Abstract
Primary Epstein–Barr virus (EBV)-associated poorly differentiated nasopharyngeal adenocarcinoma (NAC) is an extremely rare tumor. In this study, we report a case of EBV-associated poorly differentiated NAC in a 35-year-old man who presented with a clogging sensation in the right ear for 1 month. The first biopsy of the nasopharynx was suggestive of nonkeratinizing carcinoma with weak positivity for CK5/6 and p63. Based on magnetic resonance imaging of the nasopharynx and neck, chest computed tomography, abdominal ultrasound, and whole-body bone scan, the patient was diagnosed with T3N2M0 disease. After the patient received neoadjuvant chemotherapy, concurrent chemoradiotherapy, and adjuvant chemotherapy, partial remission was observed. However, reassessment after 7 months of treatment revealed tumor enlargement. Transnasal endoscopic resection was performed to remove the nasopharyngeal tumor. The postoperative immunostaining results were as follows: CK5/6 (−), p63 (−), MOC31 (+), and Ber-EP4 (+). Meanwhile, EBV-encoded RNA in situ hybridization was positive. A final diagnosis of EBV-associated poorly differentiated NAC was made. Then, the patient received chemotherapy and irradiation but died several months later because of disease progression. Our patient presented with highly malignant EBV-associated poorly differentiated NAC insensitive to chemoradiotherapy with a short survival time of 27 months.
Keywords
Introduction
Primary nasopharyngeal adenocarcinoma (NAC) is an epithelial malignancy originating from the columnar epithelium of the inner mucosal lining, crypts, glands or glandular ducts of the nasopharyngeal cavity.1–3 It is an extremely rare malignant tumor, accounting for 0.38% to 0.48% of all malignant nasopharyngeal neoplasms. 4 Few studies, including case reports and small case–control studies, on NAC have been reported.5,6 Current evidence suggests that most NACs are not associated with Epstein–Barr virus (EBV) infection. 7 To our knowledge, only one case of EBV-associated undifferentiated NAC has been mentioned in the literature. 2 In this study, we report the clinicopathological features, treatment, and prognosis of a case of EBV-associated NAC and review previously reported NAC cases in the available literature. Importantly, we sought to provide valuable information to assist clinicians in diagnosing and treating this distinct type of NAC.
Case report
In December 2017, a 35-year-old man visited the First Affiliated Hospital of Hainan Medical College (Haikou, China) with complaints of a clogging sensation in the right ear for 1 month. The histopathological findings of a biopsy specimen obtained during nasopharyngoscopy suggested an initial diagnosis of nonkeratinizing undifferentiated carcinoma of the nasopharynx (Figure 1). Immunohistochemistry revealed CK (+), CK5/6 (few +), P63 (few +), LCA (−), and a Ki67 index of 9%. The lesion was also positive on EBV-encoded RNA (EBER) in situ hybridization. Subsequently, the patient was admitted to the Second Affiliated Hospital of Sun Yat-sen University (Guangdong, China). Magnetic resonance imaging (MRI) of the nasopharynx and neck disclosed nasopharyngeal carcinoma (NPC) with lymph node metastasis. No tumor lesions were found on chest computed tomography (CT), abdominal ultrasound, and whole-body bone scan. According to the American Joint Committee on Cancer staging manual (8th edition), the patient’s initial TNM stage was cT3N2M0. Then, the patient was treated with neoadjuvant chemotherapy consisting of liposomal paclitaxel and cisplatin every 3 weeks for two cycles. The patient underwent radical intensity-modulated radiotherapy (total dose of 69.9 Gy, 2.33 Gy/fraction). In addition, the patient received one cycle of liposomal paclitaxel concurrent chemotherapy. After radiotherapy, he was treated with chemotherapy consisting of liposomal paclitaxel and cisplatin every 3 weeks for three cycles. Subsequently, MRI of the nasopharyngeal cavity and neck suggested that the nasopharyngeal mass had slightly decreased in size but the lymph nodes had significantly shrank, suggestive of a partial response according to Response Evaluation Criteria in Solid Tumours criteria. The last cycle of chemotherapy was performed in July 2018. During follow-up, the patient exhibited stable disease (SD) at 1 and 4 months after chemoradiotherapy. However, 7 months after chemoradiotherapy, nasopharyngoscopy and MRI of the nasopharynx and neck indicated progressive disease (PD; Figure 2). The patient was admitted to Hainan General Hospital (Haikou, China). Transnasal endoscopic resection was performed to remove the nasopharyngeal tumor. The postoperative pathology indicated cancer cells with high nuclear grade features arranged in nests exhibiting a cribriform pattern, necrotized tumor tissue, nuclear division, and invasive growth under microscopy. Immunohistochemistry revealed CK (some +), CK5/6 (−), CK19 (−), p63 (−), HMB46 (−), EGFR (−), HIF-1 (−), Ki-67 index of 80%, MOC31 (+), Hep (−), CD34 (−), Ber-EP4 (+), P16 (−), villin (−), and CDX2 (−), and EBER in situ hybridization was positive (Figure 3). The patient was finally diagnosed with EBV-associated poorly differentiated NAC. After surgery, the patient received second-line chemotherapy with vinorelbine/nedaplatin for six cycles. After the second and fourth cycles, the patient exhibited SD. However, after the sixth cycle, PD was observed, and the patient received third-line chemotherapy with gemcitabine/nedaplatin for two cycles. SD was observed after the second cycle (16 months after the first radiotherapy session). Given that the patient’s tumor was still large (Figure 4), the patient received re-irradiation with intensity-modulated radiotherapy (total dose of 66 Gy, 2.0 Gy/fraction). However, 3 months after re-irradiation, the patient developed frequent headaches, trismus, choking, and pneumonia and died in March 2020.

Pathological results of the first biopsy of the nasopharynx lesion. (a–c) In hematoxylin and eosin staining, the tumors exhibited nests or cords of undifferentiated small oval tumor cells with scant cytoplasm and oval hyperchromatic nuclei, as well as some vesicular nuclei. Magnification: a, ×100; b, ×200; c, ×400 magnification.
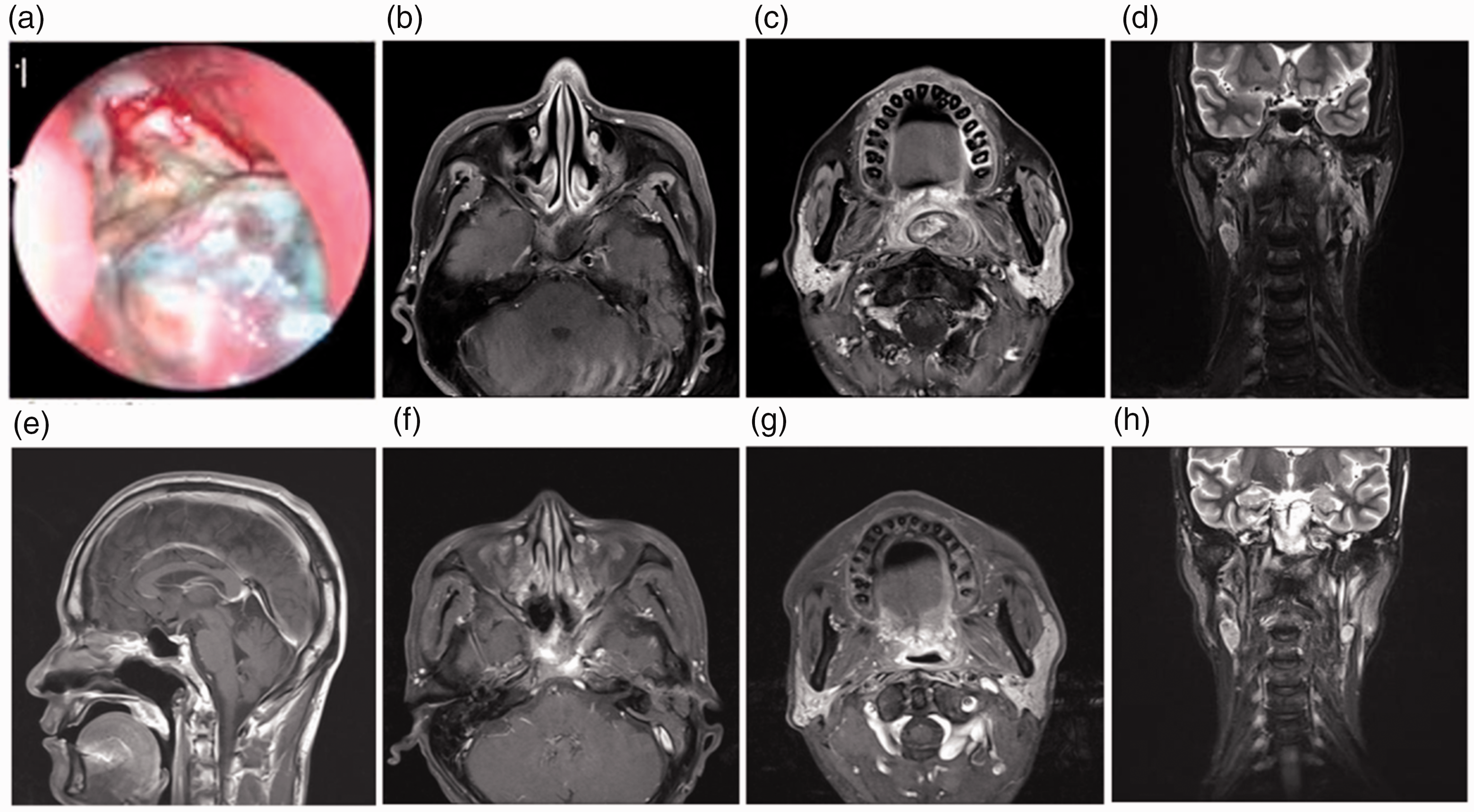
Pre- and post-surgery images of the nasopharynx lesions. (a–d) The NAC lesions involved the sphenoid sinus and oropharynx with bilateral cervical lymphadenopathy and (e–h) No tumor lesions were observed in the nasopharynx and oropharynx.

The postoperative pathological results of the nasopharynx lesion. (a–b) Hematoxylin and eosin staining revealed cancer cells exhibited high nuclear grade features arranged in nests that presented a cribriform pattern, necrotized tumor tissue, nuclear division, and invasive growth. Magnification: a, ×100; b, ×400. (c–d) Immunohistochemistry for CK 5/6 and p63 (magnification, ×100) was negative, excluding a squamous cell origin. (e–f) Immunohistochemistry for MOC31 and Ber-EP4 (magnification, ×400) was positive, suggesting a glandular cell origin. (g) EBV-encoded RNA in situ hybridization (magnification, ×400) in the nuclei of cancer cells was positive, suggesting that the tissue specimens were infected with Epstein–Barr virus and (h) On immunohistochemistry (magnification, ×100), the Ki-67 index 80%, indicating a high proliferation rate.
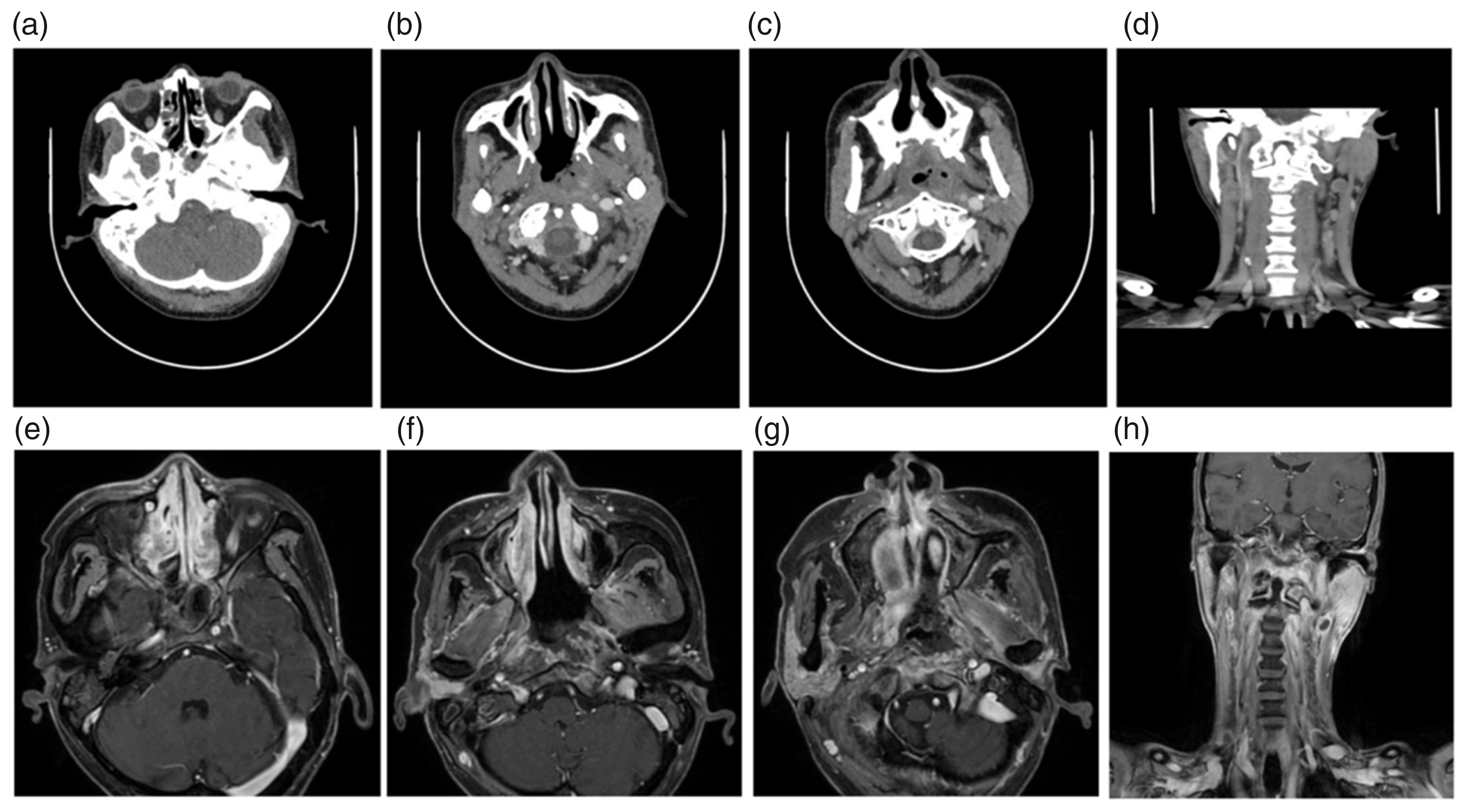
Images of the nasopharynx and neck before and after re-irradiation. (a–d) Computed tomography revealed a nasopharyngeal mass and cervical lymph node metastasis prior to re-irradiation and (e–h) On magnetic resonance imaging, the tumor size was unchanged after re-irradiation.
Written informed consent for publication of the patient’s case details and accompanying images has been obtained from his wife. The reporting of this case conforms to the CARE guidelines. 8
Discussion
According to the World Health Organization (WHO) classification, NAC is distinct from NPC, which is the most common primary malignancy in the nasopharynx diagnosed on the basis of evidence of squamous differentiation. 9 Thus, the recommended diagnosis and treatment options for NPC according to the National Comprehensive Cancer Network and Chinese Society of Clinical Oncology guidelines are not suitable for NAC. However, the incidence of NAC is extremely low, with only a few cases reported in the literature. Accordingly, the treatment for NAC is limited, and no consensus on the optimal treatment approach exists. Therefore, more research on NAC is warranted to develop therapy guidelines.
At present, the subtypes of NAC have not been specified according to the “WHO 2022” classification because of their rarity. However, there is general agreement that NAC can be classified into two pathological types: the general subtype and the salivary gland subtype. 10 Of these, the general subtype includes low-grade papillary NAC (LGNPPA) and mucinous adenocarcinoma. The salivary gland subtype includes adenoid cystic carcinoma (ACC), mucoepidermoid carcinoma, acinar cell carcinoma, low-grade polymorphic adenocarcinoma, myoepithelial carcinoma, epithelial–myoepithelial carcinoma, and hyalinizing clear cell carcinoma. Based on tumor differentiation, NAC can be classified as well-differentiated or poorly differentiated. 11 Well-differentiated NAC is characterized by glandular crypts on pathology, i.e., low-grade papillary NAC. The absence of distinct glandular crypts on pathology represents poorly differentiated NAC. In our case, histopathological examination revealed cancer cells with high nuclear grade features arranged in nests and lacking a distinct glandular crypt architecture, leading to a diagnosis of poorly differentiated NAC. Of note, the following three types can be easily misdiagnosed as NAC: (i) adenoid squamous cell carcinoma with spinous cells dissociation forming pseudoglandular patterns; (ii) vesicular nucleus cell carcinoma, known as poorly differentiated squamous cell cancer, with gland-like structure; and (iii) undifferentiated carcinoma originating from the crypt epithelium breaching basement membranes and invading neighboring tissues while retaining a visible gland-like structure. 11 Indeed, immunohistochemical markers of NAC have been largely understudied. Immunohistochemistry of LGNPPA revealed positivity for CK7, CK8, vimentin, smooth muscle actin, and pan-CK and negativity for thyroglobulin. 5 Moreover, another study of 51 patients with ACC of the nasal cavity and paranasal sinuses reported that CK8/18, CK5/6, CD117, SMA, and S100 were expressed in 100%, 100%, 94.1%, 88.3%, and 66.3% of lesions, respectively. 12 In the present case, the initial immunohistochemical findings were CK5/6 (few +) and p63 (few +), leading to a diagnosis of nonkeratinizing NPC. However, after 7 months, the second histopathological analysis revealed negativity for CK5/6 and p63, which excluded a squamous cell origin, and positivity for MOC31 and Ber-EP4, suggesting a glandular cell origin, which resulted in a final diagnosis of NAC. The reason for the difference in CK5/6 and p63 expression between the two pathological results could be that the initial pathology of the patient was NPC, which can express CK5/6 and p63. After 7 months, the pathology transformed into NAC, which generally does not express CK5/6 and p63. It is also possible that the initial pathology report was actually NAC with deterioration of the ductal epithelium, which contains basal cells that can express small amounts of CK5/6 and p63. Antigen loss might have occurred after radiotherapy and chemotherapy, resulting in negative CK5/6 and p63 expression in the second pathological analysis.
Unlike nonkeratinizing NPC, which harbors an EBV infection rate exceeding 95%, the EBV infection rate is relatively low in NAC. 13 Zong et al. 11 demonstrated that more than 95% of general subtype NACs are not related to EBV infection, and the nuclei of cancer cells are EBER-negative. In addition, Li et al. 4 and Huang et al. 10 reported six cases of LGNPPA that were not associated with EBV. By contrast, another study reported 15 cases in which the carcinogenesis of most salivary gland type NACs, including mucoepidermoid carcinoma, were potentially related to EBV. 2 These inconsistent findings could be attributable to the small sample sizes, the diverse subtype composition, and the variety of EBV probes used in those studies. Moreover, a study found that only one adenocarcinoma/undifferentiated carcinoma was positive for EBERs, in line with our findings. 2 Interestingly, for NPC, the number of EBER-positive cancer cells was associated with the degree of tumor differentiation, suggesting that increased abundance of EBER-positive cancer cells is associated with worse tumor differentiation. 14 EBV infection is linked to poorly differentiated cancer, explaining why more than 95% of general subtype NACs are not related to EBV infection, given that the general subtype with a predominance of LGNPPA is well-differentiated. In addition, this could account for the positive findings for EBER in situ hybridization in our case, suggesting this present case is not accidental. However, the interplay between malignant nasopharyngeal epithelial cells and EBV infection remains largely unclear. It has been postulated that persistent EBV infection leads to genetic mutations in nasopharyngeal epithelial cells, inducing the activation of oncogenes and inhibition of tumor suppressor genes including p16, thereby altering NF-κB signaling, PI3K/MAPK, and chromatin remodeling pathways, consequently regulating the host microenvironment and promoting cell proliferation. 15 Meanwhile, p16 inactivation and cyclin D1 pathway activation could help undifferentiated nasopharyngeal epithelial cells, allowing EBV infection to mutate. 15 Accordingly, further investigation is warranted to validate this theory.
Because of its rarity and the heterogeneity surrounding its pathological subtypes and clinical features, there is no standard treatment for NAC. The treatment and prognosis of NAC depend on the clinical stage and histological grade. 3 Radiotherapy remains the mainstay of treatment for nasopharyngeal malignant tumors given the intricacy of the nasopharyngeal anatomy, the difficulty of surgery, and poor cosmetic results. 15 However, radiotherapy is not the first-line treatment for NAC because of its insensitivity. In this respect, a retrospective analysis reported that 37.5% (6/16) of patients with early-stage NAC and 8.70% (2/23) of patients with advanced-stage were treated with surgery plus adjuvant radiotherapy, whereas the other patients received radiotherapy alone. The 5-year disease-free survival rates in these groups were 89% and 75%, respectively. 16 The 10-year data of 196 patients with ACC from the National Cancer Database revealed that the median survival for the surgery plus radiotherapy, surgery alone, and radiotherapy alone groups were 87, 74, and 62 months, respectively. 17 These results suggested that surgery plus radiotherapy was preferred for NAC. Moreover, Sun et al. 6 suggested that combined radiotherapy and surgery yielded optimal treatment outcomes for NAC. Regarding the different histological subtypes, intensity-modulated radiotherapy or surgery has been recommended as the primary treatment option for patients with ACC, whereas surgery is the first-line treatment for patients with mucoepidermoid carcinoma or general subtype NAC. Table 1 illustrates that different treatment modalities were selected for different subtypes of NAC, leading to different prognoses. However, no study has reported the specific therapeutic options of EBV-associated poorly differentiated NAC. Given our limited experience and poor awareness of NAC, the patient was first diagnosed with nonkeratinizing NPC and treated with chemotherapy and radiotherapy, resulting in partial tumor regression. However, the second pathological results revealed a diagnosis of EBV-associated poorly differentiated NAC. Then, surgery, chemotherapy, and re-irradiation were performed, but the patient eventually died. Overall survival was 27 months. The findings of this case emphasize that in patients presenting with CK5/6 (few +) and P63 (few +), immunohistochemical markers of adenocarcinoma should be assessed to dismiss the possibility of poorly differentiated NAC.
Summary of previous studies of the treatment and prognosis of NAC.

OS, overall survival; NED, no evidence of disease.
Conclusions
NAC is not associated with EBV in most cases. This report highlights our experience with a patient with poorly differentiated NAC associated with EBV infection. In our case, immunohistochemistry revealed CK5/6 (−), p63 (−), MOC31 (+), Ber-EP4 (+), and EBER in situ hybridization (+). The patient was diagnosed with highly malignant EBV-associated poorly differentiated NAC insensitive to chemoradiotherapy, and he had a short survival time of 27 months.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231173839 - Supplemental material for Epstein–Barr virus-associated poorly differentiated nasopharyngeal adenocarcinoma: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605231173839 for Epstein–Barr virus-associated poorly differentiated nasopharyngeal adenocarcinoma: a case report and literature review by Shuai Zhang, Xiao-Peng Huang, Hai-Hong Li and Ze-Tan Chen in Journal of International Medical Research
Footnotes
Author contributions
SZ and XH gathered clinical data and wrote the manuscript. HL obtained the biopsy and MRI images. ZC conceived this article, retrieved clinical data, and edited the manuscript. All authors contributed to the manuscript and approved the submitted version.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
This study involving human participant was reviewed and approved by the Ethics Committee of Hainan General Hospital (Ethical approval No.: Med-Eth-Re[2022] 737).
Funding
This work was supported by the Natural Science Foundation of Hainan Province (Grant No. 821QN0981).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
