Abstract
Background:
Lung cancer, especially non-small cell lung cancer (NSCLC), poses a significant health challenge globally due to its high mortality. Afatinib, a second-generation epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI), has shown superior efficacy over traditional chemotherapy in NSCLC treatment. However, issues like secondary resistance and adverse effects call for alternative therapies. HAD-B1, comprising 4 herbal medicines, has shown promise in lung cancer treatment in both preclinical and clinical settings. This study assesses the combination of HAD-B1 and Afatinib in advanced NSCLC patients to potentially improve outcomes by addressing the limitations of current EGFR-TKI therapies.
Method:
A randomized, open-label trial evaluated the efficacy and safety of HAD-B1 with Afatinib in 90 EGFR-mutation-positive NSCLC patients. Participants were divided into treatment and control groups, receiving Afatinib with or without HAD-B1. The study focused on the initial dose maintenance rate and disease control rate (DCR) of Afatinib, alongside secondary outcomes like survival rates and quality of life, under continuous safety monitoring.
Results:
Among the 90 participants, no significant difference was found in initial dose maintenance (60.98% in the treatment group vs 52.50% in the control, P = .4414) or DCR (80.49% vs 90.00%, P = .2283). Secondary outcomes like PFS, TTP, and OS showed no notable differences. However, physical functioning significantly improved in the treatment group (P = .0475, PPS group). The control group experienced higher rates of adverse events of special interest and adverse drug reactions (P = .01), suggesting HAD-B1 with Afatinib might enhance physical function without increasing adverse effects.
Conclusion:
Combining HAD-B1 with Afatinib potentially improves quality of life and reduces adverse events in advanced NSCLC patients. Further research is necessary to confirm the long-term benefits of this combination therapy, aiming to advance NSCLC treatment outcomes.
Trial registration:
Clinical Research Information Service (CRIS) of the Republic of Korea, https://cris.nih.go.kr/ (ID: KCT0005414).
Introduction
Lung cancer is the leading cause of cancer-related death worldwide, with 2.4 million new diagnoses and 1.8 million deaths that occurred in 2022. 1 In South Korea, there were 33 413 new cases and 18 536 deaths in 2023, 2 highlighting a significant health burden due to its high incidence rates both worldwide and in South Korea. Non-small cell lung cancer (NSCLC) is the predominant subtype, accounting for 85% of these cases. 3 Among these, approximately 10%-15% of NSCLC patients harbor epidermal growth factor receptor (EGFR) mutations. 4 In accordance with the current NCCN guidelines, the first-line treatment for patients with EGFR mutations is osimertinib. 5 However, afatinib (GIOTRIF®; Boehringer Ingelheim, Ingelheim, Germany) remains an alternative option for these patients.6,7 Afatinib may be considered for patients who have exon 19 deletions or when there is a benefit in using afatinib as the first-line treatment and reserving osimertinib for second-line therapy. 8 In a real-world clinical study, about 15% of patients with EGFR mutations received afatinib as their first-line treatment under these circumstances. 9 However, its clinical utility is hindered by adverse events (AEs), predominantly gastrointestinal and dermatologic, impacting patient quality of life (QoL) and treatment adherence. Insights from phase 3 trials underscore efficacy of Afatinib, but its safety profile, akin to first-generation EGFR inhibitors, necessitates proactive management. Notably, recent animal studies have highlighted a dose-dependent relationship between afatinib and gastrointestinal epithelial damage, suggesting persistent effects even post-treatment discontinuation. 10 Effective AE management strategies are crucial to optimizing afatinib therapy and improving patient outcomes.
Complementary and alternative medicine, particularly herbal medicine, has garnered interest as adjuvant therapy in cancer treatment, aiming to enhance therapeutic efficacy and manage side effects. In NSCLC treatment, many studies have looked at combining herbal medicine with EGFR-tyrosine kinase inhibitors (EGFE-TKIs) to improve treatment outcomes. These studies found that this combination can prolong progression-free survival (PFS), reduce side effects, and increase the 5-year survival rate.11 -14 HAD-B1, a Traditional Korean Medicine herbal formulation, has emerged as a promising adjuvant therapy for lung cancer patients in South Korea. Comprising Panax Notoginseng Radix, Panax ginseng C. A. Meyer, Cordyceps militaris, and Boswellia carteri Birdwood, HAD-B1 exhibits potential anticancer properties attributed to its constituent herbs. Panax Notoginseng Radix demonstrates anti-tumorigenic effects by modulating apoptotic markers, while Panax ginseng and Cordyceps militaris exert anticancer effects through various molecular pathways, including immune modulation and Hedgehog signaling inhibition.15-17 Boswellia carteri exhibits anti-inflammatory and anticancer effects via AKT inhibition and NF-κB downregulation. 18 HAD-B1 was additionally investigated as a formula, and it successfully suppressed the proliferation of A549-cisplatin-resistant cells in a dose-dependent manner. 19 In a preclinical study that investigated the inhibitory effect of HAD-B1 in combination with afatinib on H1975 lung cancer cells (L858R/T790M double mutation) in mice, the treatment group demonstrated significant downregulation of pERK1/2 and upregulation of p16 in the cells, as well as tumor volume reduction. 20 Finally, a multi-center, randomized, double-blind, placebo-controlled exploratory trial that investigated the efficacy, safety, and dosage-finding of HAD-B1 discovered that HAD-B1 showed partial efficacy and that the combination of afatinib and HAD-B1 of 972 mg/day was determined to be the most effective dose. 21
Therefore, in this study, the authors aim to evaluate efficacy and safety of using HAD-B1 with afatinib for locally advanced or metastatic NSCLC patients. The results of this study may suggest HAD-B1 as a potential safe and effective adjuvant therapy for NSCLC patients who are receiving EGFR-TKIs, especially afatinib.
Methods
Study Design
The clinical trial is a randomized, multi-center, open-label study to compare the safety and efficacy of HAD-B1 combined with afatinib therapy for patients with locally advanced or metastatic EGFR-mutation-positive NSCLC. The protocol was approved by Daejeon Korean Medicine Hospital IRB (IRB number: DJDSKH-20-DR-09) and was registered in the Clinical Research Information Service (CRIS) of Republic of Korea (ID: KCT0005414) on September 23, 2020. The clinical trial began on February 8, 2021 and completed on April 27, 2023. The last participant was enrolled on December 23, 2022. All participants were tracked for the entire planned 16-week duration of the treatment from their respective enrollment dates. Table 1 explains the procedures conducted at each visit (Figure 1 and Table 1).
Summary of Study Flow Chart.

Abbreviations: ECOG, eastern cooperative oncology group; EGFR, epidermal growth factor receptor; HRQoL, health-related quality of life.

Summary of study flow chart.
Study Participants
We originally aimed to recruit 142 patients, for the following reasons: the starting dose maintenance rate for afatinib in the afatinib group is assumed to be 57.6% based on the previous research, 21 while in the afatinib and HAD-B1 group, we hypothesized as 80.0%. Additionally, we hypothesized all patients to be recruited and observed for a minimum of 16 weeks. Therefore, the number of subjects in each group is 64. Considering a dropout rate of 10%, which would be 71 each group, this means a total of 142 participants. However, due to constraints imposed by the COVID-19 pandemic, as well as limitations in budget and time, the recruitment was restricted to 90 participants. The 90 patients with EGFR-mutation-positive locally advanced or metastatic NSCLC requiring afatinib therapy were enrolled from 6 centers, which are the Daejeon Korean Medicine University, Kosin University Gospel Hospital, Pusan National University, Yangsan Hospital, Konyang University Hospital, Catholic University of Korea Seoul St. Mary’s Hospital, and Ajou Universtity. All participants voluntarily agreed to participate in the clinical trial.
Participant selection was based on the following inclusion criteria:
Confirmed histopathological diagnosis of locally advanced or metastatic NSCLC (Stage IIIA-IV, per the eighth edition of the American Joint Commission on Cancer TNM staging system).
Presence of EGFR mutation requiring first-line afatinib therapy (determined by the researcher’s judgment).
Measurable disease status according to Response Criteria for Clinical Trials of Cancer (RECIST) 1.1.
Eastern Cooperative Oncology Group Performance Status (ECOG PS) score of 0-2.
Ability to orally administer the prescribed drug (as assessed by the researcher).
Age of 19 years or older.
Willingness to provide written consent for participation in the clinical trial.
Participant exclusion was based on the following exclusion criteria:
Presence of T790M mutation within the EGFR kinase domain (acquired mutation, requiring re-biopsy).
Active brain metastasis with stability of less than 4 weeks, accompanied by symptoms or leptomeningopathy, unless patients were on stable dexamethasone treatment for at least 4 weeks.
Severe or uncontrolled gastrointestinal disorder with significant diarrhea within 2 weeks of screening, such as CTC grade 2 or higher pathological diarrhea (per researcher’s assessment).
Deemed unsuitable for the trial due to interstitial lung disease, as evaluated by the researcher.
Severe hepatopathy (Child Pugh C).
Severe nephropathy (eGFR <15 ml/minute) or requiring dialysis.
Clinically significant cardiovascular disease (NYHA class 3 congestive heart failure, unstable angina, uncontrolled arrhythmia, myocardial infarction, angina within 1 year prior to study participation, etc.).
Fertile women not using effective contraception prior to the trial.
Pregnant or breastfeeding women.
Suspected or diagnosed with severe mental illnesses, substance abuse, alcoholism, etc.
Hypersensitivity to afatinib or other EGFR-targeted medications.
Hypersensitivity to HAD-B1 or its components.
Participation in other clinical trials within the month preceding this study.
Deemed unsuitable for this clinical trial based on researcher’s judgment, including severe infectious disease or organ failure.
Randomization
In this clinical trial, subjects were assigned consecutive random assignment numbers, with allocation to the treatment or control group determined by a pre-generated random assignment table. Allocation was based on an advanced assignment code, with block sizes of 4 or 8 and a 1:1 ratio for group proportions. Random distribution was performed sequentially using either SAS system B or C.1. Manufacturing followed KGMP standards, with drug labeling preceding distribution. The random number list guided allocation, with selection criteria registered sequentially. To mitigate bias, testers received blind random assignment bags, opened only at the time of assignment disclosure to subjects. Participants were assigned in a 1:1 ratio to the treatment group or control group.
Intervention
Patients in the treatment group were given afatinib (30-40 mg/day orally) combined with HAD-B1 pills (972 mg/day, twice daily orally, 486 mg each). The control group received only afatinib (30-40 mg/day orally). Any dosage modifications for afatinib was decided by the investigator’s medical judgment; however, the patients were encouraged to begin with 40 mg in anticipation of future dose reduction. HAD-B1 administration ceased if afatinib was discontinued in the treatment group, while afatinib dosage variation had no effect on HAD-B1 dosage. HAD-B1 tablets were purchased from Hanpoong Pharmaceutical Co., Ltd. (Seoul, Korea).
Outcome Measures
The primary measurement is initial dose maintenance rate for afatinib and the disease control rate (DCR). Secondary outcomes encompassed control of the PFS assessed by RECIST 1.1, overall survival (OS), objective response rate (ORR), tumor size reduction, time to progression (TTP), health-related quality of life (HRQoL), and tumor markers (Carcinoembryonic antigen, CEA) between the 2 groups. The PFS, ORR, and TTP were evaluated based on RECIST 1.1 criteria.
Safety Assessments
Throughout the study, subjects underwent continuous monitoring for adverse events, including paronychia, loss of appetite, diarrhea, stomatitis, cheilitis, rash, acne dermatitis, interstitial lung disease, and severe hepatopathy. The comprehensive safety profile assessment involved tracking treatment-emergent adverse events (TEAEs), adverse events of special interest (AESI), adverse drug reactions (ADRs), severe adverse events (SAEs), serious adverse drug reactions (SADRs), and death-related ADRs. Parameters encompassed physical examination findings, abnormal vital signs, clinical laboratory results, and shifts in ECOG PS. Treatment discontinuation rates and reasons were documented. Abnormal cases were summarized by severity (NCI CTCAE grade), drug association, and categorized by treatment group according to System Organ Class and Preferred Term. A Data and Safety Monitoring Board convened every 6 months post-study commencement to oversee safety.
Result
Demographics
In this clinical trial, 90 subjects were enrolled. The demographic breakdown of participants is as follows: 41 (45.56%) were male and 49 (54.44%) were female. The mean age was 68.99 years with a standard deviation of 10.34 years, and a median of 69.50 years. Ages ranged from 49 to 92 years. Regarding ECOG PS scores, 42 participants in the treatment group and 39 in the control group had scores of 0-1, comprising 90.00% of the total. Scores of 2 were recorded for 5 participants in the treatment group and 4 in the control group, totaling 10.00%. There was no statistically significant difference between the groups (P = 1.0000). The mean time after NSCLC diagnosis was 1.19 ± 3.37 months in the treatment group and 0.37 ± 0.72 months in the control group, with an overall mean of 0.80 ± 2.52 months. There was no statistically significant difference between the 2 groups (P = .1069) (Table 2).
Baseline Demographics.
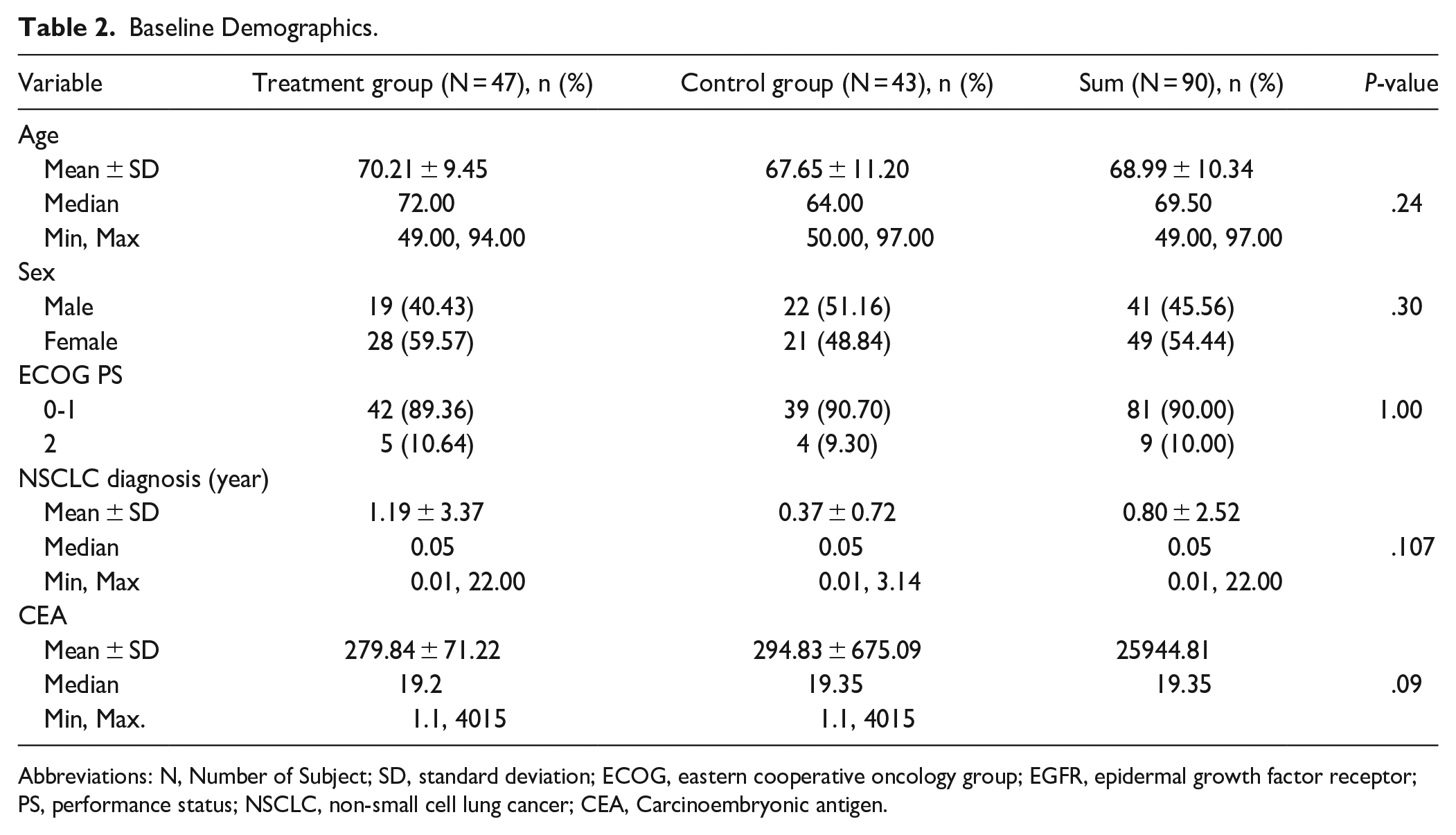
Abbreviations: N, Number of Subject; SD, standard deviation; ECOG, eastern cooperative oncology group; EGFR, epidermal growth factor receptor; PS, performance status; NSCLC, non-small cell lung cancer; CEA, Carcinoembryonic antigen.
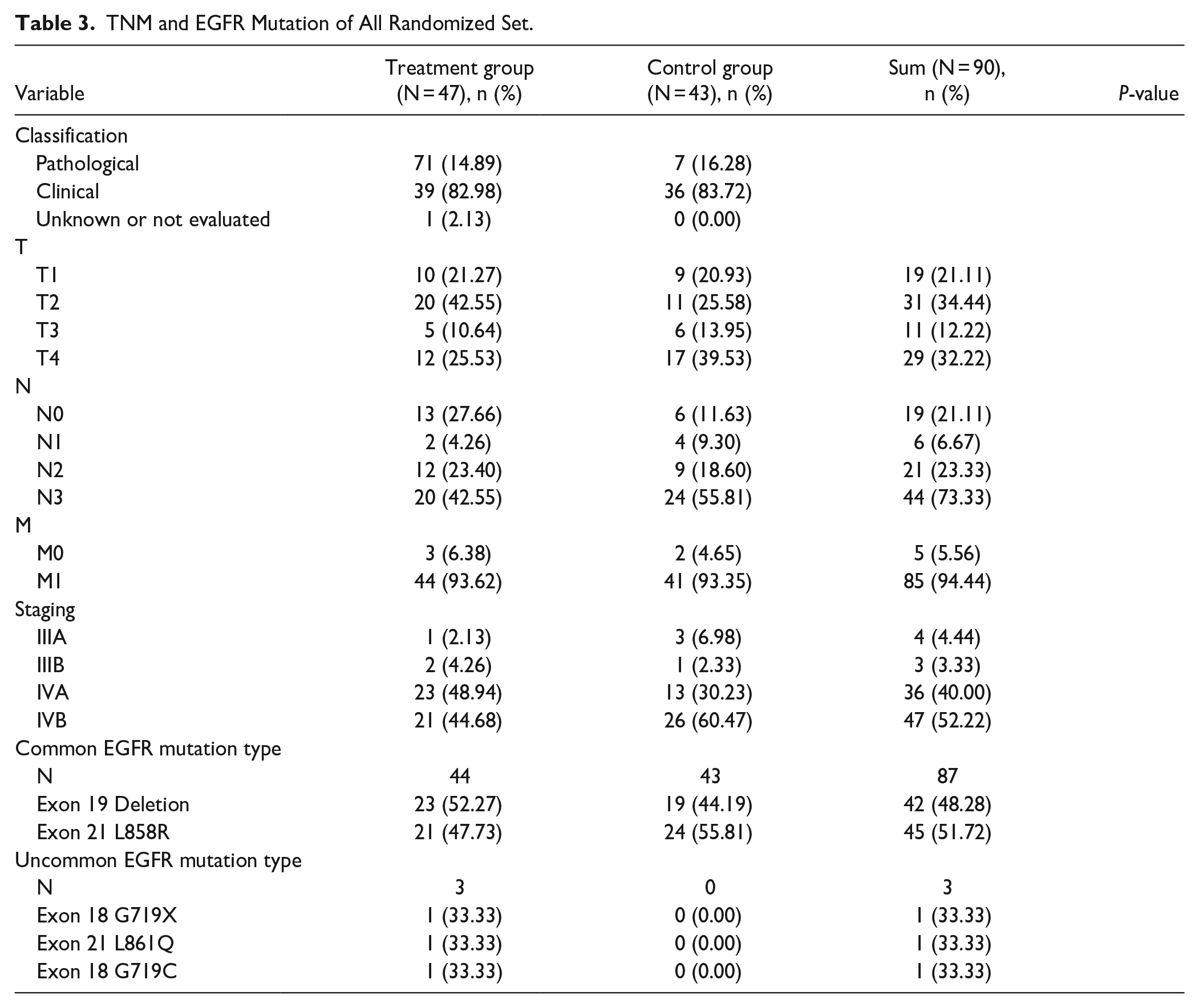
Three analysis groups were used to analyze the data. Out of 90 patients, 1 participant was excluded from the safety set (SS) due to not taking the treatment drug at least once. Out of the 89 patients in the SS group, 8 were excluded from full analysis set (FAS) group due to loss to follow up, and 14 participants were excluded from per protocol set (PPS) group due to violating the protocol, taking prohibited concomitant drugs, and randomization error (Figure 2). The majority were in advanced stages, with significant representation in Stage IV. Specifically, Stage IVA and IVB combined encompassed the majority of both groups, indicating a predominance of late-stage disease. TNM, cancer stage information, and information regarding EGFR mutations types are provided in Table 3.
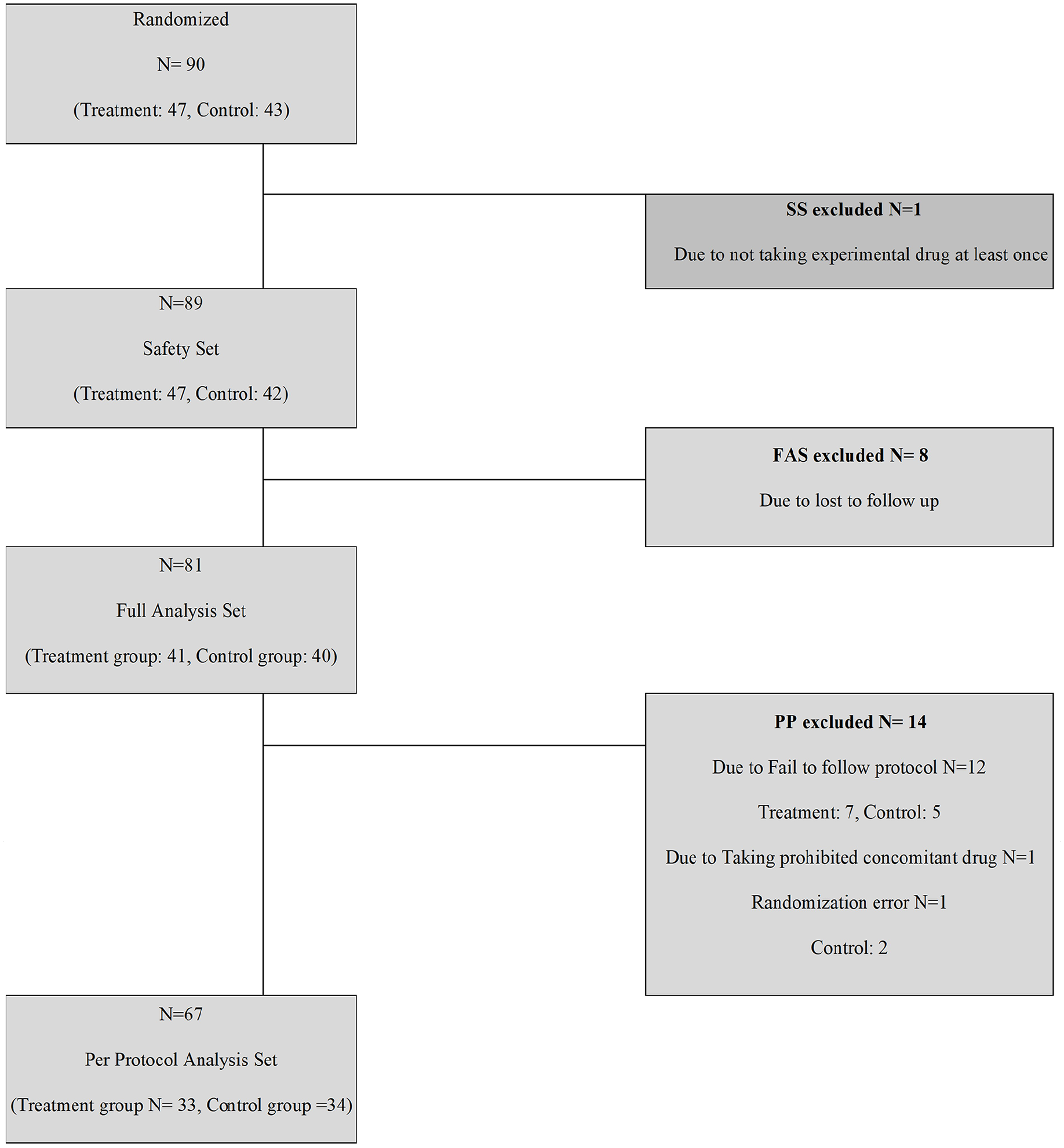
Number of participants and analysis groups.
TNM and EGFR Mutation of All Randomized Set.
Primary Outcome
In the FAS analysis group, 25 subjects (60.98%) in the treatment group and 21 (52.50%) in the control use group maintained the initial dose of afatinib, and there was no statistical significance difference (P = .4414). DCR was 80.49% in the treatment group and 90.00% in the control group. There was no statistical significance difference between the groups (P = .2283). In the PPS analysis group, 19 subjects (57.58%) maintained in the initial dose of afatinib in the treatment group and 21 (55.88%) in the control use group, and there was no statistical significance difference (P = .8888). DCR was 93.94% in the treatment group and 97.06% in the control group. There was no statistically significant difference (P = .6135) (Table 4).
FAS and PPS Group Analysis of Dose Maintenance Rate and DCR.

Abbreviations: N, Number of Subject; FAS, full analysis set; DCR, disease control rate; PPS, per protocol set.
Secondary Outcome
PFS
In the FAS group, the treatment group experienced 7 cases of disease progression (PD), 1 death, and 33 were censored. Meanwhile, the monotherapy group reported 4 PD cases, 2 deaths, and 34 censored cases. The Kaplan-Meier curve showed that the median PFS was 131 days for the treatment group, with the median for the monotherapy group not being calculable. There was no significant statistical difference between the 2 groups (P = .4932). In the PPS group, the treatment group had 2 PD cases, 1 death, and 30 were censored. The monotherapy group had 1 PD case, no deaths, and 33 censored cases. The Kaplan-Meier curve indicated that the median PFS for the treatment group was 131 days, with no calculable median for the monotherapy group. Similarly, no significant statistical difference was observed between the groups (P = .3145) (Supplemental Table 1 and Supplemental Figure 1).
TTP
In the FAS group, the treatment group experienced 7 events and 34 were censored, while the monotherapy group reported 4 events and 36 were censored. The Kaplan-Meier curve could not calculate the median TTP for either group. No significant statistical difference was found between the 2 groups (P = .3226). In the PPS group, the treatment group had 2 events and 31 were censored. The monotherapy group had 1 event and 33 were censored. The Kaplan-Meier curve again did not yield a median TTP time for either group. Similarly, no significant statistical difference was observed between the groups (P = .5605) (Supplemental Table 2).
OS
In the FAS group, there were 3 events and 38 censored cases in the treatment group, while the monotherapy group also had 3 events and 37 censored cases. The Kaplan-Meier estimated OS was 131.00 days for the treatment group, but it was not estimable for the monotherapy group. No statistical difference was observed between the 2 groups (P = .8945). In the PPS group, there was 1 event and 32 censored cases in the treatment group, compared to 0 events and 34 censored cases in the monotherapy group. The Kaplan-Meier estimated OS was not calculable. No statistical difference was found between the groups (P = .3173) (Supplemental Table 3 and Supplemental Figure 2).
EORTC QLQ-C30
In the FAS group, no statistically significant difference was observed in all categories. However, in the PPS group, the average increase in functional scales-physical functioning was 9.79 ± 19.08 in the treatment group compared to 0.61 ± 15.03 in the control group. This difference was statistically significant (P = .0475), indicating higher improvement in the treatment group regarding physical function. No statistically significant difference was found in other categories in EORTC QLQ-C30 (Table 5).
FAS Group and PPS Group Analysis of EORTC QLQ-C30.
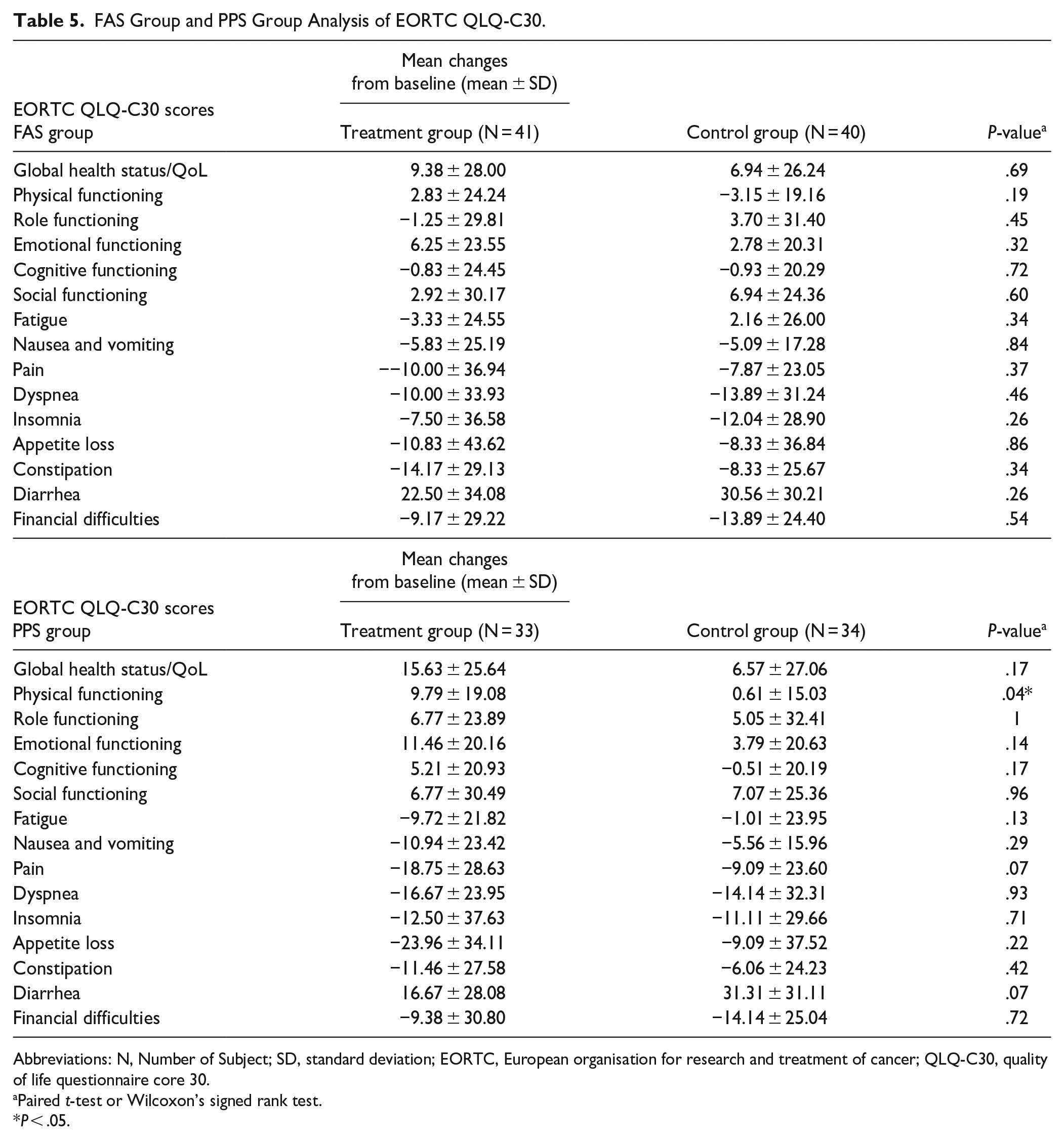
Abbreviations: N, Number of Subject; SD, standard deviation; EORTC, European organisation for research and treatment of cancer; QLQ-C30, quality of life questionnaire core 30.
Paired t-test or Wilcoxon’s signed rank test.
P < .05.
EORTC QLQ-LC 13
In both the FAS group and the PPS group, symptom scales and items including dyspnea (P = .53), coughing (P = .054), hemoptysis (.27), sore mouth (P = .95), dysphagia (P = .52), peripheral neuropathy (P = .22), alopecia (P = .19), chest pain (P = .34), pain in arm or shoulder (P = .44), other pain (P = .27) showed no statistical difference between the 2 groups (Supplemental Table 4).
HRQoL
In the FAS group analysis, the treatment group showed a mean decrease of −0.05 ± 0.37, while the control group demonstrated a change of 0.00 ± 0.22. No statistically significant difference was observed between the 2 groups (P = .6920). In the PPS group analysis, the treatment group showed an average decrease of −0.06 ± 0.24, whereas the control group showed an average increase of 0.03 ± 0.20. Again, no statistically significant difference was found between the 2 groups (P = .2214) (Supplemental Table 5).
Tumor marker (CEA)
In FAS group, the treatment group decreased by −292.89 ± 677.29 on average, and the control group decreased by −107.26 ± 230.65 on average. There was no statistical significance difference between the 2 groups (P = .7146). In PPS group, the treatment group decreased by −334.11 ± 744.34 on average, and the control group decreased by −106.82 ± 240.78 on average. Again, no statistically significant difference was found between the 2 groups (P = .6350) (Supplemental Table 6).
Safety Outcome
In this clinical trial, the treatment group, consisting of 47 participants, and the control group, with 42 participants, experienced various adverse events. Treatment-emergent adverse events (TEAEs) were reported in 42 participants (89.36%) of the treatment group, totaling 159 cases, while in the control group, TEAEs were reported in all 42 participants (100%), totaling 189 cases, marking a significant difference in the incidence rate (P = .05) (Supplemental Table 7). Events of Special Interest (AESI) occurred in 40 participants (85.11%) with a total of 95 cases in the treatment group and in all 42 participants (100%) with a total of 101 cases in the control group, demonstrating a statistically significant difference (P = .01*), with a higher incidence in the control group (Supplemental Table 8). Adverse Drug Reactions (ADRs) were noted in 40 participants (85.11%) with 125 cases in the treatment group and in all 42 participants (100%) with 125 cases in the control group, also showing a significant difference (P = .01*), indicating a higher occurrence in the control group (Supplemental Table 9). Severe Adverse Events (SAEs) were observed in 7 participants (14.89%) involving 10 cases in the treatment group and in 9 participants (21.43%) involving 13 cases in the control group, without a significant difference between the groups (P = .42) (Supplemental Table 10). Serious Adverse Drug Reactions (SADRs) occurred in 2 participants (4.26%) involving 2 cases in the treatment group and in 3 participants (7.14%) involving 3 cases in the control group, with no significant difference observed (P = .66) (Supplemental Table 11). Cases of adverse events leading to death were reported in 2 participants (4.26%) involving 5 cases in the treatment group and in 3 participants (7.14%) involving 4 cases in the control group, also without a significant difference between the groups (P = .66) (Supplemental Table 12 and Table 6).
Safety Evaluation Summary.

Abbreviation: N, Number of subject.
Paired t-test or Wilcoxon’s signed rank test.
P < .05.
Discussion
The current treatment regimen for NSCLC often leads to drug resistance and various side effect. 22 Complementary and alternative medicine, particularly herbal medicine, has emerged as a promising approach to address these challenges. Recent systematic reviews have suggested that combining herbal medicine with EGFR-TKI treatment can enhance both efficacy and tolerability.23,24 Studies have also indicated that the median PFS (mPFS) in patients receiving herbal medicine alongside EGFR-TKI was significantly longer than in those receiving monotherapy.25,26 Therefore, in this study, the authors aimed to evaluate the role of HAD-B1 as an adjuvant therapy in improving tumor control and the QOL of patients with advanced NSCLC.
In our primary outcome evaluation, no statistically significant differences were observed between the treatment group and the control group in terms of the initial dose maintenance rate for afatinib and the DCR. Similarly, secondary outcomes such as PFS, OS, TTP did not exhibit statistically significant differences between the treatment and control groups. On the other hand, the treatment group reported improved physical function in quality of life, which was statically significant (P = .04). Additionally, in drug safety evaluations, statistically significant differences were observed between the groups for ESI (P = .01) and ADR (P = .01). Overall, our finding suggests that while HAD-B1 did not demonstrate substantial benefits in terms of tumor reduction, it improved the physical functioning of NSCLC patients.
Although our original findings did not show a significantly positive effect of herbal medicine in enhancing afatinib’s dose maintenance rate or DCR, additional studies of a similar nature may support the beneficial effect of herbal medicine as maintenance therapy. One RCT that investigated mPFS with 91 NSCLC patients with EGFR mutations using Chinese herbal medicine + EGFR-TKI group and EGFR-TKI group reported 8.9 months mPFS in treatment group compared to the 12.3 months mPFS in the control group (P = .02). In addition, the mOS in the treatment group was 24.2 months compared to the 28.2 months in the control group (P = .02). 25 The constrained 16-week observation period in our study necessitates careful consideration of its potential implications. The abbreviated timeframe for data collection raises concerns regarding the comprehensive representation of the intricate dynamics inherent to the phenomenon under investigation. Specifically, the brevity of the observation period may have limited our ability to fully capture long-term trends relevant to the primary outcomes of our study. Moreover, it highlights the need for future research endeavors to employ extended observation durations, thereby fostering a more nuanced and holistic understanding of the subject matter.
Strengths of our study lie in its multi-center randomized clinical trial design, which enhances the generalizability of the findings. Additionally, HAD-B1 represents a quality-controlled herbal formula, ensuring safer use and potentially more effective treatment outcomes. 27 The study is subject to several limitations that warrant consideration. Firstly, the relatively short duration of the study period may have restricted the ability to capture longer-term treatment effects and outcomes adequately as well as restricted the maturation of the data. A longer follow-up duration would have provided a more comprehensive understanding of the sustained efficacy and safety profile of HAD-B1 in NSCLC management. Secondly, the open-label nature of the study design introduces the potential for bias, as both participants and investigators were aware of the treatment allocation. This lack of blinding could have influenced subjective endpoints and introduced confounding factors, thus affecting the robustness and reliability of the study results. Furthermore, the absence of blinding may have influenced treatment adherence and reporting of adverse events, potentially impacting the accuracy and completeness of safety assessments. Additionally, the study’s generalizability may be limited by factors such as the specific patient population enrolled, the inclusion and exclusion criteria applied, and the geographic distribution of study sites. These factors may restrict the extrapolation of findings to broader patient populations or clinical settings, thereby limiting the external validity of the study results.
In summary, the study provides valuable insights into the potential therapeutic role of HAD-B1 in NSCLC treatment. However, it is imperative to interpret the findings within the context of the inherent limitations outlined. Despite these constraints, the observed improvements in physical function and reduction in side effects associated with HAD-B1 administration underscore its potential clinical significance. Thus, addressing these limitations in future research endeavors will be pivotal for elucidating the broader therapeutic utility and clinical applicability of HAD-B1 in the comprehensive management of NSCLC.
Conclusion
In conclusion, our study provides valuable preliminary data regarding the potential role of HAD-B1 in non-small cell lung cancer, particularly in EGFR-positive patients. Significantly, our study demonstrates that HAD-B1, as an herbal medicine intervention, administered in locally advanced or metastatic NSCLC, can mitigate adverse events induced by EGFR-TKIs and enhance patients’ physical functioning. Further research is warranted to substantiate the use of HAD-B1 in conjunction with EGFR-TKIs, as well as other herbal medicines, as a maintenance therapy for NSCLC patients.
Supplemental Material
sj-docx-3-ict-10.1177_15347354241268231 – Supplemental material for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations
Supplemental material, sj-docx-3-ict-10.1177_15347354241268231 for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations by Eunbin Kwag, Soo-Dam Kim, Seong-Hoon Shin, Chulho Oak, So-Jung Park, Jun-Yong Choi, Seong Hoon Yoon, In-Cheol Kang, Mi-Kyung Jeong, Hyun Woo Lee, Sun-Hwi Bang, Ji Woong Son, Sanghun Lee, Seung Joon Kim and Hwa-Seung Yoo in Integrative Cancer Therapies
Supplemental Material
sj-jpg-1-ict-10.1177_15347354241268231 – Supplemental material for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations
Supplemental material, sj-jpg-1-ict-10.1177_15347354241268231 for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations by Eunbin Kwag, Soo-Dam Kim, Seong-Hoon Shin, Chulho Oak, So-Jung Park, Jun-Yong Choi, Seong Hoon Yoon, In-Cheol Kang, Mi-Kyung Jeong, Hyun Woo Lee, Sun-Hwi Bang, Ji Woong Son, Sanghun Lee, Seung Joon Kim and Hwa-Seung Yoo in Integrative Cancer Therapies
Supplemental Material
sj-jpg-2-ict-10.1177_15347354241268231 – Supplemental material for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations
Supplemental material, sj-jpg-2-ict-10.1177_15347354241268231 for A Randomized, Multi-Center, Open Label Study to Compare the Safety and Efficacy between Afatinib Monotherapy and Combination Therapy with HAD-B1 for the Locally Advanced or Metastatic NSCLC Patients with EGFR Mutations by Eunbin Kwag, Soo-Dam Kim, Seong-Hoon Shin, Chulho Oak, So-Jung Park, Jun-Yong Choi, Seong Hoon Yoon, In-Cheol Kang, Mi-Kyung Jeong, Hyun Woo Lee, Sun-Hwi Bang, Ji Woong Son, Sanghun Lee, Seung Joon Kim and Hwa-Seung Yoo in Integrative Cancer Therapies
Footnotes
Author Contributions
H.-S.Y. and S.J.K.: Conceptualization. S.-H.S., S.-J.P., and M.-K.J.: Methodology. S.-H.S., C.O., J.-Y.C., S.H.Y., I.-C.K., H.W.L., S.-H.B., J.W.S., S.L., and S.J.K.: Data curation. E.K., S.-D.K., and H.-S.Y.: Investigation. E.K. and S.-D.K.: Formal analysis and Writing—original draft. S.-H.S., C.O., S.-J.P., J.-Y.C., S.H.Y., I.-C.K., M.-K.J., H.W.L., S.-H.B., J.W.S., S.L., S.J.K., and H.-S.Y.: Writing—review & editing. H.-S.Y. and S.J.K.: Supervision. H.-S.Y. and E.K.: Funding acquisition. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI19C1046 and H122C1936).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
