Abstract
Introduction
Breast cancer is the most common cancer worldwide and a serious threat to human health. 1 HER2-overexpressed is a special breast cancer subtype, accounting for approximately 20%.2–4 Compared with HER2-negative patients, patients with HER2 overexpression have stronger tumor cell proliferation and invasiveness; therefore, the prognosis of these patients is worse.2,5,6 The clinical application of anti-HER2 targeted therapy has significantly improved the prognosis of these patients; however, 50% of patients still have tumor recurrence,7,8 and treatment optimization remains the focus of current research.
Neoadjuvant therapy (NAT) is an important strategy in comprehensive breast cancer treatment.9,10 Neoadjuvant therapy can improve the incidence of breast-conserving surgery and the quality of life of patients.11,12 Moreover, NAT can reduce the stage and increase the rate of radical surgery, thus improving patient prognosis.13,14 In addition, the response of patients to NAT is important for predicting their prognosis. Patients who achieve a complete pathological response (pCR) after NAT have longer overall survival (OS).15–18 According to the National Comprehensive Cancer Network (NCCN) guidelines, NAT is recommended for HER2-positive patients with clinical tumor stage ≥2 or cN positivity. Chemotherapy combined with a dual anti-HER2 blockade (trastuzumab and patuzumab) has a higher pCR rate and longer OS than chemotherapy combined with a single anti-HER2 blockade.19–21 Therefore, chemotherapy combined with a dual anti-HER2 blockade is the preferred NAT treatment scheme for these patients.
Owing to the improvement in the pCR rate and OS in the KRISTINE 22 and Neosphere 8 trials, both docetaxel + carboplatin + trastuzumab + parezumab (TCHP) and docetaxel + trastuzumab + parezumab (THP) are recommended by the NCCN guidelines as first-line treatment schemes for HER2-positive patients with NAT indicators. 23 However, comparative data regarding the efficacy and toxicity of TCHP and THP are limited. Recently, Spring et al 24 conducted a retrospective study on a small group of American patients and found that TCHP-treated patients had a higher pCR rate (63% vs 55%) than THP-treated patients. However, the incidence of treatment delay (16% vs 7%) and drug reduction (31.6% vs 13.8%) in the TCHP group was significantly higher than that in the THP. The study conducted by Hassett et al 25 on the cost-effectiveness and quality of life analysis of patients treated with TCHP and THP also showed that THP was significantly better than TCHP, which suggests that the tolerance of the TCHP scheme was worse than that of the THP scheme, and hospitalization time and economic burden increased when using TCHP. Compared with the American population, the Asian population is considered to have worse chemotherapy tolerance.26,27 However, no data comparing the efficacy and toxicity of TCHP and THP in Asian populations exist. Therefore, we conducted a single-center retrospective study on the Chinese population. This study aimed to compare the efficacy and toxicity of TCHP and THP in a NAT setting.
Material and Methods
Patients
This single-center, retrospective study was approved by the Biomedical Ethics Committee of Mianyang Central Hospital (No. S20220374-01; Add. Mianyang, Sichuan, China; 2022-12-30). The reporting of this study conforms to the STROBE guidelines. 28 HER2-positive breast cancer patients diagnosed between January 2020 and September 2022 were included. The inclusion criteria were as follows: (1) pathologically confirmed breast cancer and (2) HER2-positive, (3) patients who received NAT (TCHP or THP) before surgery, and (4) sufficient details of postoperative pathological data. Exclusion criteria: (1) patients previously associated with other tumors, (2) stage IV patients with distant metastases, and (3) patients whose NAT or surgery was performed at other centers. The patients’ details were de-identified.
Data Collection and Endpoints
Sex, age, weight, pathological type, degree of pathological differentiation, estrogen and progesterone receiver status, HER-2 status, type of taxane, primary tumor size, clinical stage, NAT regimen, NAT schedule, NAT dose, number of NAT cycles, adjuvant medication, toxicity during NAT, and efficacy of NAT of patients were collected from the medical record system. A progesterone or estrogen receptor (HR) level of >1% was defined as HR-positive. HER-2 immunohistochemical +++ and/or HER2/CEP17 ratio ≥ 2.0 by fluorescence in situ hybridization (FISH) was defined as HER2-positive. 29 The eighth edition of the American Joint Committee on Cancer Staging System for Breast Cancer was used for clinical staging. The Miller Payne system was used to evaluate the efficacy of NAT. 30 Complete pathological remission was confirmed by postoperative pathological examination, and pCR was achieved when both of the following points were met simultaneously: (A) for primary breast tumor, the disappearance of tumor, or the presence of carcinoma in situ; (B) for regional lymph nodes, the disappearance of tumor. 30 The Common Terminology Criteria for Adverse Events 5.0 was used for NAT toxicity grading. The primary endpoint was the pCR rate of the NAT, and the secondary endpoints included the NAT toxicity, the dose adjustment rate of the NAT (dose adjustment was defined as the dose reduction during the NAT or the treatment delay ≥5 days), the weight loss rate of patients after the NAT (weight loss was defined as the weight loss before surgery was more than 2.5 kg compared with that before the NAT), the use of adjuvant medication during the NAT and the completion rate of the NAT (6 cycles of new auxiliary treatment is the treatment standard in our center. If the patients did not complete 6 cycles of NAT owing to toxic reactions or their own wishes, NAT was defined as incomplete).
Statistical Analysis
The software SPSS Statistics 22.0 was used for data analysis, and the χ2 test was used to compare the differences in patients’ clinical characteristics, incidence of toxicity, dose adjustment rate of NAT, weight loss rate of patients, and the use of adjuvant medication. Considering that the retrospective study may have led to an imbalance in the baseline characteristics between the TCHP and THP groups, statistical analysis was conducted for the pCR rates, and a 95% confidence interval (CI) was given. Logistic regression analysis was used to evaluate the association between clinicopathological factors and pCR. Univariate analysis results P < .1 or factors that may be clinically significant factors were included in the multivariate analysis. Statistical significance was set at P < .05.
Results
Patient Characteristics
Between January 2020 and September 2022, 243 newly diagnosed breast cancer patients were HER-2 positive, 92 patients accepted the strategy of surgery combined with adjuvant therapy, 49 patients did not accept TCHP or THP as the NAT regimen, and 15 patients did not have complete clinical data. Finally, 81 patients were included in the study (TCHP, n = 38; TCHP, n = 43). The baseline patient characteristics are presented in Table 1. The median ages at the time of TCHP and THP were 52 years and 50 years, respectively. Patients over 60 years of age were 2 (5.3%) and 8 (18.6%) in the TCHP and THP groups, respectively. The clinical stages of the enrolled patients were stage II/III (stage III TCHP, 27 (71.1%); stage III THP, 23 (53.5%)). Patients with HER2 3+ in the TCHP and THP groups were 33 (86.8%) and 30 (69.8%), respectively. The pathological type was mainly invasive ductal carcinoma (TCHP, n = 37 (97.4%); THP, n = 41 (95.3%). More than half of the patients were histologically classified as grade 2 (TCHP, 37 (65.8%); THP, 41 (67.4%). The proportions of patients with HR-positive primary tumor >5 cm were similar between the 2 groups.
Patient Characteristics (N = 81).

Abbreviations: HR, progesterone receptor or estrogen receptor; THP, docetaxel + trastuzumab + pertuzumab; TCHP, docetaxel + carboplatin + trastuzumab + pertuzumab; FISH, fluorescence in situ hybridization.
5 (13.2%) patient in TCHP group and 15 (34.9%) patient in THP group performed FISH.
Complete Pathological Remission Rate of TCHP and THP
In all enrolled patients, the pCR rates of TCHP and THP groups were 44.7% (95% CI: 30.2%-60.3%) and 51.2% (95% CI: 36.8%-65.4%), respectively. Taking HER2 immunohistochemistry status as the stratification factor, the pCR rates of the TCHP and THP groups were 51.5% (95% CI: 35.2%-67.5%) and 60.0% (95% CI: 42.3%-75.4%) in HER2 3+ patients, respectively. For patients with HER2 < 3+, the pCR rates of TCHP and THP groups were 0% (5 patients with HER2 < 3+in the TCHP group, and no patients reached pCR) and 30.8% (95% CI: 12.7%-57.6%) (Figure 1).
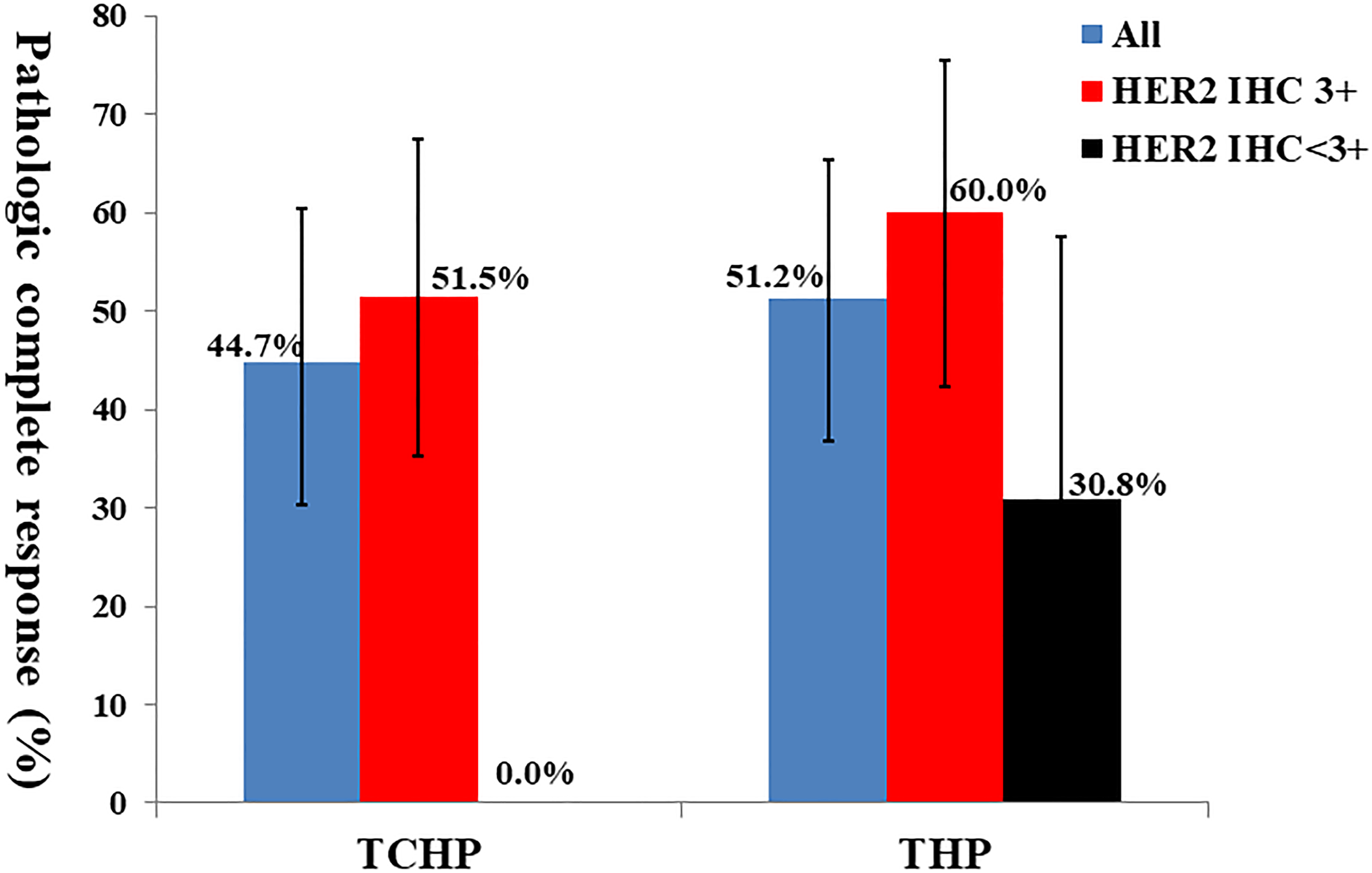
Comparison of complete pathological remission (pCR) rates between docetaxel + carboplatin + trastuzumab + pertuzumab (TCHP) and docetaxel + trastuzumab + pertuzumab (THP).
Patient Tolerance
The patient toxicities are listed in Table 2. All grades of toxicity in the TCHP and THP groups were 100% and 95.3%, respectively. The incidence of toxicity ≥ grade 3 in the TCHP group was significantly higher than that in the THP group (68.4% vs 39.5%, P = .009). Neutropenia (TCHP: 60.5%, THP: 35.7%) and asthenia (TCHP: 15.8%, THP: 7.0%) were the most common ≥ grade 3 toxicities. Four patients (10.5%) and 1 patient (2.6%) had febrile neutropenia in the TCHP and THP groups, respectively. The incidence of NAT dose adjustment was significantly higher in the TCHP group than in the THP group (26.3% vs 7.0%, P = .039) (Figure 2A). The incidence of weight loss was higher in the TCHP group than in the THP group (31.6% vs 18.6%, P = .117) (Figure 2B). Among the 81 enrolled patients, 14 did not complete 6 cycles of NAT owing to toxic reactions. The proportion of patients who underwent <6 cycles of NAT was significantly higher in the TCHP group than in the THP group (31.6% vs 4.7%, P = .004) (Figure 2C).

Comparison of dose adjustment rates, weight loss rates, and intent cycle completion rates between docetaxel + carboplatin + trastuzumab + pertuzumab (TCHP) and docetaxel + trastuzumab + pertuzumab (THP).
Toxicity.

Abbreviations: THP, docetaxel + trastuzumab + pertuzumab; TCHP, docetaxel + carboplatin + trastuzumab + pertuzumab.
P = .009, χ2 = 6.759.
Analysis of Influencing Factors of pCR
The results of the univariate analysis are presented in Table 3. HER2 immunohistochemistry status significantly impacted the pCR rate (P = .013). Subsequently, the HR status, HER2 immunohistochemistry status, pathological grade, and primary tumor size were included in the multivariate analysis. Multivariate results showed that HER2 3+ (odds ratio [OR]: 4.14, 95% CI: 1.12-15.35) and patients with primary tumor >5 cm (OR: 3.035, 95% CI: 1.12-8.26) were significantly correlated with higher pCR rate (Table 4).
Univariate Analysis: Association of Clinicopathological Factors With Pathologic Complete Response.
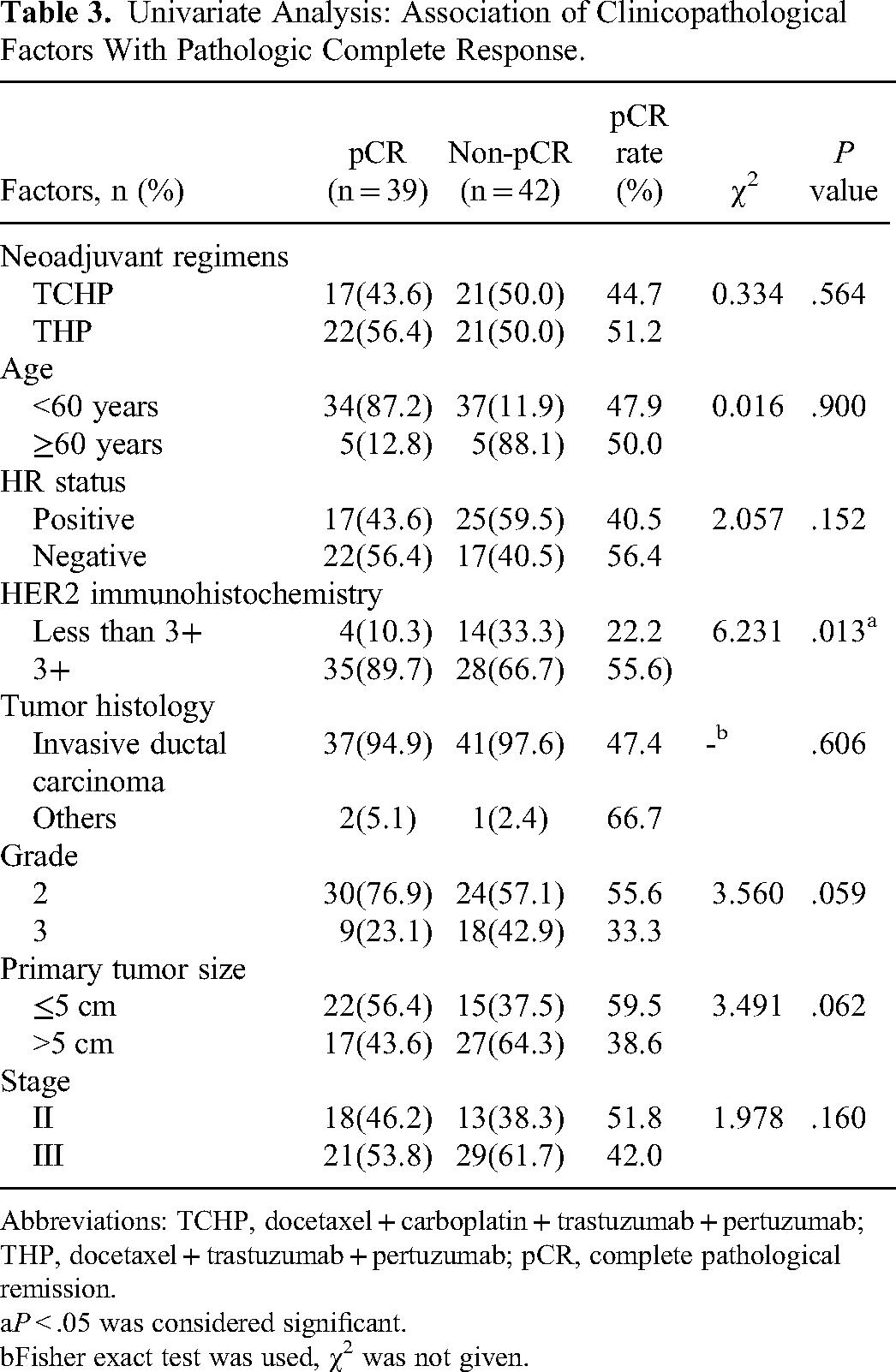
Abbreviations: TCHP, docetaxel + carboplatin + trastuzumab + pertuzumab; THP, docetaxel + trastuzumab + pertuzumab; pCR, complete pathological remission.
P < .05 was considered significant.
Fisher exact test was used, χ2 was not given.
Multivariate Analysis: Association of Clinicopathological Factors With Pathologic Complete Response.
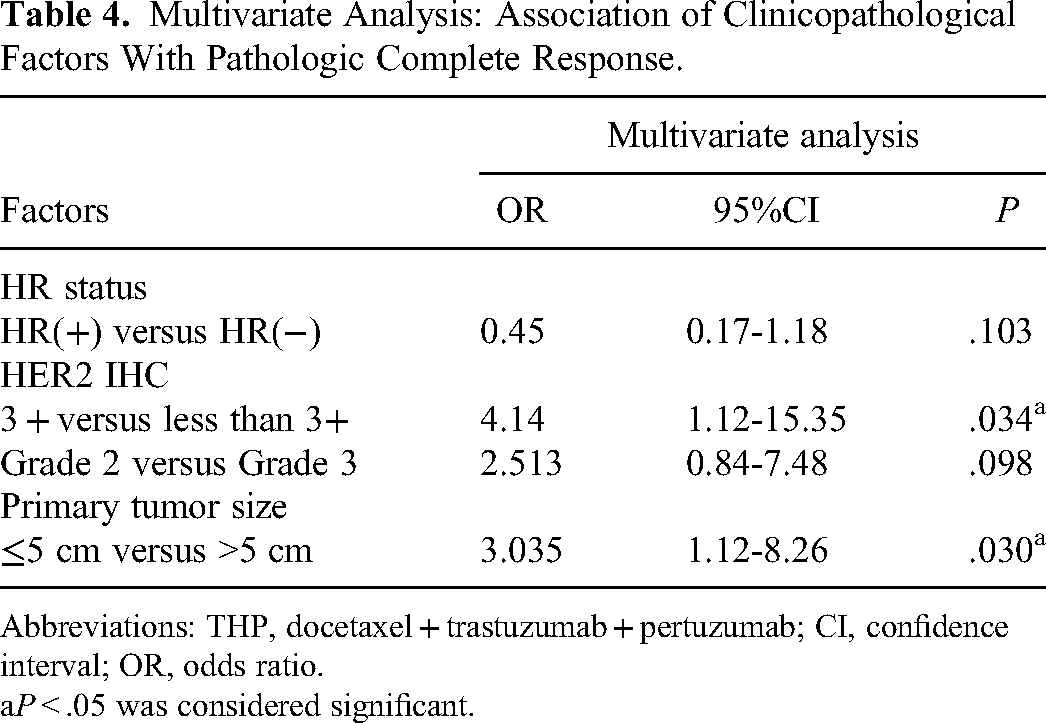
Abbreviations: THP, docetaxel + trastuzumab + pertuzumab; CI, confidence interval; OR, odds ratio.
P < .05 was considered significant.
Adjuvant Medication
The use of adjuvant medications during NAT is shown in Table 5. The proportion of patients using >30 mg of granisetron was significantly higher in the TCHP group than in the THP group (76.3% vs 9.3%, P < .001). The proportion of patients using >1500 µg recombinant human granulocyte colony-stimulating factor in the TCHP group was significantly higher than that in the THP group (52.6% vs 30.2%, P < .041). In the TCHP group, 7 (18.3%) patients used interleukin-11 to ascend platelets, 2 (5.3%) patients used erythropoietin, and 1 (2.6%) patient received a red blood cell infusion to improve anemia, which was not used in the THP group.
Comparison of Adjuvant Medications Between TCHP and THP.

Abbreviations: GM-CSF: recombinant human granulocyte colony-stimulating factor; THP, docetaxel + trastuzumab + pertuzumab; TCHP, docetaxel + carboplatin + trastuzumab + pertuzumab.
P < .05 was considered significant.
Discussion
Both TCHP and THP are recommended as first-line regimens for NAR by the NCCN guideline for HER2-positive breast cancer patients. 23 Since 2020, the Chinese Government has approved the reimbursement of trastuzumab and pertuzumab by medical insurance for NAT in HER2-positive breast cancer patients. Subsequently, these patients could afford the medical expenses of the dual-targeted anti-HER2 treatment. Therefore, we were able to recruit a certain number of patients to evaluate the clinical efficacy and toxicity of dual anti-HER2 blockade combined with chemotherapy. Our results showed that a larger proportion (18.6% vs 5.3%) of patients aged ≥60 years accepted THP, which may be because the clinical decision chose the regimen without carboplatin for safety considerations. Meanwhile, a larger proportion (71.1% vs 53.5%) of patients with clinical stage III disease accepted TCHP, possibly because the clinical decision was expected to achieve a better therapeutic effect with a stronger regimen.
In the TCHP group, our results showed that the pCR rate was 44.7%, which was slightly lower than that of the KRISTINE (55.7%) 22 and PHERGain (57.7%) 31 groups. A possible reason for this is that 31.6% of the patients in our study did not complete 6 cycles of NAT owing to toxicity. In the THP group, our results showed a pCR rate of 51.2%, similar to that reported by Spring et al; 24 however, our research results seem to be higher than those of NeoSphere (45.8%) in terms of the numerical value. 19 Possibly because in our center, the standard NAT cycle is 6, whereas, in NeoSphere's research scheme, patients only need to complete 4 cycles of NAT. The relatively small sample size may be another reason for the higher pCR rate of THP in our study.
Our study showed that neutropenia was the most common toxicity of ≥grade 3 in the TCHP and THP. Moreover, our study showed that the tolerance of patients to the TCHP regimen was lower than that in the THP group, leading to a significantly higher incidence of ≥grade 3 toxicity and a higher dosage of adjuvant medication in the TCHP group than in the THP group, leading to a higher financial burden for the TCHP regimen.25,32 Furthermore, because more patients in the TCHP group could not complete the NAT scheme, the antitumor effect may have been reduced. 33
Analysis of the factors influencing the patient's pCR rate showed that the HER2 immunohistochemical score significantly affected the patient's pCR rate. The proportion of patients who achieved pCR in the HER2 3+ population was significantly higher than that in the HER2 3+ population, which is similar to the results reported by Spring et al. 24 More importantly, we found that tumor size significantly affected the pCR rate. The primary tumor size ≤5 cm population achieved a higher pCR rate than those sized >5 cm. We speculate that this may be related to the efficiency of the anti-HER2 antibody in entering the tumor.34,35 As a monoclonal macromolecular drug, anti-HER2 antibody may be more difficult to achieve a sufficient dose to effectively kill tumor tissue in patients with a greater tumor load owing to the limitation of the blood-tumor barrier.36–40 However, our conjecture requires further validation of the pharmacokinetics of drugs in tumors.
Our study has several limitations: (1) this was a retrospective small-sample study. Thus, the sample selection may be biased. Therefore, we did not attempt to statistically analyze the pCR rates in the 2 groups of patients. (2) Because of the short period for trastuzumab and pertuzumab to be approved for medical insurance reimbursement in China, the follow-up time was insufficient, and we were unable to provide the progression-free survival and OS times of patients. (3) Power calculation was not performed to estimate the sample size selected for the study; therefore, the sample size may not have been sufficient to enable reliable statistical results.
In conclusion, our study showed that the THP regimen might be more tolerable than the TCHP regimen in NAT for HER2-positive breast cancer and did not show a lower pCR rate. However, our findings require further validation through prospective studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
This single center retrospective study was approved by the Biomedical ethics committee of Mianyang Central Hospital (No.:S20220374-01; Add: Changjiaxiang 12# Mianyang, Sichuan, China; Date:2022-12-30).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the NHC Key Laboratory of Nuclear Technology Medical Transformation (Mianyang Central Hospital) (grant nos. 2022HYX003).
