Abstract
HER2-positive breast cancer, characterized by overexpression of the human epidermal growth factor receptor 2 (HER2), accounts for approximately 15-20% of all breast cancers and is associated with aggressive tumor behavior and poor prognosis. Advances in HER2-targeted therapies, such as monoclonal antibodies (trastuzumab, pertuzumab), tyrosine kinase inhibitors (TKIs), and antibody-drug conjugates (ADCs), have significantly improved treatment outcomes in this subtype. Neoadjuvant therapy, administered before surgery, has become a cornerstone in managing HER2-positive breast cancer by improving pathological complete response (pCR) rates and survival outcomes. This review provides a comprehensive analysis of recent advancements in HER2-targeted neoadjuvant therapies, highlighting the mechanisms of action, clinical efficacy, and synergistic effects when combined with chemotherapy. Key challenges, such as treatment-related toxicities, and the potential for personalized treatment strategies based on biomarkers like tumor-infiltrating lymphocytes (TILs) and HER2-enriched subtypes, are discussed. Future directions emphasize optimizing treatment regimens to enhance efficacy while minimizing adverse effects, with novel agents such as trastuzumab deruxtecan (T-DXd) showing promise for expanding the therapeutic landscape.
Keywords
1. Introduction
Breast cancer, primarily arising from the uncontrolled proliferation of breast epithelial cells, is one of the most common malignant tumors in women. The global incidence rate of breast cancer reaches as high as 11.6%, with a mortality rate of approximately 6.6%.1,2 Conventional treatment modalities, such as mastectomy and radiotherapy, remain standard approaches for early-stage breast cancer, typically offering favorable therapeutic outcomes with manageable side effects. However, these treatments may not be equally effective for all patients, particularly those with tumors exhibiting specific molecular characteristics. Research has shown that aberrant HER2 expression (amplification or overexpression) occurs in about 15–20% of invasive breast cancers and is closely associated with aggressive phenotypes, high recurrence rates, and poor clinical prognoses.3,4 Therefore, subtyping breast cancer and clarifying its molecular features are crucial for developing more targeted and precise therapeutic strategies.
From the late 1970s to the 1980s, the rapid development of molecular biology techniques significantly advanced the research and identification processes of epidermal growth factor and its receptor. EGFR is now classified as a member of the human epidermal growth factor receptor family, specifically HER1, which also includes HER2, HER3, and HER4. HER family receptors bind with various ligands to induce homodimerization or heterodimerization, activating downstream tyrosine kinase cascades, which regulate multiple cellular processes such as proliferation, migration, invasion, and survival. These mechanisms play a central role in the onset and progression of breast cancer.5-7
HER2 is a transmembrane protein composed of 1,255 amino acids, with a molecular weight of 185 kDa. It consists of an extracellular domain, a transmembrane region, and an intracellular tyrosine kinase domain.
8
The extracellular domain is divided into four subdomains (I, II, III, and IV), which interact with epidermal growth factors or other members of the epidermal growth factor receptor family.
5
The transmembrane region anchors HER2 in the cell membrane, while the intracellular tyrosine kinase domain (amino acids 720–987) is crucial for signal transduction due to its kinase activity9,10 (Figure 1). Mechanisms of action of targeted therapies and chemotherapeutic agents in HER2-positive breast cancer treatment.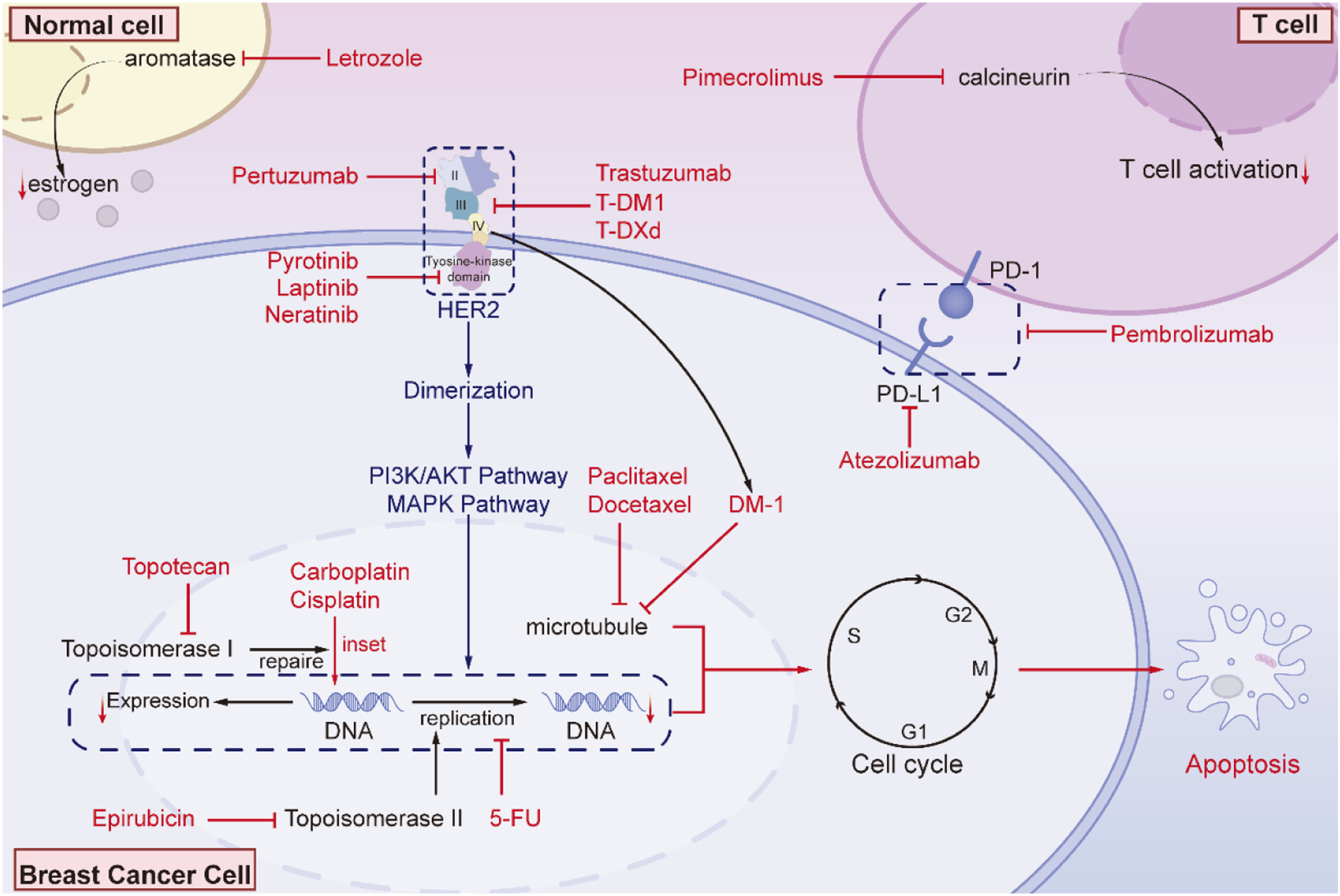
Under normal conditions, the expression of HER2 protein is tightly regulated. However, in breast cancer cells, HER2 gene amplification or protein overexpression leads to HER2 overproduction, which continuously drives rapid proliferation and metastasis of cancer cells, resulting in HER2-positive breast cancer.6,11-13 Compared to other breast cancer subtypes, this subtype tends to grow rapidly, recur frequently, and metastasize easily, leading to a lower five-year survival rate and greater invasiveness.
Neoadjuvant therapy refers to pre-surgical treatment aimed at reducing tumor size, lowering tumor staging, making inoperable tumors operable, and increasing the likelihood of breast-conserving surgery. 14 It also serves to assess the response to systemic therapy, adjust subsequent treatment plans accordingly, and provide prognostic information for patients achieving complete remission, as well as evaluating new therapies. Pathological complete response (pCR) refers to the absence of residual invasive tumor cells in the resected breast and sampled lymph nodes following neoadjuvant therapy, as determined by pathological examination. It does not necessarily indicate the complete eradication of tumor cells from the patient’s body. Studies have shown that pCR in breast cancer is significantly associated with the patient’s event-free survival and overall survival, particularly for HER2-positive breast cancer patients.15,16
Neoadjuvant therapy is also based on pCR as an alternative endpoint, accelerating the adoption of innovative therapies for HER2-positive breast cancer. Prior to the development of HER2-targeted therapies, treatment for HER2-positive breast cancer primarily relied on chemotherapy, such as taxanes and anthracyclines. 6 However, the non-selectivity of chemotherapy resulted in significant resistance and severe side effects. In contrast, HER2-targeted therapies have demonstrated significant clinical efficacy, improving both survival rates and quality of life. For example, the CLEOPATRA trial showed that dual blockade with pertuzumab and trastuzumab increased the median overall survival to 57.1 months, representing a 37% improvement over the placebo group. 17 Therefore, HER2-targeted therapies have become one of the key regimens for neoadjuvant treatment of HER2-positive breast cancer.18,19
With the advancement of molecular biology and drug development, several HER2-targeted therapies have emerged, including monoclonal antibodies, tyrosine kinase inhibitors, and antibody-drug conjugates. Trastuzumab is the first HER2-targeted drug and has shown significant efficacy in neoadjuvant therapy. When combined with pertuzumab, the therapeutic effect can be further enhanced.4,20
1.1. Trastuzumab in Neoadjuvant Therapy for HER2-Positive Breast Cancer and Its Clinical Significance
1.1.1. Efficacy Analysis of Trastuzumab Combined With Chemotherapy in Neoadjuvant Treatment for HER2-Positive Breast Cancer
Trastuzumab is a recombinant humanized monoclonal antibody specifically targeting HER2. HER2 is a tyrosine kinase receptor that is overexpressed in approximately 15%–20% of breast cancer patients. Overexpression of HER2 is closely associated with high tumor invasiveness, higher recurrence rates, and poor prognosis. Trastuzumab works by blocking HER2 receptor dimerization, thereby inhibiting downstream signaling pathways and reducing cancer cell proliferation and division. Additionally, trastuzumab can mark HER2-positive tumor cells, inducing antibody-dependent cellular cytotoxicity and promoting the internalization and degradation of the HER2 receptor, helping the immune system recognize and eliminate cancer cells, further weakening proliferation signals.21,22
Although trastuzumab has shown good efficacy in the treatment of HER2-positive breast cancer, monotherapy still faces issues of insufficient response or resistance in some patients. Therefore, it is often combined with traditional chemotherapy drugs, such as taxanes, to further enhance its anti-tumor effect. Whether this combination is due to the limitations of trastuzumab’s anti-cancer action or the insufficient control of HER2-positive breast cancer by chemotherapy agents remains an important topic of clinical and research focus. 23
When combined with chemotherapy drugs like taxanes, trastuzumab demonstrates more significant efficacy in treating HER2-positive breast cancer, notably extending PFS and OS, and has become one of the standard regimens for HER2-positive breast cancer treatment. Taxane-based drugs stabilize microtubules to prevent depolymerization, inhibiting mitosis and causing cell cycle arrest in the G2/M phase, which ultimately induces apoptosis. Cyclophosphamide, an alkylating agent, induces cell death by cross-linking DNA and disrupting its structure and function. Doxorubicin, an anthracycline antibiotic, intercalates into DNA and inhibits topoisomerase II, blocking DNA replication and transcription, leading to cell apoptosis. The antimetabolite 5-FU inhibits thymidylate synthase, preventing DNA synthesis, thus halting the cell cycle in the S phase and inducing cell death. These chemotherapy drugs act synergistically, enhancing trastuzumab’s cytotoxicity against tumor cells, significantly improving the therapeutic effect on HER2-positive breast cancer cells.24,25
In the multicenter, randomized Phase III GeparQuattro trial, 445 patients with stage II-III HER2-positive breast cancer were enrolled. 26 After adding trastuzumab to neoadjuvant chemotherapy (EC combined with taxanes), the pCR rate significantly increased to 31.7%, far higher than the 15.7% observed in the standard chemotherapy group. Moreover, no significant differences in toxic reactions were observed between the two groups. 27 Similarly, in the Phase III NOAH trial, the experimental group received trastuzumab combined with anthracycline and taxane chemotherapy (including doxorubicin, cyclophosphamide, and paclitaxel), while the control group received the same chemotherapy regimen without trastuzumab. The pCR rate in the experimental group was 43%, compared to 23% in the control group. Three-year follow-up results showed that the EFS rate in the experimental group was significantly higher than in the control group, at 71% vs. 56%. 28
These trial results strongly support the position of trastuzumab combined with chemotherapy as the standard neoadjuvant treatment for HER2-positive breast cancer. In conclusion, trastuzumab not only significantly increases the pCR rate but also plays a crucial role in improving EFS and reducing the risk of recurrence, providing a solid foundation for future HER2-positive breast cancer treatment.
1.1.2. Efficacy and Toxicity Risks of Trastuzumab Combined With Standard Chemotherapy in HER2-Positive Breast Cancer Treatment
In several single-arm studies using trastuzumab combined with standard chemotherapy drugs as neoadjuvant treatment for HER2-positive breast cancer, while high pCR rates were achieved, a number of adverse reactions were also observed, which warrant attention.
In a single-arm study conducted by Shinya Tokunaga and colleagues, 29 patients with operable stage I-IIIA HER2-positive breast cancer were included. The patients initially received four cycles of nab-paclitaxel and trastuzumab combination therapy, followed by four cycles of the FEC regimen (fluorouracil, epirubicin, and cyclophosphamide). The results showed a pCR rate of 74.0%, but a series of toxic reactions were also observed. The most common adverse effect was neuropathy, with an incidence rate of 96.6%, including a 3.4% incidence of grade 3 neuropathy. Additionally, one patient developed grade 2 interstitial pneumonia caused by albumin-bound paclitaxel or trastuzumab. During FEC treatment, common toxicities included nausea (79.3%), leukopenia (72.4%), neutropenia (72.4%), and anemia (62.1%). Furthermore, one patient experienced a grade 3 thromboembolic event. No symptomatic cardiac toxicity was observed in the study. 29
Another study enrolled 43 patients with stage II-III HER2-positive breast cancer and used the same treatment regimen. The results similarly showed risks of hematologic toxicity and neuropathy. One patient unfortunately died due to acute respiratory distress syndrome caused by febrile neutropenia and septic shock. 30
These findings suggest that although trastuzumab combined with chemotherapy significantly improves pCR rates in HER2-positive breast cancer patients, it is also associated with a higher risk of adverse reactions, particularly neurotoxicity and hematologic toxicity. Therefore, close monitoring and proactive management of these potential adverse effects are crucial in clinical practice to ensure patient safety during treatment.
1.1.3. Cost-Effective Trastuzumab Biosimilars: CT-P6 and SB3
CT-P6 and SB3 are two trastuzumab biosimilars used for the treatment of HER2-positive breast cancer. Their efficacy, safety, pharmacokinetics, and immunogenicity are similar to the reference drug trastuzumab, but they offer significantly lower costs, providing a more cost-effective treatment option.
In a Phase III equivalence trial led by Justin Stebbing and colleagues, CT-P6 demonstrated comparable efficacy and safety to the reference trastuzumab. The pCR rate for CT-P6 was 46.8%, which was within the equivalence range of the reference drug’s 50.4%, and the 6-year OS rate for CT-P6 was 96%, higher than the reference drug’s 94%. This indicates that CT-P6 provides similar efficacy to the original drug in early HER2-positive breast cancer patients, while being more cost-effective, helping to improve treatment accessibility and reduce the burden on healthcare systems.31,32
Similarly, SB3 was compared with the reference trastuzumab in a Phase III study, and the results showed that SB3 was similar to trastuzumab in terms of efficacy, cardiac safety, and long-term outcomes. After five years of follow-up, the 5-year EFS rate for the SB3 group was 82.8%, and the OS rate was 93.1%, while the trastuzumab group had an EFS of 79.7% and an OS of 86.7%. These results further confirm the feasibility of SB3 as a trastuzumab biosimilar.
Trastuzumab combined with chemotherapy has demonstrated significant efficacy in treating HER2-positive breast cancer, particularly in improving the pCR rate and enhancing EFS. However, treatment is also associated with some risks of adverse effects, particularly neurotoxicity and hematologic toxicity, which require close monitoring and active management in clinical practice. With the introduction of biosimilars, CT-P6 and SB3 provide more cost-effective treatment options, with efficacy and safety comparable to the reference trastuzumab. The use of CT-P6 and SB3 not only increases the accessibility of treatment for HER2-positive breast cancer patients but also reduces the economic burden on healthcare systems, further promoting the widespread clinical use of trastuzumab biosimilars. In the future, with further clinical validation and optimization of these biosimilars, it is expected that they will offer more widespread and economical treatment options for HER2-positive breast cancer.33,34
1.2. Lapatinib and Trastuzumab Combination in HER2-Positive Breast Cancer Treatment: Current Research Status
1.2.1. Efficacy and Mechanism of Lapatinib Combined With Trastuzumab in HER2-Positive Breast Cancer
Lapatinib is a small molecule tyrosine kinase inhibitor that exerts its effect by inhibiting the tyrosine kinase activity of both HER2 and EGFR. It competitively binds to the tyrosine kinase domains of HER2 and EGFR, blocking their phosphorylation processes and inhibiting the activation of downstream signaling pathways such as PI3K/Akt and Ras/Raf/MEK/ERK, which leads to the suppression of cancer cell proliferation and the induction of apoptosis. Unlike trastuzumab, which acts on the extracellular domain of HER2, lapatinib targets the intracellular tyrosine kinase activity, providing additional internal inhibition of signal transduction. Additionally, lapatinib decreases the number of HER2 receptors on the cell surface, enhancing the sensitivity of cancer cells to the immune system and promoting receptor internalization and degradation. In the EPHOS-B trial, 257 newly diagnosed HER2-positive breast cancer patients were treated preoperatively and randomly assigned to either no treatment, trastuzumab, lapatinib, or a combination of both. The results showed that the combination therapy group had a significantly enhanced Ki67 response, and a Ki67 reduction of ≥50% was associated with a lower risk of recurrence. Compared to the monotherapy groups, combination therapy significantly improved cancer cell suppression, indicating the important clinical significance of combining lapatinib with trastuzumab in HER2-positive breast cancer treatment. 35
1.2.2. Efficacy and Clinical Application of Trastuzumab and Lapatinib Combined With Standard Chemotherapy in HER2-Positive Breast Cancer
The combination of trastuzumab and lapatinib has shown significant efficacy in the treatment of HER2-positive breast cancer, particularly when combined with standard chemotherapy drugs such as paclitaxel or docetaxel to further improve treatment outcomes. In a Phase III randomized trial led by Lisa A. Carey and colleagues, 305 patients with stage II or III HER2-positive breast cancer were randomly assigned to different treatment groups: weekly paclitaxel combined with trastuzumab and lapatinib (THL group), trastuzumab alone (TH group), and lapatinib alone (TL group). The results showed that in 295 evaluable patients, the pCR rate in the THL group was 56%, significantly higher than the 46% in the TH group and 32% in the TL group. Particularly in hormone receptor-negative patients, the pCR rate in the THL group was 79%, compared to 54% in the TH group and 37% in the TL group. These results highlight the importance of hormone receptor status in HER2-positive breast cancer treatment and support dual HER2-targeted therapy, especially in hormone receptor-negative patients. 36 After more than seven years of follow-up, the THL group showed significantly better RFS and OS compared to the TH group, with no significant differences between the TH and TL groups. 37
Similarly, in the randomized multicenter Phase III NeoALTTO trial, six-year and ten-year follow-up data further demonstrated the significant efficacy of the combination therapy, especially in improving EFS and OS in hormone receptor-negative breast cancer patients. 38 In another Phase II randomized Cher-LOB study, 121 patients with HER2-positive stage II to IIIA breast cancer received neoadjuvant chemotherapy based on paclitaxel-anthracycline combined with trastuzumab (Group A), lapatinib (Group B), or trastuzumab plus lapatinib (Group C). The results showed that the pCR rate in Group C was the highest at 44.2%, compared to 25% in both Group A and Group B. After nine years of median follow-up, the combination of trastuzumab and lapatinib showed an improving trend in RFS (HR 0.44). Furthermore, pCR was significantly associated with better RFS (HR 0.12) and OS (HR 0.12), indicating that dual HER2-targeted therapy provides better prognostic outcomes for HER2-positive breast cancer patients. 39
1.3. Pyrotinib Combination Therapy in HER2-Positive Breast Cancer: Clinical Application, Efficacy, Mechanism, and Adverse Reactions
1.3.1. Dual-Targeted Therapy With Pyrotinib and Trastuzumab in HER2-Positive Breast Cancer
Pyrotinib is an irreversible small-molecule tyrosine kinase inhibitor (TKI) targeting members of the human epidermal growth factor receptor (HER) family, including HER1, HER2, and HER4. By inhibiting the intracellular kinase domains of these receptors, pyrotinib blocks the activation of downstream signaling pathways such as phosphatidylinositol-3-kinase/protein kinase B (PI3K/Akt) and mitogen-activated protein kinase (MAPK), thereby suppressing tumor cell proliferation and promoting apoptosis. Unlike trastuzumab, which binds to the extracellular domain of HER2, pyrotinib targets the intracellular tyrosine kinase domain and inhibits receptor signaling from within the cell. The combination of these two agents therefore enables dual blockade of the HER2 signaling pathway, resulting in enhanced anti-tumor activity. 40
The efficacy of pyrotinib-based dual-targeted therapy has been demonstrated in several clinical trials. In the multicenter, double-blind, phase III PHEDRA trial, 355 women with early or locally advanced HER2-positive breast cancer were randomly assigned to receive pyrotinib or placebo in combination with trastuzumab and docetaxel. The results showed that the pathological complete response (pCR) rate was significantly higher in the pyrotinib group than in the placebo group (41.0% vs 22.0%). 41 These findings indicate that the addition of pyrotinib can significantly enhance the efficacy of trastuzumab-based neoadjuvant therapy and provides a promising targeted treatment strategy for patients with HER2-positive breast cancer.
From a mechanistic perspective, the combination of pyrotinib, trastuzumab, and cytotoxic chemotherapy agents exerts synergistic anti-tumor effects through multiple pathways. Pyrotinib irreversibly inhibits the kinase activity of HER family receptors, while trastuzumab binds to the extracellular domain of HER2 and activates antibody-dependent cellular cytotoxicity (ADCC), thereby promoting immune-mediated tumor cell clearance. Together, these two agents provide a more comprehensive inhibition of HER2 signaling. In addition, paclitaxel, a microtubule-stabilizing agent, disrupts cell cycle progression and induces apoptosis by preventing microtubule depolymerization. Cisplatin, a platinum-based chemotherapeutic agent, forms intra- and inter-strand DNA crosslinks, thereby interfering with DNA replication and transcription and leading to tumor cell death.
Further clinical evidence supporting this therapeutic strategy was provided by the phase II randomized NeoATP trial. In this study, patients with stage IIA–IIIC HER2-positive breast cancer received neoadjuvant therapy consisting of pyrotinib and trastuzumab combined with weekly paclitaxel and cisplatin for four cycles. The overall pCR rate reached 69.81%, and an even higher pCR rate of 85.71% was observed among patients with hormone receptor-negative disease. Interestingly, the presence of PIK3CA gene mutations did not significantly affect treatment response. 42
Taken together, these findings suggest that the combination of pyrotinib, trastuzumab, and chemotherapy agents such as taxanes and platinum-based drugs demonstrates substantial clinical efficacy in HER2-positive breast cancer, particularly among hormone receptor-negative patients. This regimen therefore represents a promising and effective neoadjuvant treatment strategy.
1.3.2. Safety and Adverse Reactions of Pyrotinib Combination Therapy in Neoadjuvant Treatment for HER2-Positive Breast Cancer
Although pyrotinib-based combination therapy has demonstrated significant efficacy in the neoadjuvant treatment of HER2-positive breast cancer, treatment-related adverse events should also be carefully considered. In the phase III PHEDRA trial, the most frequently reported grade ≥3 adverse event in the pyrotinib group was diarrhea, which occurred in 44.4% of patients compared with 5.1% in the placebo group. Other commonly observed grade ≥3 adverse events included neutropenia (18.5% vs 20.3%) and leukopenia (16.3% vs 13.6%). Despite the relatively higher incidence of gastrointestinal toxicity, no treatment-related deaths were reported, indicating that the overall safety profile of pyrotinib remains acceptable and manageable in clinical practice. 41
Similar safety findings were reported in a multicenter phase II clinical study conducted by Zhenzhen Liu and colleagues. In this study, 74 patients with stage II–III HER2-positive breast cancer received a four-drug neoadjuvant regimen consisting of pyrotinib, docetaxel, carboplatin, and trastuzumab. The most common grade ≥3 adverse events included diarrhea (43.2%), anemia (37.8%), vomiting (16.2%), and thrombocytopenia (10.8%). Importantly, no treatment-related deaths occurred during the study period. 43
Overall, although pyrotinib-based regimens are commonly associated with gastrointestinal toxicity and hematologic adverse reactions, these side effects are generally manageable with appropriate supportive care. Therefore, the safety profile of pyrotinib does not diminish its clinical value as an important component of neoadjuvant therapy for patients with HER2-positive breast cancer.
1.4. Synergistic Mechanisms of Dual Targeted Therapy (Trastuzumab/Pertuzumab) Combined With Chemotherapy in Neoadjuvant Treatment for HER2-Positive Breast Cancer
The combination of trastuzumab and pertuzumab in treating HER2-positive breast cancer demonstrates powerful efficacy through several synergistic mechanisms. First, trastuzumab and pertuzumab target different extracellular domains of the HER2 receptor, preventing both homodimerization and heterodimerization, thus inhibiting downstream signaling pathways such as PI3K/AKT and RAS/MAPK. Additionally, this combination enhances ADCC, recruiting NK cells to attack tumor cells and significantly increasing the sensitivity of tumor cells to immune responses. 44
Trastuzumab and pertuzumab also reduce the number of breast cancer stem cells, decrease tumor invasiveness and metastatic potential, and induce apoptosis. These mechanisms help to improve treatment outcomes, making this combination therapy a key strategy in the treatment of HER2-positive breast cancer 45 (Figure 1).
1.4.1. Trastuzumab and Pertuzumab Combined With Taxanes Significantly Enhance Treatment Efficacy in HER2-Positive Breast Cancer
Clinical Trials for HER2-Positive Breast Cancer Treatments

H: Trastuzumab; Cis: Cisplatin; PTX: Paclitaxel; nab-PTX: nab-paclitaxel; PBO: placebo; DTX: Docetaxel; Cb: Carboplatin; PY:Pyrotinib; L: lapatinib; DOX: Doxorubicin; MTX:Methotrexate; TPCR: Total pathological complete remission rate; PP: Eligible population for the plan; mITT: modify intention to treat population; F: fluorouracil; E: epirubicin; C: cyclophosphamide.
Clinical Trial Outcomes for HER2-Positive Breast Cancer Using Trastuzumab, and Pertuzumab-Based Regiments

H: Trastuzumab; P: Paltuzumab; PTX: Paclitaxel; nab-PTX: nab-paclitaxel; PBO: placebo; DTX: Docetaxel; Cb: Carboplatin; E: epirubicin; C: cyclophosphamide; PAL: palbociclib; PLD:pegylated liposomal doxorubicin; T-DM1:Trastuzumab Emtansine; LET: Letrozole; ATEZO:Atezolizumab.
Further evidence supporting treatment optimization was provided by the Phase II NeoTOP study, which investigated the role of anthracycline-containing regimens according to TOP2A amplification status. 47 Patients received neoadjuvant trastuzumab and pertuzumab combined with chemotherapy tailored to TOP2A status. The study demonstrated similarly high pCR rates in both groups (74.4% in TOP2A-amplified vs. 71.9% in TOP2A–non-amplified patients), with favorable long-term outcomes. These results suggest that dual HER2 blockade combined with taxane-based chemotherapy can achieve high response rates regardless of TOP2A status, while the necessity of anthracycline-containing regimens remains questionable.
Overall, these studies demonstrate that the combination of trastuzumab and pertuzumab with taxanes is highly effective in treating HER2-positive breast cancer, significantly improving pCR rates and survival. Therefore, future research could further focus on exploring precision treatment regimens with less or no chemotherapy, thus maximizing efficacy while minimizing side effects.
1.4.2. Trastuzumab and Pertuzumab Dual Targeted Therapy in HER2-Positive Breast Cancer: PET-Guided Personalized Strategy for an Effective Chemotherapy-Free Regimen
The combination of trastuzumab and pertuzumab with steroid-based drugs and carboplatin or cisplatin not only inhibits HER2 signaling but also induces direct DNA damage, enhancing efficacy and improving treatment outcomes. In a study by José Manuel Pérez-García and colleagues, early HER2-positive breast cancer patients received HER2 dual blockade with or without chemotherapy. 48 The study demonstrated that an individualized strategy based on early 18F-FDG-PET evaluation could effectively select patients suitable for trastuzumab + pertuzumab dual HER2 blockade without chemotherapy. This regimen significantly improved the pCR rate in PET responders (65.6% vs. 37.9%) compared to the traditional chemotherapy-containing regimen, with no disease progression during treatment. These findings suggest that certain HER2-positive breast cancer patients could benefit from HER2 dual blockade therapy without chemotherapy.
Compared to traditional treatment regimens, the combination of trastuzumab and pertuzumab in dual-targeted therapy without platinum agents already shows a high pCR rate. 49 While platinum-based regimens (carboplatin/cisplatin) can improve pCR levels to some extent, the efficacy of dual-targeted therapy is already close to or reaches the treatment effect of platinum-containing regimens, and the optimization value of this therapeutic strategy requires further validation.
1.4.3. Management of Adverse Reactions and Optimization of Efficacy in HER2-Positive Breast Cancer Treatment With Trastuzumab, Pertuzumab, and Taxane/Platinum Chemotherapy Regimens
The combination of dual HER2-targeted therapy and various chemotherapy regimens has shown significant efficacy, but the incidence of grade 3-4 severe adverse reactions varies. Notably, cardiac toxicity and hematologic toxicity are important adverse effects that require close monitoring. In Sandra Swain’s Phase III study, 17 despite the combination of trastuzumab, pertuzumab, and docetaxel significantly extending the OS of patients, there was a significant increase in severe diarrhea (15-20%) and rash (8-12%) in the pertuzumab group, and fatal cases of neutropenia (1.2%) and myocardial infarction (0.8%) were reported, highlighting the need for monitoring cardiac and hematologic toxicity risks.
A study with a platinum-containing regimen (Brecan II study) showed that adding carboplatin significantly increased the incidence of grade 3-4 neutropenia (30.2%), raising questions about the necessity of platinum-based drugs. In the study by Lavasani and colleagues (nab-paclitaxel + dual-targeted therapy), the regimen avoided anthracyclines and carboplatin, with no cases of febrile neutropenia, suggesting a lower risk of hematologic toxicity. In the BrUOG study (wPCbTP regimen), 50 the use of divided dosing of pertuzumab significantly reduced the incidence of ≥2 grade diarrhea (40%), demonstrating the optimization value of the dosing regimen, although bone marrow suppression risks in carboplatin-containing regimens remain a concern. In the PerSeUS-BC04 trial, 51 during the FEC sequential treatment phase, non-hematologic toxicities (such as neuropathy) were significantly reduced, but pre-existing paclitaxel-related adverse effects still required management.
Notably, the Brecan II study 52 found that 31% of patients experienced grade 3 or higher toxicity, with neutropenia (30.2%) and fatigue (8.3%) being the most prominent, indicating that the blood toxicity risk with anthracycline-containing regimens (pegylated doxorubicin) is high and requires careful assessment during use.
In conclusion, the combination of trastuzumab and pertuzumab with taxane drugs and platinum-based drugs in neoadjuvant treatment for HER2-positive breast cancer significantly improves pCR and OS through various synergistic mechanisms. However, careful monitoring of cardiac function and hematologic parameters is crucial to control potential severe adverse events.
1.5. New Advances in HER2-Positive Breast Cancer Treatment: Integration of T-DM1, Atezolizumab, and Personalized Therapies
1.5.1. T-DM1 Combined With Pertuzumab for HER2-Positive Breast Cancer: Dual Benefits of Chemotherapy-Free Efficacy and Reduced Toxicity
T-DM1, as a HER2-targeted ADC, consists of trastuzumab covalently linked to the microtubule inhibitor DM1.53,54 It specifically targets HER2-positive cancer cells through trastuzumab, delivering DM1 directly to the tumor site, where it induces cell cycle arrest at the M phase and apoptosis by inhibiting microtubule polymerization. This dual mechanism of targeted delivery and cytotoxic action has been confirmed in multiple studies to have significant efficacy in the neoadjuvant treatment of HER2-positive breast cancer.
In the randomized Phase II JBCRG20 study, 55 operable HER2-positive primary breast cancer patients were divided into three treatment groups: six cycles of docetaxel, carboplatin, trastuzumab, and pertuzumab (TCbHP); four cycles of TCbHP; and four cycles of T-DM1 combined with pertuzumab (T-DM1 + P) or 4 + 2 cycles. After four cycles, patients who did not respond to the T-DM1 + P regimen were switched to anthracycline-based therapy. The results showed that the pCR rate in the T-DM1 + P group was significantly better than in the TCbHP group, with similar five-year DFS rates. In another study by Thomas Hatschek and colleagues, HER2-positive breast cancer patients were randomly assigned to either the standard treatment group (DTP: docetaxel, trastuzumab, pertuzumab, and pegfilgrastim for six cycles) or the experimental group (T-DM1 for six cycles). The pCR rates were similar between the two groups, with higher pCR rates observed in hormone receptor-negative patients. The randomized Phase II PREDIX HER2 trial 56 showed that the long-term efficacy of the T-DM1 group was comparable to the THP group (trastuzumab, pertuzumab, and docetaxel). These results indicate that the combination of trastuzumab, pertuzumab, and T-DM1 is effective in treating HER2-positive breast cancer, with pCR rates similar to traditional chemotherapy regimens. This therapy offers a chemotherapy-free alternative for certain patients.
Further exploration of T-DM1-based combinations has also been reported in the phase II TEAL trial. 57 In this open-label randomized study, neoadjuvant treatment with T-DM1 plus lapatinib and nab-paclitaxel was compared with the standard regimen of trastuzumab, pertuzumab, and paclitaxel in early-stage HER2-positive breast cancer. The experimental regimen demonstrated a significantly higher proportion of patients achieving residual cancer burden (RCB) 0–I (100% vs. 62.5%, P = 0.0035), with particularly pronounced benefit in the ER-positive subgroup. Importantly, the safety profiles were comparable between the two groups, suggesting that intensified HER2-targeted strategies incorporating T-DM1 may further improve neoadjuvant response.
In the 2023 KRISTINE trial, 58 444 HER2-positive breast cancer patients were randomly assigned to receive either neoadjuvant T-DM1 + P or TCH + P treatment. The pCR rate in the TCH + P group was significantly higher than in the T-DM1 + P group (55.7% vs. 44.4%), with an absolute difference of -11.3 percentage points (P = 0.016). The study also found a positive correlation between HER2 expression levels and immune microenvironment activity with pCR rates, highlighting the crucial role of HER2 and immune biomarkers in dual-target HER2 therapy.
1.5.2. Breakthrough Advances in New-Generation Anti-HER2 ADCs in Breast Cancer Treatment: The Application of Enhertu (T-DXd)
Enhertu (trastuzumab deruxtecan, T-DXd) is a next-generation HER2-targeted antibody–drug conjugate (ADC) consisting of an anti-HER2 antibody, a cleavable peptide linker, and a potent topoisomerase I inhibitor payload. Compared with trastuzumab emtansine (T-DM1), T-DXd exhibits improved antitumor activity through enhanced drug delivery and a strong bystander effect, enabling cytotoxic activity not only in HER2-high tumors but also in HER2-low–expressing and neighboring cancer cells.59,60 Clinical evidence further supports its efficacy in early-stage disease. In the phase III DESTINY-Breast11 trial, neoadjuvant T-DXd followed by paclitaxel plus trastuzumab and pertuzumab (T-DXd-THP) significantly improved pathological complete response (pCR) compared with dose-dense doxorubicin plus cyclophosphamide followed by THP (ddAC-THP) in high-risk HER2-positive early breast cancer (67.3% vs. 56.3%, P = 0.003), while demonstrating a more favorable safety profile with lower rates of grade ≥3 adverse events and cardiac dysfunction. These findings highlight the potential of T-DXd–based regimens to enhance efficacy while reducing the toxicity burden associated with conventional chemotherapy. 61 SHR-A1811 is another novel HER2-directed antibody-drug conjugate (ADC) comprising trastuzumab, a cleavable linker, and the novel topoisomerase I inhibitor payload SHR169265. In a global multicenter phase I trial involving multiply pretreated patients with HER2-positive advanced breast cancer, SHR-A1811 demonstrated a confirmed objective response rate of 76.3%. The recommended dose for the expansion phase was 4.8–6.4 mg/kg. 62
In breast cancer treatment, T-DXd has already shown remarkable efficacy in metastatic and refractory cases, while also demonstrating potential in early neoadjuvant therapy. Notably, in the SHAMROCK Phase II study, T-DXd showed promise in reducing the need for traditional chemotherapy, providing better treatment outcomes for patients. 63 Overall, T-DXd has become a cornerstone therapy for metastatic HER2-positive breast cancer and has largely replaced T-DM1 in advanced disease owing to its superior clinical efficacy. 64
In conclusion, T-DM1 and Enhertu represent significant advancements in the treatment of HER2-positive breast cancer. Compared to traditional chemotherapy, they offer more potent targeted therapies. T-DM1, combining trastuzumab and the cytotoxic drug DM1, demonstrates efficacy in neoadjuvant treatment, with its pCR rate comparable to that of chemotherapy regimens. Meanwhile, Enhertu, as a next-generation ADC, has a broader treatment range due to its enhanced “bystander effect,” impacting both HER2-high and HER2-low tumors as well as nearby cancer cells. T-DXd has shown promising results in both metastatic and early treatment settings, particularly in reducing the need for chemotherapy. It may represent the cornerstone of future HER2-positive breast cancer treatment, providing better survival outcomes and quality of life for patients.
1.5.3. Ki67-Dynamic Guided Letrozole Dual-Targeted Neoadjuvant Regimen: PerELISA Study Achieves Chemotherapy-Free Enhancement in HR+/HER2+ Breast Cancer
Letrozole, an aromatase inhibitor, works by inhibiting estrogen synthesis to treat hormone receptor -positive breast cancer. It enhances the efficacy of HER2-targeted therapies and reduces tumor burden, playing a critical role in neoadjuvant treatment for HER2-positive breast cancer.65,66
The PerELISA study is a Phase II multicenter trial that assessed the efficacy of a neoadjuvant treatment regimen based on Ki67 suppression after two weeks of letrozole treatment in postmenopausal HR+/HER2+ breast cancer patients. After two weeks of letrozole treatment, patients were divided into molecular responders and non-responders based on the degree of Ki67 suppression. Responders continued with letrozole + trastuzumab + pertuzumab (LHP), while non-responders switched to paclitaxel + dual-target therapy (THP). The pCR rate for molecular responders was 20.5%, while the pCR rate for non-responders was as high as 81.3%. In the HER2-enriched subtype (PAM50 classification), the pCR rate for molecular responders was significantly higher (45.5% vs. 13.8%). This study achieved the target pCR rate, suggesting that patients selected based on Ki67 suppression levels can achieve significant pCR without chemotherapy. Furthermore, the PAM50 intrinsic subtype identified patients who might not require chemotherapy. 67
The PerELISA study pioneered the “Ki67 dynamic response + PAM50 subtype dual-dimension screening” model, enabling specific subgroups (HER2-enriched/Ki67 responders) to achieve a significant pCR improvement through the letrozole dual-target regimen. This provides evidence-based support for the chemotherapy-free strategy in HR+/HER2+ breast cancer.68,69
1.5.4. Adverse Reaction Profiles and Multidimensional Monitoring Management Strategies of HER2-Targeted ADCs (T-DM1/Enhertu) in Breast Cancer Treatment
T-DM1 and Enhertu, as novel ADCs, significantly enhance the efficacy of HER2-positive breast cancer treatment while exhibiting a distinct toxicity profile compared to traditional chemotherapy. Their severe adverse reactions require targeted management. For example, in the JBCRG20 study and the research by Hatschek and colleagues, T-DM1 combined with pertuzumab significantly reduced grade 3-4 hematologic toxicities compared to chemotherapy regimens containing taxanes/platinu, such as febrile neutropenia, with a markedly lower incidence in the T-DM1 group (3.0% vs. 26.3%). Additionally, the incidence of non-hematologic toxicities such as alopecia and neuropathy was significantly lower. However, T-DM1 still carries specific risks: its DM1 payload can cause liver enzyme elevations (15-20%) and grade 2-3 thrombocytopenia (10%), requiring regular monitoring of liver function and platelet counts. 55
Enhertu, as a next-generation ADC, expands its efficacy through a “bystander effect,” but its topoisomerase inhibitor payload may pose a unique risk of interstitial lung disease (ILD). The SHAMROCK Phase II study showed a higher incidence of ILD in the T-DXd treatment group (13.6%), necessitating early detection through high-resolution CT and symptom monitoring. 63 Furthermore, the incidence of cardiac toxicity (such as a decrease in left ventricular ejection fraction) in T-DXd was lower than that of traditional anthracycline-based regimens (<2% vs. 5-10%), but regular echocardiograms every three months are still required for evaluation.
1.6. IMpassion050 Phase III Trial: Atezolizumab Combined With Neoadjuvant Chemotherapy Failed to Improve pCR Rate in HER2-Positive Breast Cancer
The PD-L1 inhibitor atezolizumab blocks the interaction between PD-L1 and PD-1, reactivating T-cell anti-tumor responses. In neoadjuvant treatment, atezolizumab combined with trastuzumab and chemotherapy can enhance the immune system’s ability to recognize and target HER2-positive breast cancer cells, thereby shrinking tumors, increasing the likelihood of surgical success and reducing the risk of recurrence.70,71
In the IMpassion050 Phase III randomized trial, 72 patients with primary tumors greater than 2 cm and lymph node positivity were randomly assigned to receive atezolizumab or a placebo. Patients first received dose-dense doxorubicin and cyclophosphamide treatment, followed by paclitaxel and HER2 dual-target therapy (PH). After surgery, they continued with atezolizumab/placebo and PH treatment for a total of one year. In both the intention-to-treat population and the PD-L1 positive subgroup, adding atezolizumab to dose-dense doxorubicin/cyclophosphamide-paclitaxel and PH did not significantly improve the pCR rate for high-risk HER2-positive early breast cancer compared to the placebo group. In the ITT population, the pCR rate was similar between the placebo group (62.7%) and the atezolizumab group (62.4%). For PD-L1-positive tumors, the pCR rate in the placebo group was slightly higher than in the atezolizumab group (72.5% vs. 64.2%).
The IMpassion050 Phase III trial indicated that adding atezolizumab to the chemotherapy + PH standard regimen did not significantly improve pCR rates, regardless of PD-L1 status, and the PD-L1 positive subgroup showed a numerical disadvantage. The standard neoadjuvant chemotherapy combined with PH regimen remains the cornerstone of treatment. 72
1.7. TOUCH Trial: Chemotherapy-free Neoadjuvant Strategy With Palbociclib and Letrozole Plus Dual HER2 Blockade
Cyclin-dependent kinase 4/6 (CDK4/6) inhibitors such as palbociclib demonstrate potent anti-proliferative activity in hormone receptor–positive breast cancer. In HR-positive/HER2-positive disease—a biologically heterogeneous subtype with relatively low pathological complete response (pCR) rates to conventional neoadjuvant chemotherapy—combining CDK4/6 inhibition with endocrine therapy and HER2-targeted agents has emerged as a promising chemotherapy-sparing strategy. 73
The phase II TOUCH trial evaluated this approach in postmenopausal patients with HR-positive/HER2-positive early breast cancer. In this international, open-label randomized study, 147 patients with tumors larger than 1 cm and node-negative or limited node-positive disease (cN0–1) were assigned to receive 16 weeks of weekly paclitaxel plus trastuzumab and pertuzumab (HP) or a chemotherapy-free regimen consisting of palbociclib and letrozole combined with HP. Among the 145 treated patients, the pCR rate was 32.9% in the paclitaxel + HP group and 33.3% in the palbociclib + letrozole + HP group, indicating comparable efficacy between the two strategies. Treatment completion rates were higher in the palbociclib group, although grade 3–4 neutropenia occurred more frequently. 74
Although the study did not confirm the predefined biomarker interaction hypothesis, the TOUCH trial provides important proof-of-concept evidence that CDK4/6 inhibitor–based, chemotherapy-free neoadjuvant therapy combined with dual HER2 blockade may represent a feasible de-escalation strategy for patients with HR-positive/HER2-positive early breast cancer.
2. Discussion
In recent years, significant progress has been made in neoadjuvant treatment for HER2-positive breast cancer, particularly in targeted therapies and combination treatments. These innovations not only open new avenues for clinical treatment but also improve the prognosis and quality of life for HER2-positive breast cancer patients. 75 This review highlights the latest advancements in neoadjuvant therapies for HER2-positive breast cancer over the past five years, with a focus on the combination of trastuzumab with standard chemotherapy regimens such as paclitaxel and docetaxel. It also examines the benefits and adverse effects associated with integrating small-molecule inhibitors, including lapatinib and pyrotinib, into these combination strategies, as well as the development and utilization of trastuzumab biosimilars.
Monoclonal antibody therapies, such as trastuzumab, significantly increase the pCR rate in HER2-positive breast cancer and greatly improve patient survival.4,76-79 Given that trastuzumab monotherapy fails to fully block downstream HER2 signaling pathways, some patients experience compensatory activation of HER2 signaling—such as through heterodimerization with EGFR or HER3—making single-agent trastuzumab insufficient to achieve the expected benefit in the neoadjuvant setting and predisposing patients to acquired resistance. For these reasons, clinical investigators have increasingly favored multi-agent combination strategies, aiming to enhance antitumor activity and achieve greater therapeutic benefit through multi-target blockade or synergistic mechanisms. Studies have shown that combining trastuzumab with conventional chemotherapeutic agents such as paclitaxel, carboplatin, and cisplatin substantially improves overall treatment outcomes in HER2-positive breast cancer.46,50,80-82 In the GeparQuattro trial, the addition of trastuzumab to the standard chemotherapy regimen significantly improved the pCR rate, and the toxicity remained within an acceptable range. 58 Jamshed Ali and colleagues also confirmed that in patients receiving neoadjuvant therapy, the combination of trastuzumab with 4 cycles of dose-dense AC and 12 cycles of paclitaxel significantly increased the pCR rate. 80 This suggests that the combination of trastuzumab with standard chemotherapy regimens could become a standard neoadjuvant treatment or foundation for HER2-positive breast cancer.
However, data from the GeparQuattro and NOAH trials indicate that even though the addition of trastuzumab to standard chemotherapy significantly improved the pathological complete response rate, the overall pCR rate remained below 50%. This limitation prompted researchers to further explore more effective treatment strategies. By incorporating pertuzumab into the trastuzumab-plus-chemotherapy backbone, a “dual-targeted” approach was established that synergistically blocks distinct extracellular domains of the HER2 receptor from multiple epitopes, thereby further improving patient prognosis and quality of life. Ulrike Nitz and colleagues confirmed the feasibility of this approach—combining the dual-targeted regimen with paclitaxel significantly improved pCR and invasive disease-free survival, with the experimental group achieving a pCR rate as high as 98%. In addition, the addition of small-molecule inhibitors such as lapatinib and pyrotinib has greatly improved pCR rates in patients with early or locally advanced HER2-positive breast cancer, particularly in those with hormone receptor-negative disease. In the TRIO-US B07 trial, the combination of trastuzumab and lapatinib resulted in a higher pCR rate compared with monotherapy, along with improved recurrence-free survival and overall survival upon follow-up, especially in patients with hormone receptor-negative breast cancer. These findings suggest a synergistic effect between trastuzumab and small-molecule inhibitors in improving survival outcomes. Notably, across existing clinical trials evaluating the addition of small-molecule tyrosine kinase inhibitors to trastuzumab-based standard chemotherapy regimens, the enrolled patient populations have almost exclusively consisted of individuals with early or locally advanced breast cancer, whereas data from patients with advanced, metastatic, or refractory disease remain extremely limited. This has constrained the application of small-molecule inhibitors in the neoadjuvant setting for HER2-positive breast cancer.
However, the improvement in efficacy has not fully resolved the issue of drug toxicity, and adverse reactions associated with neoadjuvant therapy remain a critical aspect that cannot be overlooked in clinical research. In standard chemotherapy regimens, the use of anthracyclines is associated with severe hematologic toxicities, including leukopenia, anemia, and thrombocytopenia, while the addition of platinum agents may lead to febrile neutropenia. These adverse events significantly affect patient adherence to treatment. In addition, the incorporation of small-molecule inhibitors, while improving pCR rates, has also been linked to an increased incidence of gastrointestinal adverse reactions. For instance, in the ExteNET trial, the combination of trastuzumab and neratinib resulted in transient diarrhea. These findings highlight the necessity of early prevention and symptomatic management of potential treatment-related adverse events in clinical practice.
In parallel with efforts to prevent and manage adverse events, a key area of clinical research involves developing individualized treatment strategies by carefully assessing whether and how to incorporate chemotherapy into neoadjuvant regimens based on patient-specific factors such as tumor burden, molecular subtype, immune microenvironment characteristics, and underlying comorbidities. For certain patients with low-risk profiles or poor tolerance to chemotherapy, appropriately de-escalating chemotherapy or adopting chemotherapy-free dual-targeted or targeted combination regimens may effectively avoid the majority of chemotherapy-related toxicities while maintaining therapeutic efficacy, thereby achieving an optimal balance of “low toxicity, high efficacy.”
The PerELISA study pioneered the use of a “Ki67 dynamic response plus PAM50 subtype” dual-dimensional screening model, demonstrating that patients in specific subgroups—such as those with HER2-enriched tumors who also exhibited Ki67 response—could achieve favorable pCR rates with a letrozole-based dual-targeted regimen without chemotherapy. Another study further showed that early 18F-FDG-PET assessment could effectively identify patients suitable for trastuzumab plus pertuzumab dual-targeted therapy without chemotherapy, offering another promising approach toward the goal of “low toxicity, high efficacy.”
Additionally, trastuzumab is associated with cardiac adverse reactions, which may impact long-term efficacy. The ALTTO trial showed that when trastuzumab is combined with anthracyclines, the risk of cardiac events is higher. Therefore, during treatment, clinicians must closely monitor cardiac function, blood parameters, and other patient conditions and adjust drug dosages and treatment regimens as necessary. Future research should focus on optimizing drug dosages and administration sequences, aiming for a “low-toxicity, high-efficacy” approach to achieve higher pCR rates and better prognosis and survival quality.
Meanwhile, the research on new targeted therapies remains a hot topic. ADCs, such as T-DM1, theoretically target HER2-positive cancer cells specifically while delivering cytotoxic drugs, offering precise treatment and minimizing the side effects of traditional chemotherapy. However, current trials have demonstrated mixed results regarding their actual efficacy. In the JBCRG20 study, the combination of pertuzumab and T-DM1 appeared to yield a higher pCR rate, 54 whereas in the KRISTINE trial, the T-DM1 arm showed a lower pCR rate compared with the standard regimen. Additionally, the SHAMROCK II trial suggests that another next-generation ADC, T-DXd, may show promising efficacy in metastatic and refractory cases, holding potential as an alternative treatment option for patients intolerant to neoadjuvant chemotherapy, though it is also associated with an increased incidence of interstitial lung disease. 60
However, the introduction of immunotherapy has not been as promising as expected. The IMpassion050 trial did not show an improvement in pCR rates with atezolizumab, and the efficacy in the PD-L1-positive subgroup even decreased, suggesting that HER2-positive breast cancer may not be an “immunotherapy-benefiting tumor”. 72 This highlights key gaps in current immunotherapy combination strategies, such as inaccurate biomarker selection and unclear mechanisms of action, which are worth close attention. Research has shown that immune features such as TILs and PD-L1 expression are closely associated with treatment response and can serve as key indicators for predicting the efficacy of chemotherapy-free anti-HER2 therapies in HER2-positive breast cancer patients. Moreover, the HER2-enriched feature is associated with a higher pCR rate in patients treated with trastuzumab. Edward Seung and colleagues introduced a novel targeting model that uses a tri-specific antibody targeting HER2, CD3, and CD28. This antibody can activate effector T cells, particularly CD4 cells, thereby inhibiting the growth of HER2-positive breast cancer. This approach provides a promising treatment option for HER2-positive breast cancer patients, especially those with limited response to traditional HER2-targeted therapies.
In conclusion, significant progress has been made in neoadjuvant treatment for HER2-positive breast cancer over the past five years, especially with the adoption of “dual-target” therapies that target HER2 through two mechanisms, significantly increasing pCR rates. The application of platinum agents, TKIs, and other small molecule drugs has further increased pCR rates and long-term survival, highlighting the potential of these therapies. Neoadjuvant treatment for HER2-positive breast cancer is moving toward “precision, high efficacy, and low-toxicity” approaches. Accordingly, future research can be pursued along the following directions: first, to establish stratified treatment pathways based on molecular subtyping and immune microenvironment characteristics, enabling truly individualized therapy; second, to further investigate the value of next-generation antibody-drug conjugates in chemotherapy-free strategies and to optimize their combination with traditional targeted agents and immunotherapies; and third, to integrate real-world data with clinical trial evidence to develop multidimensional efficacy-toxicity-cost assessment models that support evidence-based clinical decision-making. Through continued mechanistic innovation and clinical translation, it is expected that further improvements in pathological complete response rates will be achieved alongside meaningful gains in both quality of life and long-term survival for patients.
Footnotes
Author Contributions
Anqi Shang: Data Curation, Writing - Original Draft; Yunjing Xiong: Conceptualization, Investigation, Writing - Original Draft; Min Li: Writing-original draft, Methodology; Wanxin Tang: Resources, Supervision; Keyu Wan: Visualization, Investigation; Zekang Deng: Data Curation, Resources; Xianxian Mao: Visualization, Methodology; Haizhu Chen
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grant 82403165 from the National Natural Science Foundation of China, grant 2023A1515110952 from the Guangdong Basic and Applied Basic Research Foundation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Artificial Intelligence Statement
The authors confirm that no generative artificial intelligence (AI) tools were used in the writing, analysis, or preparation of this manuscript.
