Abstract
Purpose
This study assessed dosimetric effects of setup errors on boost target volume (PTVboost) coverage using simultaneous integrated boost (SIB) in early-stage left-sided breast cancer.
Methods
35 patients who received whole-breast radiotherapy (40.0 Gy/15 Fr) combined with a SIB to the tumor bed (48.0 Gy/15 Fr) were retrospectively analyzed. Translational-rotational coupled errors (1.0°rotation paired with 1.0 mm translation, 2.0° with 2.0 mm, 3.0° with 3.0 mm) were simulated about all axes. The D95 (dose to 95% of the PTVboost) and V95 (volume covered by 95% of the prescribed dose) were assessed through multivariate analysis to explore the relationship between PTVboost coverage and various anatomy factors, including the volume of the PTVboost (V_boost), the distance from the PTVboost centroid to the isocentre (D_iso), the mean depth from the anterior edge of PTVboost to the body surface (S_Depth), and setup errors.
Results
Under a combination of 1.0° rotation and 1.0 mm setup errors, the D95 values and V95 coverage of the PTVboost were ≥95% in all cases. However, when the error combination increased to 2.0°:2.0 mm, there was a significant decrease in coverage, with approximately 80% of the target areas exhibiting D95 and V95 values <95%. When the setup errors further increased to 3.0°:3.0 mm, D95 and V95 values were <95% in all cases. Multivariate analysis indicated that V_boost, D_iso, and S_Depth were significant predictors of target coverage.
Conclusion
PTVboost dose coverage risk were synergistically influenced by increasing D_iso, reduced V_boost, and shorter S_Depth. The multivariate model may stratify coverage risk categories using tumor anatomy and setup error magnitudes.
Introduction
Breast cancer is the most common malignant tumor among women globally, with its incidence rising in recent decades. 1 For early-stage invasive breast cancer, adjuvant radiotherapy after breast-conserving surgery is the standard treatment. Most ipsilateral breast tumor recurrences (IBTR) occur near the primary tumor site, particularly in the tumor bed.2–4 Many previous randomized trials have demonstrated that dose-escalated boost radiotherapy can reduce the risk of breast tumor recurrence by up to half.5,6 The two primary strategies for dose escalation are sequential boost (SEB) and simultaneous-integrated boost (SIB). While SEB reduces IBTR, it increases the risk of severe fibrosis.7–9 A recent phase III trial by Coles et al shows that SIB for early breast cancer is safe and reduces patient visits. 10 SIB offers three key advantages: 1) Radiobiological benefits from breast cancer's low α/β ratio, enhancing the therapeutic response to hypofractionated regimens. 11 2) Clinical benefits, including faster treatment schedules and cost-effectiveness. 12 3) Improved dosimetry, with superior dose conformation and reduced high-dose exposure to breast Planning Target Volume (PTV).13,14 These benefits make SIB a preferred option in early-stage breast irradiation.
Postoperative radiotherapy for early-stage breast cancer often uses hypofractionated whole breast irradiation (HF-WBI) due to its benefits, including shorter treatment duration, reduced radiation to normal tissues, lower costs, and better patient compliance.15,16 However, HF-WBI has drawbacks: reduced fractions lead to a smaller margin for dose deviations in critical organs (eg, heart Dmean) and target areas (PTV_boost), requiring stricter quality control and lower setup error thresholds. Additionally, higher single doses may amplify the impact of subclinical errors via the linear quadratic model. The SIB technique, with its steep dose gradient, improves conformance but increases sensitivity to setup errors. When combined with HF-WBI, the error sensitivity may worsen, risking excessive doses to organs at risk (OARs) or compromised target coverag, affecting tumor control probability and normal tissue complication probability.
Although studies by Roper et al and Zhang et al have shown that positioning errors threaten dose coverage in stereotactic radiosurgery and body radiotherapy, 17 particularly due to the synergistic of target volumes, multiple target separation and gradient positioning errors, these findings are not easily applicable to breast cancer SIB techniques. The unique features of breast cancer radiotherapy, such as respiratory motion, skin deformation, and the need to balance whole breast uniformity and high gradients at the tumor bed, make the error impact more complex. Studies on dosimetric robustness have shown that positioning errors affect PTV or CTV dose coverage, regardless of whether free-breathing (FB) or deep inspiration breath hold (DIBH) techniques are used. FB typically results in higher random errors, increasing the risk of compromised coverage. Zhong et al quantified the impact of the CIRTOG on PTVboost robustness, finding that a CIRTOG greater than 1 indicates suboptimal coverage, increasing sensitivity to positional errors. 18 However, their study has four key limitations: 1) the tumor bed volume was large, underestimating risks for “small target-high gradient” cases; 2) error simulation was conservative (maximum 2.0 mm/2.0°), lower than the commonly used clinical threshold of 3.0 mm/3.0°; 3) breast-specific anatomical parameters (eg, target depth and skin proximity) were not systematically modeled, limiting predictions for dose cold spots; and 4) the model lacked PTVboost volume (V_boost) and PTVboost centroid-to-isocenter distance (D_iso), reducing its generalizability. These limitations reduce the clinical reliability of predictive models for SIB patient stratification.
The aim of this study is to determine the dosimetric impact of setup errors on early breast cancer treated with HF-WBI using the SIB technique. By retrospectively generating new plan data under simulated error conditions, multivariate analysis methods were applied to assess the influence of several anatomy-specific parameters—V_boost, D_iso, and PTVboost anterior margin-to-surface mean depth (S_Depth)—on PTVboost dose coverage. These analyses may provide data-driven insights for clinical decision support tools and assist in the personalized selection of SIB techniques or the management of setup errors.
Methods and Materials
This retrospective study was guided by the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement to enhance methodological rigor and transparency. 19
Patients and Target Delineation
A retrospective-based study was conducted on 35 early-stage left-sided breast cancer patients who received HF-WBI with SIB between January 2022 and October 2024. The patients’ identities were anonymized, and ethical approval was granted by the Institutional Review Board. No other restrictions were applied except for the tumor bed boost volume being less than 130 cm3. Patients were positioned according to a standardized protocol using wingboard devices and customized cushions (Klarity Medical, China).All subjects were scanned on the Philips BigBore CT simulator (slice thickness of 2.5 mm, supine position) to obtain planning CT images. The acquired DICOM datasets were imported into the Eclipse™ treatment planning system (v15.5, Varian Medical Systems) for subsequent planning. The planning optimization process followed the guidelines of RTOG1005, 20 aiming for a uniform dose distribution in the whole breast while ensuring adequate coverage of the tumor bed, and minimizing radiation exposure to surrounding critical organs, such as the heart and lungs.
Automated delineation of OARs—encompassing the ipsilateral lung, contralateral lung, cardiac structures, and contralateral breast—was performed using AccuContour (Manteia Medical Technologies, China), with all contours subsequently reviewed by a senior radiation oncologist. Target volume delineation adhered to RTOG 1005 specifications, wherein an experienced radiation oncologist defined the whole-breast clinical target volume (WB-CTV) and tumor-bed boost volume (CTVboost). The latter required an integrative assessment of surgical scar characteristics, pre-/post-operative imaging findings, operative documentation, and surgical clip localization. Geometric expansion protocols applied 5-mm uniform margins to WB-CTV and CTVboost, generating respective planning target volumes, PTVb and PTVboost, with all PTVs maintained 5 mm beneath the body surface (only in rare cases, the expansion margins were adjusted locally based on clinical requirements). Spatial subtraction of PTVboost from PTVb yielded the non-boost breast planning target volume (PTVb-boost).
Treatment Plans and Evaluation Metric
For each case, dynamic IMRT plans were developed using a Varian VitalBeam system operating at 600 MU/min, employing both photon optimization and analytical anisotropic algorithms (Eclipse v15.1, Varian, USA) with a 2.5 mm isotropic calculation grid. The prescribed dose for the breast PTVb-boost was 40.0 Gy in 15 fractions, with a simultaneous integrated boost delivering 48.0 Gy/15 Fr. Treatment fields involved 6-7 tangential beams for whole breast irradiation and a dedicated boost field. Dose constraints for targets and OARs followed RTOG 1005 guidelines.
Plans evaluation were conducted using cumulative dose-volume histograms to coverage metrics, dose parameters, homogeneity index (HI), and conformity indices (CI) for both PTVboost and PTVb-boost. CI calculated using CIpaddick as (TVPIV*TVPIV)/(TV × PIV), 21 An optimal CI approaches unity for superior dose conformity. The HI was determined by (D2%-D98%)/D50%, with higher values indicating less uniform dose distribution. 21 Here, TV represents the target volume, PIV represents the prescription isodose volume, and TVPIV denotes their overlapping region. Critical organ assessments focused on ipsilateral lung and cardiac doses for the original plans, particularly analysing Dmean, V5, V20, and V30 parameters. Contralateral breast exposures were evaluated through Dmean, V5 and V20 measurements.
Setup Error and Dose Correlation Study
All rotational and translational errors were simulated and transformed in the plan isocenter using the Cartesian coordinate system and executed in Eclipse treatment planning system (TPS). Uniform translational and rotational errors were applied along the X, Y, and Z orthogonal axes, while maintaining a proportional alignment between directional translational and rotational variations (1.0 mm:1.0°, 2.0 mm:2.0°, 3.0 mm:3.0°). The methodology for Cartesian coordinate system transformations follows the established computational framework from previous studies. 17 Dosimetric analysis involved extracting the PTVboost parameters from error-incorporated plans, generating 105 error scenarios in addition to the baseline 35 plans. Dose distribution data following translational and rotational setup errors were sampled to obtain the dose metrics for PTVboost. The D95 (The percent isodose line relative to the prescription dose that covers 95% of the PTV) was recorded, with detailed documentation of each target volume and rotational angle. Additionally, V95, representing the percentage PTV volume receiving 95% of the prescribed dose, was also recorded.
The D95 data extracted from the Eclipse TPS were modeled using Generalized Estimating Equations (GEEs). This framework accounts for the correlations within different prescribed dose regions of individual patients by adjusting for the covariance matrix (eg, dose discrepancies between different fractions for the same patient). The PTVboost coverage analysis incorporated three anatomical parameters: V_boost, D_iso, and S_Depth. A multivariate regression model, incorporating interaction terms, was used to quantify the synergistic effects of anatomical parameters on D95 degradation, considering the independent combinations of translational and rotational errors. The analytical model was formalized as:
The variables in this model include the D_iso (cm), the V_boost (cm3), and the S_Depth (cm). Here, Y is the dependent variable, representing the value of D95. The parameter estimates for D_iso (parameter a), V_boost (parameter b), and S_Depth (parameter c) are provided, along with their corresponding 95% confidence intervals and
The planning data for all setup error combinations (1 mm:1.0°, 2 mm:2.0°, 3 mm:3.0°) were used to develop a multivariate logistic model via GEEs to investigate the mechanisms by which V_boost, D_iso, and S_Depth affect the dose-covered V95. The model includes planning target volume geometric characteristics (V_boost) and spatial location parameters (D_iso, and S_Depth) as covariates, and calculates the predicted probability of meeting the clinical dose criterion (V95 ≥ 95%).
In this model, D_iso, V_boost, and S_Depth are the variables in the multivariate logistic model, with corresponding regression coefficients denoted as A, B, and C, and β1 as the constant. These coefficients are reported using odds ratios (OR), probability inference boundaries (95% CI), and statistical significance indicators (P-value). Based on this logistic regression model, a systematic analysis was conducted to examine the dosimetric relationship between V_boost, D_iso, and S_Depth and V95 under different gradient errors.
Statistics
The radiotherapy plan target and OARs dosimetric parameters were described using descriptive statistics, including the mean ± standard deviation (x ± SD) and the median [range]. The dosimetric robustness analysis focused on evaluating the stability of dose coverage (D95 and V95) for the PTVboost under simulated positioning errors (translation/rotation). Statistical analysis was performed using data extracted from the treatment planning system (TPS, Eclipse v15.6), with calculations conducted using IBM SPSS Statistics 21 (IBM Corp., Armonk, USA). A significance threshold for hypothesis testing was set at 0.05. Dose distribution maps were rendered through the TPS, and statistical charts were generated using OriginPro 2024 (OriginLab, USA).
Results
In the 35 cases of this study, the median PTVb volume was 804.2 cm3 (range 462.8-1233.0 cm3), with a mean volume and standard deviation of 838.9 ± 214.9 cm3. The mean and standard deviation for the PTVboost were 73.5 ± 21.3 cm3 (71.2 cm3, range 33.5-128.66 cm3). The D_iso ranged from 2.09 cm to 6.48 cm, with a mean distance of 4.48 ± 1.29 cm, and the boost location in the central region and outer/inner quadrants. The mean distance and standard deviation for the S_Depth were 0.91 ± 0.48 cm (0.75 cm, range 0.35-2.06 cm). Detailed data are presented in Table 1. All original plans met the clinical requirements and were reviewed by senior radiation oncology specialists. The dose metrics for the target volumes (PTVb-boost, PTVboost) and critical OARs are provided in Supplementary Tables 1 and 2. Supplementary Table 3 displays the OARs dose metrics for all plans under setup error, presented as mean ± SD and median [range].
Patient Characteristics.

Abbreviations: V_boost, Boost planning target volume; PTVb, Whole breast PTV volume; PTVb-boost, Whole breast PTV volume subtracting the PTVboost; D_iso, isocenter-to-PTV_boost centroid distance; S_Depth, surface-to-target anterior margin depth; SD, Standard deviation.
Target coverage was characterized by translational-coupled rotational errors (1.0°:1.0 mm, 2.0°:2.0 mm, 3.0°:3.0 mm) applied along all orthogonal axes. The D95 and V95 values are plotted as a function of D_iso, V_boost, and S_Depth in Figures 1 and 2, respectively. The values and variations of D95 and V95 under different setup errors are indicated using a one-dimensional color coding. When the setup errors was set to 1.0° coupled with a 1.0 mm, the target coverage remained in a good state, with all PTVboosts having D95 and V95 > 95%. However, when the setup error was set to 2.0°:2.0 mm, target coverage significantly deteriorated: only 7 cases had a D95 and V95 for the boost greater than 95% (20% of cases), and these belonged to the same set of cases. Around 8.6% (3 cases) had a D95 coverage <90%, and approximately 17.1% (6 cases) had a V95 coverage <90%. When the setup error was set to 3.0°:3.0 mm (in free-breathing mode, which is within the common reference range for setup error thresholds), all cases showed a V95 and D95 for the PTVboost lower than 95%, with only 7 cases (20%) having a D95 and V95 > 95%. Among these, the minimum D95 was as low as 66.8%, and the minimum V95 was as low as 71.0%, with both belonging to different cases. Overall, target coverage deteriorated with increasing rotational error, increasing D_iso, smaller V_boost, and a shallower S_Depth. This pattern is prominently demonstrated in the cases within the red dashed ellipse region.
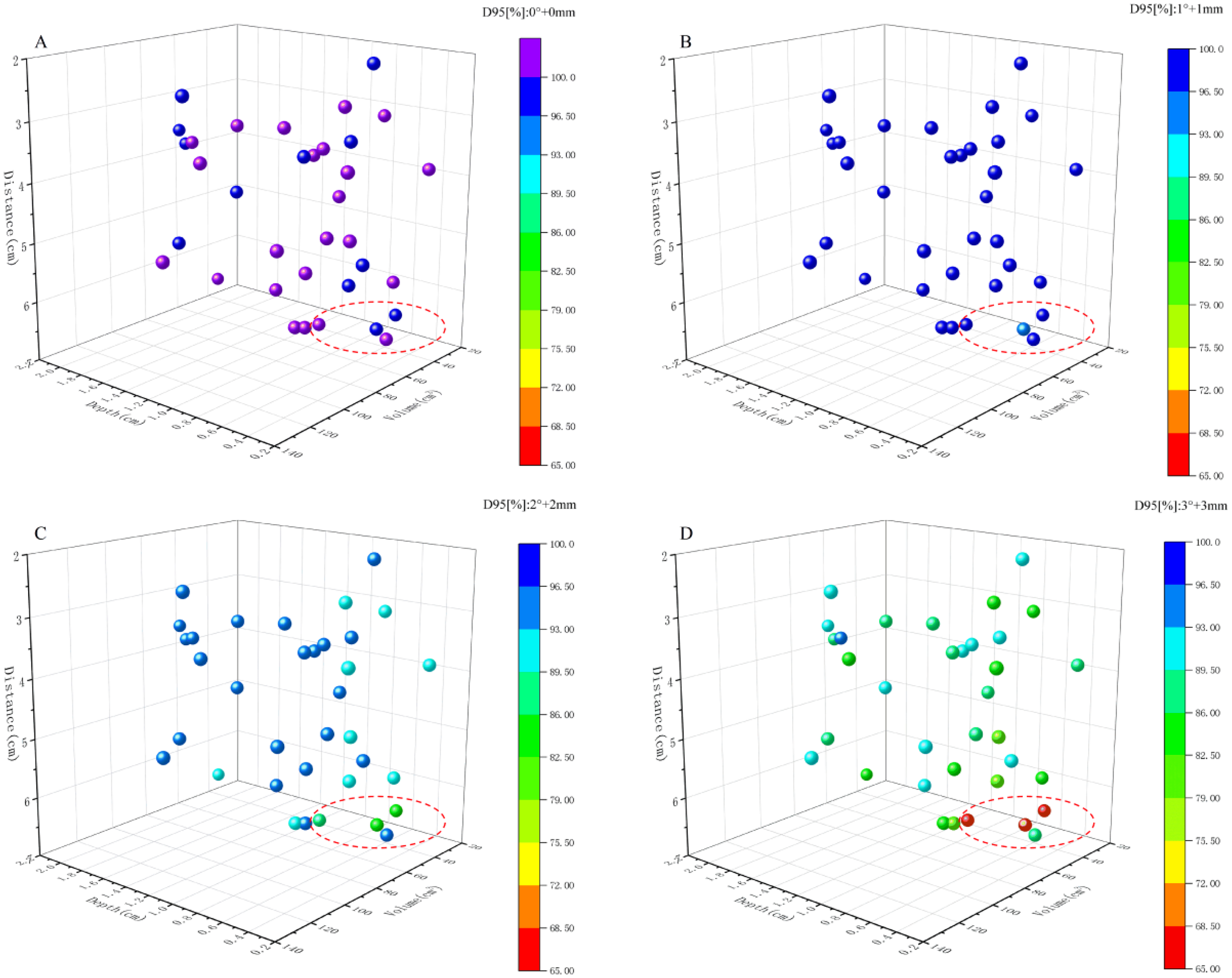
D95 Values Are Plotted as a Function of D_iso, V_boost, and S_Depth, with Variations in D95 Due to Setup Errors Represented Using a One-Dimensional Color Code. (A) Shows the D95 for All Original Plan PTVboost; (B), (C), and (D) Display the Changes in D95 Value for PTVboost Under Setup Errors of 1.0°:1.0 mm, 2.0°:2.0 mm, and 3.0°:3.0 mm, Respectively. Abbreviation: D95, Dose to 95% of the Planning Target Volume; V_boost, PTVboost Volume; and D_iso, PTVboost Centroid-to-Isocenter Distance; S_Depth, PTVboost Anterior Margin-to-Surface Mean Depth.

V95 Values Are Plotted Against D_iso, V_boost, and S_Depth, with Variations in V95 Induced by Setup Errors Visualized Through a One-Dimensional Color Coding Scheme. (A) Presents the V95 Values for All Original Plan PTVboost; (B), (C), and (D) Respectively Show the Changes in V95 Values of PTVboost under Setup Errors of 1.0°:1.0 mm, 2.0°:2.0 mm, and 3.0°:3.0 mm. Abbreviation: V95, Dose to 95% of the Planning Target Volume; V_boost, PTVboost Volume; and D_iso, PTVboost Centroid-to-Isocenter Distance; S_Depth, PTVboost Anterior Margin-to-Surface Mean Depth.
Multivariate GEE linear regression showed that D_iso, V_boost and S_Depth are strong predictors of D95 (statistically significant under all setup errors). Parameter estimates, confidence intervals, and
GEE Multivariate Linear Regression Model Is Used to Predict the D95 Value of PTVboost (Expressed as a Percentage of the Prescription Dose).

Abbreviations: CI, confidence interval; D95, dose to 95% of planning target volume; GEE, generalized estimating equations.
The model is parameterized at setup errors of 1 mm:1°, 2 mm:2° and 3 mm:3°. D95 GEE multivariate linear regression model as showed in equation (1).
The scatter plot matrix in Figure 3A reveals the linear relationships between PTV boost D95 and anatomical parameters (D_iso, V_boost, S_Depth) under different setup errors (indicating the slope, intercept, and

The Matrix Scatter Plots of D95 (Panel A) and V95 (Panel B) Demonstrate Linear Relationships between PTV Boost Dose Coverage and Anatomical Parameters (D_iso, V_boost, S_Depth) under Varying Setup Errors, with Slope, Intercept, and
In the multivariable logistic regression model, D_iso, V_boost, and S_Depth were significantly correlated with the V95 coverage <95% under a 2.0°:2.0 mm error (as presented in Table 3). Under a 1.0°:1.0 mm setup error, no model could be established, as the boost coverage exceeded 95% for all cases. Additionally, under a 3.0°:3.0 mm setup error, no model could be established due to the fact that the boost coverage was below 95% for all cases, leading to severe deterioration. The scatter plot matrix in Figure 3B reveals statistically significant linear relationships between V95 and anatomical parameters (D_iso, V_boost, and S_Depth) under different setup errors (1.0° + 1.0 mm, 2.0° + 2.0 mm, 3.0° + 3.0 mm), with regression parameters and
GEE Multivariate Logistic Regression Model for Predicting the Probability (p) That V95 of PTVboost Under 95%.
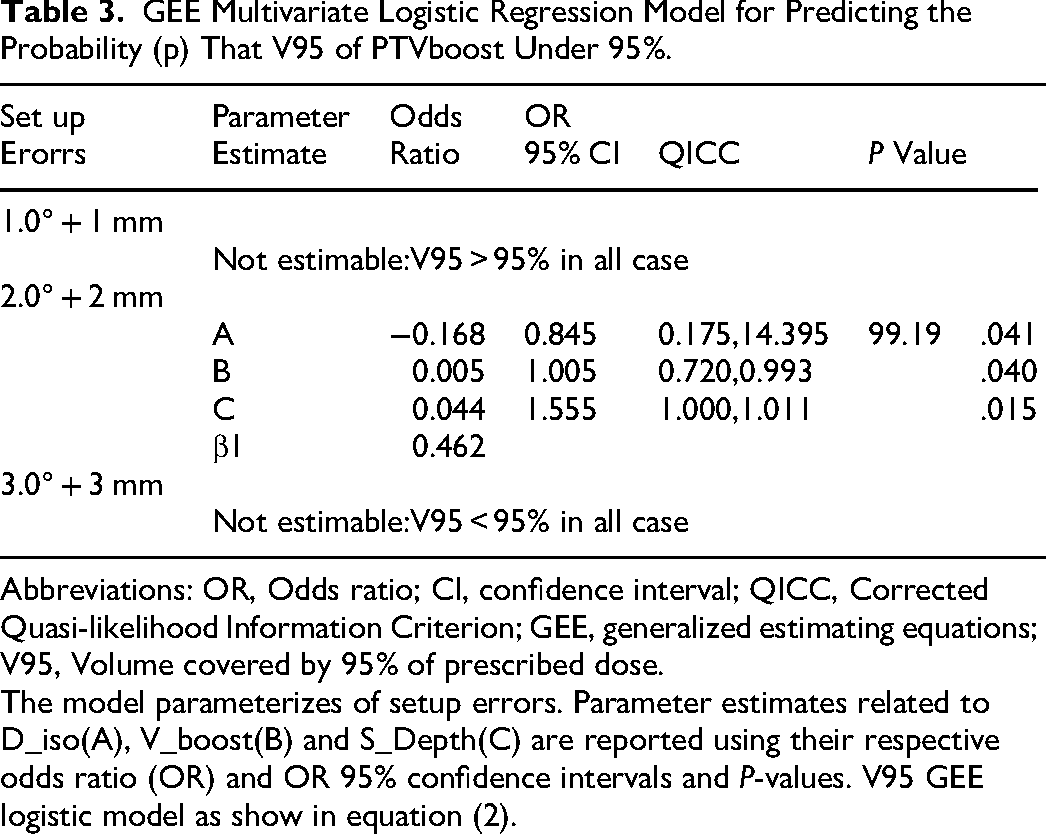
Abbreviations: OR, Odds ratio; CI, confidence interval; QICC, Corrected Quasi-likelihood Information Criterion; GEE, generalized estimating equations; V95, Volume covered by 95% of prescribed dose.
The model parameterizes of setup errors. Parameter estimates related to D_iso(A), V_boost(B) and S_Depth(C) are reported using their respective odds ratio (OR) and OR 95% confidence intervals and
As an illustrative case, Figure 4 illustrates the dosimetric impact under a 3.0°:3.0 mm setup error for a case with a PTVboost volume of 69.9 cm3, a mean depth of 0.55 cm from the anterior margin of the target to the surface, and a distance of 5.6 cm from the PTVboost centroid to the isocenter. It is located in the upper inner quadrant. Figure 4A shows the dose distribution of the original plan. Figure 4B presents the corresponding data with 3.0°:3.0 mm setup error. The dose-volume histogram in Figure 4C reports the details of the original plan and the 3.0°:3.0 mm setup error for the CTVboost and PTVboost: From the prescribed V100 perspective, the PTVboost decreases to 40.7%, and the CTVboost has approximately 16.0% of the volume experiencing insufficient dose.
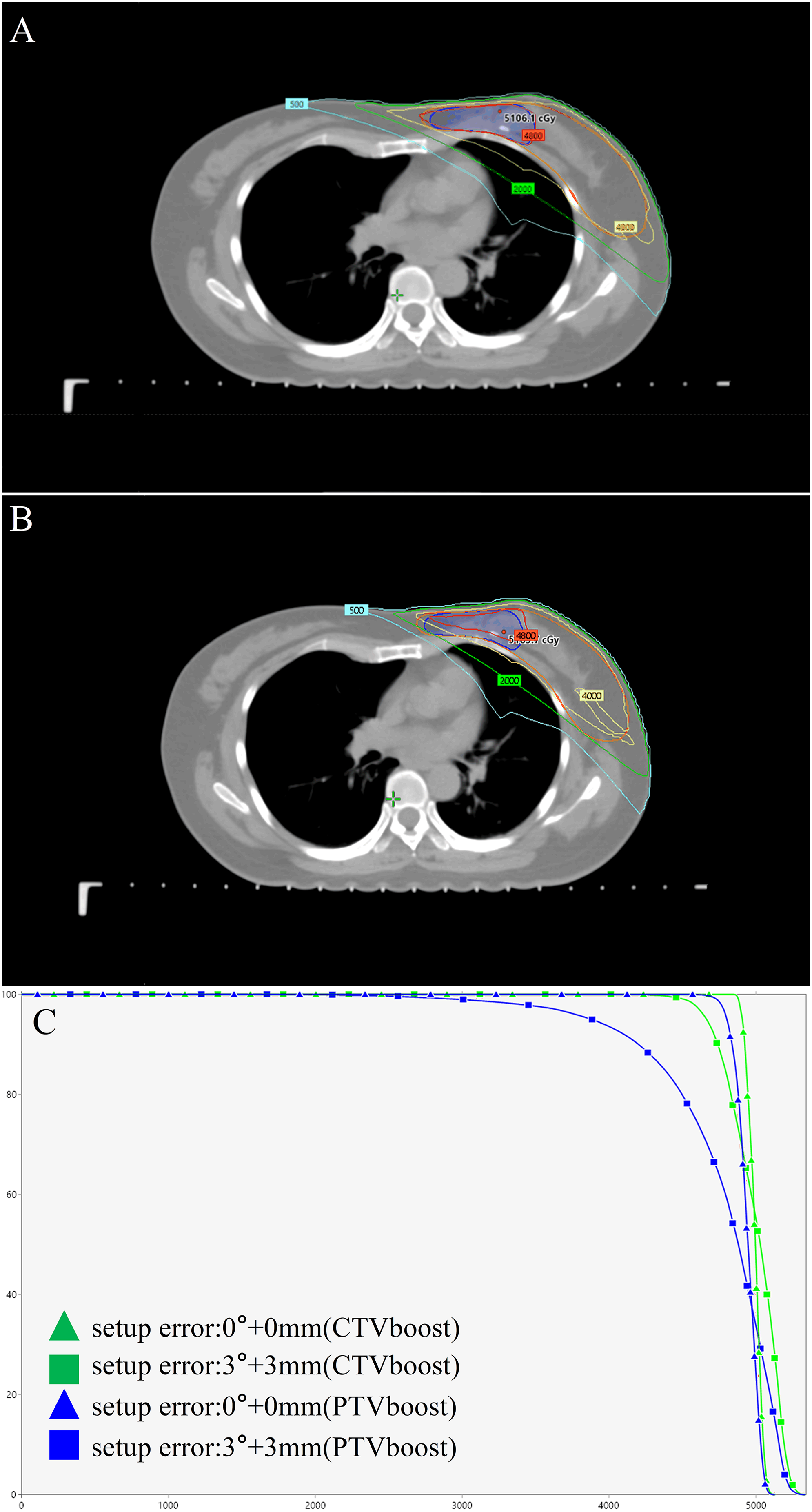
An Illustrative Case Showed the Initial Dose Distributions and the Dosimetric Effects of a 3.0° and 3.0 mm Setup Error Combination. (A) The Planned Dose Distribution Without Displacement; (B) The Dose Distribution of the Plan After Simulated Displacement; (C) The Corresponding Dose-Volume Histograms for the Boost.
Discussion
The local control of early breast cancer with HF-WBI using the SIB technique is comparable to sequential boost treatment, potentially offering similar or lower toxicity levels. 10 Our study analyzes how patient anatomical characteristics affect target coverage under setup errors. Results show that D_iso, V_boost, and S_Depth are significant factors predicting target coverage under varying positioning errors. A multivariate regression model confirms that these factors can accurately describe their effect on target coverage. These findings help determine if the SIB technique can provide a safe dose for individual patients. Except for the mean depth from the anterior margin to the target surface, which requires a specific in-house code, other anatomical parameters can be easily obtained from the planning system, allowing early evaluation of SIB suitability or necessary interventions. Additionally, the model is based on patient-specific anatomy, making it applicable to treatment equipment from any manufacturer. However, the study's small sample size (35 patients) may limit the generalizability and statistical significance of the results.
This study coupled rotational and translational errors, applying them simultaneously across all axes. While this scenario is unlikely in clinical practice, the error data quantifies the impact of combined errors (1.0°:1.0 mm, 2.0°:2.0 mm, and 3.0°:3.0 mm) on PTVboost target coverage under worst-case conditions. Setup errors were based on previous studies on setup uncertainties in breast cancer treatment. Research indicates translational and rotational errors exceeding 3.0 mm and 3.0°, respectively, may occur in free-breathing breast cancer treatments.22–24 In our clinical practice, most errors are within 3.0° and 3.0 mm, thanks to an optical surface monitoring system (OSMS) and a couch with six degrees of freedom. Additionally, a study established a 3.0° upper limit for rotational errors in breast cancer radiotherapy. 25 Thus, the simulated error range reflects extreme errors observed clinically to ensure treatment safety and accuracy. Another limitation is the discrete error settings, which may affect the accuracy of dose coverage predictions.
Breast cancer is located in the chest, where respiratory motion causes significant surface and anatomical changes that affect dose delivery accuracy.26,27 A study by Deng, Yongjin et al shows a strong correlation between body surface changes and dose variations. 28 Building on this, our research introduces the mean depth from the target area's anterior to the body surface to better assess PTVboost dose stability under setup errors. However, the mean S_Depth can be influenced by sampling precision, and its optimal use as a variable has not been fully explored. Additionally, the setup error simulations were based on the original plan's isocenter and recalculated using the original CT, without considering breathing-induced deformations, which is inconsistent with clinical practice.
This study examines the impact of setup errors with varying gradients on PTVboost dose coverage, considering the interaction between D_iso, V_boost, and S_Depth. However, we acknowledge that accurate dose delivery results from the combined influence of all factors, and other complex elements may still significantly affect dosimetry. The residual respiratory motion from original CT imaging at different phases is inconsistent. Although RTOG1005 recommends a 5 mm margin for CTVboost to PTVboost expansion, clinical practice requires adjustments in certain areas. Additionally, according to our predictive model, the SIB technique can maintain favorable PTVboost dose coverage when setup errors are controlled within 1.0°:1.0 mm. This can be achieved using OSMS (Optical Surface Monitoring System), though it may prolong dose delivery time and reduce patient compliance. When larger setup error thresholds are applied, the use of SIB may require methods such as online patient-specific quality assurance for fraction-level dose evaluation and dynamic positional adjustments to ensure adequate dose coverage. An ideal approach would involve using respiratory management techniques, such as DIBH, 29 to control the respiratory phase during beam delivery, reducing setup errors both within and between fractions. Future research will focus on incorporating patient anatomy, critical organ dose constraints, respiratory motion management, and planning implementation techniques.
Conclusion
In conclusion, SIB for early breast cancer can reduce treatment duration while maintaining efficacy. However, clinical use indicates that rotational and translational errors may compromise PTVboost dose coverage. To mitigate this, positioning accuracy should be validated through system checks before clinical use. Once spatiotemporal uncertainties are identify, statistical models incorporating patient-specific variables (eg, D_iso, V_boost, and S_Depth) may help estimate coverage probability, aiding adaptive planning or serving as quality assurance for positioning accuracy thresholds.
Supplemental Material
sj-docx-1-tct-10.1177_15330338251386512 - Supplemental material for Anatomy-Based Multivariate Model Predicts Boost Coverage Robustness of Dose-Escalated Simultaneous Integrated Boost Radiotherapy in Early Breast Cancer
Supplemental material, sj-docx-1-tct-10.1177_15330338251386512 for Anatomy-Based Multivariate Model Predicts Boost Coverage Robustness of Dose-Escalated Simultaneous Integrated Boost Radiotherapy in Early Breast Cance by Mengyuan Wang, MSc, Changyou Zhong, MSc, Xiao Luo, MSc and Jian Li, PhD in Technology in Cancer Research & Treatment
Footnotes
Ethical Approval Statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Central Hospital of Guangdong Provincial Nongken(25119). Informed consent or a substitute for it was obtained from all patients for being included in the study.
Author Contributions
Mengyuan Wang: Methodology, investigation, writing – original draft
Xiao Luo: Data curation, formal analysis.
Changyou Zhong: Investigation and data analysis.
Jian Li: Conceptualization, project administration, writing – review, and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Science and Technology Foundation of Guangdong Province, (grant number B2025924).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
