Abstract
Keywords
Introduction
Neoadjuvant therapy has emerged as a standard-of-care for patients with HER-2 positive breast cancer, or for patients with and without pathologically proven clinically node-positive at diagnosis (cN(+)). 1 Total pathological complete response (total pCR), defined as complete eradication of invasive cancer in the breast and axillary lymph nodes (ypT0/is ypN0) after neoadjuvant therapy, was strongly associated with higher overall survival (OS) and event-free survival (EFS), particularly in HER-2 positive patients.2,3 Total pCR (ypT0/is ypN0) was related to a better prognostic outcome of OS and EFS,4,5 compared to the eradication of invasive cancer in the breast alone (ypT0/is).
As for cN(+) patients, eradication of tumors from axillary lymph nodes, known as axillary pCR (ypN0), was of high prognostic value,6,7 and even had a greater effect on EFS than breast pCR (ypT0/is). 8 Moreover, axillary downstaging (cN(+) → ypN0) may also allow the omission of conventional axillary lymph node dissection (ALND) after neoadjuvant therapy, enabling less invasive axillary staging procedures to minimize the risk of surgical complications.9,10 Previous studies showed that sentinel lymph node biopsy (SLNB) was technically feasible as an alternative to ALND in cN(+) patients after neoadjuvant therapy 11 and that the false-negative rate of SLNB was the lowest among HER-2 positive subtype. 12
In conclusion, the achievement of total pCR (ypT0/is ypN0) and the conversion of cN(+) to ypN0 after neoadjuvant therapy are important prognostic factors and may influence de-escalation surgical strategies. Interestingly, a recent meta-analysis found that the HER-2 positive subtype was associated with the highest axillary pCR rate in cN(+) patients compared to other breast cancer subtypes, 13 implying that the HER-2 positive subtype should be considered in the first place. Therefore, achieving ypN0 and total-pCR in HER-2 positive cN(+) patients were of great significance.
Neoadjuvant therapy for HER-2 breast cancer has recently been fully explored, and plenty of meta-analyses have already reached a consensus on therapeutic efficacy. The addition of trastuzumab to conventional chemotherapies can significantly increase pCR rate, 14 and considerable pCR benefit has been achieved with dual-target therapy (lapatinib/pertuzumab plus trastuzumab) compared with single-target therapy (lapatinib/trastuzumab).15–17 In addition, a recent network meta-analysis strongly recommended trastuzumab plus pertuzumab with carboplatin-contained chemotherapy, which was ranked first by the surface under the cumulative ranking curve (SUCRA) analysis in terms of pCR rate. 18 Novel regimens such as trastuzumab emtansine (T-DM1)19–21 and trastuzumab biosimilar (bio-H),22,23 were frequently enrolled in clinical trials and presented discrepant outcomes between varied therapy regimens. An up-to-date network meta-analysis confirmed that T-DM1 combined with lapatinib ranked top in achieving total pCR, compared with conventional single and dual-target therapy. 24
As for lymph node conversion, a randomized phase III trial (PEONY) indicated that adding pertuzumab to trastuzumab significantly improved axillary pCR rate versus trastuzumab alone in patients with cN(+) confirmed by aspiration prior to neoadjuvant therapy. 25 Another retrospective controlled trial comparing neoadjuvant chemotherapy alone, single-target, and dual-target showed a prominent effect on lymph node downstaging in single or dual-target therapy. 26 However, the neoadjuvant regimen of lymph node conversion in HER-2 positive breast cancer has not been well-researched, and no meta-analysis has been conducted; furthermore, studies of the efficiency of neoadjuvant therapy considering clinical lymph node status were also limited. Among the various anti-HER2 targeted agents listed above, which should be the priority for patients with clinically node-positive at diagnosis? Which chemotherapeutic agent is more effective, carboplatin or anthracycline? Does the rank of regimen efficiency on total-pCR differ between cN(+) and cN(−) patients? Whether the pre-adjuvant nodal status would influence the choice of anti-HER2 agents? Although there was a previous meta-analysis focused on the impact of clinical lymph node status on pCR, 27 their study only involved a small sample size of 4 RCT trials and used a non-uniform definition of pCR, which made their conclusion unreliable. Therefore, this paper aimed to provide an overall review of neoadjuvant regimens for lymph node downstaging and to indirectly compare the total-pCR by various neoadjuvant regimens with network meta-analysis in HER2-positive patients according to their clinical lymph node status.
Materials and Methods
Literature Searching Strategies
Literature searching was conducted comprehensively and systematically in electronic databases of PubMed, Cochrane, Embase, Scopus, and Web of Science. Specific search strings for PubMed were listed as examples (Supplemental File 1). References to the Chinese Society of Clinical Oncology (CSCO) Breast Cancer Guidelines 2022 28 related to HER-2 positive breast cancer neoadjuvant therapy were also carefully reviewed.
Selection Criteria
Inclusion Criteria
Participants: Patients with HER2-positive breast cancer, diagnosed as clinically node-positive (cN(+)) via palpation or medical imaging, with or without pathological evidence.
Interventions and comparisons: Different neoadjuvant therapies were employed in 2 or more treatment arms.
Outcomes: Axillary pCR(ypN0) or total pCR(ypT0/is ypN0).
Study type: Phase II/III randomized controlled trials or case-control studies.
Exclusion Criteria
Case reports, systematic reviews, abstracts, comments, animal trials, and single-arm studies.
Incomplete or missing data of pCR rates for cN(+) patients.
Publications derived from the same clinical trial.
Definition of Outcomes
Axillary pCR was defined as the absence of residual invasive disease in the axilla by pathological examination (ypN0), and total pCR was defined as the elimination of invasive cancer cells in both breast and axilla (ypT0/is ypN0). Articles that provided outcomes different from our above definition, such as breast pCR (ypT0/is) and strict pCR (ypT0 ypN0), were excluded.
Literature Screening and Data Extraction
Relevant articles were sorted using Endnote X9 software and duplicates were excluded. Two investigators independently screened the full text in accordance with the inclusion and exclusion criteria, and any disagreements were discussed with a third investigator. Data were extracted from the included studies as follows: title, first author's name, trial phase, publication date, details of intervention (including cycle, drug, and duration), the definition of lymph node status, the number of cN(+) and cN(−) patients enrolled, and outcomes (axillary pCR and total pCR). The result of data extraction was rechecked by a third investigator.
Risk of Bias Assessment
Two evaluators independently performed a quality assessment for each included RCT trial from 7 perspectives and 3 levels using Review Manager 5.3 software, with disagreements resolved by discussing with a third investigator. Visualization of the assessment results was conducted through Review Manager 5.3. As for case-control studies, the Newcastle–Ottawa Quality Assessment Scale (NOS) was used to evaluate the quality of the included controlled trials.
Statistical Analysis
Network Meta-Analysis
In order to integrate direct and indirect comparisons of various neoadjuvant therapies for HER2-positive breast cancer, a network meta-analysis was conducted using STATA 15.0 software. Network evidence plots, funnel plots, forest plots, and the SUCRA were completed successively. The overall ranking of the regimen was estimated by SUCRA for each method, with higher SUCRA values being associated with higher pCR rates. The funnel plot was used to evaluate the publication bias of the involved literature.
Traditional Meta-Analysis
However, data extracted from articles that belonged to cN(+) patients and with axillary pCR as outcomes were insufficient to form a network meta-analysis, thus the Review Manager 5.3 software was employed to perform a traditional meta-analysis. The relative risk (RR) and 95% CI acted as effect size indicators for the dichotomous variables. The heterogeneity test was mainly determined by I2 and the random-effects model was employed.
Our study was reported according to the relevant Equator network guideline (https://www.equator-network.org/reporting-guidelines/prisma/), named ‘The PRISMA 2020 statement: an updated guideline for reporting systematic reviews’. 29 After preliminary searches, the protocol was registered with PROSPERO (registration number: CRD42022365517).
Results
Study Selection
A total of 1508 potentially relevant literature were presented by the initial search. After the layer-by-layer screening, 17 trials were considered suitable for the meta-analysis. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flowchart outlines the study selection procedure, see Figure 1.

Flowchart describing the process of literature filtering.
Characteristics of Involved Literature
Finally, our study included 11 Phase II/III randomized controlled trials and 6 double-arm retrospective trials published between 2010 and 2021, involving 37 treatment arms, with a total of 4747 patients (Supplemental File 2).25,26,30–44 The definition of axillary lymph node status before neoadjuvant therapy varied between trials, with 12 trials using clinical node status detected by ultrasound and radiography, and 5 trials using pathological node status confirmed by the aspiration/biopsy, while both definitions were allowed in our study. Besides, 2 trials had patients with clinically negative lymph node status that underwent sentinel node biopsy before the start of neoadjuvant therapy, which had no impact on our study samples.
Three trials evaluated the efficacy of lapatinib in neoadjuvant therapy, 2 trials evaluated the efficacy of trastuzumab biosimilars, and another 1 focused on T-DM1.
Thirteen trials involving 2662 cN(+) patients had an outcome of total pCR, and 7 trials involving 2054 cN(+) patients had an outcome of axillary pCR. As for total pCR, 804 cN(−) patients were available in 8 of the 13 trials, which enabled further analysis of different pre-neoadjuvant lymph node statuses.
Bias and Quality Assessment
As for RCT trials (n = 11), most of the literature described the method of randomization, such as random permuted blocks and biased-coin algorithm, with only 3 studies not mentioning their specific methods. As a few of the trials (3/11) adopted double-blind designs, performance bias that did not affect the outcomes might exist. Three out of 8 open-label trials declared that pathological assessment was masked to keep detection bias to a minimum. For incomplete result data, 4 pieces of literature had unclear dropout cases, which may affect the true results. In particular, the remaining 38 pieces of literature had no missing outcome data. The risk of bias assessment of the involved literature is illustrated in Figure 2.

Risk of bias for all included RCTs (n = 11).
As for retrospective controlled studies (n = 6), the quality of the literature can support the meta-analysis, as all studies had NOS scores of 5 or more. The NOS score for included controlled studies is illustrated in Table 1.
The Newcastle–Ottawa Quality Assessment Scale for Included Controlled Studies (n = 6).

Axillary pCR in cN(+) Patients of Direct Comparisons
Seven trials25,26,32,34,36,38,39 presented lymph node conversion for patients diagnosed with positive lymph nodes who underwent neoadjuvant therapy and reached axillary pCR (pN0). Four studies directly compared chemotherapy alone with single-target therapy, and 3 studies directly compared single-target therapy with dual-target therapy. However, 1 of 7 trials 32 include 2 arms that were both single-target therapy and were therefore excluded from our meta-analysis.
After pooled analysis, single-target therapy was significantly better than chemotherapy alone; no significant difference was found between single-target therapy and dual-target therapy. These results are shown in Figure 3.

Direct comparisons of axillary pCR in cN(+) group. (a) Chemotherapy alone versus single-target therapy. (b) Single-target therapy versus dual-target therapy.
Total pCR in cN(+) Patients in Network Meta-Analysis
Thirteen articles25,26,30–33,35,37,40–44 reported total pCR in pre-neoadjuvant lymph node-positive patients, involving 10 RCT trials. The dot size represents the sample size using the intervention, and the line thickness indicates the number of trials comparing the 2-point treatment intervention. Figure 4(a) depicts the network of total pCR for cN(+) patients in 10 regimen groups. The funnel plot in Figure 4(b) indicates publication bias, revealing that most of the scatter points are located on both sides of the vertical line.
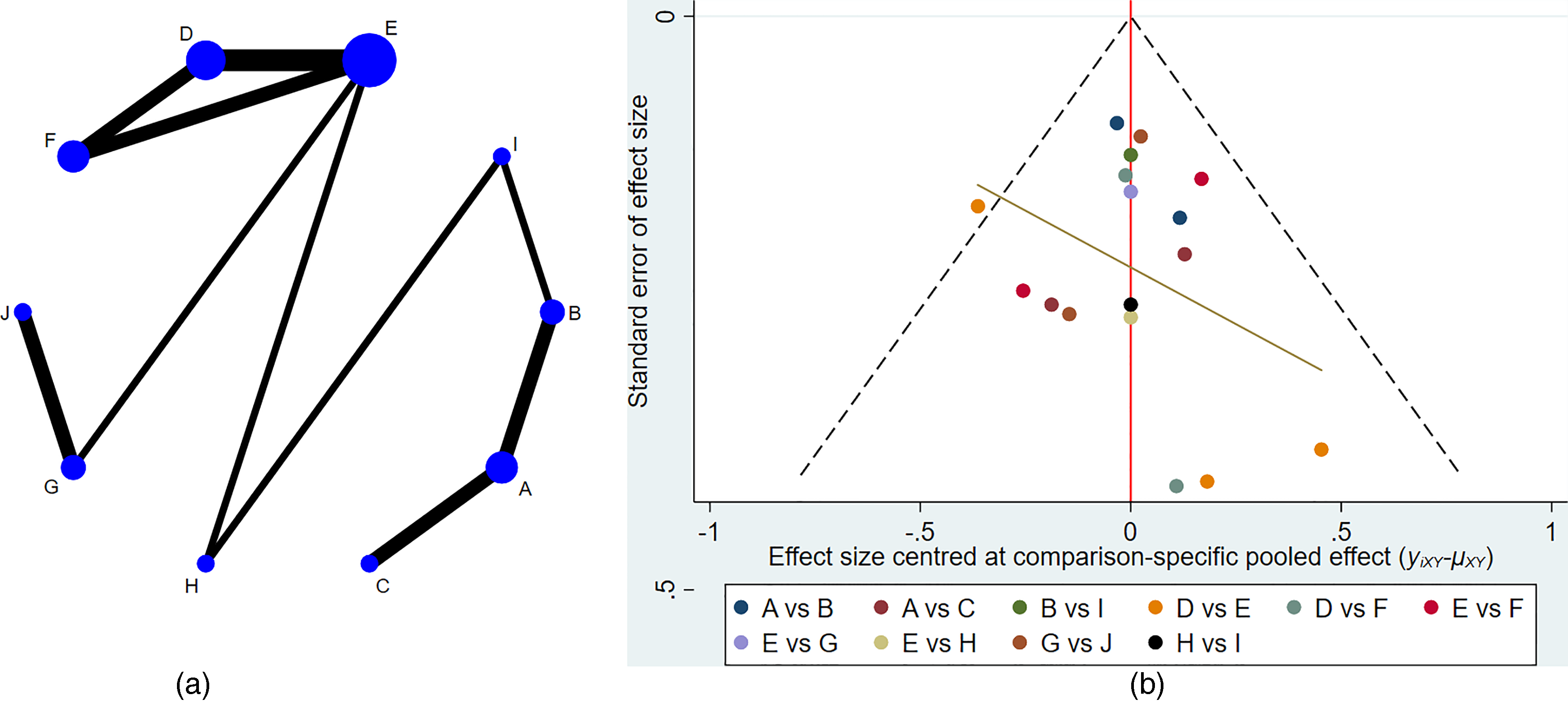
Total pCR in cN(+) patients of network meta-analysis. (a) Network of total pCR in 10 regimen groups. (b) Funnel plot. Regimen groups: A: chemotherapy (without carboplatin or with anthracycline) + trastuzumab + pertuzumab, B: chemotherapy (with carboplatin) + trastuzumab + pertuzumab, C: T-DM1 alone or chemotherapy (paclitaxel alone) + trastuzumab, D: chemotherapy (with anthracycline) followed by lapatinib, E: chemotherapy (with anthracycline) followed by trastuzumab, F: chemotherapy + trastuzumab + lapatinib, G: chemotherapy (with anthracycline) concurrent with trastuzumab, H: chemotherapy (with carboplatin) + trastuzumab, I: chemotherapy alone, J; chemotherapy (with anthracycline) concurrent with bio-trastuzumab.
Table 2 shows network comparisons for the 10 regimen groups, presenting a total of 45 pairwise comparisons, 17 of which are statistically significant. The rank of SUCRA in 10 regimen groups is shown in Figure 5. Forest plots and the results of SUCRA can be seen in Supplemental File 3.

Total pCR rates ranking for 10 regimen groups in cN(+) patients. Regimen groups: Cb + HP: chemotherapy (with carboplatin) + trastuzumab + pertuzumab, (without Cb/with A) + HP: chemotherapy (without carboplatin or with anthracycline) + trastuzumab + pertuzumab, Cb + H: chemotherapy (with carboplatin) + trastuzumab, H + Lap: chemotherapy + trastuzumab + lapatinib, T-DM1/T + H: T-DM1 alone or chemotherapy (paclitaxel alone) + trastuzumab, A concurrent with bio-H: chemotherapy (with anthracycline) concurrent with bio-trastuzumab, A followed by H: chemotherapy (with anthracycline) followed by trastuzumab, A concurrent with H: chemotherapy (with anthracycline) concurrent with trastuzumab, chemotherapy alone: chemotherapy alone, A followed by Lap: chemotherapy (with anthracycline) followed by lapatinib.
Network-Comparison RRs with 95% CIs Among 10 Regimen Groups of Total-pCR in cN(+) Patients.

Values in bold represent statistically significant differences.
Regimen groups: Cb + HP: chemotherapy (with carboplatin) + trastuzumab + pertuzumab, (without Cb/with A) + HP: chemotherapy (without carboplatin or with anthracycline) + trastuzumab + pertuzumab, Cb + H: chemotherapy (with carboplatin) + trastuzumab, H + Lap: chemotherapy + trastuzumab + lapatinib, T-DM1/T + H: T-DM1 alone or chemotherapy (paclitaxel alone) + trastuzumab, A concurrent with bio-H: chemotherapy (with anthracycline) concurrent with bio-trastuzumab, A followed by H: chemotherapy (with anthracycline) followed by trastuzumab, A concurrent with H: chemotherapy (with anthracycline) concurrent with trastuzumab, chemotherapy alone: chemotherapy alone, A followed by Lap: chemotherapy (with anthracycline) followed by lapatinib.
Chemotherapy combined with trastuzumab and pertuzumab (one with carboplatin and the other with anthracycline) were ranked as the top two by the SUCRA analysis. Trastuzumab in combination with pertuzumab was significantly better than trastuzumab or bio-trastuzumab or T-DM1 alone [(without Cb/with A) + HP versus A followed by H 4.38 (1.51,12.71), A concurrent with bio-H 2.41 (1.81,3.21), T-DM1/T + H 1.58 (1.16,2.17)]. Trastuzumab in combination with lapatinib was significantly better than trastuzumab or lapatinib alone [H + Lap versus A followed by H 1.36 (1.08,1.73), A followed by Lap 2.95 (1.35,6.41)]. Dual-target therapy that combines H and P was superior to the combination of H and lapatinib according to the SUCRA rankings. The time of trastuzumab employed did not show a statistically significant impact on total pCR [A followed by H versus A concurrent with H, 1.06 (0.79,1.43)]. However, lapatinib performed poorly in cN(+) patients, as chemotherapy (with anthracycline) followed by lapatinib ranked poorly, and was significantly inferior to chemotherapy (with anthracycline) followed by trastuzumab [A followed by Lap versus A followed by H, 0.46 (0.22,0.97)].
Regarding chemotherapy agents, carboplatin performed significantly better than anthracycline combined with single-target therapy [Cb + H versus A followed by H 1.76 (1.05,2.94), A concurrent with H 1.87 (1.03,3.39)]. The use of anthracycline lowered the SUCRA of total pCR, indicating that the addition of anthracycline to chemotherapy might not improve the outcome.
Total pCR in cN(−) Patients in Network Meta-Analysis
Eight RCT trials25,30–33,35,41,44 reported total pCR in pre-neoadjuvant lymph node-negative patients. However, 2 arms of 1 trial 44 failed to join our regimen groups, and therefore the trial was excluded from our network analysis. The network of total pCR in 5 regimen groups for cN(−) patients is described in Figure 6(a). The funnel plot in Figure 6(b) indicates the publication bias.

Total pCR in cN(−) patients of network meta-analysis. (a) Network of total pCR in five regimen groups. (b) Funnel plot. Regimen groups: A: chemotherapy (paclitaxel alone) + trastuzumab + pertuzumab, B: chemotherapy + trastuzumab or T-DM1 alone, C: chemotherapy (with anthracycline) followed by lapatinib, D: chemotherapy (with anthracycline) followed by trastuzumab, E: chemotherapy + trastuzumab + lapatinib.
The network comparisons of 5 regimen groups are shown in Table 3, presenting a total of 10 pairwise comparisons without statistically significant. The ranking of SUCRA in 5 regimen groups is shown in Figure 7. The forest plot and the results of SUCRA can be seen in Supplemental File 4.

Total pCR rates ranking for five regimen groups in cN(−) patients. Regimen groups: H + Lap: chemotherapy + trastuzumab + lapatinib, A followed by H: chemotherapy (with anthracycline) followed by trastuzumab, T/A/Cb concurrent with H or T-DM1: chemotherapy + trastuzumab or T-DM1 alone, T + HP: chemotherapy (paclitaxel alone) + trastuzumab + pertuzumab, A followed by Lap: chemotherapy (with anthracycline) followed by lapatinib.
Network-Comparison RRs with 95% CIs among 5 Regimen Groups of Total-pCR in cN(−) Patients.

Regimen groups: H + Lap: chemotherapy + trastuzumab + lapatinib, A followed by H: chemotherapy (with anthracycline) followed by trastuzumab, T/A/Cb concurrent with H or T-DM1: chemotherapy + trastuzumab or T-DM1 alone, T + HP: chemotherapy (paclitaxel alone) + trastuzumab + pertuzumab, A followed by Lap: chemotherapy (with anthracycline) followed by lapatinib.
Trastuzumab in combination with lapatinib was ranked at the top by the SUCRA analysis. In the cN(−) group, the duration of trastuzumab employment and the addition of anthracycline or not had no significance on total pCR. Same as the cN(+) group, the worst outcome was seen with chemotherapy (with anthracycline) followed by lapatinib. However, trastuzumab or T-DM1 alone performed better than chemotherapy (paclitaxel alone) + trastuzumab + pertuzumab, in contrast to the primacy of pertuzumab in cN(+) groups.
Discussion
With the upgrading of neoadjuvant therapy for HER-2 positive breast cancer, various chemotherapeutic and targeted agents were recommended due to their efficacy in breast pCR or total pCR. However, axillary pCR and axillary lymph node conversion (cN(+) → ypN0) were always forgotten by researchers. Although comprehensive studies included axillary pCR as the primary endpoint, they analyzed the impact of breast pCR, pretreatment node status, 45 and molecular subtype 13 on axillary pCR, ignoring the effect of the neoadjuvant regimen. We confirmed the advantages of single-target therapy, while there was no significant difference between single-target therapy and dual-target therapy.
The lack of significance may due to the variety of chemotherapeutic agents, as Cha 26 and Murthy 39 failed to unify chemotherapy strategy between their single and dual-target therapy groups. In addition, there were clinical trials focusing on the selection of chemotherapy. Buzdar 32 found that fluorouracil, epirubicin, and cyclophosphamide (FEC-75) followed by paclitaxel plus trastuzumab had a significantly higher lymph node downstaging rate compared with paclitaxel plus trastuzumab followed by FEC-75 plus trastuzumab, whereas Dominici 6 found no significant difference in the rate of axillary conversion between patients who received trastuzumab concurrently and no anthracycline. Moreover, hormone receptor (HR) status may influence the rate of axillary downstaging, as confirmed in 2 retrospective studies. One study suggested that the axillary downstaging rate was higher in HR(−) patients compared with HR(+) patients in TCbH regimen, 46 and another study employed the TCbHP regimen and came to the same conclusion that HR(−) patients were more likely to achieve lymph node conversion. 47 Therefore, besides targeted therapy, which was confirmed in our study, the chemotherapy regimen still needs further exploration in axillary downstaging, and HR status should be taken into consideration.
HER-2 positive breast cancer with clinically lymph node-positive at diagnosis was considered a high-risk disease pattern. Our study confirmed that the total pCR rate was significantly lower in the cN(+) patients than in the cN(−) patients by paired t-test (t = −2.7415, p = 0.0139), consistent with the findings of the previous meta-analysis of HER-2 positive breast cancer 27 and multivariate analysis that did not consider molecular subtype. 48 Therefore, our study is the first to employ network meta-analysis on cross-comparing various neoadjuvant regimens and successfully put forward the optimal effective therapy for cN(+) HER-2 positive patients.
Similar to other studies focusing on HER-2 positive patients regardless of clinical axillary status, our study also proved that dual-target therapy had its advantages. 16 Moreover, we found that in cN(+) patients, dual-target therapy that combines H and P is superior to the combination of H and lapatinib according to the SUCRA ranking, which is also consistent with the overall HER-2 positive population in the previous studies. 49 Notably, our single-target therapy in cN(+) patients differed from the overall HER-2 positive population in the previous meta-analysis in several ways. Firstly, we found that the timing of trastuzumab employment showed no statistically significant impact on total pCR [A followed by H versus A concurrent with H], whereas Wu claimed that pCR rate was significantly higher in the group receiving the concurrent use of trastuzumab and anthracycline-based neoadjuvant chemotherapy than in the non-concurrent group. 50 Secondly, we noted that T-DM1 alone ranked higher than anthracycline-based chemotherapy followed by or concurrent with trastuzumab in cN(+) patients, yet the SUCRA ranking of pCR between T-DM1 and anthracycline + trastuzumab in overall HER-2 positive population remained controversial.18,24 These 2 novel conclusions suggested that T-DM1 may be given priority to neoadjuvant therapy in cN(+) patients when dual-target was not possible. In contrast, for the anthracycline-based chemotherapy combined with trastuzumab, the timing of trastuzumab employment requires no consideration.
It is important to further explain that in the network analysis of total-pCR in cN(+) patients, our study chunked T-DM1 alone and chemotherapy (paclitaxel alone) + trastuzumab into 1 regimen group; while theoretically, the antibody-drug conjugate may increase the drug concentration in tumor cells, resulting in superior efficacy. ‘KAITLIN’, a phase III study, demonstrated that there was no significant difference in invasive disease-free survival (IDFS) between T-DM1 + pertuzumab and taxane + trastuzumab + pertuzumab as adjuvant therapy for cN(+) patients. 51 Therefore, it is feasible to consider T-DM1 alone and chemotherapy (paclitaxel alone) + trastuzumab as a unified regimen when analyzing cN(+) patients.
The selection of a high-efficiency chemotherapy regimen is particularly important in some regions where dual-target drugs are not available and only single-target regimens are available. In this case, our study confirmed that a carboplatin-based regimen can result in significantly better total pCR than an anthracycline-based regimen [Cb + H versus A followed by H, Cb + H versus A concurrent with H]. According to SUCRA values, the chemotherapy (with carboplatin) + trastuzumab regimen group ranked second only to dual-target therapy of trastuzumab and pertuzumab, and even higher than dual-target therapy with trastuzumab and lapatinib, implying that carboplatin-based chemotherapy may make up for the deficiency of targeted therapy to a certain extent. The favor of carboplatin and the deprecated of anthracycline were also evident in neoCARH, a phase II RCT trial directly comparing carboplatin-containing versus anthracycline-containing neoadjuvant chemotherapy in combination with trastuzumab for cN(+) patients. 33 And the TRAIN-2 trial (phase III RCT trial) demonstrated that the addition of anthracycline to the TCbHP regimen may bring down the total pCR rate in cN(+) patients. 44
Interestingly, the SUCRA ranking of cN(−) patients was different from that of cN(+) patients. Trastuzumab combined with lapatinib was ranked at the top, and trastuzumab or T-DM1 alone even performed slightly better than trastuzumab combined with pertuzumab, contrary to pertuzumab's first place in cN(+) groups. By comparing the results of SUCRA analysis between cN(+) and cN(−) patients, our study suggested that the selection of the efficient neoadjuvant regimen depends on the clinical lymph node status at diagnosis. When using agents in combination with trastuzumab, cN(+) patients are more likely to benefit from pertuzumab treatment, while cN(−) patients are in favor of lapatinib. To our knowledge, only 1 previous meta-analysis investigated the impact of clinically nodal status on pCR in HER2-positive breast cancer 27 ; however, they concluded that cN(+) patients favored lapatinib treatment, whereas cN(−) patients benefited more from pertuzumab treatment, which was the exact opposite of our findings. The reason are as follows: (1) Twice as many trials were included in our study; (2) The previous study did not have a uniform definition of pCR (ypT0/is ypN0 or ypT0/is or ypT0 ypN0), but our study strictly restricted the definition of total pCR (ypT0/is ypN0), excluding trials using other definitions as outcomes. (3) In previous studies, there was large heterogeneity in the included trials that could not be fully explained. Moreover, we used network meta-analysis instead of traditional meta-analysis. Network meta-analysis could conduct a more detailed grouping, and perform an overall ranking via pairwise comparison. Therefore, our study is more reliable and may be endowed with a clinical reference value.
Nowadays, personalized medicine has been proposed, and it is of great importance to select the right medicine for each individual. To achieve precision medicine in breast cancer neoadjuvant therapy, plenty of clinical drug trials incorporated subgroup analysis to study the impact of clinicopathological data on treatment efficiency, including HR status, age, tumor grade, tumor size, node status, clinical stage, etc Up to now, only HR status has already been adequately analyzed and has been able to guide the selection of neoadjuvant regimen in HER-2 positive breast cancer.18,27,52 Compared to other previously published meta-analyses, our study has included the maximum number of clinical trials and involved a sufficiently large sample size of HER-2 positive breast cancer with the specific pre-neoadjuvant axillary status. The quantitative and visual ranking of the various regimens was performed using a network meta-analysis. Our study was able to offer a proposal for the appropriate neoadjuvant regimen according to lymph node status.
However, there are still some limitations to our study. Firstly, due to the limited number of eligible clinical trials, lapatinib was the only 1 that represented tyrosine kinase inhibitors (TKIs), pyrotinib, and neratinib were unable to be involved in our study. Here we would like to introduce the molecular cross talk between key molecular players in HER2-positive breast cancer microenvironment with respect to administration of TKIs. HER2 is a transmembrane glycoprotein with tyrosine kinase activity in its endo-domain. In cancer microenvironment, extracellular ligands bind to other receptor tyrosine kinases (RTKs), such as EGFR, HER3, or HER4, resulting in HER2 homodimerization or heterodimerization with another member of RTK's family. The dimerization catalyzes the transfer of phosphate groups from ATP to tyrosine residues on protein substrates. 53 Then the downstream signal was activated, including PI3K/PIP3/AKT/mTOR signal pathway, RAS/RAF/MEK/MAPK signal pathway, and JAK/STAT3 signal pathway. 54 Excessive activation of the above signaling pathways may facilitate cell survival, proliferation, differentiation, motility, invasion, migration, adhesion, and angiogenesis. 55 Lapatinib has a homologous structure with ATP and can competitively bind to the intracellular ATP binding domain of both EGFR and HER2, thus inhibiting tyrosine kinase phosphorylation and blocking downstream signaling. 56 Analogously, neratinib binds and inhibits EGFR, HER2 and HER4, as do its active metabolites (M3, M6, M7, M11). 57 However, pyrotinib and neratinib, as irreversible TKIs, were mechanistically more effective than the reversible TKI lapatinib and performed better in clinical trials. 58 Clinical trials showed that pyrotinib plus trastuzumab achieved total pCR up to 75% in cN(+) patients, 59 while cN(+) patients are more likely to benefit from neratinib therapy to achieve 5-year survival. 60 And these results suggested that pyrotinib and neratinib should be taken into consideration in future analyses of cN(+) HER-2 positive.
Secondly, our study stopped at the primary outcome of complete pathological response due to limited data. As for the prognostic outcome, Asaoka 61 and Xie 62 reached a consensus that cN(+) was found to be the most significant predictor of cancer recurrence in patients with acquired breast pCR. Besides, cN(+) and HER2-positive subtype were 2 clinicopathological prognostic factors that were significantly associated with unfavorable outcomes in terms of DFS. 61 Therefore, there is still much room for improvement in the post-surgery adjuvant therapy of cN(+) HER-2 positive breast cancer to reach better prognostic outcomes.
Nowadays, immunotherapy is extensively regarded as an important part of adjuvant therapy for triple-negative breast cancer. Recently, more and more investigators broke the constraint of cancer subtype, and the latest research applied programmed cell death protein 1 (PD-1) inhibitor to HER-2 positive breast cancer, despite the results remaining controversial. In the neoadjuvant therapy setting, IMpassion050 Trial randomly assigned patients with cN(+) 1:1 to atezolizumab/placebo with dual-target therapy, and the results showed that the addition of atezolizumab did not increase pCR rates versus placebo in the intension-to-treat or PD-L1 positive populations. 63 However, Neo-PATH, a multi-center, single-arm phase 2 study, applying atezolizumab, and docetaxel with dual-target therapy, confirmed that the pCR rate was higher in patients with positive PD-L1 expression at baseline. 64 In the adjuvant therapy setting, the addition of PD-1 inhibitor showed a possible survival advantage in patients with PD-L1 expression, but no improvement in progression-free survival in the overall trastuzumab-resistant, advanced, HER-2 positive populations.65,66 In general, immunotherapy may have a possible therapeutic efficacy on HER-2 positive patients with PD-L1 expression, while the high-quality clinical trials remained limited and were unable to join in our meta-analysis.
Conclusions
In summary, the addition of anti-HER2 targeted agents to chemotherapy can significantly improve the axillary downstaging rate. Chemotherapy combined with trastuzumab and pertuzumab had the highest efficacy in cN(+) patients, as they were ranked in the top two by the SUCRA analysis. Considering that carboplatin performed significantly better than anthracycline in single-target conditions, TCbHP is strongly recommended as the preferred choice for the neoadjuvant treatment in cN(+) HER-2 positive breast cancer. Since lapatinib and trastuzumab ranked top in the cN(−) group, our study looked to discover the potential value of TKI in cN(−) patients. Although no significant differences were found in the cross-comparisons, further analysis is needed in the future.
Supplemental Material
sj-pdf-1-tct-10.1177_15330338221150325 - Supplemental material for Axillary Downstaging and the Impact of Clinical Axillary Status on Efficacy of Neoadjuvant Therapy for HER2-Positive Breast Cancer: A Network Meta-Analysis
Supplemental material, sj-pdf-1-tct-10.1177_15330338221150325 for Axillary Downstaging and the Impact of Clinical Axillary Status on Efficacy of Neoadjuvant Therapy for HER2-Positive Breast Cancer: A Network Meta-Analysis by Yunzhao Luo, Hongchuan Jiang, Chengjiang Liu and Chao Zhang in Technology in Cancer Research & Treatment
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank Feng Xu for his valuable help on the statistical advice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors. An ethics statement is not applicable because this study is based exclusively on published literature.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Beijing Science and Technology Project (grant number ID161100000816002) to Hongchuan Jiang.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
