Abstract
Introduction
Breast radiotherapy is associated with a higher risk of cardiac diseases. Although deep inspiration breath-hold (DIBH) reduces the heart dose, it is underutilized. The selection of proper candidates for DIBH remains an unresolved issue. This study compared dosimetric parameters between free breathing (FB) and DIBH, monitored myocardial enzymes, and aimed to identify factors that can predict cardiac injury thus developing a method to identify proper patients for DIBH.
Methods
This is a prospective cohort study, enrolling 58 patients with left-sided breast cancer following breast-conserving surgery. All patients underwent computed tomography scans in both FB and DIBH states. A comparative analysis of dosimetric features between DIBH and FB was conducted. Myocardial enzyme was monitored until six months post-radiation therapy. T-tests were used to assess differences between the DIBH and the FB. Pearson correlation and receiver operating characteristic (ROC) analysis was conducted to identify factors associated with the subclinical acute cardiac injury.
Results
The mean heart dose (MHD) of the DIBH group significantly dropped as compared to the FB group (3.81 Gy vs 1.65 Gy p = 0.001). Cardiac V40, V30, V25, V10, and V5 volumes also significantly reduced. 9(15.51%) patients exhibited increased myocardial enzyme, with cTnI being the most sensitive indicator. The heart dose was a predictor for the cardiac enzyme's elevation. The ROC curve analysis revealed an area under the curve of 0.6. With an MHD threshold of 2 Gy, both sensitivity and specificity exceeded 0.7.
Conclusion
DIBH significantly diminishes radiation exposure to the heart and LAD compared with FB. Cardiac enzyme analysis facilitates the early detection of cardiac injury following radiation therapy. An MHD threshold of less than 2 Gy is associated with a reduced risk of subclinical cardiac injury, potentially obviating the need for DIBH, which optimizes clinical efficiency and economic viability.
Background
Breast cancer is among the most prevalent malignancies in women, and radiation therapy constitutes a critical component of the therapeutic regimen. Adjuvant radiotherapy can improve local control and overall survival in breast cancer patients undergoing breast-conserving surgery (BCS) and mastectomy. Nonetheless, as survival rates for patients with breast cancer continue to increase, radiation-related cardiotoxicity is emerging as a salient concern. Left-sided breast cancer patients undergoing radiotherapy exhibit an augmented risk of long-term cardiotoxicity, which includes a spectrum of cardiac conditions such as cardiac insufficiency, heart failure, valvular heart disease, and diverse arrhythmias, especially coronary artery disease.1–6 Darby et al demonstrated that the incidence of major coronary artery events increased by 7.4% for every 1 Gy increase in mean heart dose (MHD). 7 In addition to MHD, other factors contribute to cardiotoxicity, including fraction size, left anterior descending branch (LAD) dose, and left ventricular dose, preexisting cardiovascular risk factors, and prior cardiovascular disease.8,9
The delayed onset is an important feature of radiation-induced cardiotoxicity, with the risk of cardiotoxicity increasing progressively over time. This suggests that early subclinical cardiac dysfunction may be detectable long before the overt clinical manifestations of cardiac disease are apparent. 5 Following cardiomyocyte injury, the release of specific cardiac enzymes into the circulatory system is well-documented. Among these enzymes, creatine kinase (CK), its MB isoenzyme (CK-MB), and cardiac troponin (cTn) are commonly identified. Notably, cTnT exhibits a high degree of specificity for cardiac muscle tissue. 10 Prior studies have indicated that cTnI levels in patients undergoing chemotherapy can detect cardiotoxicity in the subclinical phase before a decrease in left ventricular ejection fraction (LVEF). 11 Few studies have examined changes in the heart during and immediately after radiation therapy, with biomarkers, echocardiography (ECHO), and strain imaging, demonstrating the value of these screening modalities in identifying radiation-related cardiotoxicity.12–15 D'Errico et al reported higher N-terminal pro-brain natriuretic peptide (NT-proBNP) levels in patients 5–22 months post-radiotherapy compared to pre-treatment levels. 12 Furthermore, the correlation between cardiac enzymes and prognosis in other diseases suggests that early alterations in biomarkers after radiation may indicate long-term cardiotoxicity. A study revealed that elevation of CK-MB after percutaneous coronary intervention (PCI) was associated with a higher 2-year mortality rate. 16 The same results were obtained for one-year mortality. 17 Recent studies have demonstrated that statin therapy significantly reduces the risk of major adverse cardiovascular events in breast cancer patients treated with radiation therapy. 18 Collectively, these findings underscore the importance of early monitoring for cardiotoxicity and the implementation of effective interventions to manage radiation therapy-induced cardiotoxicity, potentially leading to a survival benefit for patients.
As the recognition of the impact of cardiotoxicity on patient survival deepens, there is an active pursuit among researchers to mitigate this effect. Strategies such as heart shielding, partial breast irradiation, and deep inspiration breath-hold (DIBH) techniques have been implemented to reduce the radiation dose to the heart.19,20 Among these, DIBH technique has emerged as one of the most effective methods. It capitalizes on the patient's breath-holding motion to deliver radiation therapy in intermittent intervals of chest wall/ breast movement, effectively separating the chest wall from the heart and LAD, thereby diminishing the radiation dose to the heart. Although a plethora of studies has demonstrated the dosimetric advantages of using DIBH, its implementation in clinical practice remains limited. A survey conducted by van der Laan et al revealed that only 19% of Europeans practitioners used DIBH techniques in their clinical routines. 21 Another study by Desai et al also reported that in the UK, only 43% of doctors regularly employed DIBH for cardiac protection during radiation therapy. 22 The underutilization of DIBH technique can be attribute to increased planning and treatment time and increased workload for departments. Consequently, in treatment-intensive centers, the identification of beneficiaries for the DIBH technique has emerged as a salient issue, and it is the focus of the present research
In this study, we first conducted a comparative analysis of dosimetric parameters between free breathing (FB) and DIBH techniques. Furthermore, we monitored actual clinical changes in myocardial enzymes and endeavored to reveal the dosimetric and clinical factors associated with subclinical cardiac injury, aiming to select optimal patients for the DIBH technique and improve clinical outcomes.
Methods
This is a prospective cohort study and it enrolled patients diagnosed with early-stage left-sided breast cancer at our institution from January 2021 to October 2022. The eligibility criteria included histopathological confirmation of early-stage left-sided breast cancer, undergoing BCS with a requirement for whole-breast radiotherapy, and the ability to hold their breath for at least 35 s. Patients were excluded if they were pregnant, had a history of prior breast or chest wall radiotherapy, or failed to perform DIBH. Ethical considerations were strictly adhered to, with all participants providing written informed consent, and the study protocol was approved by the institutional ethics committee. All patient details had been de-identified. The reporting of this study conforms to STROBE guidelines. 23
Simulation Positioning and Treatment Plan Design
During simulation process, patients were positioned in the supine position. The breast was stabilized using a breast bracket, a supportive device designed to immobilize the breast tissue and facilitate accurate radiotherapy delivery. Both arms were positioned overhead and extended, forming at least a 90-degree angle. The contours of the breast tissue and surgical incision were marked with lead wires. Non-contrast-enhanced CT scans were performed using a Siemens Somatom Emotion 16-slice CT scanner for each patient, both in FB and DIBH conditions. The two sets of CT images were transferred to our treatment planning system for further analysis and plan generation. The treatment planning was executed using Elekta Versa HD machine and Monaco TPS v.5.11 software. The target volume and organs at risk (OAR) contouring were performed in accordance with the Radiation Therapy Oncology Group (RTOG) guidelines. The contouring of heart substructures followed the University of California, Michigan Heart Atlas. 24 Intensity-modulated radiotherapy (IMRT) was used with four tangent fields, and a multileaf collimator was utilized for the irradiation to minimize the heart dose. For each patient, two treatment plans were formulated: one under DIBH and the other under FB conditions. Both plans prescribed a total dose of 42.6 Gy to the breast using 6 MV-x-rays, administered in 16 fractions over four weeks with a fraction size of 2.65 Gy per fraction. A supplementary boost dose was administered to the tumor bed, with a total dose ranging up to 53 Gy in 20 fractions, using an electron beam with a specific energy level determined based on the initial depth of the tumor. Despite the development of two plans, the DIBH technique was exclusively utilized in the treatment of all patients involved. The planning acceptability criteria were as follows: 100% of the planning target volume of the breast (PTV breast) must be covered by at least 95% of the prescribed dose range, and the dose to any 2cc volume within the high-dose region must not surpass 110% of the prescribed dose.
Cardiac Enzyme Monitoring
Cardiac enzyme tests were conducted on patients two weeks before radiotherapy, throughout the treatment, and at 3–6 month intervals post-treatment. The results were meticulously collated and subjected to rigorous analysis. In this study, any abnormal elevation beyond the normal range in any of the three cardiac enzymes that emerged after radiotherapy was defined as a subclinical acute cardiac injury.
Statistical Analysis
Data analysis was performed using IBM SPSS software version 25. Descriptive data were presented as mean, median, standard deviation, or percentage. Paired t-tests were used for continuous numerical variables to assess differences between the DIBH technique and the FB in specific anatomical or dosimetric parameters. For each anatomical and dosimetric parameter, the mean differences between FB and DIBH were recorded. Pearson's correlation was used to analyze patient-related parameters. The correlation between MHD in DIBH and FB states was investigated using linear correlation analysis. Receiver Operating Characteristic (ROC) analysis was conducted to identify factors associated with the occurrence of subclinical acute cardiac injury event, potentially aiding in the selection of patients for DIBH. A p-value of less than 0.05 was considered statistically significant.
Results
Patients and Characteristics
A total of 58 patients with left-sided breast cancer were included in this study, and 116 sets of CT scan images were obtained. The median age of the patients was 43.8 years (range: 27-63 years), and the body mass index (BMI) ranged from 17.64 to 29.36. In terms of medical histories, 4 patients exhibited a history of hyperlipidemia, 2 patients had diabetes, while 1 patient had hypertension. Notably, none of the patients had a documented history of coronary heart disease or familial cardiac disease. The tumor locations were as follows: 25 in the upper inner quadrant, 13 in the lower inner quadrant, 15 in the upper outer quadrant, 3 in the lower outer quadrant, and 2 in the central region. All patients underwent BCS and sentinel lymph node dissection, with negative margins. Among the 58 patients, 46 were diagnosed with invasive carcinoma and 12 with in situ carcinoma, with tumor diameters ranging from 0.2–5 cm. The number of sentinel lymph nodes removed ranged from 1 to 10. Of all patients, 54 were lymph node-negative,whereas 4 had micrometastases. Chemotherapy was administered to 23 patients, including 17 who received cardiotoxic drugs (doxorubicin, epirubicin). Endocrine therapy was provided to 47 patients and 15 received targeted therapy with trastuzumab. See Table 1 for details.
Baseline Characteristics of Patients.

Dosimetric Comparison
In comparison with FB, DIBH significantly reduced the MHD, with values of 3.81 Gy and 1.65 Gy, respectively (p = 0.001). Additionally, DIBH was effective in reducing the volume of the heart exposed to various dose thresholds, including heart V40, V30, V25, V10, and V5. In terms of the LAD, DIBH led to a significant reduction in both the mean (Dmean) and maximum (Dmax) radiation doses received; the Dmean decreased from 20.29 Gy to 7.14 Gy (p = 0.001), and the Dmax from 42.73 Gy to 25.81 Gy (p = 0.001). For the Right Coronary Artery (RCA), although DIBH induced a reduction in Dmean and Dmax, no statistical significance was observed, with Dmean values of 1.22 Gy and 0.72 Gy (p = 0.109), and Dmax values of 3.14 Gy and 1.65 Gy (p = 0.051), respectively. See Table 2 for details.
Dosimetric Comparison Between FB and DIBH Treatment Plan.

FB = free breathing; DIBH = deep inspiration breath-hold; heart dmean = mean dose to the heart; heart V40, V30, V25, V10, V5 = volume percentage of the heart receiving 40 Gy, 30 Gy, 25 Gy, 10 Gy, 5 Gy, respectively; LAD dmean = mean dose to the left anterior descending artery; LAD dmax = maximum dose to the left anterior descending artery; RCA dmean = mean dose to the right coronary artery; RCA dmax = maximum dose to the right coronary artery.
The Effect of DIBH on Cardiac Injuries
Among the 58 patients, a significant elevation in cardiac enzyme levels, exceeding the normal limits (cTnI ≤ 0.034 µg/L,CKMB-mass ≤ 5 µg/L, NT-proBNP ≤ 125 pg/mL), was documented in 15.51% of the cases (n = 9), predominantly occurring at 4 weeks post-radiotherapy, and persisted for 3 to 6 months before returning to normal levels. Serial assessment of cardiac enzyme levels was conducted at baseline, during treatment, and at 1, 3, and 6 months post-treatment for all enrolled patients. Nine patients experienced an elevation in cardiac enzymes (two during treatment, eight at 1 month post-treatment, and three at 3 months post-treatment); however, all patients returned to normal levels at 6 months post-treatment. cTnI was the most sensitive cardiac enzyme, with elevation observed in 5 patients, while an elevation of NT-proBNP was observed in 4 patients. See Table 3 and Figure 1 for details.

Dynamic changes of cardiac enzymes in patients.
Dynamic Changes of Cardiac Enzymes in Patients.
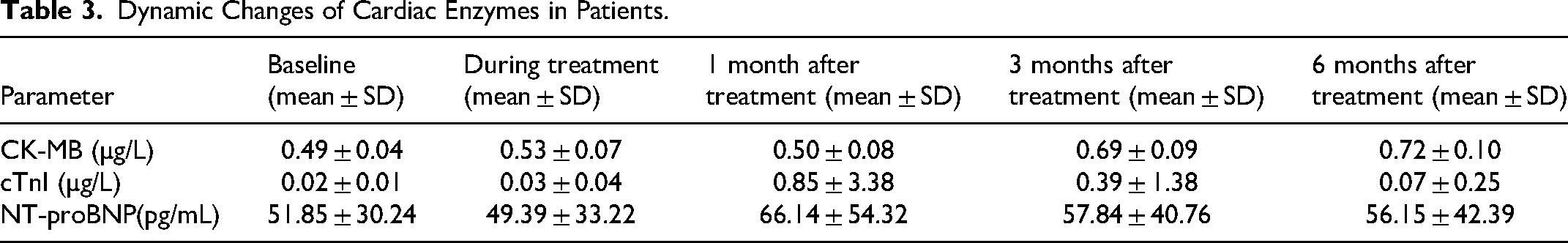
Analysis of Factors Influencing Cardiac Injuries
Among the 58 patients, 9 patients experienced subclinical cardiac injuries. The odds ratio or relative risk was utilized, and a cross-tabulation and binary logistic analysis were conducted to examine the factors associated with the occurrence of such injuries. The results showed no statistically significant correlation between the occurrence of cardiac injuries and factors including patient age, BMI, hyperlipidemia, hypertension, diabetes, chemotherapy, use of cardiac toxicity chemotherapy drugs, and targeted therapy. See Table 4 for details.
Analysis of Factors Influencing Cardiac Function.

BMI = body mass index.
Relationship Between Incidence of Cardiac Injuries and Dosimetry Analysis
The ROC curve analysis indicated that the MHD had a moderate predictive accuracy for identifying subclinical cardiac damage, with an area under the curve (AUC) of 0.6 (Figure 2A, Table 5A). When the 2Gy MHD was used as the cutoff value, both sensitivity (SE) and specificity (SP) exceeded 0.7, indicating that this cutoff value offers satisfactory diagnostic efficacy (Table 5B). Subclinical cardiac injury was less frequently observed in cases where the MHD was below 2Gy. Furthermore, a linear regression analysis assessed the relationship between MHD in the DIBH and FB conditions. The results demonstrated a substantial correlation between MHD across these two states (Pearson coefficient 0.874,p < 0.05 Figure 2B), indicating that MHD under DIBH can be predicted based on that in FB, thereby facilitating the screening of patients with DIBH.

ROC analysis of the relationship between the incidence of cardiac events and dosimetry. (A) ROC analysis of the relationship between the incidence of cardiac events and dosimetry (heart dmean, heart V5, heart V10, heart V25, heart V30, heart V40, LAD dmean, LAD dmax, RCA dmean, RCA dmax. (B) Scatter plot of heart dmean dose.
Analysis of the Relationship Between Cardiac Injuries Incidence and Dosimetry.

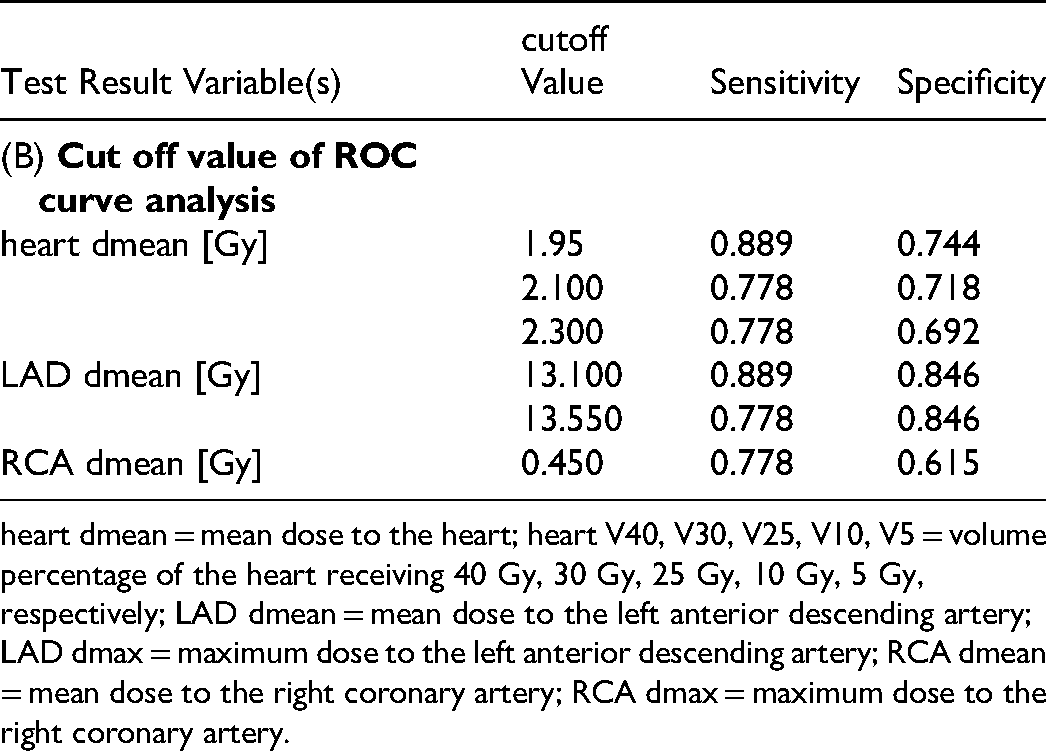
heart dmean = mean dose to the heart; heart V40, V30, V25, V10, V5 = volume percentage of the heart receiving 40 Gy, 30 Gy, 25 Gy, 10 Gy, 5 Gy, respectively; LAD dmean = mean dose to the left anterior descending artery; LAD dmax = maximum dose to the left anterior descending artery; RCA dmean = mean dose to the right coronary artery; RCA dmax = maximum dose to the right coronary artery.
Discussion
The present study elucidated the dosimetric superiority of DIBH technique in patients with left-sided breast cancer following BCS. Compared to the FB, the DIBH technique can reduce the dose to organs at risk (OAR), including the heart and LAD. The results showed a significant reduction in the MHD, maximum heart dose, and heart V5, V10, V25, V30, and V40. The mean and maximum LAD doses were also significantly reduced. While the mean and maximum RCA doses showed a decreasing trend, these differences were not statistically significant. These results are consistent with many previous studies.25,26 Smyth et al conducted a systematic review that included 10 studies on the benefits of DIBH in reducing measured mean heart and left ventricular dose. 27 The range of MHD under FB conditions was from 2.3 Gy to 6.9 Gy, while under DIBH it was from 1.3 Gy to 3.9 Gy. DIBH reduced the MHD by up to 3.4 Gy and the maximum LAD dose by up to 14.1 Gy.
The majority of extant studies have focused on dosimetric analysis and emphasized the reduction of heart and LAD doses. In contrast, the present study focuses on the practical application of DIBH and dynamically monitored changes in cardiac enzymes in patients to whom DIBH was applied. We analyzed the levels of cardiac enzymes in 58 patients at baseline, during treatment, and at 1, 3, and 6 months post-treatment. cTnI emerged as the most sensitive marker with elevations primarily observed 4 weeks after radiotherapy and recovery to normal levels after 3–6 months. The rationale for employing cardiac enzyme markers to detect radiation-induced cardiac injury lies in the sequential pathophysiological effects of radiotherapy. Initially, radiotherapy inflicts damage upon cardiac capillaries, precipitating myocardial ischemia. Subsequently, this ischemia leads to impairment of myocardial cells, enhanced membrane permeability, and alterations in cellular architecture. These cellular-level changes culminate in releasing large molecular weight substances from the compromised cells into the circulatory system, thereby serving as a biomarker for myocardial damage. 8 However, given the limited myocardial cell damage incurred by a single radiation therapy session, traditional markers such as CK-MB and electrocardiographic assessments exhibit diminished sensitivity. While cTnI is an early appearing, long-lasting marker that is only present in myocardial cells with high specificity, and is widely used in the diagnosis of minor myocardial injury in hypertension and coronary heart disease.28–30 Moreover, researches have detected a persistent elevation in cTnI levels even one month after radiotherapy.31,32 It also has been suggested that cTnT can serve as a biomarker of cardiac damage after radiation therapy, and is associated with myocardial damage and potential cardiac events. 33 Additionally, the elevation in cTnI observed in the present study further underscore the promise of cTnI as an early monitoring modality.
ROC curve analysis was applied,revealing that when the MHD value was below 2 Gy, the incidence of subclinical cardiac injury was low, with both SE and SP exceeding 0.7. Limited research has been devoted to the investigation of early subclinical cardiac injury and the determination of cardiac limiting doses. This study bridges that gap in knowledge. Furthermore, our study revealed that when free FB was employed, MHD for 9 patients was already notably low (<2 Gy), and no significant decrease was observed upon the application of DIBH. This indicates that effective patient selection is valuable for clinical practice. Studies have already attempted to establish predictive models based on anatomical and dosimetric parameters. 34 Nevertheless, there remains a paucity of clinical recommendations that can be applied. The present findings support that patients can be treated without DIBH when the MHD of FB status is lower than 2 Gy. This is due to two factors. First, the possibility of cardiac injury is less when MHD is below 2 Gy. Secondly, a correlation has been identified between the cardiac dose of DIBH and FB status, suggesting that an MHD of less than 2 Gy under FB conditions may not result in a substantial reduction in cardiac dose when the DIBH technique is employed.
The present study has several limitations. Firstly, the size of our study cohort is relatively small but similar to most published studies on DIBH. Secondly, we did not report any clinical outcomes or analysis related to normal tissue complication probability (NTCP). Additionally, although a correlation was identified between the cardiac dose of DIBH and FB, a predictive model for the dose of cardiac irradiation under DIBH was not established. In future studies, we will aim to develop improved dose prediction models to assist in the screening of patients with DIBH.
Conclusions
Based on the dose comparison described in our study, we conclude that DIBH significantly reduces the radiation dose to the heart and LAD compared to FB. Moreover, cardiac enzymes may serve as a tool for the early detection of cardiac injury following radiotherapy. The measurement of cardiac enzymes shows that the cardiac dose can predict subclinical damage to the heart after radiotherapy. Additionally, when the MHD value is below 2 Gy, the incidence of cardiac injury is low, with acceptable SE and SP. Patients with an MHD below 2 Gy may forgo the DIBH technique, potentially reducing the number of patients requiring DIBH, thereby improving clinical efficiency, and reducing clinical costs, all while preserving the overall benefit to patients.
Footnotes
List of Abbreviations
Acknowledgements
None
Ethical Approval and Consent to Participate
The Institutional Review Board (IRB) of Peking Union Medical College Hospital (PUMCH) reviewed the protocol on 2020-10-27. This prospective study follows a sound scientific rationale and aligns with ethical principles. The protocol has been reviewed and approved by the IRB. IRB code is ZS-2643. In 2021, an amendment was submitted and approved on 2021-8-9. We reapplied for ethical approval for data collected from 2023 onwards. The initial review resulted in approval with modifications (I-23YJ070). The re-review led to the approval of the study, with the latest approval number I-23PJ116. All patients have signed the informed consent form. Address of the Ethics Committee: No.1 Shuaifuyuan, Dongcheng District, Beijing, China.
Authors’ Contributions
SJ and ZK were responsible for data collection and drafted the manuscript;
MJB, MXY, YB, HXR participated in the design of the study;
SJ performed statistical analysis and data interpretation;
HK and ZFQ designed the study and revised the manuscript;
All authors read and approved the manuscript.
Funding
This research is funded by the following grants: The National Key R&D Program of China (Grant No. 2023YFC2411504) from the Ministry of Science and Technology of China; The National High-Level Hospital Clinical Research Funding (Grant No. 2022-PUMCH-B-116); Peking Union Medical College Hospital Outstanding Young Talent Development Program (Grant No. UHB04712); The National High-Level Hospital Clinical Research Funding [2022-PUMCH-A-099].
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
