Abstract
Background
RAD51 is a central protein involved in homologous recombination, which has been linked to cancer development and progression. systemic inflammatory indicator markers such as neutrophil-to-lymphocyte ratio and lymphocyte-to-monocyte ratio have also been implicated in cancer. However, the relationship between Rad51 and these inflammatory markers in esophageal cancer patients undergoing esophagectomy is not yet understood.
Methods
We retrospectively observed 320 esophageal cancer patients who underwent esophagectomy. We collected clinical characteristics, postoperative complications, and survival analysis data and analyzed the relationship between Rad51 expression, inflammatory markers, and prognosis.
Results
We found significant linear relationships among the inflammatory markers. There were also close relationships between Rad51 expression and neutrophil-to-lymphocyte ratio or C-reactive protein. Patients with low lymphocyte percentage were more likely to have low Rad51 expression (P = .026), high C-reactive protein (P = .007), and high neutrophil-to-lymphocyte ratio (P = .006). Low lymphocyte-to-monocyte ratio was associated with poor overall survival and was an independent prognostic factor (HR = 2.214; 95% confidence interval: 1.044-4.695, P = .038). In patients without lymph node metastases, low albumin (HR= 0.131; 95% confidence interval: 0.025-0.687, P = .016), high neutrophil-to-lymphocyte ratio (HR = 0.002; 95% confidence interval: 0.000-0.221, P = .009), and high Rad51 expression (HR = 14.394; 95% confidence interval: 2.217-97.402, P = .006) were associated with poor overall survival.
Conclusions
Our study found a close correlation between elevated Rad51 expression and inflammatory markers. High Rad51 expression, high neutrophil-to-lymphocyte ratio, and low lymphocyte-to-monocyte ratio are associated with lower survival rates. The combined assessment of Rad51 and inflammatory markers can be useful for preoperative assessment and prognostic evaluation in esophageal squamous cell carcinoma patients.
Introduction
Esophageal cancer (EC) is the eighth most common cancer and the sixth leading cause of cancer death worldwide. 1 China has a high incidence of EC, especially the esophageal squamous cell carcinoma (ESCC) subtype, which accounts for more than 90% of all EC cases.2,3 Despite advances in surgery, radiotherapy, chemotherapy, and targeted therapy for EC, the survival prognosis for ESCC remains poor. This is mainly because nearly half of the patients have lymph node metastasis at diagnosis, and the overall 5-year survival rate is low. Therefore, it is crucial to accurately predict patients’ survival and tailor the therapy to their needs. Several potential prognostic indicators of EC have been identified, such as histological features (tumor length, grading, differentiation, invasion depth, and lymph node classification) and nutrition or inflammation-based factors. Inflammation plays an important role in cancer development and progression by altering the tumor microenvironment. In recent years, some blood inflammatory biomarkers or scores have been shown to be predictive of survival for various cancers, 4 including ESCC. These include C-reactive protein (CRP), albumin (ALB), body mass index (BMI), lymphocyte percentage (LYM%), neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), and platelet-to-lymphocyte ratio (PLR).
It is well known that inflammation and cancer are closely linked. Inflammation can increase the risk of cancer and promote its growth and spread through various mechanisms, such as stimulating cell division, preventing cell death, enhancing blood vessel formation, facilitating metastasis, suppressing immunity, and so on.
A novel concept is emerging, suggesting that acquired genetic instability in cancer cells can result from the dysregulation of crucial DNA repair pathways attributed to inflammation. The presence of chronic inflammation within the tumor microenvironment promotes tumor growth through the production of cytokines and heightened macrophage activity. 5 Within the tumor microenvironment, macrophages release pro-inflammatory cytokines and contribute to genomic instability by generating reactive oxygen species (ROS) and reactive nitrogen species, thereby inducing various forms of DNA damage such as oxidized bases, single-strand breaks, and double-strand breaks. 6 These DNA lesions are subsequently repaired through distinct DNA repair pathways. The interplay between inflammation and DNA damage acts synergistically to induce mutations that drive cancer progression and recurrence. Moreover, many cancers exhibit an inflammatory microenvironment that facilitates tumor growth. 7
RAD51 plays a pivotal role as a mediator in facilitating homologous base pairing and strand exchange during the process of homologous recombination (HR)-mediated DNA repair in mammalian cells.8,9 Its primary function involves identifying and invading homologous DNA sequences to enable accurate and timely DNA repair. RAD51 is required to protect human cancer cells from transcription-replication conflicts and protects genomic regions that are prone to amplification in human cancers. Maintaining accurate DNA repair and replication processes is essential for preserving genomic stability and preventing cancer. Elevated levels of RAD51 protein have been observed in numerous cancer cell lines and primary tumors. 10 Previous bioinformatic analyses have provided supporting evidence for the potential clinical significance of RAD51 in cancer management, including cancer diagnosis, prognosis, and therapeutic prediction.
The interactions between inflammation and HR in cancer are complex. 11 DNA damage responses, repair pathways, and tolerance mechanisms can trigger more inflammation in a positive feedback loop. Conversely, inflammatory transcription factors can protect DNA from damage by neutralizing reactive chemicals with antioxidants and boosting DNA repair pathways. 12
Previous studies have systematically reviewed the role of some inflammation and nutrition-based indicators in the prognosis of EC. 10 However, no studies have explored the relationship between RAD51, the key mediator of HR and the inflammation status and postoperative outcomes of ESCC patients.
The purpose of this study was to evaluate the prognostic value of combining RAD51 expression and systemic immune inflammation indicators in EC patients.
Material and Methods
Patients
In this retrospective study, we consecutively collected and analyzed 320 patients who underwent esophagectomy and lymph node dissections from April 1, 2007, to December 31, 2015. The inclusion criteria were: (1) pathologically confirmed ESCC diagnosis; (2) no acute or chronic inflammation before surgery or any treatment that affected blood indicators; and (3) no neoadjuvant radiotherapy or radiochemotherapy before surgery. We excluded patients who had: (1) infectious disease or other inflammatory conditions; (2) incomplete resection; or (3) missing or incomplete medical records. We have de-identified all patient details. The reporting of this study conforms to STROBE guidelines and REMARK guidelines.13,14 This study was approved by the Ethics Committee of Huashan Hospital, Fudan University.
Clinical Data
We performed preoperative staging for all patients using barium meal test, fibro-gastroscopy, chest and abdomen computed tomography (CT), and neck and retroperitoneal lymph node ultrasound. Sixteen patients also received whole-body positron emission tomography (PET)-CT scanning. We assessed all patients for their physiological ability to undergo esophagectomy. These assessments included pulmonary function test, cardiac function test, and nutritional assessment. We performed transthoracic esophagectomy via left thoracotomy with 2-field lymphadenectomy for 186 patients with tumors in middle or lower thoracic esophagus and no evidence of lymph node involvement in the superior mediastinum or neck region. We performed McKeown 3-hole esophagectomy with three-field lymphadenectomy for 53 patients with tumors in the middle or upper thoracic esophagus and possible lymph node metastasis in the superior mediastinum or neck region.
We used the AJCC eighth edition guidelines for TNM staging. We followed up patients every 6 months for the first 3 years and then annually. Overall survival (OS) was calculated as the time from surgery start to the time of any cause of death or the last follow-up. Disease-free survival (DFS) was the time from surgery start to the date of relapse or death or the last follow-up. To diagnose recurrent or metastatic disease, endoscopy, CT scans, PET-CT scans, and radionuclide bone scans were conducted as needed. The patients’ weight and height were measured during their first hospitalization, and their BMI was calculated by dividing their weight in kilograms by their height in meters squared. Serum ALB levels were measured using automated equipment, and patients with acute or chronic active liver disease were excluded. Serum CRP levels were measured using a latex turbidimetric immunoassay. Routine hematology tests and the complete blood count test were carried out within two weeks prior to surgery. Two milliliters of ethylenediaminetetraacetic acid anticoagulated peripheral blood and 2 mL plasma anticoagulated by sodium citrate as well as serum samples were collected before breakfast. The median time between blood inflammation indicators tests and histological examination was 5 days (range, 2-9 days). The NLR was calculated as the ratio of neutrophils to lymphocytes, the PLR was calculated as the ratio of platelet counts to lymphocyte counts, and the LMR was calculated as the ratio of lymphocyte counts to monocyte counts. All reagents and equipment were convenience-validated and standardized in the central clinical laboratory. Patients were initially treated with total parenteral nutrition during the first few days after surgery. From the second or third postoperative day, patients received an initial dose of 5 to 10 kcal/d/kg of enteral nutrition via a feeding tube, which was gradually increased to the full dose of 25 to 30 kcal/d/kg. Some patients with low BMI or low serum ALB levels received a full dose of enteral nutrition from one week before surgery to the tenth day after surgery.
Tissue Microarray and Immunohistochemistry
The study also conducted TMA and immunohistochemistry on 90 cases of ESCC using the anti-RAD51 primary antibody. All these tissue samples were obtained from the HuaShan Hospital, Fudan University. Sections were cut at 4 µm, mounted on silane-coated slides, and subjected to antigenic retrieval in EDTA [pH 8.0] for 2 min at 105 °C. IHC was done using the anti-RAD51 primary antibody (SC 8349, Santa Cruz Biotech; 1:40 dilution, 2 h incubation at 37 °C) and the MACH 2™ Polymer Detection system according to the manufacturer's (Biocare Medical). The immunoreactive score system based on the staining intensity and area was visualized using a scoring system from 0 to 12, was determined as the multiplication of a percentage of labeled thyrocytes score (0, <5% labeling; 1, 5%-25%; 2, 26%-50%; 3, 51%-75%; 4, >75% of labeled thyrocytes), and an intensity score (0, no staining; 1, weak; 2, mild; 3, strong staining), where low and high expression were defined as scores of 0 to 7 and 8 to 12, respectively.
Statistical Analysis
All statistical analysis was performed using SPSS 22.0. The study used Spearman's rank-order correlation coefficient for nonparametric data and the Kaplan-Meier method and log-rank test for univariate analysis of survival. For multivariate analysis, the study used Cox-regression model to estimate hazard ratios (HRs) with 95% confidence intervals (CIs) and identify independent prognostic factors. The level of significance was set to P < .05.
Results
Overall Survival and DFS With Regard to Patient Characteristics, Comorbidities, and Postoperative Complications
The clinical characteristics and 5-year survival outcomes of the patients are summarized in Table 1. The OS rates at 1 year, 3 years, and 5 years were 85.8%, 58.9%, and 51.9%, respectively. The DFS rates at 1 year, 3 years, and 5 years were 76.6%, 54.8%, and 48.8%, respectively.
Patient Characteristics and Univariate Analysis.
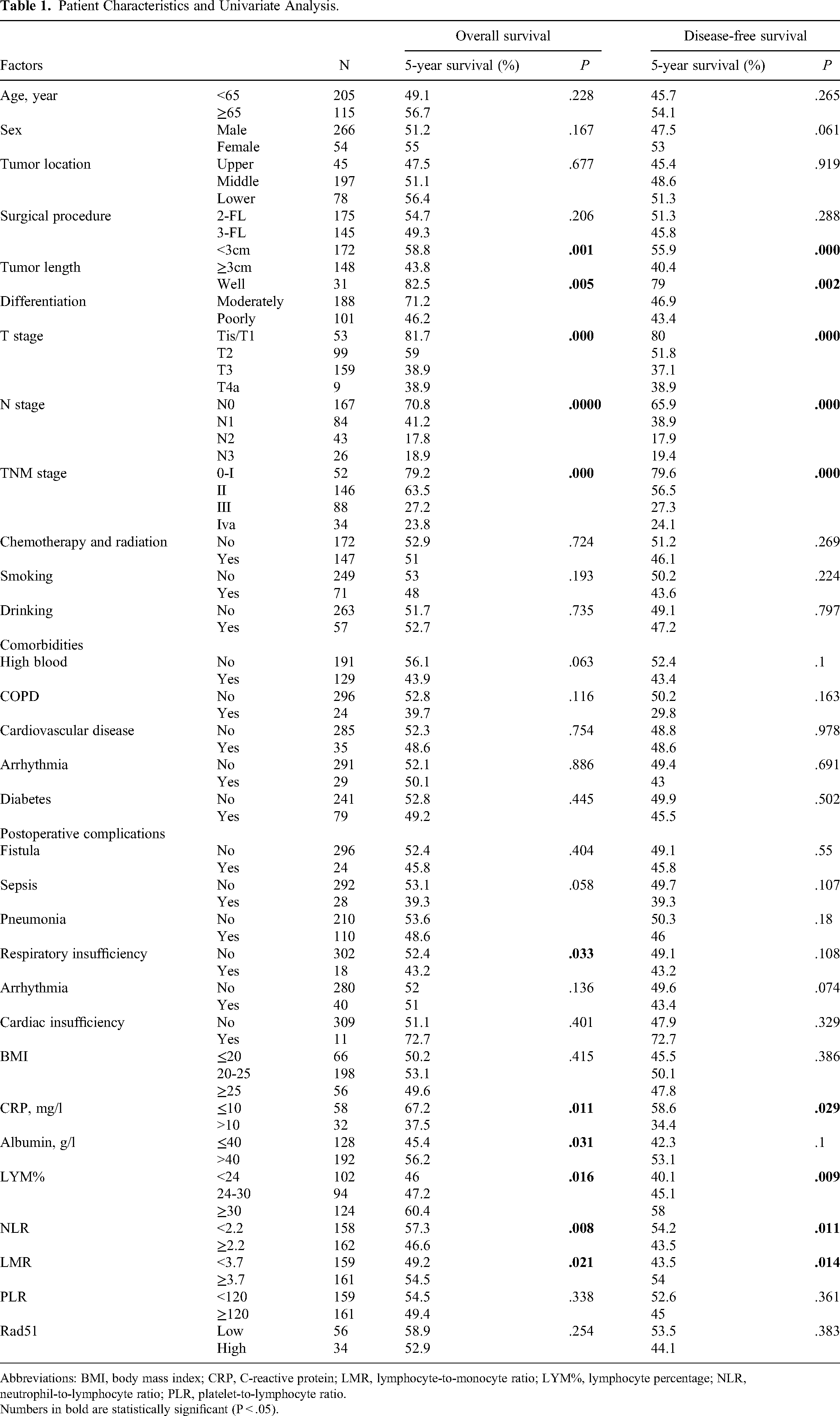
Abbreviations: BMI, body mass index; CRP, C-reactive protein; LMR, lymphocyte-to-monocyte ratio; LYM%, lymphocyte percentage; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Numbers in bold are statistically significant (P < .05).
Univariate survival analysis was performed and revealed that the patients’ tumor length (P = .001 and P < .001 for OS and DFS, respectively), differentiation (P = .005 and P = .002 for OS and DFS, respectively), T-stage (both P < .001), N stage (both P < .001), and TNM stage (both P < .001) were significantly associated with both OS (Figure 1) and DFS. However, for preoperative comorbidities and postoperative complications, only postoperative respiratory insufficiency was significantly associated with OS (P = .033), but not DFS (P = .108).

Kaplan-Meier overall survival curves of esophageal cancer patients with curative resection stratified by clinicopathologic features.
Overall Survival and DFS With Regard to Rad51, Pretreatment Inflammation-Based Indicators
Positive Rad51 immunochemical staining was mainly found on the nucleus of ESCC cells. Among the 90 cases, Rad51 had a low expression in 56 cases and in the rest 34 cases Rad51 was highly expressed. In this study, we conducted a univariate analysis of inflammation-based prognostic indicators (Figure 2) and found that patients with low CRP (P = .011 and P = .029, respectively) and low NLR (P = .008 and P = .011, respectively) groups exhibited better OS and DFS, as did those with high LYM% (P = .016 for OS and P = .009 for DFS, respectively) and high LMR (P = .021 for OS, and P = .014 for DFS, respectively). In addition, the high ALB group also had better OS (P = .031).

Kaplan-Meier overall survival curves of esophageal cancer patients with curative resection stratified by inflammation-based indicators.
We also investigated the relationship between Rad51 expression and OS and found no significant association (P = .254). However, upon further analysis, we found that Rad51 was a worse OS factor in the N0 group (P = .006).
Patient Characteristics With Regard to Rad51 and Inflammation-Based Indicators
Table 2 shows the patient characteristics with respect to Rad51 and inflammation-based indicators. Spearman's ρ indicated that patients with high NLR were more likely to have a large tumor size (P < .001), poor differentiation (P = .033), advanced T-stage (P = .005), and N-stage (P = .024). Patients with low ALB tended to have advanced N-stage (P < .001).
Associations Among Characteristics, Rad51, Albumin, CRP, NLR, and LMR.

Abbreviations: CRP, C-reactive protein; LMR, lymphocyte-to-monocyte ratio; LYM%, lymphocyte percentage; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio.
Numbers in bold are statistically significant (P < .05).
Smoking and drinking patients were more likely to have low LYM% (P = .038 and P = .011, respectively). Furthermore, patients with low LYM% were more likely to have a large tumor size (P < .001). Hypertension was more frequent in high NLR patients (P = .048), and diabetes was more common in low ALB patients (P = .044).
Interestingly, we explored the relationship between Rad51 expression and inflammation-based indicators and found that patients with low LYM% tended to have low Rad51 expression (P = .026), low ALB (P = .014), low LMR (P < .001), high CRP (P = .019), and high NLR (P < .001). We observed a close relationship between Rad51 expression and NLR or CRP. Low Rad51 expression was more likely to be seen in patients with low LYM% (P = .026), high CRP (P = .007), and high NLR (P = .006).
Univariate analysis revealed that low Rad51 expression (for N0 patients), low LYM%, low ALB, low LMR, high CRP, and high NLR are worse prognostic factors for ESCC patients. These findings were consistent with the Spearman correlation analysis, which identified significant linear relationships between Rad51 expression and inflammation-based indicators.
Multivariate and Subgroup Analysis
Variables that showed significance in the univariate analysis, including tumor length, differentiation, T-stage, TNM-stage, ALB, NLR, LMR, LYM, CRP, and respiratory insufficiency, were included in a multivariate analysis (Table 3). TNM-stage (HR = 1.745; 95% CI 1.109-2.746; P = .016), LMR (HR = 2.214; 95% CI 1.044-4.695; P = .038), LYM (HR = 0.466; 95% CI 0.221-0.981; P = .044), and respiratory insufficiency (HR = 5.037; 95% CI 1.037-24.463; P = .045) were identified as independent prognostic factors.
Multivariate Prognostic Analysis for Overall Survival.
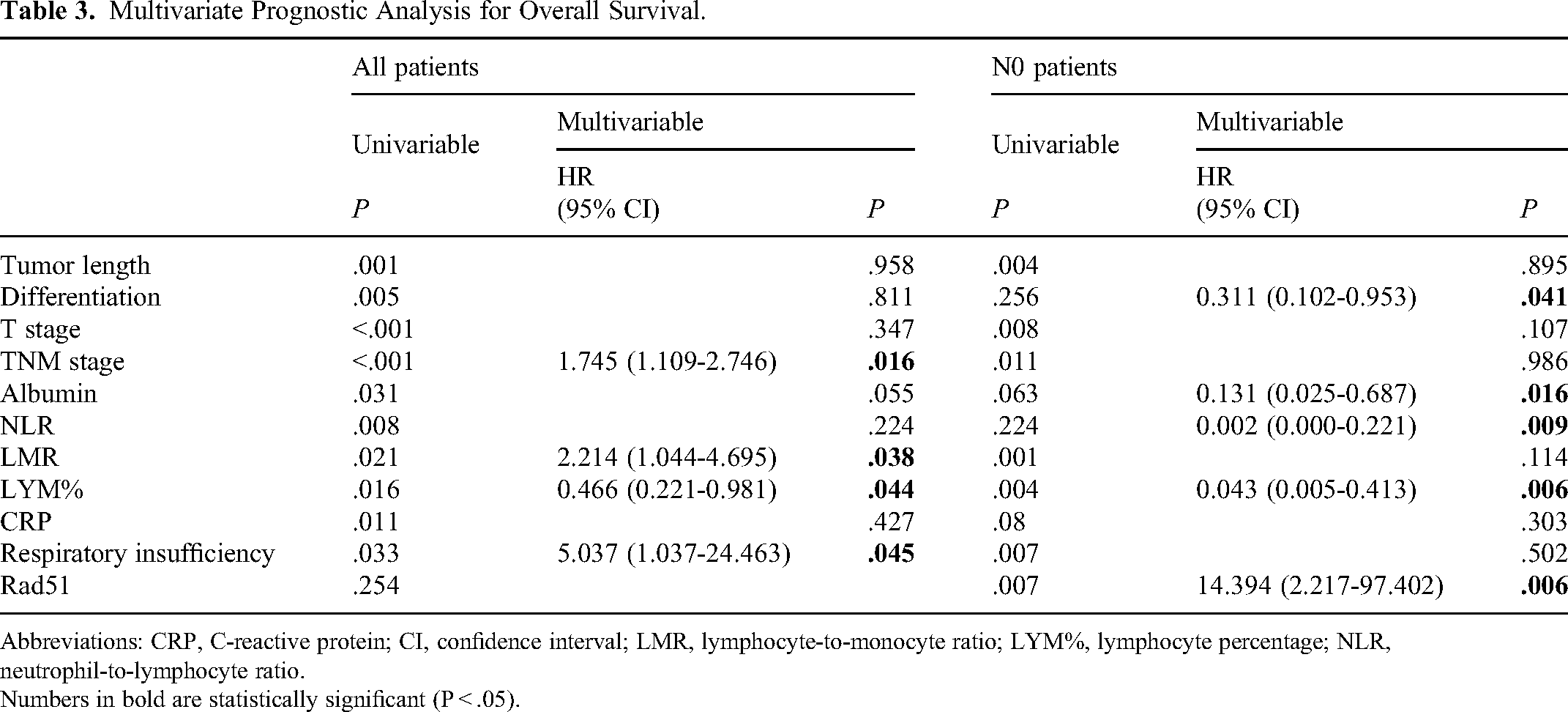
Abbreviations: CRP, C-reactive protein; CI, confidence interval; LMR, lymphocyte-to-monocyte ratio; LYM%, lymphocyte percentage; NLR, neutrophil-to-lymphocyte ratio.
Numbers in bold are statistically significant (P < .05).
To evaluate the impact of BMI and REE on different tumor stages, we divided patients into subgroups based on N-stage (N0 vs N1-4). Patients with higher Rad51 expression had significantly better OS in the N0 subgroup (P = .006), but there was no statistically significant difference in the N1-4 subgroup (P = .364). Multivariate analysis of subgroups revealed that differentiation (HR = 0.311; 95% CI 0.102-0.953; P = .041), ALB (HR = 0.131; 95% CI 0.025-0.687; P = .016), NLR (HR = 0.002; 95% CI 0.000-0.221; P = .009), LYM (HR = 0.043; 95% CI 0.005-0.413; P = .006), and Rad51 expression (HR = 14.394; 95% CI 2.217-97.402; P = .006) were independent prognostic factors for N0 patients.
Discussion
Inflammation plays a crucial role in the development and progression of several malignant tumors, and it has been linked to various steps involved in tumorigenesis, including cellular transformation, promotion, survival, proliferation, invasion, angiogenesis, and metastasis.4,12 The tumor's inflammatory microenvironment is characterized by the presence of host leukocytes in both the supporting stroma and tumor areas. Tumor-infiltrating lymphocytes may contribute to cancer growth and spread and to the immunosuppression associated with malignant disease. Peripheral blood cell analysis provides an opportunity to establish a prognostic prediction model based on inflammatory-related indicators. The NLR, PLR, and LMR are all essential indicators of systemic inflammation in the body, which can be used to assess the nutritional status of patients and may serve as biomarkers for the tumor diameter, lymph node metastasis status, TNM staging, and prognosis of tumor patients. Previous studies have systematically reviewed the role of these inflammation-based indicators in the prognosis of various tumors.
As a simple and inexpensive biomarker that reflects systemic inflammation and immune status, previous studies have thoroughly investigated the role of NLR in tumors. On the one hand, tumors are often accompanied by systemic inflammation activation, and neutrophil elevation is itself a marker of tumor progression. 15 Activated neutrophils can secrete vascular endothelial growth factor and leukotrienes, which are important factors supporting tumor occurrence and progression and facilitating colonization by cancer cells with higher tumorigenic potential. Neutrophils can cause DNA damage and gene mutation through ROS, nitric oxide, microRNAs, and matrix metalloproteinase 9, which induce carcinogenesis. Neutrophils can also eliminate senescent cells through interleukin-1 receptor antagonist and thus promote cancer progression. On the other hand, lymphocytes, especially CD4+ and CD8+ T cells, mediate innate immunity, which is the fundamental effector cell for infiltrating and killing tumors. The ratio of neutrophils to lymphocytes reflects the dynamic balance state of tumor progression and inhibition of innate immunity.
Recently, a particularly strong association between NLR and patient outcomes has been demonstrated in a variety of cancers. 16 One study analyzed the prognostic value of NLR in 32 types of cancer using a large database and found that high NLR was associated with worse survival in most cancers. Some studies have also found that high pre-treatment NLR is significantly associated with tumor grade, advanced stage, lymphovascular space invasion, lymph node metastasis, and recurrence in various cancers.16,17
For EC, the impact of NLR on long-term outcomes remains controversial. 18 While some studies have not found positive predictive values for NLR in EC cancer.19,20 Several other studies have suggested that elevated NLR is linked to poor survival outcomes, treatment resistance, tumor progression, and recurrence in patients who underwent surgery, chemotherapy, or chemoradiotherapy.21,22 Our study found that higher NLR correlated with larger tumor size, poorer differentiation, deeper invasion, advanced N stages, and advanced TNM stages of the tumor. Lymphocyte-to-monocyte ratio may be a useful biomarker for predicting the risk of developing EC.
However, the optimal cutoff value and the underlying mechanisms of NLR in EC are still unclear. In our study, significant differences in survival were observed when the NLR cutoff value was set at 2.2. The optimal cutoff value of NLR may vary depending on the study design, such as predicting pathological response to neoadjuvant chemotherapy or survival after recurrence or curative surgery.22,23
Lymphocytes and monocytes are two types of white blood cells that play a crucial role in the immune system's defense against infections and diseases. The ratio of lymphocytes to monocytes, known as LMR, is a crucial indicator of inflammation and immune system function. A high LMR ratio indicates a strong immune response against cancer cells, which may prevent cancer development and progression. Several studies have suggested a possible association between LMR and EC.
One study investigated the relationship between LMR and response to chemotherapy in patients with EC. The study included 204 patients with advanced EC who underwent chemotherapy. The results showed that patients with a high LMR had a better response to chemotherapy than those with a low LMR. The researchers suggested that both LMR and NLR could be useful biomarkers for predicting the response to chemotherapy in EC patients. 24 Another meta-analysis including 26 studies of 8586 ESCC patients found that high NLR and PLR and low LMR were associated with poor OS/cancer-specific survival and event-free survival and malignant phenotypes such as deeper depth of invasion (T), positive lymph node metastasis (N), and advanced TNM stage. 25
Our findings indicate that patients with a history of smoking and drinking are more likely to have a low LMR, and that those with a high LMR have a better OS rate than those with a low LMR. Our research suggests that LMR could be a useful biomarker for predicting the survival of patients with EC who undergo surgery.
Previous studies have demonstrated that HR undergoes spontaneous dysregulation in cancer cells, including esophageal adenocarcinoma. 26 This dysregulation contributes to the accumulation of ongoing genomic changes. 27 Inflammation has been associated with the production of specific factors and activities, such as growth factors, cytokines, and deaminase, which further disrupt DNA repair mechanisms, including HR, and compromise genome stability. Additionally, inflammation is known to induce oxidative stress, which has the potential to disrupt genomic integrity and stability, thereby promoting cancer development. 10 It is important to note that chronic inflammation, triggered by either intrinsic or extrinsic factors, can significantly contribute to oncogenesis. Conversely, cancer itself can also induce an inflammatory response. Rad51 is a protein involved in DNA repair by HR and plays a crucial role in DNA repair and recombination. In cancer cell lines, the inhibition of RAD51 resulted in suppressed HR-related strand exchange activity, as well as reduced occurrence of spontaneous DNA breaks and genomic instability induced by DNA damaging agents in live cell fractions. 28 Both Rad51 and inflammation have been implicated in cancer development and progression. Several studies have reported that RAD51 expression is altered in EC. However, limited research has specifically explored the relationship between Rad51 and inflammation in ESCC. Studies in other types of cancer have suggested that Rad51 expression may be influenced by inflammation. For instance, in EAC, breast, and colon cancer elevated expression of RAD51 correlated with genes involved in inflammation and HR activity, thus contributing to chemoresistance, immune dysregulation, and poor prognosis in cancer. 10
Inflammation may also play a role in EC through the activation of NF-κB, a transcription factor that regulates genes involved in immune response, inflammation, and cell survival. NF-κB has been shown to be upregulated in EC and is associated with poor prognosis. 29 In addition, activation of NF-κB has been shown to increase Rad51 expression in types of cancer cells, leading to enhanced DNA repair and resistance to chemotherapy. 30 Our study found that elevated expression of RAD51 correlated with inflammatory-related indicators including NLR, LYM%, and CRP. Further research is needed to fully elucidate the mechanisms underlying the relationship between Rad51 and inflammation in EC.
Patients with digestive tract malignancies are more likely to suffer from hypoalbuminemia, which has been attributed to increased catabolism, obstruction of the digestive tract, and the systemic inflammatory response. 4 The combined use of ALB and serum CRP has been introduced by the Glasgow prognostic score,31,32 and the combined use of ALB and lymphocyte counts has been mentioned by Onodera's prognostic nutritional index. Hypoalbuminemia has been shown to correlate with ideal body weight, weight loss, body cell mass, and poor prognosis in cancer patients. 33
There are several limitations to our study. Firstly, it is a retrospective study, and the sample size is not large. Secondly, we explored the relationship between Rad51 and inflammatory-related indicators, but the underlying mechanisms are still unclear.
Conclusion
In conclusion, our study is the first to explore the relationship between Rad51 and inflammatory-related indicators. We found that high RAD51 expression, high NLR, and low LMR are associated with low survival rates. It is advisable to use the combined assessment of RAD51 and inflammatory-related indicators for better preoperative assessment and prognostic evaluation in EC patients.
Footnotes
Abbreviations
Authors’ Note
Ethics Statement: This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Huashan Hospital, Fudan University (address: No.12 Middle Wulumuqi Road, Shanghai, China; approval number: 2022-614; date: August 15, 2022). All individual participants agreed the study and signed informed consent.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
