Abstract
Introduction
Chemoradiotherapy (CRT) is important to the esophageal cancer (EC) management. However, the predictive value of lymphocyte-related parameters, such as lymphocyte count (L), neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), and platelet-to-lymphocyte ratio (PLR), is not yet fully understood. Moreover, chemotherapy agents like fluorouracil and cisplatin may have an impact on lymphocyte dynamics. This meta-analysis aims to evaluate the prognostic value of these parameters in EC patients undergoing concurrent CRT (eg, radiotherapy combined with fluorouracil and cisplatin), particularly in the context of specific chemotherapy regimens.
Methods
Electronic databases were comprehensively searched up to September 2023 for research that assesses the prognostic role of lymphocyte-related indicators in EC patients undergoing CRT. Combined Hazard Ratios (HR) were estimated with a random-effects model, supplemented by meta-regression and subgroup analyses for enhanced insights.
Results
Of the 41 studies selected for qualitative evaluation, 22 were eligible for meta-analysis. These results revealed that increased pre-NLR (HR = 1.87, 95% CI = 1.55-2.26), lower pre-LMR (HR = 1.94, 95% CI = 1.36-2.77), lower dur-L (HR = 1.56, 95% CI = 1.28-1.90), and higher post-NLR (HR = 1.95, 95% CI = 1.08-3.51) predicted poorer overall survival (OS). Lower pre-LMR (HR = 1.73, 95% CI = 1.14-2.65) and lower dur-L (HR = 1.39, 95% CI = 1.14-1.69) were significant predictors of worse progression-free survival (PFS). The predominant chemotherapy regimen analyzed was fluorouracil combined with cisplatin, which significantly influenced lymphocyte counts and ratios during treatment.
Conclusions
Our meta-analysis indicates that pre-treatment NLR, pre-treatment LMR, during-treatment L, and post-treatment NLR are valuable prognostic biomarkers for EC undergoing CRT, particularly in those treated with fluorouracil and cisplatin. Further investigations are warranted to explore their prognostic implications and therapeutic potential.
Introduction
As one of the most aggressive cancers, esophageal cancer (EC) ranks seventh in terms of cancer-related morbidity and sixth in cancer-related mortality, respectively.1,2 For patients with locally advanced EC ineligible for surgery, concurrent chemoradiotherapy (CCRT) is the standard treatment.3,4 Nevertheless, the prognosis of CRT for EC remains suboptimal, exhibiting five-year overall survival (OS) rates from 25% to 47%. 5 In recent times, some novel techniques such as gene expression assays and circulating tumor marker assays have been introduced with a view to improving the prognosis prediction of EC. 6 However, these approaches are costly and not widely accessible in routine clinical practice.
Emerging evidence suggests a relationship between tumor prognosis and host's systemic inflammation, with lymphocytes playing a critical role. 7 Evidence indicates that lymphocytes are the cells most susceptible to radiation within the hematopoietic network. Indeed, it has been demonstrated that even a small fraction of 2 Gy can result in the death of half of the irradiated lymphocytes. 8 Moreover, a study evaluating the hematological toxicity of radiotherapy (RT) in solid malignant tumors revealed that individuals diagnosed with EC exhibited the second lowest levels of lymphocytes at the conclusion of RT, surpassed only by those with head and neck cancer. 9 The presence of radiation-induced lymphopenia (RIL), an inexpensive, readily accessible, and routinely tested biomarker, is considered a poor predictor in different types of cancer. 10 Furthermore, additional prognostic value has been identified in other peripheral blood components such as neutrophils,11,12 platelets, 13 and monocytes. 14 Therefore, lymphocyte-related parameters that integrate lymphocytes with other blood components, consisting of neutrophil-to-lymphocyte ratio (NLR),15–18 lymphocyte-to-monocyte ratio (LMR),19,20 and platelet-to-lymphocyte ratio (PLR)21,22 that obtained from various time points, may provide more predictive information about the prognosis of EC undergoing CRT. Nevertheless, the impact of these parameters on prognosis in EC receiving CRT, particularly at different time points around CRT, remains controversial.
There is limited agreement on the prognostic significance and clinical value of these parameters obtained from disparate temporal assessments in EC patients undergoing CRT. The objective of this study was to conduct a systematic review and meta-analysis to assess the effect of lymphocyte-related parameters, as measured before, during, or after CRT, on OS and progression-free survival (PFS) among EC patients.
Material and Methods
As a systematic review article, this study does not require the approval of an ethics/review committee. This systematic review followed the best practices of PRISMA and AMSTAR.23,24 The study protocol registration in PROSPERO (https://www.crd.york.ac.uk/) has been completed with CRD42023466165.
Search Strategy
An extensive search was carried out via databases up to September 20, 2023. Additional relevant articles were found through a review of the references lists of the included studies. Details of the search methodology are provided in Supplementary Note 1.
The Inclusion and Exclusion Criteria
The studies selected should adhere to the following requirements: (1) the research is a cohort analysis involving EC patients; (2) CRT should be included in the patients’ treatment regimen; (3) peripheral blood lymphocyte-related parameters before, during, or after CRT have been recorded; (4) it provides data for estimating Hazard Ratio (HR) and 95% Confidence Interval (CI) for OS or PFS.
Studies were excluded when the following situations existed: (1) the study includes fewer than 10 patients; (2) it is not written in English; (3) there are no defined cutoff thresholds for lymphocyte-related parameters.
Assessment of Study Quality and Risk of Bias
The literature review was conducted following the PRISMA 2020 flowchart. 23 Duplicate records from various databases were removed. Potentially relevant records were selected after title and abstract, and their full texts were then evaluated against the inclusion and exclusion criteria. When significant study population overlap existed, the study that reported the biggest group of participants was selected. In addition, reference sections of the included papers were examined for additional research. Included studies were appraised for quality with Newcastle-Ottawa Quality Assessment Form for Cohort Studies (NOS). 25 This tool evaluates studies using three modules, comprising eight items, which include the selection of participants, comparability, and assessment of outcomes. Additionally, Review Manager Version 5.4.1 was utilized to evaluate the risk of bias. Two reviewers carried out the review and quality assessment. Disagreements were settled by consensus.
Data Extraction
Information was collected from each included study, as detailed below: first author, sample size, age, follow-up time, treatment regimen, radiation dose, concurrent chemotherapy rate, chemotherapy drug, surgery rate, time point based parameters and their cutoff points, and HR (95%CI) for prognostic analysis. Incomplete or ambiguous information was recorded accurately, with no presumptions. Two reviewers conducted the information extraction.
Statistical Analysis
In the event that a parameter is reported by a minimum of three studies, the combined HR and 95% CI for OS and PFS were estimated. If included studies have both univariate and multivariate outcomes, the multivariate outcomes are preferred for pooling. As models with random and fixed effects produced similar outcomes under low heterogeneity, the random model was used. Egger's test was employed to detect publication bias, with a P-value < .05 indicating potential bias. The trim and fill methods were used to address publication bias. To ensure the reliability of the statistically significant pooled outcomes, a sensitivity analysis was employed. To identify heterogeneity within the studies, the Higgins I² test was utilized. Heterogeneity was classified as non-significant (I² = 0%–25%), low (I² = 25%–50%), moderate (I² = 50%–75%), and high (I² > 75%). For parameters with more than three included studies, we will further explore the sources of heterogeneity. For parameters with more than five included studies, meta-regression and subgroup analysis were further conducted. Subgroup analyses were conducted on the following variables: publish year, type of study, sample size, concurrent chemotherapy rate, or surgery rate. In the present meta-analysis, forest plots served to show individual research and their pooled results.
Stata software version 15.1, along with the metan, metareg, metainf, metabias, and metatrim packages, was applied for this meta-analysis. The threshold for statistical significance was established at P < .05 (two-tailed), unless specified differently.
Results
Study Selection
As shown in Figure 1, the search yielded 920 records, which included 157 studies in PubMed, 681 in Embase, and 82 in the Cochrane Library. Following the removal of 144 duplicate records, 776 records of titles and abstracts were reviewed, and 90 articles progressed to full-text review. According to inclusion and exclusion criteria, 49 studies were further removed, with 41 studies remaining for the systematic review. Hand checking the references of the selected studies and relevant reviews did not yield further eligible studies. Then, we retained 24 studies,15–22,26–41 of which two studies did not meet the requirement of no less than 3 studies for pooled analyses on one single parameter.38,39 Eventually, 22 studies met the criteria for quantitative meta-analysis, allowing for the pooling of HR.15–22,26–37,40,41
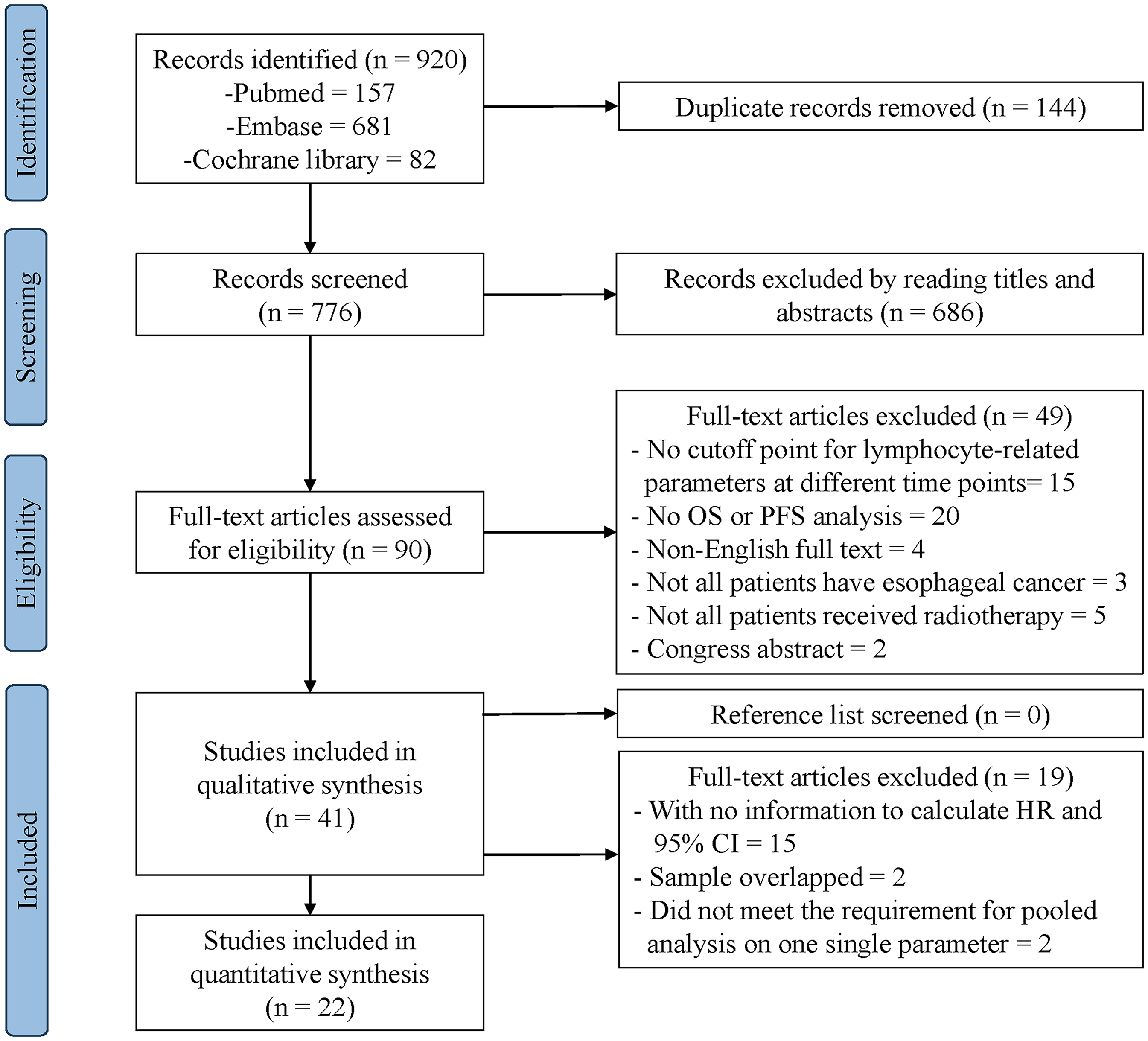
PRISMA Diagram Showing Selection of Articles for Review.
Studies Characteristics
Studies characteristics are showed within Table 1. Supplementary Table 1 shows these parameters and HR for each included study. Published between 2014 and 2023, the 24 studies featured sample sizes larger than 200 in just six cases. All studies included CRT as part of a treatment regimen with doses between 40 and 70 Gy, with the majority of chemotherapy regimens being fluorouracil plus cisplatin. The most frequently parameters are NLR prior to CRT (pre-NLR) and lymphocyte counts during CRT (dur-L). The parameters that enable quantitative meta-analysis include: lymphocyte counts before CRT (pre-L) for OS, pre-L for PFS, pre-NLR for OS, pre-NLR for PFS, LMR before CRT (pre-LMR) for OS, pre-LMR for PFS, dur-L for OS, dur-L for PFS, and NLR after CRT (post-NLR) for OS. Furthermore, there was insufficient data for meta-analysis on studies investigating other lymphocyte-related parameters, such as PLR before CRT (pre-PLR), prognostic nutritional index before CRT (pre-PNI), and systemic immune-inflammation index before CRT (pre-SII). These details are still available in Table 1 and Supplementary Table 1 for reference.
The Characteristics of Each Included Study.

Quality Assessment
Study quality was moderate to good overall, with only minor to no concerns regarding the selection of the study population. For the purposes of comparison, with the exception of lymphocyte-related parameters, the majority of the data presented in each included study were comparable between the various groups. With regard to the evaluation of outcomes, the majority of the literature described the methods of outcome measurement and provided follow-up data sources. However, it should be noted that five of the included studies did not specify the duration of follow-up,19,21,30,31,37 while other studies reported a median follow-up duration ranging from 11.4 to 65.5 months. The lack of clarity regarding the follow-up period, or the fact that it was insufficiently long, gave rise to concerns regarding the potential for study attrition bias. Furthermore, only a limited number of studies (4/24) prospectively explored the prognostic association between lymphocyte-related parameters and EC.15,22,28,32 The lack of clear inclusion/exclusion criteria in 3 of 24 studies raised concerns about participant selection.16,17,31 Supplementary Table 2 shows the NOS quality assessment results, indicating relatively high quality (score ≥7) for all included studies. Supplementary Figures 1 and 2 present the bias assessment results for the studies included.

Meta-Analysis of Impact of Lymphocyte-Related Parameters on OS.
Prognostic Value
For OS analysis, a significantly worse OS was associated with lower pre-LMR (HR = 1.94, 95%CI = 1.36-2.77, Figure 2 and Table 2)19,20,22 and lower dur-L (HR = 1.56, 95%CI = 1.28-1.90, Figure 2 and Table 2).26,27,29,32–37 Conversely, a significantly better OS was associated with lower pre-NLR (HR = 1.87, 95%CI = 1.55-2.26, Figure 2 and Table 2)15–18,21,28,30,35,41 and lower post-NLR (HR = 1.95, 95%CI = 1.08-3.51, Figure 2 and Table 2).16–18,31 Furthermore, a trend towards statistical significance was observed in the association between decreased pre-L and reduced OS (HR = 1.25, 95%CI = 0.97-1.61, Figure 2 and Table 2).32,33,37
Meta-Analysis of Parameters Affecting EC Prognosis.

Abbreviations: EC, esophageal cancer; HR, hazard ratio; pre-L, lymphocyte counts before chemoradiotherapy; pre-NLR, neutrophil-to-lymphocyte ratio before chemoradiotherapy; pre-LMR, lymphocyte-to-monocyte ratio before chemoradiotherapy; dur-L, lymphocyte counts during chemoradiotherapy; post-NLR, neutrophil-to-lymphocyte ratio post chemoradiotherapy; OS, overall survival; PFS, progression-free survival.
For PFS analysis, both pooled analysis of low pre-LMR versus high pre-LMR (HR = 1.73, 95%CI = 1.14-2.65, Table 2 and Figure 3)19,20,22 and low dur-L versus high dur-L (HR = 1.39, 95%CI = 1.14-1.69, Table 2 and Figure 3)27,32,33,36,40 showed a statistically significant inferior PFS. Furthermore, a trend towards statistical significance was observed for the association between high pre-NLR and poor PFS (HR = 1.36, 95%CI = 0.98-1.88, Figure 3 and Table 2).18,22,28,30 No statistically significant association between low pre-L and PFS was found based on the existing researches (HR = 1.28, 95%CI = 0.82-2.00, Table 2 and Figure 3).22,33,40

Meta-Analysis of Impact of Lymphocyte-Related Parameters on PFS.
Heterogeneity and Subgroup Analysis
As illustrated in Table 2, the majority of analyses exhibited non-significant to moderate heterogeneity, with the exception of the analysis of post-NLR for OS, which demonstrated considerable heterogeneity (I² = 86.5%). Meta-regression and subgroup analysis were conducted to explore a relationship between dur-L and OS, as well as a correlation among pre-NLR and OS. The meta-regression findings are presented in Tables 3 and 4, while the results for this subgroup analysis are presented within Supplementary Figure 3.
Subgroup and meta-Regression Analysis of dur-L for OS.

Abbreviations: dur-L, lymphocyte counts during chemoradiotherapy; OS, overall survival; HR, hazard ratio; 95% CI, 95% confidence interval; CHT, chemotherapy.
P value: significance of difference between subgroup stratified HR.
Subgroup and meta-Regression Analysis of pre-NLR for OS.
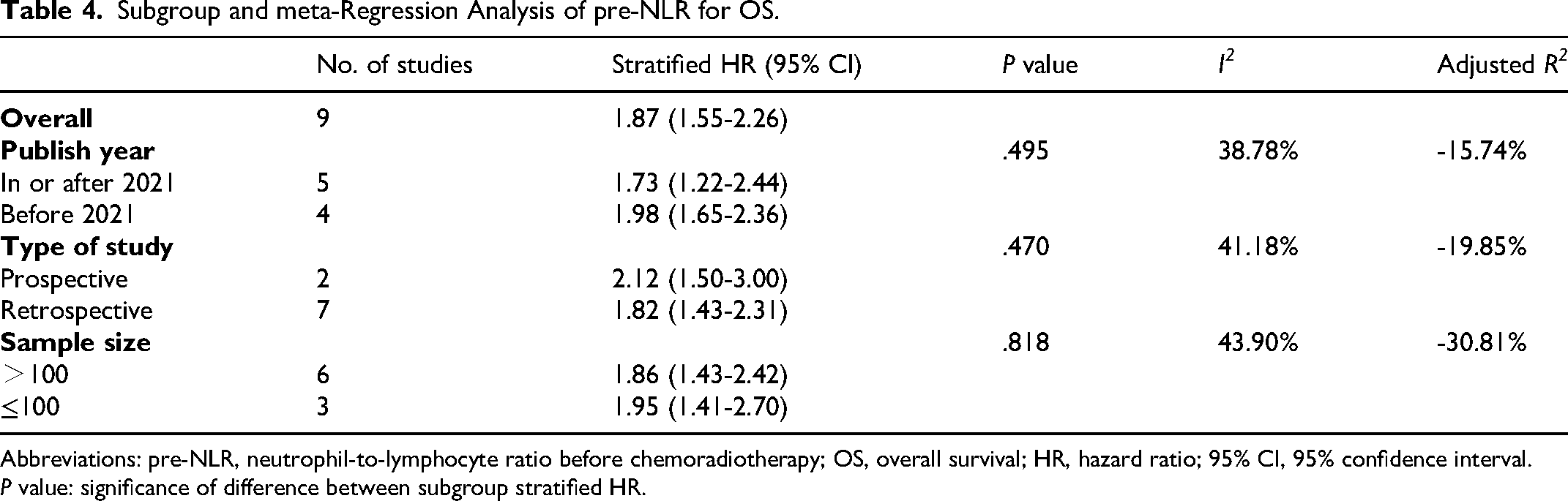
Abbreviations: pre-NLR, neutrophil-to-lymphocyte ratio before chemoradiotherapy; OS, overall survival; HR, hazard ratio; 95% CI, 95% confidence interval.
P value: significance of difference between subgroup stratified HR.
Meta-analysis of dur-L for OS revealed moderate overall heterogeneity (I² = 56.8%). As demonstrated in Table 3, HR estimates differed significantly between studies with larger (>200) and smaller (≤200) sample sizes (P = .022). This subgroup explained all of the between-study variance (R2 = 100%), leaving a residual heterogeneity (I2) of 31.53%. No significant differences were identified in the subgroup HR estimates for studies published in or after 2022 versus those published before 2022 (P = .718), studies including more than 60% versus less than 60% of patients receiving concurrent chemotherapy (P = .349), or studies including people who underwent surgery versus those who did not (P = .339). The overall heterogeneity for the meta-analysis of pre-NLR for OS is low (I2 = 36.5%). Despite the application of meta-regression and subgroup analysis, the sources of heterogeneity remained unidentified. As demonstrated within Table 4, no significant differences were discovered in this prognostic association between pre-NLR and OS across studies published in or after 2021 versus those published before 2021 (P = .495), studies of prospective versus retrospective design (P = .470), or studies with sample size >100 versus ≤100 (P = .818).
Following the removal of a single study that may have contributed to heterogeneity, 37 the meta-analysis of dur-L for OS revealed a notable reduction in heterogeneity and a stable prognostic association (I² = 31.5%, HR = 1.48, 95%CI = 1.27-1.71, Supplementary Figure 4). Following the exclusion of one study that may have been a source of heterogeneity, 32 the meta-analysis of dur-L for PFS demonstrated that low dur-L remained significantly associated with inferior PFS, accompanied by a notable reduction in heterogeneity (I² = 25.8%, HR = 1.28, 95%CI = 1.06-1.55, Supplementary Figure 4). In the meta-analysis of pre-NLR for OS, the exclusion of a single study that might have caused heterogeneity 30 resulted in a significant decrease in heterogeneity. However, the new pooled results displayed that high pre-NLR was still linked to worse OS within EC patients (I² = 0%, HR = 2.00, 95%CI = 1.73-2.32, Supplementary Figure 4). Similarly, following the exclusion of the same study, 30 the meta-analysis suggested that elevated pre-NLR was obviously linked to poor PFS, with a notable reduction in heterogeneity (I² = 0%, HR = 1.56, 95%CI = 1.28-1.90, Supplementary Figure 4). In the meta-analysis of post-NLR for OS, the exclusion of one study that may have contributed to heterogeneity 31 resulted in a notable reduction in heterogeneity. The revised pooled results continued to indicate that high post-NLR was associated with inferior OS (I² = 59.5%, HR = 1.48, 95%CI = 1.00-2.19, Supplementary Figure 4).
Publication Bias and Sensitivity Analysis
The Egger's test results showed no significant publication bias in most analyses (Table 2), with the exception of the meta-analysis of pre-LMR for PFS (P = .026) and the meta-analysis of low dur-L for OS (P = .009). Accordingly, the trim and fill methods were employed on the two analyses. The trim and fill analysis for pre-LMR and PFS showed that there was no change in the pooled HR (Supplementary Figure 5). This is likely due to the inclusion of only three studies in the analysis. With regard to the trim and fill analysis assessing dur-L for OS, this pooled result continued to indicate a significant correlation at the statistical level (HR = 1.38, 95% CI = 1.12-1.70, Supplementary Figure 5).
A sensitivity analysis was conducted for all statistically significant pooled results, and the new pooled results of pre-NLR for OS, dur-L for OS, and dur-L for PFS were found to be robust (Supplementary Figure 6 and Supplementary Tables 3-5). However, the new pooled results of pre-LMR for OS, pre-LMR for PFS, and post-NLR for OS were no longer statistically significant (Supplementary Figure 6, Supplementary Table 6-8). This may be attributed to the limited number of included studies.
Discussion
With the rising morbidity and mortality rates of EC, and the growing importance of CRT in its management, there has been a significant increase in the number of studies exploring the prognostic potential of RIL as a monitoring tool for patients with diverse cancer types.10,42 In addition, previous meta-analyses have also indicated the prognostic value of NLR, 43 LMR 44 and PLR 45 in other types of cancer. However, the prognostic significance of these lymphocyte-related parameters in EC remains underexplored. Furthermore, concerns may be raised regarding the consistency of the prognostic value of these parameters when assessed at different time points. 46
In the current systematic review and meta-analysis, we summarized the prognostic significance of lymphocyte-related parameters in EC patients who have undergone CRT as part of their treatment regimen. The findings indicated that elevated pre-NLR, reduced pre-LMR, reduced dur-L and elevated post-NLR could all result in worse OS among EC patients. In particular, dur-L showed the most consistent prognostic value across different analyses. A cutoff value of 0.2 × 109/L was the most common threshold used in studies evaluating the prognostic value of dur-L and EC. For NLR, the most common cutoff values fall between 2 and 3. Furthermore, the current meta-analysis indicated that decreased pre-LMR and decreased dur-L could lead to inferior PFS. Moreover, the correlation between pre-NLR and PFS was also statistically significant after the exclusion of one study that significantly contributed to heterogeneity.
Similarly, Dai et al 47 have conducted a meta-analysis and identified a correlation between severe RIL and a poorer prognosis in EC patients. However, this study only analyzed the prognostic impact of lymphocyte counts and did not elucidate the correlation between other lymphocyte-related parameters and prognosis in EC, nor did it investigate their prognostic value at various time points around CRT. Our meta-analysis revealed a robust correlation between the prognosis of EC and lymphocyte-related parameters, particularly the LMR and the NLR before CRT, lymphocyte count during CRT, and the NLR post CRT. In other words, a relative decrease in lymphocyte counts before, during, or after CRT may be associated with an unfavorable prognosis for EC.
Among the included studies, there was considerable inconsistency in the concurrent chemotherapy and surgery rates observed in the included population, which may have contributed to potential population selection bias. However, for the meta-analysis of dur-L for OS, no significant differences in subgroup HR estimates were found for studies including >60% versus ≤60% patients receiving concurrent chemotherapy, as well as for comparisons between studies involving surgically treated patients and those without surgery. Similarly, in a meta-analysis examining the correlation between RIL and the prognosis of solid tumors, the authors observed no significant difference between studies that included patients who received concurrent chemotherapy and studies that included patients who did not. 46 With regard to surgical intervention, one study demonstrated that an elevated NLR was linked to poorer OS within EC patients receiving CCRT, but not in those who were surgically treated. 48 Furthermore, given that lymphocyte counts demonstrated no significant alteration following neoadjuvant chemotherapy but exhibited a pronounced decline after CRT, 49 it can be inferred that CRT may exert an important influence on the correlation between lymphocyte-related parameters and EC prognosis.
However, the underlying biological mechanism remains unclear. Recently, many studies have also focused on tumor-infiltrating lymphocytes (TIL) and their prognostic correlation with EC patients undergoing CRT, and they found TIL could also serve as a prognostic biomarker for clinical outcomes.50–54 TIL have also been shown to be prognostic in other cancer, such as lung cancer, 55 cholangiocarcinoma, 56 and breast cancer. 57 As the immune system principal component, lymphocytes are pivotal in anti-tumor immunity, influencing the occurrence and development of tumors.58,59 It is inevitable that EC patients receiving CRT will experience radiation exposure to the cardiopulmonary blood pool, which includes a significant percentage of circulating lymphocytes.60–62 Given the radiation sensitivity of lymphocytes, significant fluctuations in peripheral blood lymphocyte counts may occur around CRT, 8 potentially compromising the immune system's role in identifying and destroying malignant cells. 63 Moreover, the reduction in peripheral blood lymphocytes may have a considerable effect on TIL within the tumor microenvironment (TME), ultimately leading to a compromise in the antitumor immune response64,65 and, consequently, an adverse prognosis for patients with EC (Figure 4).

Graphical Abstract of the Association Between Lymphocyte-Related Parameters and EC Prognosis. Abbreviations: RT = radiotherapy; 5-FU = fluorouracil; CDDP = cisplatin.
The findings of the present meta-analysis require further investigation into which EC patients receiving CRT are at high risk to suffer from the decrease in lymphocyte counts, as well as the exploration of potential strategies to prevent or reverse this decrease. A number of clinical and dosimetric factors were identified as being associated with the incidence of lymphopenia. Studies found that performance status, pretreatment lymphopenia, older age, lower tumor site, and larger tumor size were defined as significant predictors of treatment-induced lymphopenia in EC.32,66 In addition to clinical factors, Xu et al 32 observed that the Criteria for Adverse Events Grade 4 (G4) lymphopenia in EC patients was associated with larger planning target volume (PTV), higher lung volume receiving more than 10 Gy (V10), and heart V10. In a randomized trial, the authors observed a significant link between larger PTV and increased cardiopulmonary radiation exposure and increased incidence of G4 lymphopenia in patients who underwent intensity modulated photon radiation therapy (IMRT) or proton beam therapy (PBT). 67 Furthermore, a significant reduction in G4 lymphopenia was observed with PBT compared to IMRT, suggesting a potential clinical benefit. There is a theory that these dosimetry factors may be associated with lymphopenia due to radiation exposure to the whole cardiopulmonary blood pool, which comprises a sizable portion of circulating lymphocytes.60–62 A recent study observed a strong association between a higher effective dose to circulating immune cells (EDIC) and severe lymphopenia in EC patients treated with CCRT. 68 Furthermore, an increased number of fractions has been linked to an elevated risk of severe lymphopenia in other tumors, including lung cancer 69 and hepatocellular carcinoma.70,71 In the future, it would be beneficial to consider PTV, lung V10, heart V10, RT techniques, EDIC model, and RT fractures in the design of the RT plan for EC. These factors may prove to be important measurements in avoiding a decrease in lymphocyte counts and therefore improving the prognosis of EC patients.
In addition to focusing on the associated risks, other studies are also investigating strategies for the prevention or restoration of lymphopenia. For example, one method is lymphocyte harvesting and reinfusion, which has been demonstrated to be both practical and safe in patients with high-grade gliomas. 72 Furthermore, preclinical data have demonstrated that interleukin 7 (IL-7) has the potential to enhance the circulating T cell population. 73 And it was observed that administering IL-7 to mice following irradiation may facilitate recovery from RIL and enhance the prognosis. 74 Studies have shown that IL-7 levels, both pre- and post-RT, significantly correlate with lymphocyte counts in hepatocellular carcinoma patients undergoing RT, indicating IL-7's potential role in both the development and recovery from lymphopenia, which could improve prognosis, particularly given the link between higher pre-RT lymphocyte counts and improved OS and PFS in these patients.75,76 Moreover, in a trial that included patients with metastatic breast cancer, recombinant IL-7 administration before chemotherapy was found to increase lymphocyte counts. 77 Additionally, a more favorable treatment response was observed in a limited sample size. It would be beneficial to conduct further evaluation of the impact of IL-7 on RIL and outcomes in EC in a larger cohort in the future.
In the current meta-analysis, with the exception of post-NLR,16–18,31 most of the lymphocyte-related parameters included in the studies were assessed prior to or during CRT. The prognostic impact of late lymphocyte-related parameters after CRT remains unclear. It would be beneficial for future research to dedicate more attention to the prognostic impact of post-RT lymphocyte-related parameters in EC. Furthermore, only a limited number of studies have investigated the prognostic impact of lymphocyte-related parameter recovery. One study included in the meta-analysis demonstrated that lymphocyte count recovery after CRT is unable to improve the poor 5-year OS in patients experiencing G4 lymphopenia during CRT. 27 Conversely, another study included in our meta-analysis indicated that for patients with a high NLR during CRT, NLR recovery after CRT was associated with better OS and PFS. 18 Therefore, researchers in the future should also focus on the prognostic effect of lymphocyte-related parameters recovery after CRT. Nevertheless, these findings suggest that preventing a reduction in lymphocyte levels before, during, or after CRT may be beneficial for EC prognosis.
There were some limitations in the current study. First, there existed certain heterogeneity in the current meta-analysis. This included inconsistencies in the thresholds applied to each parameter and variations in the concurrent chemotherapy rate and surgery rate. Further work should define reproducible cutoff points for which these may be of most value. Second, due to the predominantly retrospective design of the included studies, limitations such as missing data, loss to follow-up, and potential confounding must be considered. And it is not possible to infer a potential causal relationship between lymphocyte-related parameters and EC prognosis based on the current meta-analysis, which means more evidence from prospective studies are required. Third, we have to acknowledge that there may be some inclusion bias because we only consider English language articles, which may lead to missing important information from non-English articles.
Conclusion
In conclusion, prior to CRT, high NLR and low LMR are both linked to worse OS of EC patients, and low LMR is also related to inferior PFS. During CRT, low lymphocyte count is observed to reduce both OS and PFS. Furthermore, post CRT, high NLR is related to unfavorable OS. The most frequently used chemotherapy regimen was fluorouracil plus cisplatin. The application of these inexpensive and readily available prognostic biomarkers may help clinicians to more precisely select lymphocyte-related parameters at different time points around CRT, enabling early risk stratification and proactive intervention in patients with EC. Further prospective, large-scale investigation are asked to explore whether an improvement of these parameters could represent a novel target for EC treatment and to enhance comprehension of its underlying mechanisms.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251341431 - Supplemental material for Prognostic Significance of Dynamic Lymphocyte Changes in Esophageal Cancer Patients Receiving Fluorouracil-Cisplatin Combined with Radiotherapy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-tct-10.1177_15330338251341431 for Prognostic Significance of Dynamic Lymphocyte Changes in Esophageal Cancer Patients Receiving Fluorouracil-Cisplatin Combined with Radiotherapy: A Systematic Review and Meta-Analysis by Cong Zhang, Zhi Yang, Jie Li and Lina Zhao in Technology in Cancer Research & Treatment
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 82272941).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
