hallmarks. Research on the use of comprehensive integrative approaches can contribute to the development of systems of multitargeted treatment regimens and would help clarify the combined effect of these approaches on cancer outcomes.
Keywords
Integrative medicine is an approach to health and healing that “makes use of all appropriate therapeutic approaches, health care professionals, and disciplines to achieve optimal health and healing.” 1 The foundation of integrative medicine in cancer is lifestyle therapies, emphasizing diet and physical activity. Use of traditional herbs, nutraceuticals, and phytochemicals is common in integrative medicine. In addition, the study of the connection of the mind and emotions (biobehavioral factors) to cancer outcomes has recently uncovered startling evidence on how emotional states and stress directly or indirectly affect cancer-related physiological pathways. Much of what is termed “integrative oncology” entails individual complementary therapies such as massage or meditation that are separate from—or perhaps concurrent with—conventional treatment but are not methodically combined in a full multidimensional system of treatment. Implementation of comprehensive integrative oncology, however, is a complete system that employs specific lifestyle interventions and nutraceuticals strategically to combat disease in a broad-spectrum fashion by addressing symptoms and treatment side effects, and attacking molecular pathways of progression and terrain factors. 2 Such therapy also includes individualization of interventions based on assessments of molecular profiles, metabolic terrain factors, and other lifestyle and clinical variables. In this commentary, we briefly review some of the impacts of elements of comprehensive integrative oncologic care with the potential to affect the biology and outcomes of cancer.
Lifestyle approaches contribute to molecular target medicine in unique ways. Integrative approaches modify multiple cancer-related molecular targets through specific lifestyle interventions. Such interventions are not simple to implement, but they promote overall health, and they are arguably less expensive per patient than current molecular target therapies. 3 Basic principles of diet and exercise therapies have received sufficient research that the American Cancer Society has recently issued guidelines for nutrition and exercise. 4 We briefly review the status of research on diet, exercise, and biobehavioral therapies as they relate to the cancer hallmarks and to a variety of molecular targets associated with them, as well as cancer-related variables including survival, immunity, weight change, and depression.
The concept of the cancer hallmarks was published by Douglas Hanahan and Robert A. Weinberg in 2000. 5 The hallmarks “constitute an organizing principle that provides a logical framework for understanding the remarkable diversity of neoplastic diseases.” The hallmarks are the biological capabilities that cells acquire during malignant promotion and progression that support the function of malignant tissue. Hanahan and Weinberg initially proposed 6 hallmarks: sustained proliferative signaling, evading growth suppressors, activating invasion and metastasis, enabling replicative immortality, inducing angiogenesis, and resisting cell death. Subsequent to its publication, this concept gained widespread recognition. In 2011, the same authors expanded the concept of the hallmarks. 6 Added were 2 enabling characteristics that permitted cells to acquire the 6 hallmarks, and 2 new hallmark capabilities. The enabling characteristics were genomic instability and tumor-promoting inflammation; the new hallmarks were deregulating cellular energetics and avoiding immune destruction. The hallmarks that are of greatest relevance and interest in integrative therapies, which could be characterized as “metabolic” hallmarks, are genomic instability, inflammation, deregulated energetics, and immune evasion. These hallmarks are all involved with general metabolic processes and are among the hallmarks that might be more directly targeted through diet and lifestyle measures.
The hallmarks that are most directly related to integrative diet therapy are deregulated metabolism and inflammation. Optimal integrative nutrition after cancer diagnosis is quite similar to a cancer prevention diet. 7 Major features include a diet that is high in vegetables (especially high-nutrition vegetables such as the crucifers 8 and alliums 9 ), fruits, and whole grains and low in refined carbohydrates, red and processed meat, and in fats and oils containing omega-6 fatty acids. Suitable proteins include plant proteins, omega-3 enriched eggs, or egg whites and fish. Avoiding weight gain and, in cases of obesity or overweight, weight loss under supervision of a cancer-trained dietitian may be important. A diet of this type can help reduce inflammation, oxidation, glycemia, and other adverse features associated with the typical Western diet. 10
Observational studies in cancer patients have assessed dietary patterns in relation to survival and recurrence of disease. Such studies usually divide diet patterns into “healthy” diets that are high in vegetables and fruits, versus “unhealthy” diets high in red meats and refined grains. For example, postmenopausal breast cancer patients in Germany who consumed healthy versus unhealthy diets had reduced overall mortality and breast cancer mortality. 11 Stage 3 colon cancer patients who consumed a Western diet after diagnosis had higher risk of recurrence and death than those with healthy diets. 12 Diets high in fruits and vegetables improved survival in ovarian cancer patients, while patients consuming high red/processed meats and milk had reduced survival. 13
Randomized trials are also found in the literature. Two randomized trials of diet in early breast cancer—one of a low-fat diet and one of a low-fat diet high in vegetables, fruit, and fiber—had differing results.14,15 Subjects on the low-fat diet lost weight and had reduced recurrence risk, in particular, for estrogen receptor (ER) negative patients. 14 Subjects on the diet emphasizing vegetables, fruit, and fiber, who were unsuccessful in reducing fat consumption relative to controls, did not lose weight and evidenced no significant difference in recurrence or mortality. 15 Notably, a high-fat diet may inhibit expression of SIRT3, a critical sirtuin that reduces development of the metabolic syndrome. 16 This suggests that the reduction of fat tissue through diet may be an important mechanism of dietary interventions. A trial in “watchful waiting” prostate cancer patients randomized to a low-fat vegetarian diet and lifestyle program versus conventional care found reduced prostate-specific antigen (PSA) levels in the diet group and increased levels in the usual care group. 17
Tables 1 and 2 suggest that the impact of diet on laboratory tests related to molecular targets and cancer hallmarks may be significant. Table 1 shows the effects of various diets on inflammation through effects on C-reactive protein (CRP). Most trials are randomized studies showing that whole grains and low glycemic, low or high protein, low fat, low carbohydrate, or Mediterranean diets can reduce CRP. Red meat, a high glycemic index diet and a control low-fat diet (which was not adhered to by most of the control group participants and was thus in fact a high fat diet), did not reduce CRP.
Effect of Diet on C-Reactive Protein and Related Variables.

Abbreviations: CRP, C-reactive protein; GL, glycemic load; GI, glycemic index; RCT, randomized controlled trial; CVD, cardiovascular disease; TNFR, tumor necrosis factor receptor.
Only 21% of the control diet group achieved reduction of fat calories to <35% of daily intake. 23
Effects of Diet on Cell Signaling Pathways.
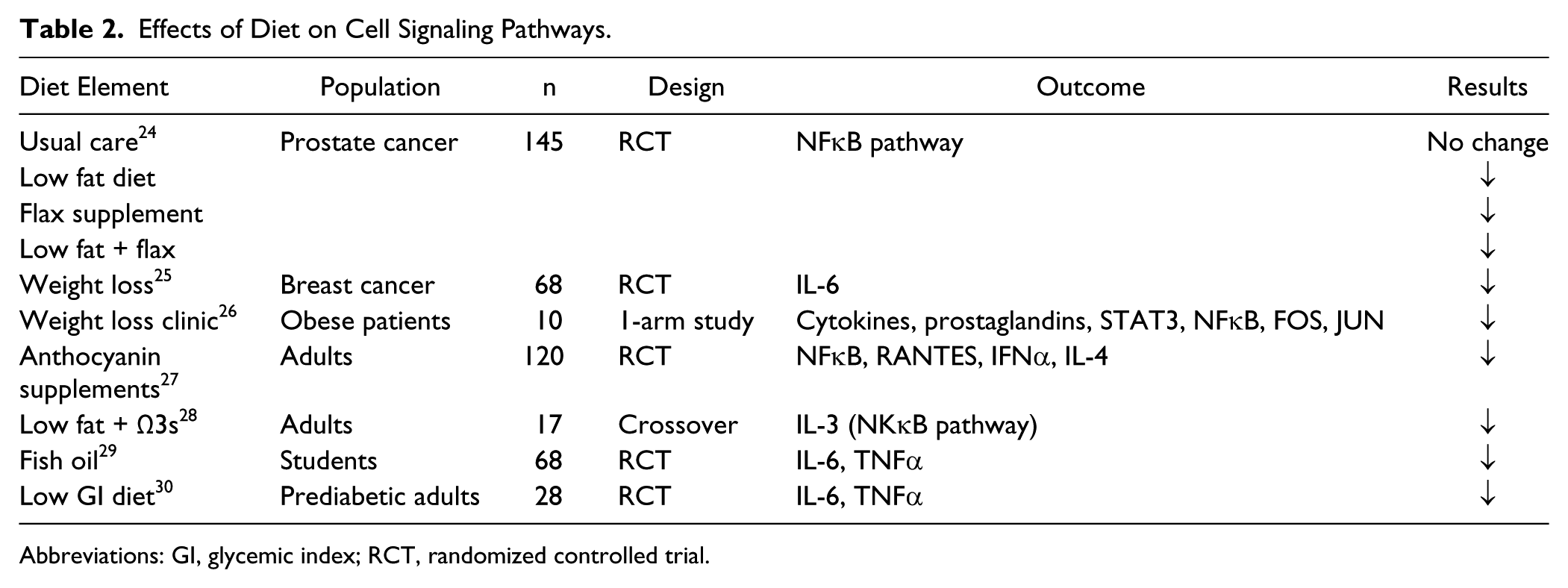
Abbreviations: GI, glycemic index; RCT, randomized controlled trial.
Table 2 shows the effects of diet on a variety of other molecular markers relevant to inflammation. Two studies show effects of dietary supplements (anthocyanin and fish oil), and the others show effects of dietary interventions. Only 2 of the studies were conducted in cancer patients, but similar effects on inflammatory markers are likely to be observed in healthy adults. These studies indicate that a variety of manipulations can alter cytokines and molecular markers. Dietary manipulation may be a means to effect large-scale change in biomarkers related to a number of cancer hallmarks.
Investigation of the connection between mind and emotions and cancer progression has given rise to the field of biobehavioral oncology or psychooncology. Randomized trials and mechanistic studies suggest that specific strategies for coping with the stresses of cancer can affect its biology. For instance, patients randomized to receive palliative care beginning at the time of lung cancer diagnosis lived longer than those who received such care only when close to death. The palliative care intervention consisted only in an average of 4 visits to discuss topics such as pain control and quality of life. 31 Several lines of evidence point to the hypothalamic-pituitary axis and the sympathetic-adrenal system as mediators for bodily impacts on stress that could affect survival. 32 In part, these may be affected by changes in circadian rhythmicity, documented to affect angiogenesis. 33 Additionally, behavioral influences on cellular immune response, angiogenesis, inflammation, chemotherapy resistance, and other processes have been documented.34,35 Stress hormones may shield cancer cells from anoikis, a form of programmed cell death that retards metastasis. 36 Table 3 lists randomized controlled trials and observational studies that indicated impacts of biobehavioral interventions and social-psychological conditions that affect disease outcomes and molecular targets of interest in cancer control.
Selected Biobehavioral Studies and Cell Signaling Pathways.

Abbreviations: EMT, epithelial-mesenchymal transition; RCT, randomized controlled trial.
The relevance of physical activity for cancer risk and cancer survival has begun to receive substantial research attention in the past decade. While designing interventions that successfully promote exercise is challenging, Table 4 shows that studies are succeeding in modifying cancer-related outcomes. In general, there are fewer studies dealing directly with molecular targets and exercise. The largest numbers of exercise-related studies deal with immunity and with effects of exercise on hormones in hormonally related cancers. However, exercise studies do examine other molecular targets, as well as effects on characteristics of patients that can be expected to influence multiple targets. In particular, the meta-analysis by Fong et al 45 provides a strong rationale for the use of exercise by breast cancer patients by reducing IGF-1, body weight, body mass index, and depression. In addition to improvements in variables such as fatigue and quality of life, exercise boosts capacity for daily activity. A review of exercise and cancer risk reduction cites convincing evidence of decreased risk of colon cancer with increased physical activity, probable evidence for postmenopausal breast and endometrial cancers and suggestive evidence for lung, prostate, ovary, gastric, and pancreatic cancers. Proposed mechanisms include reductions in adiposity, sexual and metabolic hormones, insulin resistance, and inflammation, with increases in immune function. 46 An anti-inflammatory effect of exercise might help prevent cancer cachexia and counteract muscle catabolism by reducing protein degradation. 47 However, it is difficult to assure that patients achieve exercise goals. 48
Selected Studies of Exercise and Cancer.

Abbreviations: RCT, randomized controlled trial; BMI, body mass index.
In summary, studies on effects of diet, biobehavioral strategies, and exercise on molecular targets related to cancer have been, and continue to be conducted, with promising results. Dietary interventions have been the most thoroughly studied, with an emphasis on markers related to inflammation predominating in the studies reviewed. The effects of biobehavioral strategies have been examined for a diverse set of targets. Exercise studies have emphasized immunity and hormonal signaling related to cancer. Lifestyle interventions may be ideal for altering a large number of targets in ways that will be beneficial to both cancer prevention and cancer control. Certainly further research conducted to evaluate the impact of an integrative system of multitargeted treatment regimens would help clarify the combined effect of these approaches on cancer outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
