Abstract
Introduction
Breast cancer remains a leading cause of cancer mortality despite being potentially curable when detected early, particularly in low- and middle-income countries where access to screening is limited. This is largely driven by operational gaps, including limited access to screening and delays in diagnosis and treatment. JULIETA is a portable bioimpedance spectroscopy device designed to identify electrical tissue patterns associated with potentially malignant findings and to prioritize women for further diagnostic evaluation. This study assessed the performance of a hierarchical algorithm integrated into JULIETA to distinguish findings without malignant potential (BI-RADS 1-2) from those with malignant potential (BI-RADS ≥3), using mammography as the reference standard.
Methods
A cross-sectional observational study with prospective data collection was conducted between May and July 2024 in four Colombian cities. Adult women undergoing screening or follow-up mammography were evaluated with JULIETA prior to imaging. Impedance-derived features, breast density estimates, and individual risk scores were used to retrain a hierarchical classifier combining Random Forest and SVM-RBF models, using an 80/20 stratified split and cross-validation.
Results
A total of 1350 women were recruited (mean age 56.5 ± 8.0 years); 67% were BI-RADS 1-2 and 21% BI-RADS 4. After data cleaning, 673 breasts (469 women) were included. The model achieved 73% sensitivity, 76% specificity, 65.5% positive predictive value, and 82.1% negative predictive value.
Conclusion
JULIETA is a feasible, safe, and reproducible noninvasive bioimpedance pre-screening tool that may enable scalable triage and support earlier detection and improved equity when integrated into public health pathways.
Introduction
Breast cancer is the most common malignant neoplasm affecting women worldwide. According to the World Health Organization, it is the most commonly diagnosed cancer in women worldwide and a leading cause of cancer-related mortality among women, with a disproportionate burden in low- and middle-income countries. 1 .
In Colombia, the most recent data from the High-Cost Account (CAC) indicate that, as of January 1, 2024, a total of 140,096 cases of breast cancer in women had been reported. In addition, between January 2,2023 and January 1, 2024, 9615 new cases were diagnosed, making breast cancer the most frequently reported cancer among the 11 priority cancers in the country. 2
From 2015 to 2023, morbidity and mortality trends reported by the CAC revealed a sustained increase in the prevalence of breast cancer. In 2023, specifically, there was an 8.03% increase in prevalence. At the same time, the proportion of newly diagnosed cases reached its highest level since records began. 3 This is paradoxical, considering that breast cancer, when detected in its early stages, is potentially curable and associated with substantial improvements in survival rates among patients. 4 Achieving this benefit requires not only timely detection, but also access to confirmatory diagnosis and treatment in a timely manner. In this context, mammography has established itself as the gold standard for breast cancer screening.5–8Mammographic screening is associated with reduced breast cancer mortality, with larger reductions among women who participate regularly in organized screening programs. 9
However, despite advances in diagnostic and therapeutic technologies, mortality from this disease remains high, especially among women in marginalized and vulnerable communities. These populations often lack timely and adequate access to healthcare services, as many women are unable to undergo screening mammograms due to limited healthcare infrastructure or financial and geographical barriers.10–12
To address these limitations, bioimpedance has emerged as a promising alternative and complementary tool, allowing healthcare professionals to quickly detect benign or malignant abnormalities in breast tissue. This technique provides quantifiable parameters related to the electrical conductivity and capacitance of tissues.13–16 In normal breast tissue, moderate variations in impedance values are observed, reflecting the heterogeneity of cellular components. In contrast, pathological tissues—especially malignant ones—show a significant increase in capacitance and conductivity, resulting in a decrease in impedance.14,17–20
In this context, JULIETA is an innovative portable device currently under development that applies bioimpedance technology to detect electrical signatures associated with changes in breast tissue and cellular composition. The palm-sized unit is connected via cables to a standardized electrode interface designed to ensure consistent positioning during acquisition. Specifically, electrodes are applied using a flower-shaped applicator centered on the nipple, with synthetic “petals” housing commercially available adhesive electrodes that contact the skin and capture impedance measurements across the breast.
As part of its technological development, the prototype device's software and algorithm must be capable of interpreting both the reactance and resistance components of the bioimpedance signals and correlating them with the findings of mammographic readings. This correlation is essential for distinguishing at-risk patients based on the bioimpedance analysis of breast tissue.
The viability of the JULIETA as a non-invasive, easy-to-use, and reliable device with reproducible results for detecting breast tissue alterations using bioimpedance depends on its software accurately interpreting conductance signals. Because the performance of the algorithm can be affected by signal acquisition conditions, it is necessary to retrain the model by incorporating scenarios with gel-based electrodes. In this context, the present study evaluates whether, in adult women (≥18 years) undergoing screening or follow-up mammography, evaluation with JULIETA and its retrained hierarchical algorithm adequately distinguishes between findings with no potential for malignancy (BI-RADS 1-2) and those with potential for malignancy (BI-RADS ≥3), using mammography as the reference standard, and quantifies its performance through sensitivity, specificity, positive predictive value, and negative predictive value.
Methods
Study Design
A cross-sectional study with prospective data collection was conducted in accordance with STARD reporting standards in a real clinical setting to evaluate the performance of a portable bioimpedance-based device for breast tissue assessment. The study involved collecting bioimpedance measurements alongside the corresponding mammographic classification to evaluate and retrain the hierarchical algorithm integrated into the device in terms of its ability to differentiate between benign and potentially malignant findings. The algorithm's performance was quantified using sensitivity, specificity, positive predictive value, and negative predictive value. Data were collected between May and July 2024.
As a reference standard, mammograms were interpreted by experienced radiologists using the same edition of the BI-RADS classification system. Image interpretation was performed blindly to the JULIETA results. Systematic double reading of all mammograms was not conducted; instead, each examination was read by one radiologist according to center workflow and availability. When diagnostic uncertainty arose, the BI-RADS category was established by consensus between both radiologists. Prior to modeling, bioimpedance signals were calibrated for magnitude and phase using a frequency-dependent gain factor and intrinsic phase correction (AD5933). 21 Analyses were restricted to the 5–100 kHz range to mitigate low-frequency noise, and signal integrity was ensured through automated hardware, connection, and rule-based quality checks. Features were normalized using Min–Max scaling. Classification thresholds followed clinical BI-RADS criteria, grouping BI-RADS 1–2 as class 0 and BI-RADS ≥3 as class 1; outputs assigned to class 1 were considered positive under a hierarchical ensemble decision rule.
The study protocol was reviewed and approved by an institutional ethics committee. This approval covered all participating study sites under a centralized ethics review process. Written informed consent was obtained from all participants prior to enrolment. The study was conducted in accordance with the Declaration of Helsinki (1975), revised in 2024.
The presentation of this cross-sectional study complies with the STROBE guidelines. 22
Study Population
The study population consisted of women with fully developed breast tissue who underwent screening or follow-up mammograms at four urban centers in Colombia affiliated with a national diagnostic services provider in Colombia.
The inclusion criteria were women aged ≥18 years who had a clinical indication for screening or follow-up mammography. Women with a prior Tru-Cut breast biopsy and benign histopathology were also eligible, provided they had no deep skin lesions. Exclusion criteria included: lack of written informed consent; current pregnancy or breastfeeding; history of breast cancer or lumpectomy; the presence of breast prostheses, implants, or metal components in the thorax; cardiac or cerebral pacemakers; breast radiotherapy in the last year; scars, dermatitis, or other skin conditions in the area where the electrodes were placed that could exacerbate irritation or discomfort ; or metal piercings on the breasts o chest.
Sample Size
Participants were recruited consecutively at participating centers from among women with confirmed mammography appointments, using non-probabilistic convenience sampling. Those who attended screening or follow-up mammography during the study period (May –July 2024) were included, according to predefined inclusion and exclusion criteria. The target sample size was set at 1050 participants to ensure adequate representation across diagnostic categories, given the expected higher proportion of normal BI-RADS scores. A total of 1350 women were recruited during the study period.
A predefined data cleansing process was applied to ensure completeness and traceability between the mammography result (gold standard) and impedance measurements. At the patient level, records that were incomplete or contained duplicate entries were excluded from the study. Subsequently, the dataset was structured at the breast level, given that the analytical objective focused on clinically relevant findings in women with BI-RADS ≥3, which are usually unilateral. At this stage, measurements lacking confirmed laterality, BI-RADS 0 findings (due to their inconclusive nature), and records with missing values in essential variables (NaN in the risk score) were excluded.
Recruitment Process
Patients were selected consecutively based on confirmed mammography appointments. The research team made an initial telephone invitation using a standardized script, followed by active recruitment at participating centers until the target sample size was reached. A 15-day pilot phase was conducted at the Medellín site to estimate the recruitment rate. Technical and administrative training sessions were provided for operational staff, including specific instructions on the proper use of the JULIETA device and communication strategies led by the investigator team.
Recruitment was carried out in four cities in Colombia, using various support tools, including educational infographics on breast self-examination and cancer prevention, staff identification badges with the prototype logo, and thank-you posters given to participants after their participation.
Measurement Instruments
Each participant was evaluated with the JULIETA device before undergoing mammography, as medically indicated. Measurements were performed by technicians trained independently of mammography personnel. The results of both procedures were collected and analyzed independently and blindly. The JULIETA device is attached to the skin over the nipple, with six electrodes evenly distributed around each breast (Figure 1). Thirty measurements per breast were taken at multiple frequencies (5000-100,000 Hz), using gel-based electrodes approved for medical use.

Representative image of the flower-shaped element with petal-shaped extensions attached to the skin and housing the electrodes.
A connectivity test was performed before each session to ensure the functionality of the system. The estimated time per patient was 1 min (30 s per breast). The device operates on an internal low-voltage battery, is not connected to the mains, and poses no risk of electric shock. Additional technical specifications are available in the logistics annex.
Data Sources
Data were consecutively collected and managed through an anonymous database designed by the research team, stored in a secure, restricted-access electronic repository. All participant records were de-identified prior to analysis; no direct identifiers were included in the study database, and no identifiable patient information is reported. Two main data sources were used: (i) primary data from bioimpedance measurements obtained with the prototype device and patient-reported information, and (ii) secondary data corresponding to mammography results, extracted from the diagnostic provider's information system and subsequently recorded by radiologists in a standardized template provided by the research team. Mammograms were performed and interpreted locally by a responsible radiologist at each of the four participating centers, in accordance with international recommendations and guidelines (American College of Radiology, ACR BI-RADS; European Society of Breast Imaging, EUSOBI) and following a standardized protocol across sites. Both sources were integrated daily into the study database.
Quality Control of Acquisition and Handling of Missing Data
To ensure data reliability, a structured quality control and missing data management strategy was applied prior to model development. This included a hardware verification test comparing measured impedance values against internally stored reference standards validated under controlled conditions, as well as an electrode connectivity check designed to detect disconnections or poor contacts. The latter relied on multi-frequency measurements and outlier detection to flag signal patterns consistent with abnormally high impedances. After measurement, records with technical acquisition failures (eg, electrode disconnections or unstable contacts), incomplete values in key variables (risk score and breast density), or invalid signal patterns were excluded from the analysis. BI-RADS 0 cases were also excluded due to their inconclusive diagnostic nature. In cases of minor data loss related to wireless transmission, limited interpolation (up to five non-consecutive values per test) was allowed; this affected less than 5% of the dataset and introduced errors below 1% relative to expected signal values.
Considerations by Electrode Type and Bipolar Configuration
In the initial development phase, the algorithm was trained using gel-free electrodes. Since the device's performance had to be consistent with different acquisition inputs, exploratory evaluations were performed with different types of electrodes. Although preliminary tests with gel-free electrodes were satisfactory, a significant decrease in performance was observed when gel electrodes were incorporated. This behavior suggests that, in the context of a bipolar measurement configuration, variations in the electrode-skin interface can modify signal characteristics and affect classifier stability.
This finding prompted a comprehensive review of the reliability of the system and the quality of the signal, which included (i) a review of the theoretical basis, revisiting the Cole–Cole model as the expected representation of impedance measurement; (ii) validation of the signal in different body regions to verify the system's ability to discriminate between tissue types; (iii) reevaluation of the initially considered operating range (1-100 kHz), identifying high noise levels below 5 kHz and adjusting the measurement range accordingly; (iv) reviewing and improving phase and magnitude calculations through more appropriate calibration of the AD5933 21 ; (v) updating the design of the support (“star”) to standardize the fixation and distance between electrodes; and (vi) improving the design and manufacture of cables with the aim of reducing noise and variability.
In parallel, the gel electrodes underwent additional internal testing, and no significant variability was observed under the acquisition of verifications implemented. However, given that the measurement is performed in a bipolar configuration, the possible influence of electrode contact impedance on the recorded signal is acknowledged. To mitigate variability introduced by gel-based electrodes during retraining—particularly given the bipolar measurement configuration—a combined hardware, signal processing, and modeling strategy was implemented. Impedance magnitude and phase were calibrated using a frequency-specific gain factor and intrinsic phase correction based on reference resistive loads. Signal preprocessing excluded frequencies below 5 kHz due to increased noise susceptibility, restricting analysis to the 5–100 kHz range. At the modeling stage, feature-wise Min–Max normalization, class-balanced weighting, and genetic hyperparameter optimization were applied to improve robustness against contact-related impedance shifts. Records showing evidence of suboptimal acquisition were excluded prior to model training and validation.
Risk and Mitigation
The JULIETA device is safe and delivers very low intensity microcurrents with strictly controlled parameters for use on the skin. Patient feedback was monitored in real time to interrupt the procedure in case of any discomfort. In rare cases, the electrodes may cause mild skin irritation. Medical-grade electrodes with national and international regulatory approval were used. Suitable for sensitive skin, latex-free, with hypoallergenic adhesive. Basic post-procedure care and follow-up phone calls were provided, activating institutional support protocols if necessary. Each site had a backup device. In case of failure, the devices were sent to the Medellín site for technical adjustments and recalibration.
Statistical Analysis
The study population was characterized using descriptive statistics, and differences between cities were explored. Supervised machine learning was used to develop the classification model based on Impedance-derived features, using characteristics derived from electrical impedance curves measured at 96 frequencies (30 combinations), including statistical descriptors of the signal, Cole–Cole parameters (in different electrode combinations),breast density estimates, and individual risk scores.
The dataset was split into 80% for training and 20% for validation, preserving class proportions across BI-RADS categories. Feature selection and hyperparameter optimization were performed exclusively within the training set using 5-fold stratified cross-validation to prevent information leakage.
Feature selection was conducted using an optimized Random Forest model, from which the 20 most relevant features were retained. The main selection criterion was the maximization of the macro F1-score using genetic algorithms hyperparameter tuning. Model selection was guided by the maximization of the macro F1-score, chosen to balance sensitivity and specificity under class imbalance.
The final algorithm followed a hierarchical ensemble strategy aimed at generating a binary prioritization outcome (Prioritize —BI-RADS ≥3— vs Do not prioritize —BI-RADS 1-2—). The master node implemented a StackingClassifier combining Random Forest, SVM with radial basis function kernel, CatBoost, K-nearest neighbors, and Gaussian Process classifiers, with Logistic Regression as the meta-model. Two auxiliary submodels (anomaly and benign anomaly) employed SVM classifiers with radial basis function kernels and class-weight balancing. To prevent data leakage, synthetic data were generated exclusively within the training set to balance class distributions, for the benign anomaly submodel.
The overall model performance was assessed on the validation set using confusion-matrix–derived metrics (sensitivity, specificity, PPV, and NPV). All analyses were performed in Python 3.10 using standard machine learning libraries.
Results
Clinical and Sociodemographic Characteristics of the Study Population
During the study period, 1350 patients were recruited. After the patient-level screening process, 74 records were excluded due to incomplete information and 111 due to duplicates or missing files, resulting in 1165 patients with mammography (reference standard) and bioimpedance measurements available. Since the outcome of interest was defined at the breast level— given that clinically relevant findings in women with BI-RADS 3 + usually occur unilaterally—the set was structured by breast, for a total of 2330 measurements. At this stage, 470 breasts without confirmed laterality, 76 cases with low-quality signals, 36 records classified as BI-RADS 0 due to their inconclusive nature, and 1075 records with missing values in essential variables (NaN in the risk score) were excluded. Consequently, the final analytical set for model training and validation included 673 breasts, corresponding to 469 women (Figure 2).

Participant flow and data set refinement at the patient and breast level.
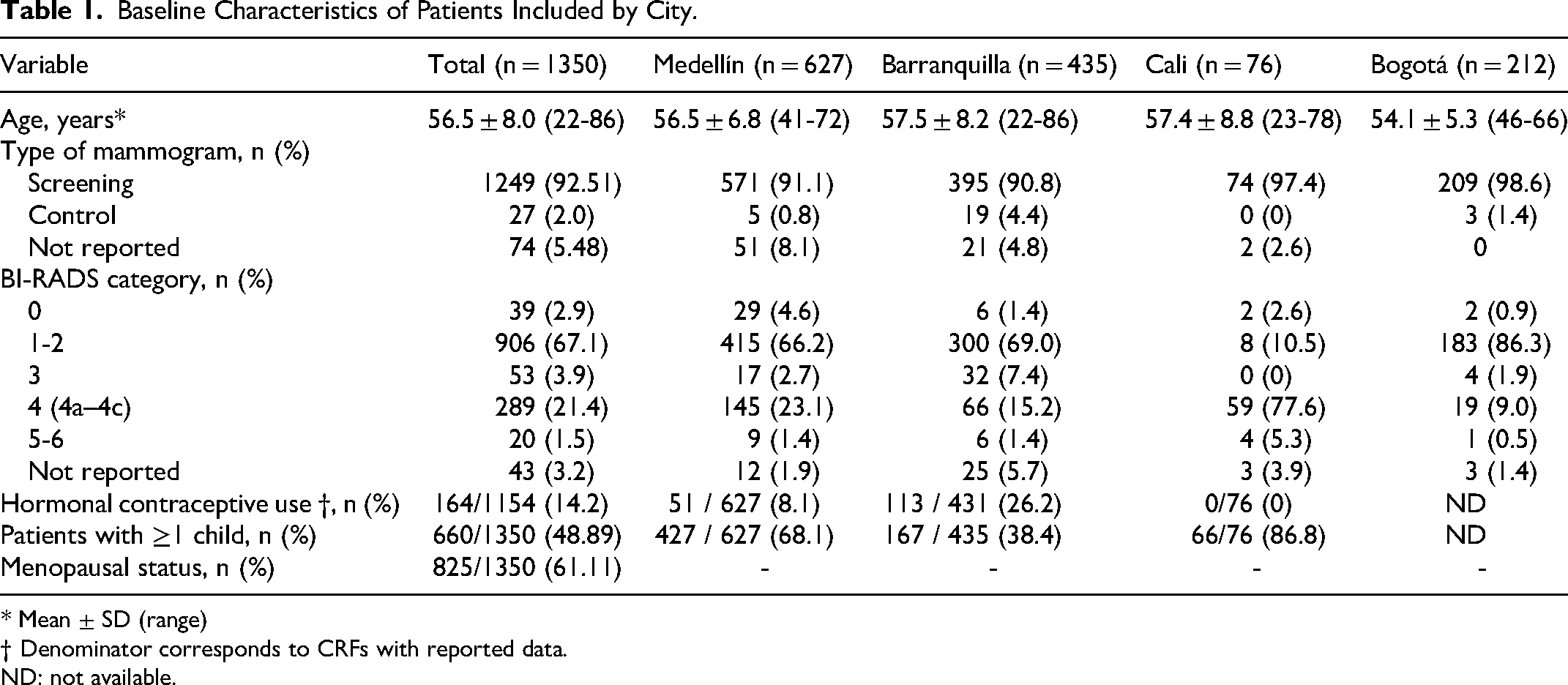
Baseline characteristics were described for all women initially recruited (n = 1350). The mean age was 56.5 ± 8.0 years (range: 22-86 years). The main indication for mammography was screening, which accounted for 92.5% of cases (n = 1249); 2.0% (n = 27) were performed for follow-up, and 5.5% (n = 74) had no reported indication.
Regarding BI-RADS classification, most patients were categorized as BI-RADS 1–2 (906/1350; 67%). Suspicious lesions (BI-RADS 4) accounted for 21% of the sample, while categories indicating high suspicion or histological confirmation (BI-RADS 5-6) were rare (10/1350; 1.5%). The specific distribution by city showed notable differences: in Cali, 77.6% (n = 59) of cases were classified as BI-RADS 4, while in Bogotá, 86.3% (n = 183) were classified as BI-RADS 1–2.
Hormonal contraceptive use was reported by 14.2% of participants, with the highest frequency observed in Barranquilla (26.2%, n = 113). In terms of parity, 48.9% of patients (n = 660) had one or more children (n = 167). Menopausal status was reported in 61.1% of participants (n = 825) (Table 1).
Baseline Characteristics of Patients Included by City.
* Mean ± SD (range)
† Denominator corresponds to CRFs with reported data.
ND: not available.
Development of the Patient-Based Classification Model
To develop the patient-based model, the data were initially divided into two sets: a training set and a validation set. Subsequently, an optimization process was carried out to improve predictive capacity, which included selecting the most informative variables, constructing base classifiers, and designing a hierarchical model.
Data set
The development of the models was based on a dataset that complies with the characteristics of electrical impedance and allowed the extraction of the Cole-Cole parameters obtained and selected from different combinations of electrodes, breast density, and each woman's risk of developing breast cancer. To obtain this value, the most relevant risk factors for breast cancer were identified from the literature, along with their respective relative risks. Each factor was then assigned a corresponding weight by multiplying it by its relative risk; the results were added together and finally averaged to obtain a single value.
Classification Strategy
Each model aimed to classify the signals obtained into different BI-RADS categories according to their electrical characteristics, although each responded to a specific separability criterion (Table 2).
Classification of Models Based on BI-RADS Categories.

Note: + indicates grouped categories (eg, BI-RADS 3 and above). N/A = not applicable.
Model Construction
Three basic models and one hierarchical model were designed. The master model was trained without synthetic data, starting with a Random Forest optimized by genetic algorithms and stratified cross-validation across five partitions. From this process, the 20 most relevant features were selected, which served as input for a set classifier.
In addition, the anomaly model used an architecture similar to the master model, but employing only RBF kernel SVM as the final classifier, optimized using genetic algorithms with stratified cross-validation.
Meanwhile, the benign anomaly model incorporated synthetic data into the construction to balance the classes, while maintaining the feature selection logic with Random Forest and subsequent classification with radial SVM.
Finally, the healthy hierarchical model was conceived as a hierarchical ensemble integrating the three previous classifiers, with the aim of progressively discriminating between patients with low risk, high risk benign chances and high risk of potentially malignant changes.
A hierarchical structure of the combination of models is shown below (Figure 3). In this approach, the processed data passes through a first model whose output determines which model is applied to which patient, thus generating a decision system based on their experiences.

Decision-making framework of the combined healthy model.
To obtain performance indicators, confusion matrices were constructed. In this case, a transition was made from the confusion matrix of classes 0, 1, and 2 to that of two classes for the healthy hierarchical version, joining the elements of the matrix and assembling the second one.
Validation and Metrics
Validation was performed using cross-validation on five partitions stratified by BI-RADS category, using macro F1 maximization as the objective function. In this process, confusion matrices and classification reports were calculated, allowing the sensitivity and specificity for each classification model to be estimated.
Modeling
The healthy hierarchical model demonstrated robust performance during training, as evidenced by the confusion matrix and diagnostic metrics (Figure 4, Table 3). The validation results are summarized through the corresponding confusion matrix (Figure 5) and key performance indicators such as sensitivity, specificity, PPV, and NPV (Table 4).
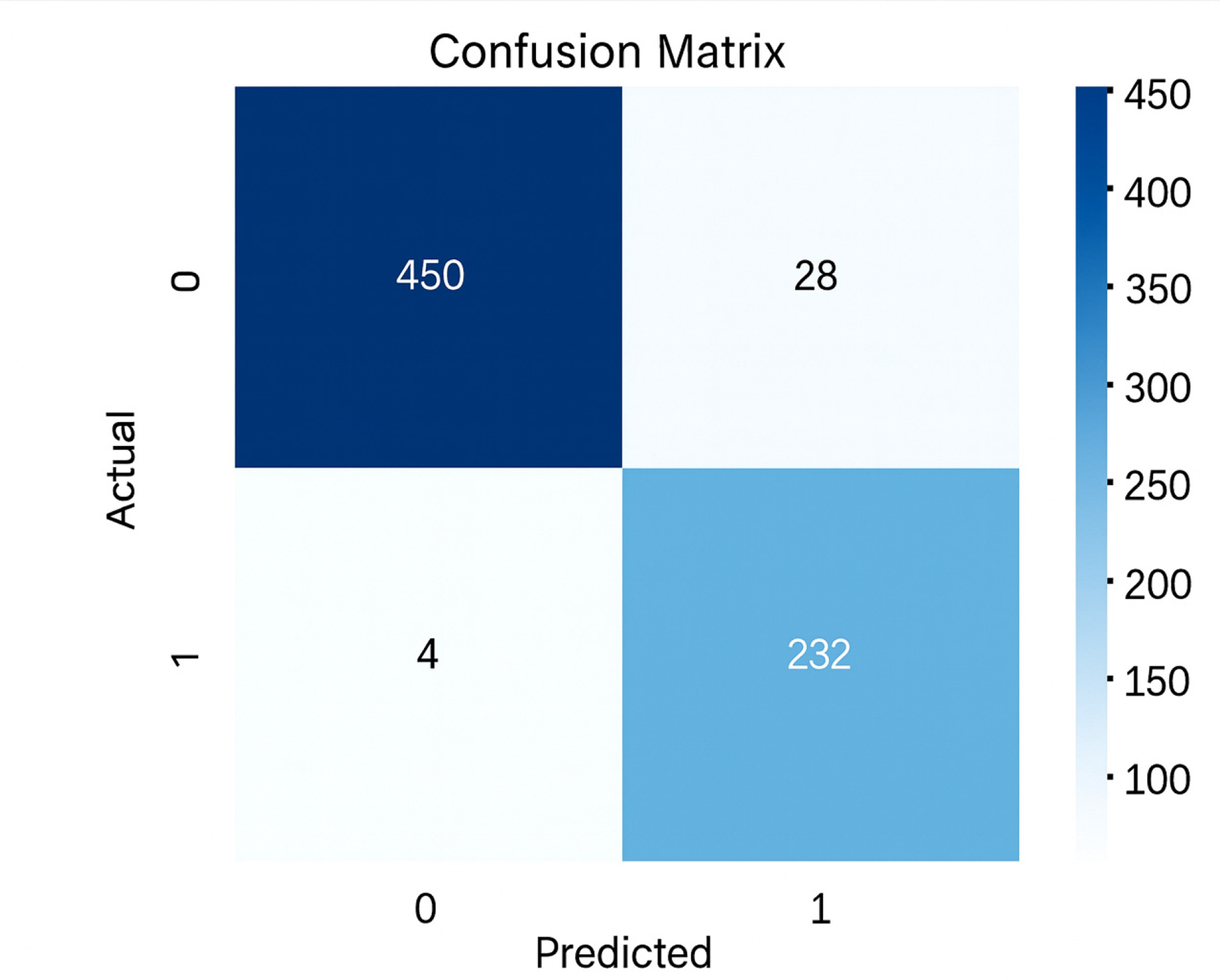
Confusion matrix of the healthy hierarchical model during training.
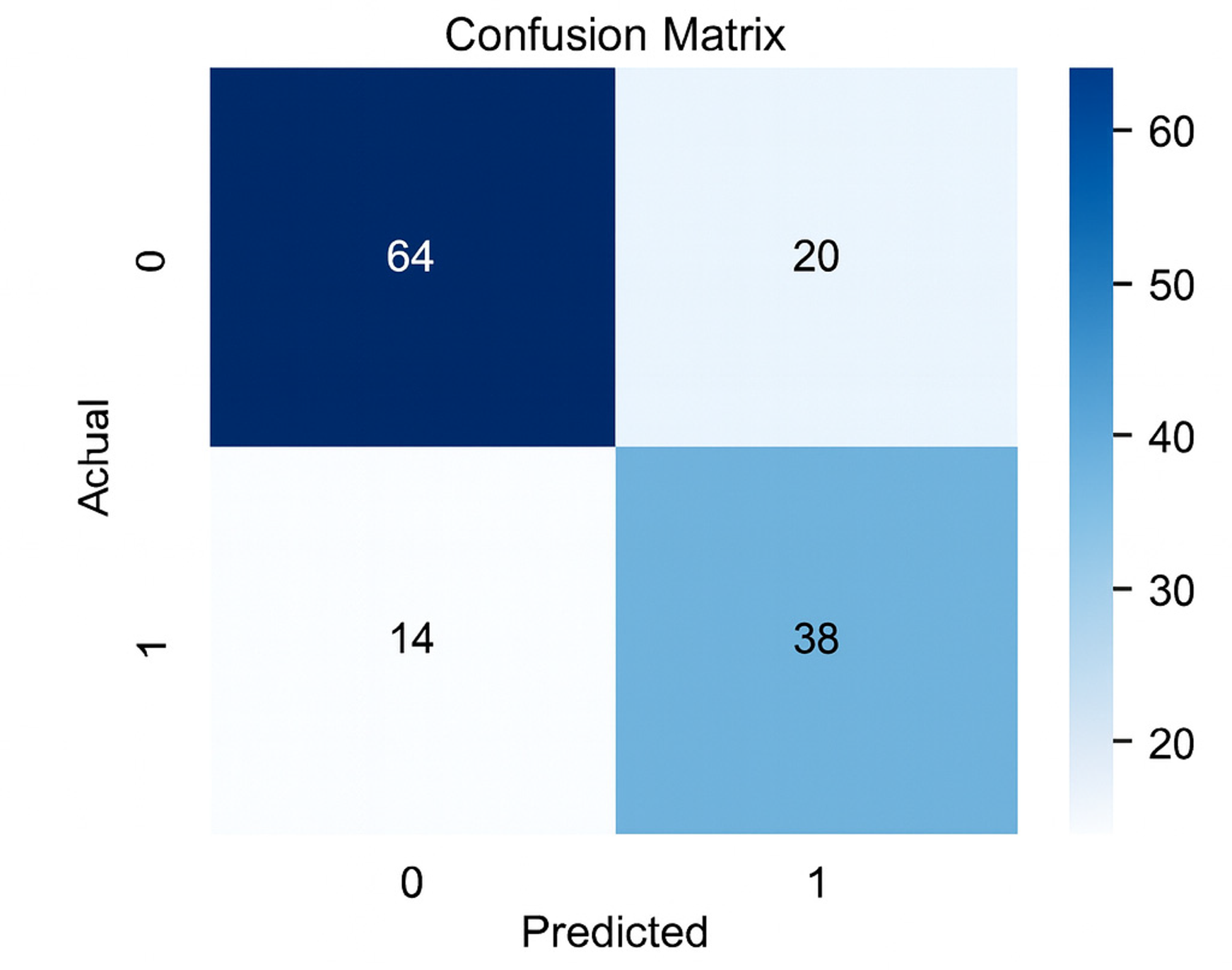
Confusion matrix of the healthy hierarchical model during validation.
Performance Metrics of the Healthy Hierarchical Model During the Training Walk.

Performance Metrics of the Healthy Hierarchical Model in the Validation Step.

Notably, the model achieved a balanced sensitivity of 73% (95% CI: 61-85%) and a specificity of 76% (95% CI: 67-85%) in validation.
In addition to estimating the performance indicators of the hierarchical healthy model and submodels, the performance of the hierarchical healthy model was evaluated by BI-RADS category based on the proportion of correct and incorrect classifications (Table 5). In this analysis, the performance observed for BI-RADS 3 was 25% (1/4 cases). Given the small sample size in this category (n = 4), this estimate is imprecise and highly variable and should therefore be interpreted with caution.
Performance by BI-RADS Category Based on Correct and Incorrect Classifications.

Although the primary endpoint corresponds to the hierarchical healthy model, the performance of the individual submodels was also evaluated to contextualize their respective contributions prior to integration. The master model achieved an overall accuracy of 60% (recall class 0: 54%, recall class 1: 58%, recall class 2: 63%), serving as the initial screening stage. The anomaly detection submodel yielded a sensitivity of 73% and a specificity of 71%f. Finally, the benign anomaly submodel, showed a sensitivity of 70% and a specificity of 67%. Confusion matices and metrics for all submodels are provided in the Supplemental Material (Supplemental Figure 1-3 and Supplemental Tables 1–3)
Discussion
This study demonstrates the consistent performance of the JULIETA device as a preselection tool in the Colombian context. In a cohort of 673 breasts, corresponding to 469 women a sensitivity of 73% and a specificity of 76% were achieved in distinguishing low-risk findings (BI-RADS 1-2) from those with higher suspicion (BI-RADS 3-5). This performance was achieved using a hierarchical ‘healthy’ model that integrates the three preceding classifiers to progressively discriminate among patients at low risk, at high risk of malignant changes, or at high risk of benign changes. In this approach, the processed data goes through a first model whose output determines which model is applied to which patient, thus generating a decision system based on their experiences. The physiological basis of the technique—EIS—supports these results, as it is a non- nvasive, radiation-free method capable of differentiating the electrical properties of breast tissue.
Our findings are consistent with published evidence. A 2020 meta-analysis confirmed statistically significant electrical differences between benign and malignant tissue, supporting SIE as a promising screening strategy. 23 Similarly, Stojadinovic et al reported the detection of 29 cancers using EIS, 66% of which were intangible, with more than half in women under 50 years of age; in the subgroup under 40 years of age, sensitivity was 50% and specificity was 90%, and a positive test was associated with a 4.5-fold increased likelihood of carcinoma. 24 Taken together, these data reinforce the potential usefulness of the JULIETA device in younger women, a group for whom routine mammography is not recommended and in whom clinical diagnosis often occurs at more advanced stages.
The relevance of this approach is particularly evident in Colombia, where mammography coverage among eligible women is estimated to be less than 50%, contributing to delayed diagnoses and a substantial decrease in five-year survival—from nearly 100% to ∼30%—when diagnosis is delayed.25,26 In this context, the prototype device is positioned as a strategic complement to reinforce early detection and reduce access gaps in rural or marginalized populations.11,12,27 Its safety, ease of use, portability, and automatic identification of abnormalities allow for initial evaluation in communities historically excluded from conventional screening, optimizing timely referral for confirmatory testing.
However, the device does not replace mammography or histopathological confirmation. A positive result must be followed by imaging or biopsy, and its sensitivity implies a margin of false negatives; therefore, it should not be used as the sole criterion in high-risk patients. Initial development revealed sensitivity to electrode type: an algorithm trained with dry electrodes showed reduced performance when gel electrodes were used, presumably due to signal variation. This limitation was addressed by retraining the network with mixed data; after adjustment, sensitivity and specificity stabilized at approximately 80%–85% regardless of consumable. In addition, the classification strategy evolved toward a binary scheme (prioritize vs not prioritize) implemented using a hierarchical model that provided greater stability. Challenges remain in detecting very small lesions and finely differentiating benign from malignant abnormalities, areas that will likely improve with larger and more diverse datasets. In terms of safety, no serious adverse events were reported, and skin irritations were mild; use is contraindicated in patients with pacemakers, metal implants, or other conditions that may interfere with measurement.
From an operational and ethical perspective, integrating the device into primary care and outreach campaigns seems feasible, if data confidentiality is protected, staff receive adequate training, and the scope of the test is clearly communicated—any abnormal results require confirmation—to avoid false alarms or false reassurances. This implementation, consistent with the principles of minimal risk and respect for autonomy, can contribute to health equity by bringing early detection to those with less access without adding significant risks or logistical barriers.
Looking ahead, improvements are anticipated from increasing the sample size and incorporating additional bioimpedance parameters (eg, phase analysis or tissue anisotropy) to refine the discrimination between benign and malignant lesions. Priorities include multicenter and longitudinal studies to validate performance in diverse populations, as well as clinical evaluations exploring its usefulness in periodic surveillance of survivors or women with high-risk benign lesions, complementing interval screening between mammograms.
Based on the limitations observed for BI-RADS 3, the main point of friction—discrimination between BI-RADS 2 and 3—was identified as a priority. Beyond sample size, this distinction represents a recognized clinical challenge, characterized by interobserver variability and overlapping imaging features, which may translate into similar electrical tissue patterns. Importantly, BI-RADS 3 is associated with a very low probability of malignancy (<1% in large follow-up series), 28 with most lesions ultimately downgraded to BI-RADS 2 after stability over time, as reported in screening ultrasound and mammography studies. 29 In this context, an alternative binary prioritization framework (BI-RADS 1-2–3 vs BI-RADS 4-5) should be studied in future work. This approach aligns with the intended clinical use of JULIETA as a prioritization tool and, as it is an accessible and radiation-free technology, it would also allow for more frequent follow-up of non-prioritized cases to identify any developments in a timely manner.
At the public health level, integration into national programs through mobile units and links to telemedicine could streamline diagnostic pathways. At the same time, regulatory progress will require evidence of cost-effectiveness and impact on health outcomes. In terms of external validity, these results should be interpreted in the context of an urban clinical setting with access to mammography. Although the multicenter design improves sample heterogeneity, generalization to rural or resource-limited settings—where infrastructure, risk profile, follow-up logistics, and acquisition quality may differ—requires specific evaluation. Future studies should validate the algorithm's performance and JULIETA's operational feasibility in rural and low-complexity settings.
In summary, the prototype device emerges as a viable complementary tool for breast cancer prescreening: its moderate accuracy, safety, and operational simplicity allow for the prioritization of patients requiring diagnostic confirmation, with the potential to improve early detection and help close gaps in settings with low mammography coverage.
Conclusions
The findings of this study support the viability of the prototype device as a safe, non-invasive, and portable bioimpedance-based device capable of identifying women at high risk for abnormal breast tissue. By retraining the algorithm with gel-based electrodes and integrating a hierarchical classification framework, the model achieved balanced sensitivity and specificity, reinforcing its potential as a complementary pre-screening tool rather than a replacement for mammography.
In the Colombian context—where access to breast imaging remains limited—the prototype represents an innovative and scalable approach to expanding early detection coverage and promoting health equity. Continued development through multicenter validation, the inclusion of larger and more heterogeneous datasets, and the optimization of artificial intelligence modules will be essential to consolidate diagnostic reliability and regulatory readiness.
Ultimately, the prototype exemplifies how biomedical engineering and artificial intelligence can converge to strengthen breast cancer screening strategies at the population level in low- and middle-income settings, contributing to earlier detection and better survival outcomes.
Supplemental Material
sj-pdf-1-tct-10.1177_15330338261422902 - Supplemental material for Portable Electrical Impedance Prescreening for Breast tissue suspicious for malignancy: Model Optimization and Clinical Performance of the Julieta Device in a Multicenter Cross-Sectional Study in Colombia
Supplemental material, sj-pdf-1-tct-10.1177_15330338261422902 for Portable Electrical Impedance Prescreening for Breast tissue suspicious for malignancy: Model Optimization and Clinical Performance of the Julieta Device in a Multicenter Cross-Sectional Study in Colombia by Maria Andrea Negret, Valentina González, David Grajales, Maria Alejandra Velez, Maria Alejandra Yepez, Valentina Agudelo, Sergio Lopez and Clara Piedrahita in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgements
The authors used an artificial intelligence tool (ChatGPT) to improve the language and readability of the manuscript. All changes were reviewed and approved by the authors, who take full responsibility for the content.
ORCID iDs
Ethics Statement
In compliance with Resolution 8430 of 1993 from the Colombian Ministry of Health, this study was classified as minimal risk, as it did not involve intentional intervention or modification of the biological or social variables of participants, using only a non-invasive device for breast bioimpedance measurement.
The study was reviewed and approved by the Ethics Committee and Good Clinical Practices in Health Research of SURA (CEI-Sura); this approval covered all participating study sites under a centralized ethics review process (approval Acta No. 127 [reference F-CEI024, Version 01], approved on 13 March 2024). Written informed consent was obtained from all participants prior to study procedures.
Informed Consent/Patient Consent
Participant inclusion was conducted following pre-screening via telephone and the acquisition of written informed consent, in accordance with the Declaration of Helsinki and applicable national regulations.
Author Contributions
Study conception and design, data collection, analysis and interpretation of results, and manuscript preparation: Maria Andrea Negret-Noreña, Valentina González-Sicard, David Grajales, Maria Alejandra Vélez-Clavijo, Maria Alejandra Yépez, Valentina Agudelo-Vargas, Sergio López-Isaza, and Clara Piedrahita-Montoya.
All authors reviewed the results and approved the final version of the manuscript.
Funding
This study was conducted as a collaborative effort between SalvaHealth and Seguros SURA Colombia.
SalvaHealth was responsible for the technological development of the Julieta device, and Seguros SURA supported the clinical implementation and patient recruitment.
No external or commercial funding was received for this work.
Declaration of Conflicting Interests
María Andrea Negret-Noreña, Valentina González-Sicard, David Grajales, María Alejandra Vélez-Clavijo, María Alejandra Yépez, Valentina Agudelo-Vargas, and Sergio López-Isaza are employees of SalvaHealth, Medellín, Colombia, the company that designed and developed the Julieta device.
The remaining author declares no competing interests.
Data Availability
The data supporting the findings of this study were collected prospectively from participants recruited at diagnostic centers in Medellín, Barranquilla, Cali, and Bogotá, Colombia. Data sources included bioimpedance measurements obtained using the Julieta device, mammographic assessments interpreted by certified radiologists and a structured questionnaire capturing sociodemographic information completed by each participant.
Due to the sensitive nature of the data, individual-level datasets cannot be publicly shared. De-identified data may be made available upon reasonable request to the corresponding author, subject to approval by Seguros SURA's Ethics and Good Practices in Health Research Committee.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
