Abstract
The upregulation of both HSP70 and HSP90 frequently compromises the effects of thermotherapy. The co-inhibition of HSP70/HSP90 may be preferable to enhance the effects of thermotherapy on nasopharyngeal carcinoma cells. The changes of HSP70 and HSP90 were detected after thermotherapy in human nasopharyngeal cancer cell HNE1. 17-Dimethylaminoethylamino-17-demethoxygeldanamycin (17-DMAG) and quercetin were used to inhibit the activity of HSP90 and HSP70. The enhanced effects were evaluated in vitro and in vivo. Both HSP70 and HSP90 were upregulated promptly in HNE1 after thermotherapy. Single inhibition of HSP70 resulted in overexpression and delayed descent of HSP90. The co-inhibition of HSP70/HSP90 with quercetin plus 17-DMAG significantly increased apoptosis in hyperthermia-treated HNE1 cells both in vitro and in vivo. The co-inhibition of HSP70/HSP90 synergistically sensitizes nasopharyngeal carcinoma cells to hyperthermia.
Introduction
Thermotherapy has proven an effective approach in cancer therapy. The temperatures used in heat treatments are in the range of 40°C to 45°C in the case of locoregional treatment and up to 42°C in the case of whole body hyperthermia. However, hyperthermia exposure induces synthesis and overexpression of heat shock proteins (HSPs). HSPs behave as molecular chaperones, which act as the primary cellular defense against damage to the proteome, initiating refolding of denatured proteins and regulating degradation after severe protein damage. 1 HSPs protect cells both by limiting the effects of protein-damaging agents through protein chaperoning and refolding and by directly blocking the cell-death pathways.2-5
HSP expression is tightly controlled in normal human cells, whereas it is often dysregulated in tumor cells. Aberrant expression of HSP90, HSP70, HSP27, and HSP40 either individually or in combination has been widely reported in human malignant tumors of various origins.6-11 More important, these HSPs are inducible to a variety of stresses, especially hyperthermia exposure. Among the HSPs, HSP70 and HSP90 play more important roles and have been linked to cancer resistance to stress-mediated apoptotic signals.8,12-14 The upregulation of HSPs frequently compromises the effects of thermotherapy, although targeting HSPs has proven to be an effective strategy to reverse the thermotolerance of cancer cells.13,15,16 However, the single inhibition of HSP90 may induce the upregulation of HSP70 and the induction of HSP70 may ultimately limit the efficacy of HSP90 inhibitors under certain circumstances. 8 Likewise, it can be speculated that the single inhibition of HSP70 may affect the changes of HSP90 in cancer thermotherapy. Therefore, co-inhibition of HSP70/HSP90 may be necessary for better inhibitory efficacy in this situation.
Nasopharyngeal carcinoma (NPC) is one of the most common cancers among Chinese or Asian ancestry. High-dose radiotherapy with adjunctive chemotherapy is the primary treatment of NPC, 17 whereas hyperthermia can be an additive strategy as a sensitizer to improve the potential of the radiotherapy or chemotherapy.18-20 Here, we investigated the changes of the levels of HSP70 and HSP90 and the effects followed by HSP90 or HSP70 inhibition with 17-dimethylaminoethylamino-17-demethoxygeldanamycin (17-DMAG), an HSP90 inhibitor, and quercetin, an HSP70 inhibitor, on NPC cell line HNE1 in response to hyperthermia both in vitro and in vivo.
Materials and Methods
Cell Line and Culture
The human NPC cell line HNE1 (purchased from ATCC) was maintained in Dulbecco’s Modified Eagle Medium (DMEM; Invitrogen, Carlsbad, CA) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Invitrogen, Carlsbad, CA) and antibiotics. The culture was maintained in 95% air-humidified atmosphere containing 5% CO2 at 37°C.
Hyperthermia Treatment
HNE1 cells (2 × 105) were seeded in RPMI-1640 supplemented with 5% FBS in 6-well plates for 24 hours. The cells were pretreated for 10 minutes at increasing temperatures (38°C, 40°C, 41°C, 42°C) and then exposed to heat by placing the cell culture plates into a 42°C, 5% CO2 incubator for 2 hours. For pretreatment of 17-DMAG and quercetin, the agents were added into the culture media 1 hour before the heat treatment at a final concentration of 17-DMAG (10 µmol) and quercetin (100 µmol). After hyperthermia, the culture media were replaced by DMEM supplemented with 10% FBS. The treated cells were incubated at 37°C for further experiments. Cells were harvested at various times to detect the level of HSP70/HSP90.
Detection of HSP70/HSP90 by Real-Time Polymerase Chain Reaction
Total RNA was extracted with TRI reagent according to the manufacturer’s instructions (Sigma-Aldrich, St. Louis, MO). The amount and quality of RNA were quantified by measurement of absorbance at 260 and 280 nm in a UV spectrophotometer. Reverse transcription was carried out using 500 ng of total RNA treated with RT-PCR kit (Takara Bio Inc., Shiga, Japan). The resulting polymerase chain reaction (PCR) products were used to perform real-time PCR. The sequences of the forward and reverse primers for HSP70 and HSP90 mRNA quantification were designed and synthesized by Takara (Table 1). Real-time PCR was performed with ABI PRISM 7000. All measurements were carried out in triplicate. Controls of no template cDNA were included in the PCR experiments. The melting curve was used to check if the amplification products had the expected melting temperature. The mRNA encoding glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a reference to normalize the mRNA of HSPs. The expression levels of HSP70 and HSP90 were quantified as the ratios between their mRNA levels and GAPDH mRNA. We checked that the GAPDH variation coefficient Ct was below 10%. This variation is very low compared with the differential Ct between target mRNA and reference mRNA. Consequently, we can exclude the possibility that the observed variation pattern in HSP expression is because of the variation of the reference GAPDH mRNA itself.
Real-time PCR Primers

Abbreviations: PCR, polymerase chain reaction; HSP, heat shock protein; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Western Blot
HNE1 cells were harvested via trypsinization and washed in cold phosphate-buffered saline at 0, 2, 4, 8, 12, and 24 hours after hyperthermia; total cellular protein was extracted. Protein concentration was measured by the Bradford Protein Assay Kit (Bio-Rad Laboratories, Hercules, CA) according to the manufacturer’s instruction. The antibodies anti-HSP70 and anti-HSP90 (Santa Cruz Biotechnologies, Santa Cruz, CA) were used to determine the expression of HSP70/HSP90. The 2 antibodies were diluted at 1:1 000. The transferred polyvinylidene fluoride membranes were incubated at 4°C for 12 hours. The protein bands were visualized using an enhanced chemiluminescence detection system (Pierce, Rockford, IL).
Flow Cytometry Assay
HNE1 cells, including both attached cells and floating cells, were harvested 24 hours after hyperthermia. Flow cytometric analysis was performed to identify sub-G1 cells/apoptosis cells. Briefly, cells were suspended in 1 mL hypotonic fluorochrome solution containing 50 µg/mL propidium iodide in 0.1% sodium citrate plus 0.1% Triton X-100 and were analyzed by a flow cytometer. Apoptosis cells appeared in the cell cycle distribution as cells with DNA content less than that of G1 cells.
Hyperthermia of Established HNE1 Tumor Models
Exponential growth phase HNE1 cells (5 × 106) were subcutaneously inoculated into the right legs of nude mice (6-8 weeks, female). Treatment was started when the volume of the tumor reached 50 mm3. Mice were grouped into 6 groups (n = 4) randomly as follows: untreated, hyperthermia, 17-DMAG plus hyperthermia, quercetin plus hyperthermia, 17-DMAG plus quercetin, and 17-DMAG plus quercetin together with hyperthermia. The 17-DMAG (15 mg/kg) and quercetin (5 mg/kg) were administered as described previously,21,22 and the protocol was adjusted to combine with thermotherapy. The 17-DMAG (intravenous) and quercetin (intraperitoneal) were given once a day for 6 days on days 1 to 6. For better heat response in vivo, the heat treatments with 43°C for 2 hours were performed 2 hours after administration. The mice were then fixed. Heat treatments were performed through dipping the tumor-loading legs into preheated water. All mice were treated twice on days 2 and 5 in a week and then sacrificed. Tumors were harvested, weighed, and fixed with formalin for further experiments.
TUNEL Assay
Tumor tissues were removed from tumor-bearing nude mice 48 hours after the heat treatment. Then cellular apoptosis analysis was performed using a terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay according to the manufacturer’s instructions (Roche Diagnostics, Indianapolis, IN).
Results
Increased Expression of HSP70 and HSP90
We first investigated the expression of HSP70 and HSP90 following hyperthermia. The real-time PCR showed that the expression of both HSP70 and HSP90 upregulated quickly and reached a peak 4 hours after heat treatment. Then, the levels of HSP70 and HSP90 descended and reached the normal level at 24 hours (Figure 1A). Similar results were observed for protein expression of HSP70 and HSP90 (Figure 1B). The prompt upregulation of HSPs suggests that inhibitors targeting HSPs should be administered before or at an early stage of heat treatment. In this study, HNE1 cells were pretreated with the inhibitors 17-DMAG and quercetin before heat treatment in the following experiments.

HSP70/HSP90 expression in HNE1 cells in response to thermotherapy
Inhibition of HSP70 Results in Prolonged Upregulation of HSP90
A previous report has shown that single inhibition of HSP90 induces the upregulation of HSP70. 8 In this study, we investigated whether single inhibition of HSP70 also induced the upregulation of HSP90. Unexpectedly, the results of real-time PCR revealed that the level of HSP90 did not significantly increase in spite of the inhibition of HSP70. However, a prolonged upregulation of HSP90 was observed both in mRNA and protein levels (Figures 2A and 2B). Therefore, we speculated that co-inhibition of HSP70/HSP90 may promote better effects of thermotherapy in NPC.

Inhibition of HSP70 results in mild upregulation and delayed descent of HSP90 after thermotherapy (*
Co-Inhibition of HSP70/HSP90 Sensitizes HNE1 Cells to Hyperthermia Both In Vitro and In Vivo
There are some inhibitors that have demonstrated preclinical efficacy in mouse xenograft models and even in clinical trials.8,23,24 The compound 17-DMAG is an HSP90 inhibitor that is derived from the prototypical ansamycin natural product HSP90 inhibitor geldanamycin, and now undergoing clinical trials.8,16 Another natural product, quercetin, is an HSP70 inhibitor that has been reported as a hyperthermia sensitizer by inhibiting the synthesis of HSP70 in a variety of tumor cell lines.23,25,26
To further investigate our hypothesis, we used 17-DMAG and quercetin to co-inhibit HSP70 and HSP90 as described above. The heat treatments were then performed. The HNE1 cells were harvested for flow cytometry analysis 24 hours after heat treatment. The results showed that the heat treatment at 42°C for 2 hours in vitro only induced less than 20% of HNE1 cells apoptosis, which suggests thermotolerance of HNE1 cells. However, either quercetin or 17-DMAG alone or 17-DMAG plus quercetin significantly increased the heat-induced apoptosis of HNE1 cells. Nevertheless, the 17-DMAG plus quercetin combined with hyperthermia resulted in more intensive cytotoxicity in comparison with 17-DMAG or quercetin alone plus heat treatment (Figure 3,
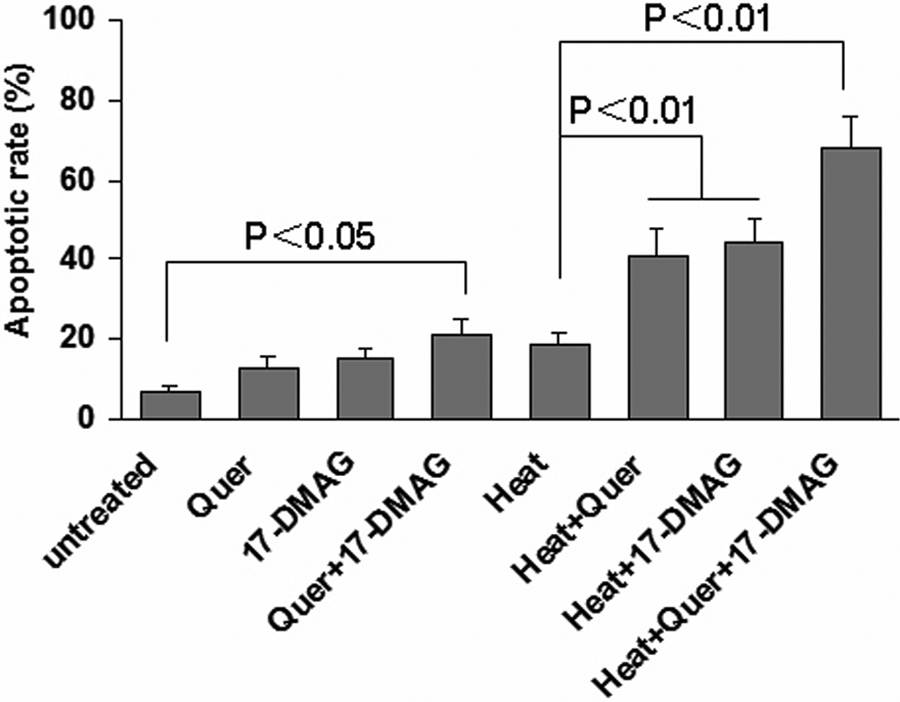
Co-inhibition of HSP70 and HSP90 increased HNE1 cell apoptosis induced by thermotherapy in vitro
In vivo, the weight of tumors from the group treated with 17-DMAG plus quercetin combined with hyperthermia showed a significant difference when compared with that of the group treated with hyperthermia alone, although the tumor-loaded mice were treated twice a week. Owing to the short period of treatment, significant differences were not found between 17-DMAG or quercetin alone plus heat treatment and heat treatment alone (Figure 4A). Therefore, we concluded that the co-inhibition of HSP70/HSP90 showed more intensive activity in sensitizing HNE1 cells to heat treatment. To confirm this further, we detected cellular apoptosis with a TUNEL kit. The combination of co-inhibition of HSP70/HSP90 and heat treatment resulted in significantly increased tumor cell apoptosis (Figures 5A and 5B). In addition, the treatments with 17-DMAG or quercetin alone without hyperthermia were also investigated. Both of the 2 groups showed similar tumor growth and little apoptosis with the untreated group (data not shown). Unlike the in vitro effects, the combination of 17-DMAG plus quercetin without hyperthermia did not display significant antitumor activity in vivo; this may be attributed to decreased dosage.

Co-inhibition of HSP70 and HSP90 plus thermotherapy inhibits growth of HNE1 cell xenografts in vivo

Co-inhibition of HSP70 and HSP90 plus thermotherapy induces more intensive apoptosis in vivo
Discussion
Thermotherapy has been used as an effective strategy to improve the potential of the radiotherapy or chemotherapy for NPC.18-20 Both the primary lesions and the most frequent metastases of neck lymph nodes are superficial, and it is thus potentially feasible to exert local heat treatment. Normally, heat stress can induce death signals that trigger programmed cell death. However, HSPs are critical components of the cancer cell’s defense mechanism against injury associated with heat stresses. Heat stress frequently results in overexpression of HSPs in cancer cells. The apoptotic program is inhibited when HSPs are elevated in cancer cells. Elevated HSP expression can also enhance tumor cell viability and impart increased resistance to subsequent chemotherapy and radiotherapy, thereby antagonizing the apoptotic effects of these treatments. 27 Inhibition of HSPs can thus break down the thermotolerance of cancer cells and subsequently increase their apoptosis.3,16,28 Our study supports the view that the co-inhibition of HSP70/HSP90 can get more intensive sensitizer effects.
Thermotolerance depends on the ability of HSPs to function as molecular chaperones and prevent aggregation. Besides, HSP70 acts as a wide spectrum inhibitor of the cell death pathways in a caspase-independent way by interacting with the lysosomal membrane and preventing the activity of hydrolytic lysosomal enzymes. 29 HSP90 can promote autonomous growth and leads to resistance to chemotherapy and hyperthermia. 30 In this study, we first investigated the expression of the 2 important HSPs—HSP70 and HSP90—and found constructional expression of both HSP70 and HSP90 and quick upregulation of them after heat treatment in HNE1 cells. This suggests that previous or early inhibition of HSPs may be necessary and more effective. Moreover, previous studies have shown that inhibition of HSP90 induces expression of HSP70.8,31,32 Here, we found that inhibition of HSP70 resulted in the delayed downregulation of HSP90 in spite of not increasing the level of HSP90. The co-inhibition of HSP70 and HSP90 plus heat treatment induced significantly increased apoptosis. This suggests that the implications of not only the inhibition of HSP70 activity but also the simultaneous inhibition of HSP90 result in more extensive apoptosis after thermotherapy.
As known, HSPs also play important roles in the immune system as carriers of tumor antigens.33,34 HSPs are able to form complexes with peptide antigens in the cytoplasm, which enhances antigen presentation in host antitumoral immunity. Especially of interest is whether HSP70 or HSP90 can help present antigens to antigen-presenting cells in different ways and thus induce antitumor immunity.35-38 In the current study, the possible immune effects of co-inhibition of HSP70 and HSP90 were excluded because of the immune defaults of nude mice. Nevertheless, the antitumor effects of co-inhibition of HSP70 and HSP90 and subsequent sensitizing activity to thermotherapy are attractive for further application.
In the current study, quercetin and 17-DMAG were used to inhibit HSP70 and HSP90. To understand the change of HSP90 following the inhibition of HSP70, 100 µM quercetin was adopted for better inhibition in vitro. Quercetin at this concentration resulted in effective inhibition of HSP70 in vitro as described previously. 39 The inhibition of HSP70 further resulted in delayed descent of HSP90. However, a lower dose of quercetin (5 mg/kg body weight, intraperitoneal) can effectively inhibit the expression of HSP70 in vivo.22,40,41 Therefore, the dosage of 5 mg/kg quercetin was selected for in vivo experiments. Nevertheless, the optimal dosage both of quercetin and 17-DMAG for sensitizing thermotherapy in further clinical application remains to be explored.
In summary, our study not only suggests that the changes of the HSP expression followed by thermotherapy are complicated and interactive but also supports the view that the co-inhibition of HSP70 and HSP90 is a preferable strategy for antagonizing activity of HSPs in tumor thermotherapy.
Footnotes
The author(s) declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article:
This study received financial support from the Inner Mongolia Medical College Major Research Projects Funded Project (NY2006ZD009) and the Natural Science Foundation of Inner Mongolia (2009MS1107).
