Abstract
Keywords
Introduction
Lung cancer is the most common cause of cancer death worldwide, 1 and most patients have advanced-stage disease at diagnosis. Despite treatment-related advances, the prognosis of lung cancer remains poor with a 5-year survival rate of 4% in distant-stage disease.2,3 Epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) therapy is a promising treatment for non–small cell lung cancer (NSCLC). Specific mutations in the tyrosine kinase domain of the EGFR gene are associated with favorable EGFR-TKI therapy-related clinical outcomes 4 in patients with NSCLC. Most mutations are present in exons 18 to 21 of the EGFR gene, 5 and are frequently observed in lung adenocarcinoma. 6 EGFR mutations are found in <10% of non-Asian NSCLC patients 7 and in 30% to 50% of East Asian patients. 8 Missense mutations in exon 21 (L858R) and in-frame deletions within exon 19 are the most frequently occurring EGFR-TKI-sensitive mutations (80%) in patients with NSCLC. 9 EGFR mutations in patients with lung cancer are associated with a favorable response to the administration of EGFR-TKIs, 10 such as gefitinib, 11 erlotinib, 12 and afatinib, 13 versus standard chemotherapy (CT).
Metfromin (N′,N′-dimethylbiguanide) has been a standard drug for the treatment of type 2 diabetes mellitus (T2DM) for more than 50 years. A lower cancer-related mortality has been noted in T2DM cancer patients with metformin use compared with those with sulfonylurea and insulin use. 14 Recently, metformin was observed to decrease the incidence of lung cancer in T2DM patients 15 and was associated with a decreased mortality in T2DM lung cancer patients receiving CT.16,17 The synergistic effect of metformin and EGFR-TKI was reported recently in a retrospective clinical study. 18
In this study, we proposed that metformin use may enhance the effect of EGFR-TKI and prolong survival in T2DM patients with lung cancer receiving this therapy. A nationwide population-based study was conducted to determine the effect of metformin use in patients with T2DM and lung cancer receiving EGFR-TKI therapy.
Material and Methods
Data Source
The National Health Insurance (NHI) is a compulsory program for all Taiwan residents. The Taiwan National Health Insurance Research Database (NHIRD)—a comprehensive health care database that covers nearly the entire 23.7-million-strong population of this country—was used in our study. Data on patients’ characteristics, such as sex and date of birth, and information regarding admissions and outpatient visits, including date of admission, date of discharge, dates of visits, and up to 5 discharge diagnoses or 3 outpatient visit diagnoses, were collected. The International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes were used for diagnosis. All patients with “catastrophic illnesses” were also included in this database. The Ethics Review Board of Chang Gung Memorial Hospital, Chiayi Branch, Taiwan, approved this study (201600067B1). The data used in this study were analyzed anonymously in accordance with strict confidentially guidelines and regulations regarding personal electronic data protection. The requirement for informed consent was waived by the institutional review board.
Study Cohorts
We identified all patients with DM (ICD-9-CM 249-250) and an initial and primary diagnosis of lung cancer (ICD-9-CM 162) between January 1, 2004, and December 31, 2012, from the NHIRD. Patients who underwent EGFR-TKI therapy (gefitinib or erlotinib) were included, while those diagnosed with other types of cancer, aged <40 years, or with insulin use were excluded. Since insulin is used in the treatment of type 1 DM, all the patients in our study were T2DM patients. The metformin cohort comprised patients who had taken metformin for more than 28 cumulative defined daily doses after a diagnosis of lung cancer and with the use of EGFR-TKI (Figure 1). All participants were followed-up till the end of 2013.
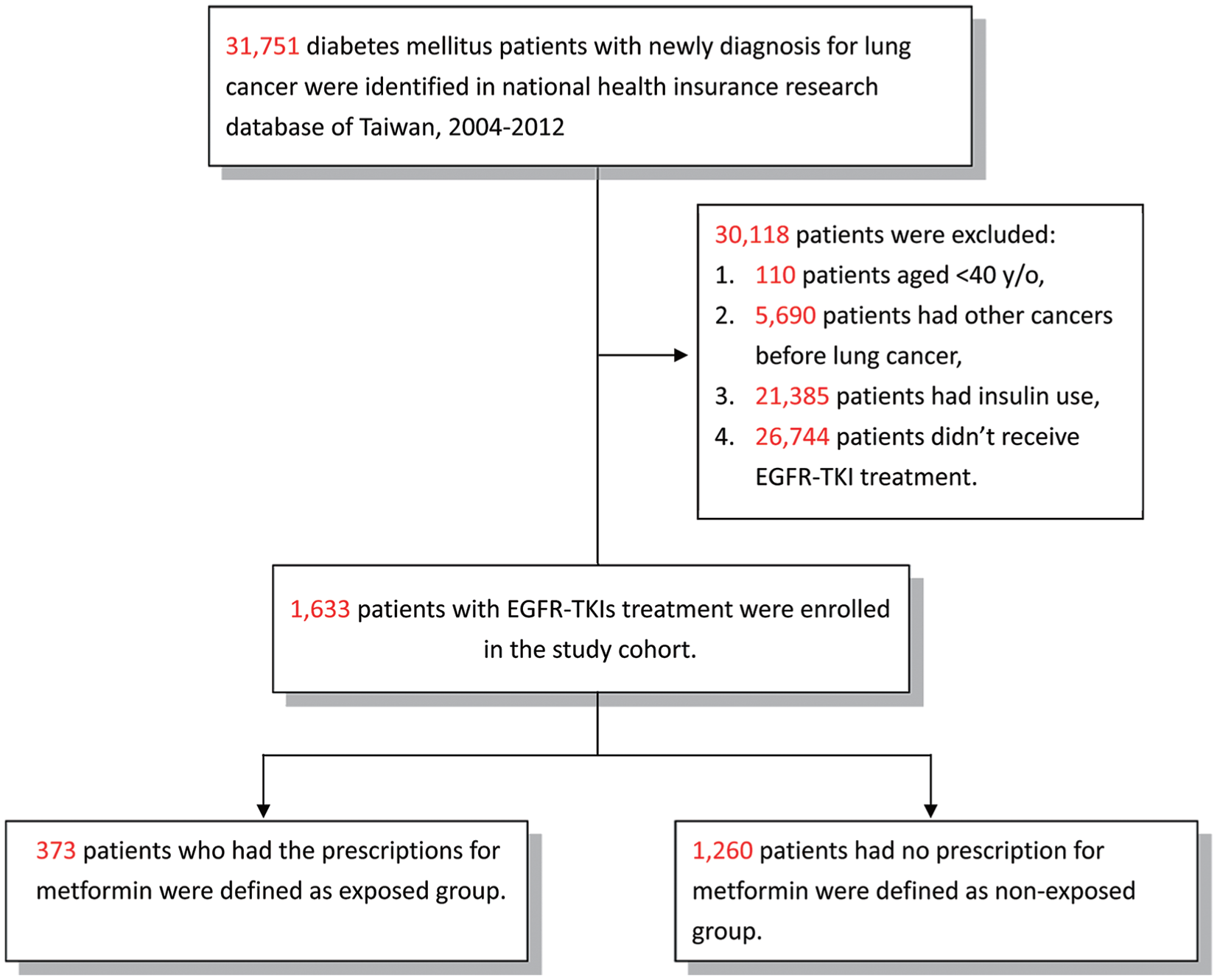
Flowchart of the patient enrollment process of the metformin cohort and matched non-metformin cohort.
Demographic Variables and Comorbidities
Demographic variables, including age, sex, income for the estimation of insurance payment, and urbanization of the participants’ residential areas, were included. Monthly income was categorized as follows: ≤NT$15 840, NT$15 841 to $25 000, and ≥NT$25 000. The urbanization level was categorized as “very high,” “high,” “moderate,” or “low,” based on the population density. 19 Hypertension (ICD-9-CM 401-405), coronary artery disease (ICD-9-CM 414-419), stroke (ICD-9-CM 430-438), renal insufficiency (ICD-9-CM 585, 586), chronic obstructive pulmonary disease (COPD; ICD9-CM 491, 492, 496), and smoking-related disorders (ICD9-CM 305.1, 491.2, 492.8, 496, 523.6, and V15.82) were included as comorbidities. Radiotherapy (RT), CT, or both (concurrent chemoradiotherapy [CCRT]) were also included in our study. EGFR-TKI responders were defined as patients who received EGFR-TKI therapy for more than 90 days; the remaining patients were defined as nonresponders. 20 The CT regimens before EGFR-TKI therapy were also included in this study.
The EGFR-TKIs used were approved by the NHI in November 2007 (gefitinib) and June 2008 (erlotinib) for the treatment of stage IIIB or IV lung cancer as the second-line therapy for lung adenocarcinoma and the third-line therapy for NSCLC (erlotinib), and in June 2011 and November 2013, as the first-line therapy for lung adenocarcinoma with EGFR mutations (gefitinib and erlotinib, respectively), in Taiwan. The performance of imaging studies and the application of EGFR-TKI therapy every 3 months were requested in patients who received EGFR-TKI therapy. EGFR-TKI therapy was declined by the NHI once progressive disease was observed. Since the results of imaging studies were not available in the NHIRD, we alternatively defined progression-free survival (PFS) in our study as the interval from the beginning to the end of EGFR-TKI therapy. All patients receiving second-line gefitinib and erlotinib therapy in our study had adenocarcinoma, and those receiving third-line erlotinib therapy had NSCLC. Since data on the EGFR mutation status are not available in the NHIRD, EGFR-TKI responders were used as surrogates of EGFR mutations in our study. Overall survival (OS) was defined as the time from diagnosis to any cause of death or the time of censoring at the last follow-up.
Statistical Analysis
The differences in the demographic characteristics and comorbidities between the metformin and non-metformin cohorts were examined by the χ2 test. The hazard ratios (HRs) and 95% confidence intervals (CIs) of the risk of death for metformin users compared with the comparison cohort were examined by Cox proportional hazard regression analysis. Survival analysis was performed using Kaplan-Meier analysis and a log-rank test. A multivariate Cox proportional hazards model was used to determine the risk factors of mortality in patients with lung cancer and their adjusted HR within the metformin cohort. All analyses were conducted using SAS statistical software (Version 9.4; SAS Institute, Cary, NC).
Results
Differences in Demographic Characteristics and Comorbidities Between the Metformin and Non-Metformin Diabetes EGFR-TKI Lung Cancer Cohorts
A total of 1633 patients with T2DM and lung cancer, undergoing EGFR-TKI therapy from 2004 to 2012, were included in our study. Of these patients, 373 patients were enrolled in the metformin cohort and 1260 patients in the non-metformin cohort. The metformin cohort had a significantly higher presence of hypertension, less COPD, renal insufficiency, and smoking-related disorders than the nonuser cohort (Table 1). The metformin cohort also had a significantly higher proportion of patients without CT or/and RT, and with gefitinib use. In the non-metformin cohort, 103 of the 1260 patients received oral antidiabetic agents (Table S1, available online) after the diagnosis of lung cancer. The average years from the diagnosis of T2DM to the diagnosis of lung cancer in the metformin (6.7 ± 4.7 years) and non-metformin (6.9 ± 4.3 years) cohorts were not significantly different (P = .419).
Characteristics of NSCLC Patients Undergoing EGFR-TKI Therapy.

Abbreviations: NSCLC, non–small cell lung cancer; EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor; IQR, interquartile range; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CT, chemotherapy; RT, radiotherapy; CCRT, concurrent chemoradiotherapy.
P < .05.
Clinical Variables and HRs of Death in Diabetes Patients With Lung Cancer Undergoing EGFR-TKI Therapy
The risks of death in the metformin user and nonuser cohorts were then compared with the different clinical variables. In the univariate analysis, a reduced risk of death was observed in the metformin users (HR: 0.69, 95% CI: 0.59-0.81, P < .001), those from very high urbanization areas (HR: 0.74, 95% CI: 0.60-0.92, P = .0056), and the EGFR-TKI responder group (HR: 0.34, 95% CI: 0.30-0.39, P < .001). An increased risk of death was observed in patients with male sex (HR: 1.60, 95% CI: 1.42-1.81, P < .001), a monthly income of 15 841 to 25 000 NT$ (HR: 1.20, 95% CI: 1.04-1.39, P = .0134), stroke (HR: 1.21, 95% CI: 1.06-1.38, P = .0042), smoking-related disorders (HR: 1.37, 95% CI: 1.19-1.57, P < .001), CCRT (HR: 1.23, 95% CI: 1.01-1.51, P = .044), and RT (HR: 1.65, 95% CI: 1.26-2.17, P < 0.001; Table 2).
Comparison of Hazard Ratios.

Abbreviations: HR, hazard ratio; CI, confidence interval; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CT, chemotherapy; RT, radiotherapy; CCRT, concurrent chemoradiotherapy; EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor.
P < .05.
After adjustment for metformin use, age, sex, urbanization, income, hypertension, stroke, CAD, COPD, smoking-related disorders, CT/RT, EGFR-TKI response, and the regimens used before EGFR-TKI therapy, a reduced risk of death was still observed in the metformin users (HR: 0.73, 95% CI: 0.62-0.85, P < .001), those from very high urbanization areas (HR: 0.78, 95% CI: 0.62-0.98, P = .0321), COPD (HR: 0.81, 95% CI: 0.69-0.96, P = .0124), and the EGFR-TKI responders (HR: 0.34, 95% CI: 0.30-0.39, P < .001). An increased risk of death was observed in patients with male sex (HR: 1.41, 95% CI: 1.24-1.61, P < .001), stroke (HR: 1.16, 95% CI: 1.01-1.33, P = .0409), smoking-related disorders (HR: 1.36, 95% CI: 1.14-1.63, P = .0009), and RT (HR: 1.46, 95% CI: 1.11-1.93, P = 0.0075; Table 2).
HRs of Death in Subpopulations Treated With Metformin
The risk of death after metformin use was then evaluated in subpopulations of diabetic lung cancer patients with EGFR-TKI therapy. All subpopulations of metformin users stratified by sex, age, hypertension, stroke, CAD, COPD, and renal insufficiency before EGFR-TKI treatment had a significantly reduced risk of death (Table 3). A significantly reduced risk of death was also observed in patients without smoking-related disorders, and patients with CCRT, CT, RT, gefitinib, or ≤1 CT regimen before EGFR-TKI therapy (Table 3).
Adjusted Hazard Ratios of Mortality in Subpopulations Treated With Metformin.

Abbreviations: HR, hazard ratio; CI, confidence interval; CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CT, chemotherapy; RT, radiotherapy; CCRT, concurrent chemoradiotherapy; EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor; NSCLC, non-small-cell lung cancer.
HRs were adjusted for sex, age, urbanization, income, hypertension, stroke, CAD, COPD, renal insufficiency, smoking-related disorders, CT/RT, CT regimens used before EGFR-TKI therapy.
P < .05.
PFS and OS in the Metformin and Non-Metformin Diabetes Patients With Lung Cancer Undergoing EGFR-TKI Therapy
PFS and OS were also evaluated among the metformin users and nonusers. Metformin use was associated with a significantly longer median PFS (9.2 months, 95% CI: 8.6-11.7 vs 6.4 months, 95% CI: 5.9-7.2, P < .001; Figure 2), and OS (33.4 months, 95% CI: 29.4-40.2 vs 25.4 months, 95% CI: 23.7-27.2, P < .001; Figure 3).

Progression-free survival curve of the metformin and non-metformin cohorts.

Overall survival curve of the metformin and non-metformin cohorts.
Discussion
In this retrospective, nationwide, longitudinal cohort study, we observed that metformin use was associated with a decreased risk of death, and prolonged PFS and OS in patients with T2DM and lung cancer receiving EGFR-TKI therapy.
The underlying mechanism of how metformin enhances the effect of EGFR-TKIs in lung cancer has been explored in several studies. Metformin increases the sensitivity of EGFR-TKI-resistant lung cancer cells to erlotinib or gefitinib through the inhibition of interleukin-6 signaling and reversal of epithelial-mesenchymal transition. 21 Metformin has antitumor effects via the inhibition of the mTORC1 through either AMP kinase-dependent or independent signaling pathways, 22 and also inhibits the PI3K/AKT/mTOR signaling pathway. 23 Since the aberrant activation of the PI3K/AKT/mTOR signaling pathway is one of the mechanisms behind the acquired resistance to EGFR-TKI therapy in patients with adenocarcinoma and EGFR mutations, 24 the use of metformin in combination with an EGFR-TKIs could produce a synergistic antitumor effect on lung cancer cells.
In our study, an increased risk of death was observed in patients with male sex, stroke, COPD, smoking-related disorders, and RT. These findings are similar to those of previous studies. Male sex has a negative impact on EGFR-TKI therapy outcomes compared with female sex. 25 An increased risk of stroke was reported in patients with lung cancer. 26 The administration of RT may imply a more advanced stage of lung cancer, such as the presence of brain or bone metastasis, and the prognosis of such patients is poor. 27 Smoking is a risk factor of NSCLC and is associated with a lower response to EGFR-TKI therapy. 28
As data on the EGFR mutation status is not available in the NHIRD, we alternatively used EGFR-TKI responders as surrogates for EGFR mutations.20,29 In the subgroup analysis, EGFR-TKI responders had a reduced risk of death in the metformin cohort, implying that metformin may have protective effects mainly in lung patients with EGFR mutations. The protective effect of metformin was also more prominent in patients with first-line or second-line gefitinib use, owing perhaps to the smaller number of patients with erlotinib use, or the different effects of metformin on gefitinib or erlotinib. The protective effect of metformin was also more prominent in patients who received CT, RT, or both, which is consistent with the findings of previous studies in which metformin enhanced the effect of CT 30 and RT. 31
DM is a poor prognostic factor in lung cancer patients.29,32 In our study, metformin users had a prolonged OS of up to 33.4 months. The OS in our study is similar to that observed in previous studies focusing on the first-line treatment of patients with EGFR mutation-positive advanced NSCLC (23.6 to 30.5 months).12,33 This implies that metformin use enhances the therapeutic effects of EGFR-TKI in patients with T2DM.
Our study has some limitations. Data on lung cancer stage, pathology, symptoms, physical status, smoking status, and genetic factors are not available in the NHIRD. The immortal bias could be a confounding factor in our study. In our study, patients in both the metformin and non-metformin cohorts received EGFR-TKI therapy after the diagnosis of lung cancer. We also reviewed our data and observed that no patients died in the non-metformin cohort within the 28 days immediately after the diagnosis of lung cancer. As a result, we assumed that the effects of immortal bias in our study could be minimal. Nevertheless, further prospective randomized controlled trials are needed to verify our findings.
Conclusions
In conclusion, our study showed that metformin use potentially enhances the therapeutic effect and decreases the mortality in T2DM patients with lung cancer receiving EGFR-TKI therapy. Our results suggest that in T2DM lung cancer patients with EGFR-TKI therapy, metformin could be the preferred oral hypoglycemic agent.
Supplemental Material
Table_S1 – Supplemental material for Metformin Prolongs Survival in Type 2 Diabetes Lung Cancer Patients With EGFR-TKIs
Supplemental material, Table_S1 for Metformin Prolongs Survival in Type 2 Diabetes Lung Cancer Patients With EGFR-TKIs by Ming-Szu Hung, Min-Chun Chuang, Yi-Chuan Chen, Chuan-Pin Lee, Tsung-Ming Yang, Pau-Chung Chen, Ying-Huang Tsai and Yao-Hsu Yang in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors would like to thank the Health Information and Epidemiology Laboratory (CLRPG6G0042) for the comments and assistance in the data analysis. This study was based on the National Health Insurance Research Database provided by the Central Bureau of National Health Insurance, the Department of Health, and managed by the National Health Research Institutes. The interpretation and conclusions contained herein do not represent those of the Bureau of National Health Insurance, Department of Health, or National Health Research Institutes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant (CLRPG6G0042) from Chang Gung Memorial Hospital, Chiayi Branch.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
