Abstract
EGFR mutations comprise a sizeable portion of non-small cell lung cancers. While the most common EGFR mutation consists of exon 19 in-frame deletions and exon 21 point mutations, rare EGFR mutations have become a more frequent occurrence. Currently, no clinical guidelines exist for the treatment of such mutations. In this case, we see a 68-year-old non-small cell lung cancer male patient with a history of smoking presenting with a rare exon 20 R776H EGFR mutation who demonstrates a response to Osimertinib, further exploring potential standard treatments for patients with rare EGFR mutations.
Introduction
Lung cancer is the second most common cancer by incidence and the leading cause of cancer-related deaths. Non-small cell lung cancer (NSCLC) comprises the majority (84%) of clinical lung cancer cases. 1 Specific cases of NSCLC can be categorized depending on the specific oncogenic driving mutation. One such mutation is that in epidermal growth factor receptors (EGFR).
EGFR mutations comprise a sizeable (32.3%) portion of NSCLC cancers. Specific incidence rates vary across ethnicities. EGFR mutations are more common within Asian population (38.1%) than in European (14.1%). 2 The most common EGFR mutations consist of exon 19 in-frame deletions and exon 21 point mutations. 3 However, rare mutations are found in around 12% of NSCLC cases. 4 In this case, we see a patient with a rare R776H mutation and document a response to Osimertinib.
Case presentation
A 68-year-old male, heavy smoker, with a family history of lung cancer presented to the hospital after recent admission to the emergency department (ED) for respiratory distress, cough and hemoptysis. The patient had imaging completed in the ED including a computed tomography angiography chest which revealed a 1.24 cm and two 7 mm spiculated masses in the right middle and lower lobe, with right upper lobe collapse. Pulmonary was consulted and the patient underwent endobrachial ultrasound scan and liquid biopsies of stations 4 L, 4 R and 7. Abdominal and pelvic imaging was completed which revealed mild thickening in the rectosigmoid junction with diverticulosis of the sigmoid. The patient noted 40 lb unintentional weight loss over the year but otherwise outside of the aforementioned respiratory issues, review of systems was negative.
The liquid biopsy was prepared in the standard fashion according to Guardant360 instructions for plasma 5 and cfDNA isolation and following CLSI GP41 6 blood draw guidelines. Biopsies revealed positive malignant cells characteristic of metastatic pulmonary adenocarcinoma at lymph node stations 4, 4 L and 4 R. (Figures 1 and 2). Guardant360 next-generation sequencing detected mutations EGFR R776H (0.2%), BRCA2 N1201fs (0.4%), TP53 R282W (5.7%), FBXW7 D527fs (2.9%), NTRK3 Q662H (2.2%) and FGFR3 P798Q (2%) (Table 1).

(a) Lung FNA, highly cellular smears from aspirate (Diff Quik stain, low power). (b) Cohesive cluster of malignant cells with pleomorphic nuclei and vacuolated cytoplasm (Diff Quik stain, high power). (c and d) groups of malignant cells with atypical nuclei and vacuolated cytoplasm (H&E stain, low and intermediate power).

Immunohistochemical stains. (a and b) The neoplastic cells display positive cytoplasmic/membranous pankeratin stain (AE1/AE3, intermediate and high power). (c) Napsin A is positive with granular cytoplasmic stain (Napsin A, high power), (d) Thyroid Transcription Factor 1 ( TTF-1) is expressed as strong and diffuse nuclear staining (TTF-1, high power).
Guardant360 liquid biopsy results.

Positron emission tomography/computed tomography (PET/CT) staging scans were conducted revealing stage IV non-small cell lung carcinoma including a confluent large mass right upper lobe with bronchial obstruction, and central necrosis which could be consistent with a post-obstructive component. Additional fluorodeoxyglucose avid lymph nodes including cervical, supraclavicular, and mediastinal with the largest nodal disease burden at the R hilum and L supraclavicular/cervical stations. Multiple pulmonary nodules were also seen. After consulting the literature and presenting the patient to tumor board, Osimertinib was considered the beneficial treatment option and treatment began.
To date, the patient has shown a response. A staging CT 6 months after treatment commencement demonstrated an interval decrease in size of some bilateral pulmonary nodules and mediastinal lymph nodes. Based on RECIST v1.1 criteria, 7 a right upper lobe nodule measured 8.4 cm × 6.2 cm which previously measured 11.5 cm × 9.1 cm, a right lower lobe nodule posteriorly measured up to 1.1 cm which previously measured up to 1.2 cm. A significant decrease was also seen in the size of left lower lobe nodule. No further new sites of disease were identified (Figure 3(a) and (b)). A CT scan 3 months after the initial shrinkage demonstrated stability from the lung standpoint.
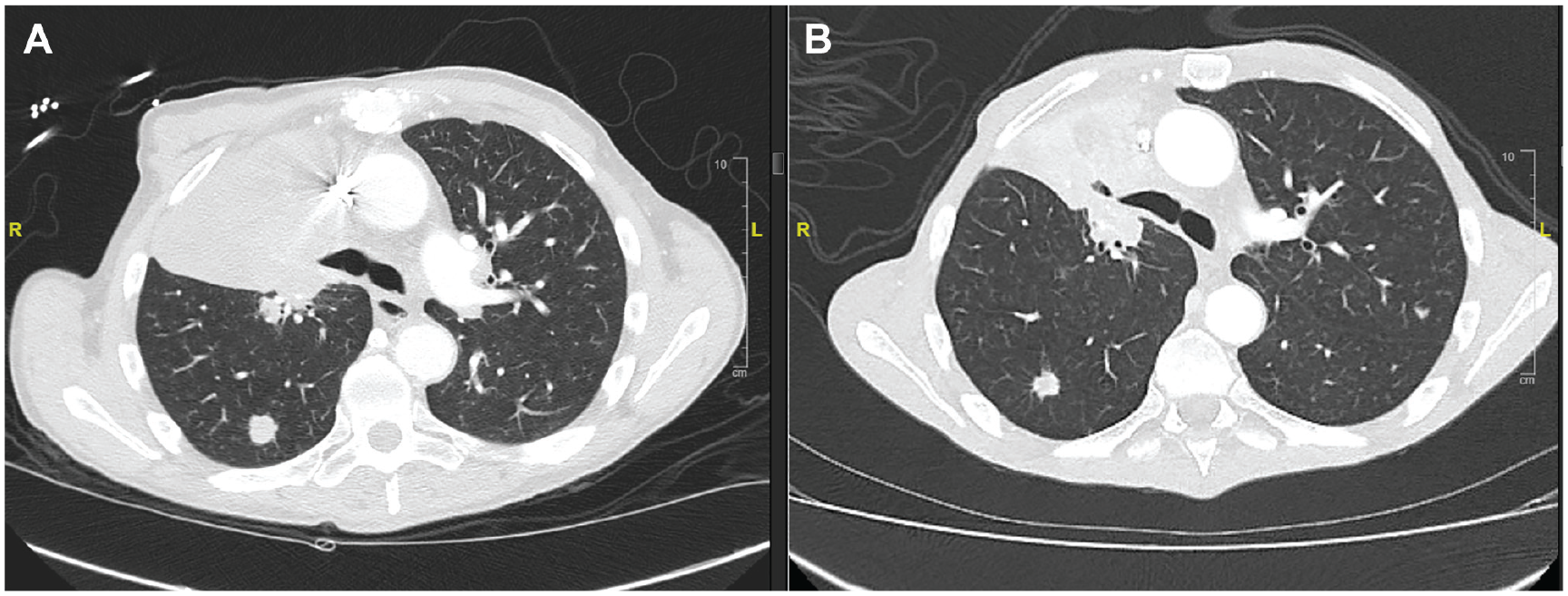
(a) Chest CT before Osimertinib initiation. (b) Chest CT 6 months after Osimertinib initiation.
Discussion
Within this case study, we strive to document the response of a patient with a rare R776H EGFR mutation to Osimertinib. According to the literature, an exon 20 missense mutation, the R776H mutation, has only been documented 12 times within the COSMIC database. This mutation has also been seen to co-occur with other rare EGFR mutations such as EGFR G724S 3 and has been observed as a germline mutation with transmission between generations. 8
In all documentation of the R776H EGFR mutation, the treatment schedule has followed the use of tyrosine kinase inhibitors (TKIs). A study demonstrated that EGFR G724S/R776H mutations showed good response, particularly towards Afatinib 3 which was also seen in the case of the germline mutation, where both a mother and daughter responded well to Afatinib and other treatments with TKIs. 8 Furthermore, studies have documented the effectiveness of second and third-generation TKIs such as Osimertinib in the treatment of this mutation. 9
Current research has also demonstrated that second and third-generation TKIs are more effective than first-generation TKIs in the treatment of rare EGFR mutations in NSCLC. 9 The phase III FLAURA trial which compared Osimertinib versus Gefitinib/Erlotinib demonstrated that progression free survival (PFS) was significantly improved in the Osimertinib arm (18.9 vs 10.2 months) with a higher duration of response (17.2 vs 8.5 months). 9 Regarding the R776H mutation itself, a retrospective study demonstrated PFS of 11 and 10 months in two patients with R776H mutations who were treated using Osimertinib. 10 Therefore, Osimertinib could prove to be an effective treatment for increasing the survival outcomes of patients with R776H mutations. For patients refractory for Osimertinib and chemotherapy, future directions point towards Amivantamab and Lazertinib which demonstrate encouraging antitumor activity in the aforementioned populations. 11
Through mutation genotyping, more and more EGFR rare mutations are being identified. However, no current clinical guidelines exist to guide the treatment of such mutations. Therefore, by deepening our understanding of the effectiveness of different treatments in survival outcomes for rare EGFR mutations, clinical guidelines can be developed to better standardize treatment for these individuals.
Regarding the limitations of this study, liquid biopsy has been shown to have limited sensitivity, so repeat sampling for driver mutations may need to be conducted in the future. Furthermore, while the EGFR R776H mutation in this case may be a primary driver based on current clinical data points, other case reports have mentioned the possibility that the EGFR R776H mutation may be germline. 8 Therefore, it may be appropriate to consider thorough/comprehensive germline testing in addition to somatic testing.
Conclusion
Overall, we see a 68-year-old NSCLC male patient with a history of smoking with a rare exon 20 R776H EGFR mutation who demonstrates a response to Osimertinib. As there are no current clinical guidelines for the treatment of rare exon 20 mutations this case can serve to deepen our understanding of optimal treatments for such mutations and consider the use of TKIs for these patients. Looking ahead, more patients with rare EGFR mutations need to be examined to further increase our understanding of the efficacy of current treatments, and to develop new effective treatments that are individualized for specific rare EGFR mutations.
Footnotes
Acknowledgements
We would like to acknowledge the patient for allowing the study to be published.
Author contributions
Joyce Cui: Writing original draft, review & editing. Sana Tabbara: Data curation, visualization. Juskaran Chadha: Data curation, supervision, writing: review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for anonymized patient information to be published in this article.
