Abstract
Breast cancer is the most common malignancy and the second most common cause of cancer-related mortality in women. Triple-negative breast cancers do not express estrogen receptors, progesterone receptors, or human epidermal growth factor receptor 2 and have a higher recurrence rate, greater metastatic potential, and lower overall survival rate than those of other breast cancers. Treatment of triple-negative breast cancer is challenging; molecular-targeted therapies are largely ineffective and there is no standard treatment. In this review, we evaluate current attempts to classify triple-negative breast cancers based on their molecular features. We also describe promising treatment methods with different advantages and discuss genetic biomarkers and other prediction tools. Accurate molecular classification of triple-negative breast cancers is critical for patient risk categorization, treatment decisions, and surveillance. This review offers new ideas for more effective treatment of triple-negative breast cancer and identifies novel targets for drug development.
Introduction
Breast cancer is the most frequently diagnosed cancer in women.1–3 It has diverse features; based on the expression of estrogen receptors (ERs), progesterone receptors (PRs), and human epithelial growth factor growth factor receptor 2 (HER2), it is categorized as hormone receptor-positive (ER + , PR + , HER2−), HER2-enriched (ER−, PR−, HER2 + ), or triple-negative (ER−, PR−, HER2−).4,5
The number of women with breast cancer worldwide increased sharply from 1.05 million in 2000 to 2.09 million in 2018; meanwhile, the number of breast cancer-related deaths increased from 370,000 in 2000 to 520,000 in 2012 but decreased to 310,000 in 2018. 6 The incidence of and mortality rates for female breast cancer vary across countries. According to the Global Cancer Observatory, 7 its incidence is highest in Australia and New Zealand (Figure 1A), whereas mortality is highest in Melanesia followed by Western Africa (Figure 1B). From 2003 to 2015, incidence and mortality were much higher in the United States and Australia than in China. During this period, incidence tended to increase in all 3 countries, while mortality declined slightly in the United States and Australia and increased in China. Hence, breast cancer incidence and mortality are highly related to ethnicity.8–10

Approximately 15% to 20% of all breast cancers are triple-negative breast cancers (TNBCs). TNBC is most common in premenopausal women aged <40 years 11 and is highly aggressive, with an earlier age of onset, greater metastatic potential, and poorer clinical outcome than those of hormone receptor-positive and HER2-enriched breast cancers. 12 Generally, patients with TNBC have a shorter survival time, and 46% have extensive metastases. The lungs, liver, brain, bone, and supraclavicular lymph nodes are the most common locations of distant metastases, and patients with metastases have a survival time <2 years. 13
In general, molecular-targeted therapy has limited effect on TNBC because of TNBC's distinct phenotype. Consequently, chemotherapy is the most common treatment; however, standard postoperative adjuvant chemoradiotherapy is ineffective, 14 and tumor recurrence is likely. For patients with TNBC, the mean time to relapse is approximately 19 to 40 months, with a 75% death rate during the first 3 months after relapse. 15 Timely development of novel therapies and targets is therefore critical.
Owing to advances in genomics, epigenetics, and transcriptomics, a better understanding of TNBC is being achieved. We searched databases such as PubMed, ScienceDirect, and Springer for relevant studies published in the last 10 years using the following keywords in combination: TNBC, subtypes, classification, treatment, and genetic biomarker. Here, we review and summarize the classification, treatment, and biomarkers of TNBC, with the aim of identifying novel approaches and strategies for TNBC therapy. Figure 2 summarizes the key points discussed in this study.
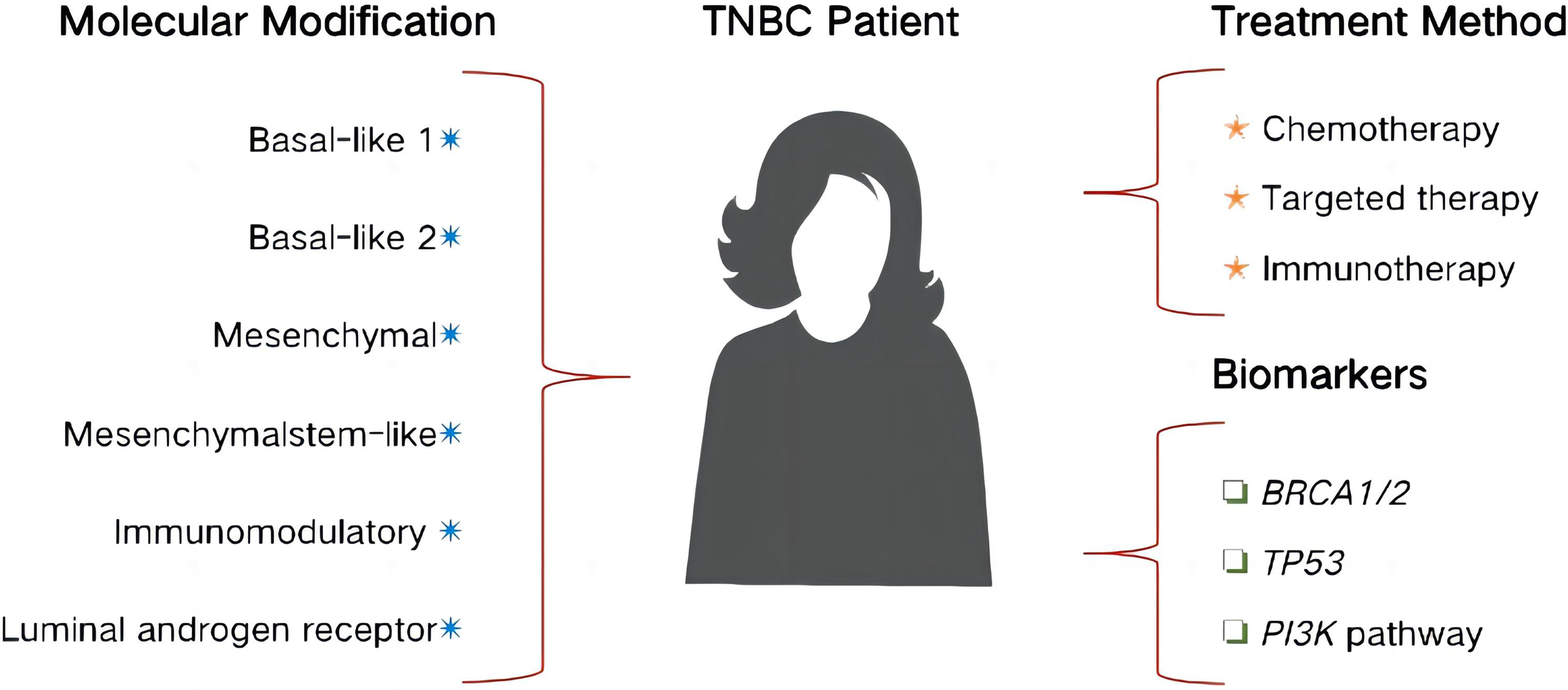
Brief summary of the review.
Molecular Classification
Identification of the Molecular Subtypes of TNBC
In 2011, expression profiling of samples from 587 TNBC patients classified 6 TNBC molecular subtypes: basal-like 1 (BL1), basal-like 2 (BL2), mesenchymal (M), mesenchymal stem-like (MSL), immunomodulatory (IM), and luminal androgen receptor (LAR).4,15–17 These subtypes were subsequently regarded as models for clinical treatment. In 2016, after discovering that the IM and MSL subtypes originated from infiltrating lymphocytes and tumor-associated stromal cells, the classification was updated to 4 subtypes: BL1, BL2, M, and LAR. 18 In addition, in 2015, Burstein categorized the following 4 subtypes via whole-genome sequencing of 198 TNBC samples: LAR, M, basal-like immunosuppressive, and basal-like type II immune-activated. 19 Currently, there is no unified diagnostic standard for molecular typing of TNBC internationally. Although there are different categorizations, we discuss the 6 subtypes identified in 2011.
Basic Characteristics of Each TNBC Subtype
The BL1 and BL2 subtypes are vulnerable to DNA-damaging drugs and express higher levels of DNA damage-response proteins than that of the other subtypes. Breast cancers with the BL1 subtype express high nuclear levels of Ki-67 and thus are highly proliferative. In comparison, those with the BL2 subtype have aberrantly activated growth factor signaling pathways and increased myoepithelial marker expression.20,21
Owing to highly active signaling pathways linked to cell migration, breast cancers with the M subtype are also known as metaplastic breast cancers. The M subtype is also associated with extracellular receptor interactions and overexpression of genes associated with cell differentiation. Characteristic features of the MSL subtype include low expression of cell proliferation-related genes and high expression of stemness genes and genes participating in angiogenesis and growth factor signaling pathways.
The IM subtype is distinguished by increased expression of immunological signaling genes, relating to antigen processing and presentation, and activation of immune-related signaling pathways. 22 Jézéquel divided the IM subtype into C2 and C3 groups according to the level of the immune response; in general, C3 patients were younger than C2 patients and had a better prognosis. 23 The LAR subtype lacks ERs but has active hormonal signaling pathways, and androgen receptors (ARs) and other downstream targets are overexpressed. The AR mRNA level is 9 times higher in the LAR subtype than in the other TNBC subtypes. 24 On imaging, the LAR subtype of TNBC often presents as high-density shadows and irregular masses, with accompanying calcification. 25
Current molecular classification of the TNBC subtypes largely reflects mRNA levels of targeted genes and is limited by mismatches between mRNA and protein levels. 26 Future investigations should focus on the molecular subtypes of TNBC in combination with the clinical classifications.
Therapeutic Targets Based on Molecular Typing
Molecular subtypes help reveal the occurrence and development of TNBC. Many experts and scholars have proposed a variety of different classification methods, the purpose of which is to further clarify the molecular characteristics of the different TNBC subtypes and thus better guide clinical treatment.
Poly-ADP ribose polymerase (PARP) inhibitors and genotoxic compounds are potential therapeutics for the BL1 subtype. 27 Several PARP inhibitors currently in clinical trials, including olaparib, veliparib, and rucaparib, show encouraging results. Cancer cells in BL1 individuals are sensitive to cisplatin therapy, and mechanistic target of rapamycin (mTOR) inhibitors and growth factor inhibitors are also promising therapeutic targets. 28
The M subtype is more resistant to chemotherapeutic medicines than that of other subtypes 29 and thus may be susceptible to mTOR inhibitors or drugs that target the epithelial–mesenchymal transition. Effective treatments for the MSL subtype include phosphoinositide 3-kinase (PI3K) inhibitors and antiangiogenic agents.30,31 Potential treatments for the IM subtype include programmed cell death protein 1 (PD-1), programmed death ligand 1 (PD-L1), and other immune checkpoint inhibitors.32,33 Expression of PD-L1 in tumor cells or its presence in the tumor microenvironment indicates high levels of tumor-infiltrating lymphocytes and positively correlates with the triple-negative status in breast cancer. 34 Anti-AR medication is a possible treatment option for the LAR subtype. 35 Indicative of potential sensitivity to such treatment, antiandrogen drugs (eg, bicalutamide and abiraterone) prolonged the tumor-free survival of patients with the LAR subtype in the study by Gerratana. 36
Identification of the molecular subtypes of TNBC and their clinical features not only guides the development of targeted therapies but also helps determine the histological characteristics and prognosis of patients. For example, the tendency of the LAR subtype to metastasize and spread and the relatively high immune activity of the IM subtype indicate that the IM subtype has a better prognosis than that of the LAR subtype.
Table 1 summarizes the features of the TNBC subtypes and potential treatment methods. 37
TNBC Subtypes and Potential Treatment Methods.

Abbreviations: TNBC, triple-negative breast cancer; BL1, basal-like 1; BL2, basal-like 2; M mesenchymal; MSL, mesenchymal stem-like; IM, immunomodulatory; LAR, luminal androgen receptor; PARP, poly ADP-ribose polymerase; mTOR, mechanistic target of rapamycin; PI3K, phosphoinositide 3-kinase; PD-1, programmed cell death protein; PD-L1, programmed cell death ligand.
Treatment of TNBC
Despite its challenges, surgery remains the traditional treatment method for TNBC. Radiation can enhance locoregional control in patients with breast cancer following breast-conserving surgery, with a beneficial effect on long-term survival. 38 Radiation therapy strongly correlated with better outcomes in patients with early-stage TNBC in a national database study in New Zealand. 39
Chemotherapy
Owing to its tendency to metastasize and recur, TNBC has few therapeutic options and a poor prognosis. Because TNBCs do not express ERs, PRs, or HER2, endocrine treatments and targeted medications are relatively ineffective.40,41 Chemotherapy is the primary therapeutic option for TNBC; patients with TNBC respond better to chemotherapy than that of patients with other types of breast cancer.42,43 Previous studies have shown that chemotherapy has a considerable advantage in the neoadjuvant setting and metastatic environment.44,45 Using a suitable chemotherapy regimen is critical for TNBC patients to obtain satisfactory treatment outcomes. 46 Taxane, anthracyclines, cyclophosphamide, and cisplatin are the recommended drugs for TNBC treatment. 47 In patients with the BL1 and BL2 subtypes of TNBC, taxane-based chemotherapy significantly improved the clinical ease rate, whereas other chemotherapy agents (eg, platinum agents and fluorouracil) affected TNBC outcomes to different degrees. 15
Chemotherapy remains the mainstay of treatment for early and locally advanced TNBC. Adjuvant chemotherapy with cytotoxic agents is performed postoperatively to eradicate microscope cancerous lesions. However, recurrence and metastasis within only a few months after completion of adjuvant chemotherapy are likely. Several clinical studies have shown that systemic chemotherapy is more effective when administered before versus that after surgery. Moreover, preoperative chemotherapy helps guide adjuvant treatment by enabling a more precise prognostic estimation and smaller surgical interventions.
As evidence of the importance of standard chemotherapy, a novel study found that TNBC patients with a germline BRCA mutation who received neoadjuvant cisplatin monotherapy less frequently had a pathologic complete response than those who received conventional chemotherapy with platin derivatives. 48 Although taxanes and anthracyclines are regarded the most effective agents, almost all women with metastatic TNBC have a poor prognosis.
Targeted Therapy
Owing to the significant heterogeneity of TNBCs, targeted therapy is challenging. 49 Personalized targeted treatments can be effective in 60% to 70% of patients with TNBC who incompletely respond to chemotherapy.50,51 Targeted therapy may include but is not limited to PARP and mitogen-activated protein kinase (MEK) inhibitors and antiandrogens. 52 Epidermal growth factor receptors and ER-ɑ36 are also potential targets. 15
PARP is a nuclear enzyme that catalyzes the movement of ADP-ribose from NAD + to target proteins. Its primary role is to stabilize the genome, repair DNA, and regulate cell cycle progression, apoptosis, and transcription. PARP inhibitors suppress DNA repair activity and induce apoptosis. 53 They greatly improve the treatment benefits of chemotherapy, increase tumor synthetic lethality in individuals with BRCA1/2 gene mutations, and target “BRCA-like” and DNA repair deficiencies. PARP inhibitors are often used for the treatment of BRCA1/2-deficient tumors; in this setting, they have considerable anticancer effects28,54,55 that far exceed those for BRCA1-expressing tumors. BRCA1/2 mutations are found in up to 19.5% of TNBC patients and are most common in Black and Hispanic populations. 56
ARs are expressed in normal and cancerous breast tissues. Expression levels vary greatly in breast cancer tissues, 57 with positive expression in approximately 10% to 15% of TNBC patients. As noted above, the LAR subtype of TNBC is sensitive to AR antagonists; thus it is regarded AR-positive.58–61 In a previous study, targeted antiandrogen therapy achieved good results in patients with the LAR subtype of TNBC. 62 In another study, application of the AR inhibitor enzalutamide had a clinical benefit rate of 25% in AR-positive patients with TNBC. 63 More investigations addressing AR function in tumors should be performed to facilitate further development of the antiandrogen therapy.
MEK inhibitors are also used in targeted TNBC therapy. Numerous cell lines prepared from TNBCs are sensitive to MEK inhibition in vitro.64,65 MEK activation stably amplifies the expression of c-Myc, a key oncogene product, in 30% of patients with TNBC, 66 and single-agent MEK inhibition interrupts c-Myc degradation in TNBC. MEK inhibitors combined with other compounds applied in clinical studies to improve TNBC treatment.67,68
Immunotherapy
The critical involvement of the immune system in cancer has been recognized for decades. Owing to exceptional anticancer activity and curative potential, immunotherapy has emerged as the major cancer treatment option in recent years.69,70 The immune checkpoint system helps prevent tumor cells from being detected and degraded by the host immune system; thus, blocking this system is a promising antitumor immunity strategy.71,72 TNBCs tend to produce neoantigens, which can be detected by the adaptive immune system owing to increased genomic instability and mutational load.
Compared with other breast cancer types, TNBCs have more tumor-infiltrating lymphocytes, as well as higher expression of PD-L1 protein and mRNA. 73 Both PD-L1 and PD-1 play a vital role in immunological tolerance control. In a 2017 study, the PD-L1 monoclonal antibody atezolizumab (Tecentriq) had a long-term impact on approximately 10% of patients with TNBC. 74 Increasing responsiveness to anti-PD-1/PD-L1 therapy will help reduce the death rate, reassuring patients with TNBC. Other useful immunotherapies include chimeric antigen receptor T-cell therapy, which effectively inhibits the actions of folate receptor alpha-expressing TNBC cells.75,76
Treatment Guidelines and Latest Trends
There are numerous national guidelines for TNBC treatment. The National Comprehensive Cancer Network advises patients with TNBC who received preoperative systemic therapy to consider receiving 6 to 8 cycles of capecitabine as adjuvant systemic therapy following radiotherapy. 77 In addition to chemotherapy, the European Society for Medical Oncology suggests that PD-L1-positive TNBC patients should also receive atezolizumab in combination with nab-paclitaxel (Abraxane) or the PD-1 inhibitor, pembrolizumab (Keytruda). 78 In a recent study, pembrolizumab combined with chemotherapy significantly extended event-free survival in patients with high-risk early-stage TNBC. 79 KEYNOTE-522, a phase 3, randomized, double-blind trial examining pembrolizumab for early-stage breast cancer, has been approved. This study promotes the development of breast cancer immunotherapy and thus raises the expectations of patients with TNBC.
Recently, many new drugs have been approved for TNBC treatment. Trastuzumab deruxtecan (T-DXd) is an antibody-drug conjugate composed of an anti-HER2 antibody and a cytotoxic topoisomerase I inhibitor. T-DXd exhibited durable antitumor activity and has been approved by the U.S. Food and Drug Administration for treatment of patients with HER2-positive breast cancer. 80 Sacituzumab govitecan is a novel antibody-drug conjugate composed of a humanized IgG1 antibody targeting trophoblast cell surface antigen 1 and SN-38, the metabolically active product of the chemotherapeutic drug irinotecan. It has recently been approved by the U.S. Food and Drug Administration for treatment of unresectable TNBC that has undergone at least 2 metastatic disease interventions. Its safety and efficacy were confirmed in the ASCENT phase III clinical trial, and its approval provides a new option for the management of solid tumors. 81 Multiple clinical trials are currently evaluating its potential use as a neoadjuvant therapy for TNBC and an adjuvant therapy in combination with immunotherapy for various solid tumors.
Genetic Biomarkers
Prognostic indicators such as tumor size and lymph node metastases are insufficient for personalized treatment of patients with early-stage breast cancer. 82 Therefore, it is essential to identify molecular biomarkers and verify their usefulness as targets in personalized treatments and as indicators of clinical efficacy although achieving this is difficult owing to the lack of therapeutic targets.83,84 Nevertheless, the following targets hold promise for patients with TNBC: PARP, insulin-like growth factor binding protein, PD-L1, interleukin-8, and glucocorticoid receptors. Three additional biomarkers are discussed below.
BRCA1/2
The protein products of BRCA1 and BRCA2 regulate numerous processes including transcription, the cell cycle, and cell proliferation and differentiation. In addition, they repair DNA via homologous recombination to maintain genetic integrity and DNA stability. 85 Approximately, 15% of patients with TNBC harbor a germline BRCA mutation.86,87 Patients with sporadic TNBC often share characteristics with BRCA1/2 mutation carriers; these include homologous recombination repair (HRR) deficiencies (sometimes referred to as “BRCAness”). Germline BRCA1/2 mutations increase susceptibility to platinum agents, PARP inhibitors, and other DNA-damaging drugs. 88 Immunotherapy combined with PARP inhibitors is being studied and appears to be a promising method of treatment for TNBC patients with BRCA1/2 mutations. Figure 3 takes the genetic biomarker BRCA as the example to illustrate how it is involved in HRR.

BRCA involve in homologous recombination repair (By Figdraw).
TP53
The protein product of the TP53 gene is critical for maintaining homeostasis and genomic integrity during DNA repair and apoptosis. 89 TP53 is one of the most frequently mutated genes in breast cancer: TP53 mutations are present in nearly 80% of all TNBC cases, 90 62% of basal-like TNBC cases, and 43% of non-basal TNBC cases. TP53 mutations greatly increase genetic instability and cause numerous cytogenetic alterations, resulting in a greater likelihood of loss of heterozygosity.91,92 Given its prevalence in patients with TNBC, TP53 is an attractive prospective target for anticancer therapy.
PI3K Pathway
The PI3K/AKT/mTOR pathway is associated with cell proliferation, differentiation, and survival. Its activity is increased in all types of breast cancer including TNBC. 93 PI3K pathway mutations/alterations are specific to the different subtypes of TNBC. Approximately 30% to 50% of TNBC patients have phosphatase and tensin homolog (PTEN) abnormalities. 94 PTEN negatively regulates PI3K signaling, 95 and its deficiency accelerates tumor cell proliferation, resulting in poor prognosis. The components of the PI3K/AKT/mTOR pathway are potential therapeutic targets in TNBC; thus, PI3K and AKT inhibitors may be useful. 96
Discussion
In recent years, the application and development of a variety of omics techniques have stimulated TNBC research. Epigenetics is the study of heritable changes in gene expression without any changes in the nucleotide sequence. Classical epigenetic mechanisms include histone modification, chromatin remodeling, DNA methylation, gene silencing, and expression of non-coding RNAs. Epigenetic changes in the expression of TNBC biomarkers are an important area of study. Genomics and transcriptomics also have broad applications in TNBC research. Genome and transcriptome sequencing analyses have shown that the molecular phenotypes of patients with TNBC vary. A recent study characterized breast cancers as “HER2-low expression” (immunohistochemistry score of 1+ or 2+ with negative in-situ hybridization results) or “HER2-zero expression” (immunohistochemistry score of 0). We suggest that HER2-low breast cancer has the potential to become a separate subtype in future.97
With the advancement of various technologies in omics, further information on suitable genetic biomarkers for TNBC is likely forthcoming, along with more accurate classification of the TNBC subtypes. Such information will aid the development of new and effective drugs for individualized treatment of patients with TNBC.
Conclusions
TNBC encompasses a diverse set of malignancies characterized by multiple genetic abnormalities. Recent progress in the investigation of TNBC subtypes, treatments, and genetic markers was reviewed to provide a theoretical basis for future studies aimed at improving TNBC outcomes. Current research on TNBC focuses on identifying genes that can serve as therapeutic targets or prognostic markers. Researchers predict that several novel biomarkers will be discovered in forthcoming decades, hopefully with encouraging results. TNBC patients have impaired immune systems, and their prognosis is worse than that of those with other types of breast cancer. Therefore, effective treatment of TNBC requires the use of multiple treatment approaches; these include a combination of recently developed immunotherapy methods, whose widespread use is likely in the future. In addition, analysis of genomic data is an effective method for subtyping and devising subtype-guided therapies. Developments in molecular biology, immunology, and computer networks will help clinicians offer personalized therapy to patients with TNBC more promptly and with enhanced precision.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This article is a review article and it does not involve related animal and patient studies.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
